Abstract
Three-dimensional (3D) bioprinting is a promising and rapidly evolving technology in the field of additive manufacturing. It enables the fabrication of living cellular constructs with complex architectures that are suitable for various biomedical applications, such as tissue engineering, disease modeling, drug screening, and precision regenerative medicine. The ultimate goal of bioprinting is to produce stable, anatomically-shaped, human-scale functional organs or tissue substitutes that can be implanted. Although various bioprinting techniques have emerged to develop customized tissue-engineering substitutes over the past decade, several challenges remain in fabricating volumetric tissue constructs with complex shapes and sizes and translating the printed products into clinical practice. Thus, it is crucial to develop a successful strategy for translating research outputs into clinical practice to address the current organ and tissue crises and improve patients’ quality of life. This review article discusses the challenges of the existing bioprinting processes in preparing clinically relevant tissue substitutes. It further reviews various strategies and technical feasibility to overcome the challenges that limit the fabrication of volumetric biological constructs and their translational implications. Additionally, the article highlights exciting technological advances in the 3D bioprinting of anatomically shaped tissue substitutes and suggests future research and development directions. This review aims to provide readers with insight into the state-of-the-art 3D bioprinting techniques as powerful tools in engineering functional tissues and organs.
Keywords
Introduction
Organ shortage is a global issue that has intensified over decades due to increasing population aging, disease, trauma, or surgery. 1 Organ transplantation is one of the major procedures to replace non-functioning/malfunctioning organs. However, the availability of organ donors is limited. 2 Tissue engineering (TE) and regenerative medicine (RM) have been successfully applied in both research and clinical areas, with great progress in permanent replacement and cell-free tissue scaffolding in the past few decades.3–5 However, the fabrication of living and functional artificial tissue or organ substitute with bionic spatial complex, heterogeneous structure, intercellular interactions, and blood supply is difficult to achieve by traditional TE methods. 6
TE was integrated with multidisciplinary additive manufacturing (AM) technology to generate bio-engineered 3D structures that mimic the biological and functional complexity of native tissues/organs7–13; the method is called biofabrication or bio-additive manufacturing. Three-dimensional (3D) bioprinting is a remarkable technique used in TE biofabrication. With high precision, it controls the spatial distribution of biomaterial inks, cells, and biomolecules in predefined engineering constructs by a computer-aided design and manufacturing (CAD & CAM) process.14,15
In recent times, various 3D bioprinting techniques, such as extrusion-based inkjet, laser-assisted stereolithography (SLA), and digital light processing (DLP) techniques, have been developed to construct TE substitutes. Several research and reviews have been published covering different bioprinting techniques.14,16–23 Moreover, the number of articles published in peer-reviewed journals has constantly increased over the past few years (see Figure 1). These techniques fabricated 3D acellular scaffolds or implants with high-precise complex geometries capable of controlling the function of adherent cells in vitro and even growth tissue in vivo.
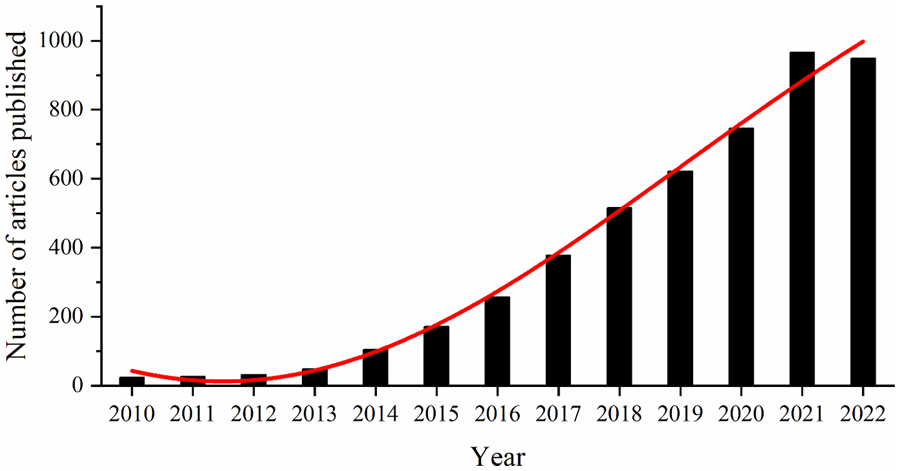
Time series of publications related to “3D printing and tissue engineering.” The statistical data for the last 13 years were collected from the Web of Science Core Collection database.
However, bioprinting of living and functional cellular constructs at clinically relevant dimensions remains elusive, encountering hurdles, such as the transition of materials from synthetic to low-viscosity biologically functional materials, integration of printed tissue with physiological vasculature network, incorporation of various cell types to recapitulate complex organ biology, and mechanical stability of cell-laden structures for long-term incubation for tissue maturation.21,24–28 Recently, substantial progress has been made in the biofabrication of living cell-laden, anatomical-shaped, and volumetric TE constructs by developing functional biomaterials. These biomaterials have advantages, such as good printability and cell-laden capability; innovation and improvement of the fabrication strategies; functional induction of specific cells or stem cells.12,29–35 This article reviews the current development in 3D bioprinting of tissue analogs and their translational implications.
Unmet need in biofabrication of cell-laden volumetric constructs
Biofabrication of living and functional volumetric constructs is a fated path for TE and RM toward clinical applications. Moreover, the anatomical-shaped tissue structures usually have complex and heterogeneous features and vascularized channel networks to avoid central necrosis. These features require high shape fidelity and the addition of perfusable channels or open macropores in the fabricated scaffolds.12,21,36,37 During biofabrication, the cells must be suspended homogeneously in an aqueous and soft biomaterial matrix environment, which supports the cell suspension. 38 Such a living cell-biomaterial blend is termed bioink. Bioink has strict requirements for physicochemical properties, such as maintaining the viability of cells during and after processing while providing an adequate extracellular matrix environment to support cellular functions.
On the other hand, the materials used to print acellular structures are used as surgical guides, permanent implants, or cell-free scaffolds for clinical TE applications and defined in vitro models for cell seeding. The combination of cell-free printing materials is termed biomaterial inks. The definition of bioinks and their distinction from biomaterial inks is explained by Groll et al. (See Figure 2(a)). 39 In this review, 3D bioprinting (or cell-laden printing) processes are described using the term bioink, while biomaterial inks are used in 3D printing.

The main challenge of 3D bioprinting cell-laden volumetric structures is limited by the biofabrication window. 38 In other words, a restricted range of material properties is suitable for printability with high shape fidelity and supporting cell function, as shown in Figure 2(b). The biofabrication window mainly describes inkjet- and extrusion-based 3D bioprinting techniques, 40 which the dependency of shape fidelity and thus resolution to the polymer concentration and cross-linking density in extrusion bioprinting, while cell viability exhibited the opposite relevantion with these factors. This concept has since then widely been appreciated in the biofabrication community.24,29,34,41 Even for the relatively new lithography-based bioprinting technologies, such as SLA and DLP, the bioinks are usually composed of a low-viscosity photo-cross-linkable hydrogel precursor 42 and photosensitive resin materials. 25 However, the contradiction between the stiffness of photo-cross-linked scaffold and the viability of cells in scaffolds remains. Thus, bioprinting functional volumetric constructs still have significant limitations due to the lack of bioinks and process techniques. Recent works to address these challenges can be categorized into five technical solutions for the construction of cell-laden scaffolds at clinically relevant dimensions.
Current technical solutions
The dimension or volume of fabricated scaffolds must be specified for 3D bioprinting of the clinically-relevant scalable constructs. In this work, the volume of fabricated cell-laden constructs >1 cm3 (⩾10 mm × 10 mm × 10 mm, L × W × H) is defined as clinical substitutes for application, termed as a volumetric scaffold. Here, the selected and summarized 3D bioprinting strategies for volumetric scaffolds do not form or describe the cell-laden volumetric or macroscopic structure in their studies, but in estimation, these techniques should be able to fabricate cell-laden volumetric constructs. According to their forming method and process, all the potential manufacturing process strategies for 3D bioprinting volumetric scaffold were classified into five technical solutions: Assisted bioprinting, multichannel multi-materials extruded bioprinting, freeform bioprinting, volumetric bioprinting, and in vivo bioprinting. The definition, principle, and relevant research of each technical solution are described in this section. The assisted bioprinting strategy consists of four different approaches: printing environment-assisted strategies, crosslinking-assisted strategies (including pre-crosslinking bioink and in situ crosslinking approaches), internal-enhancing strategies, and coaxial-assisted strategies. They are listed in Table 1; a short introduction about this part was presented. For more details, please refer to the article written by Kilian et al. 35 Each strategy is discussed in detail in the following sections.
Summary of assisted bioprinting strategies for biofabrication of volumetric structure and corresponding cases.

Assisted bioprinting strategy
Extrusion-based bioprinting is one of the most studied biofabrication processes. It can be considered the most promising and straightforward printing technique to fabricate TE substitutes or in vitro disease models.12,51–53 Post-fabrication, the bioprinted structures could be solidified through physical, chemical, or photo-crosslinking. 26 However, as mentioned in section 2, cells or cell aggregates are usually suspended in the aqueous and soft biomaterial to obtain low-viscosity bioink with high cell viability. However, it is difficult to print volumetric constructs directly. Using the assisted printing strategy to solve this problem improved printability. Herein, assisted bioprinting technical solutions are summarized. These solutions enhance the printability of cell-laden bioinks via different strategies to make the extruded cell-laden filaments construct volumetric scaffolds via strand-by-strand and layer-by-layer methods.
Four different effective assisted strategies are listed in Table 1:
(1) Printing environment-assisted strategies enhance the printability or self-supporting of bioinks by providing some specific dispensing environments (such as temperature, magnetic, light, etc.). In particular, this strategy is applied for temperature-sensitive biomaterials, which could cause immediate temporary stabilization of the extruded strands.
Duan et al. mechanically fabricated robust alginate/gelatin hydrogel valve conduits with anatomical architecture and directly incorporated dual cell types in a regionally constrained manner via this strategy, utilizing the thermo-sensitivity of gelatin. 54 Also, controlling the environment’s temperature enhanced the printability of human chondrocyte-laden gelatin methacryloyl bioink. 55 Although the printability of bioinks could be improved, the applications of this strategy are limited by the requirement of responsive biomaterials (such as the thermal-sensitive biomaterials), temperature control, and stability of printing environment.
(2) Crosslinking-assisted strategies include pre-crosslinked bioink and in situ crosslinking approaches. In the pre-crosslinking approach, bioinks could be partly crosslinked before extrusion by mixing crosslinkers into the bioinks or photo-crosslinking them to enhance the viscosity and supportability of the bioinks. 56 For instance, Hazur et al. developed pre-crosslinked alginate-based bioinks by adding CaCO3 to improve the printability for low-concentrated alginate-based bioinks (2% w/v), fabricating 3D volumetric scaffold (the height of around 5 mm) with open macroporous structure while maintaining high cell viability. 45 However, the pre-crosslinking degree of bioinks is an important factor for this strategy, which can easily block needles.
In the in situ crosslinking approach, the bioinks are crosslinked when extruded from the nozzle, forming strong and stiff strand structures to enhance printability. Commonly, chemical- and photo-crosslinking methods are applied. Tabriz et al. reported a partially crosslinked alginate hydrogel printed on the z-axis-movable platform in CaCl2. The printed structure could be crosslinked completely in situ along the z-axis in the bath. This biofabrication process assisted in forming volumetric structures, like large branched vascular structures. 57 Additionally, Ouyang et al. used the in situ photo-crosslinking approach to fabricate hollow tube and nose structures while maintaining highly encapsulated cell viability. 41 However, since the printed cell-laden structures can be exposed to crosslinking agents for long periods during printing, the effect of cell viability and the risk of DNA damage need to be assessed. 58
(3) Internal-enhancing strategies involve increasing or enhancing the viscosity of bioinks by increasing the concentration of biomaterials or adding other supporting biomaterials. Even though the pre-crosslinking approach can also be classified in this category, the internal enhancing strategy typically focuses more on the physical viscosity enhancement of the adjusting biomaterials. Schütz et al. developed alginate (Alg)/methylcellulose (MC) bioinks with good printability, maintaining high cell viability. 47 The main component, MC of the bioink, was mixed with low-concentrated Alg (3%, w/v) to enhance the viscosity to fabricate scaffolds in clinically relevant dimensions. During the crosslinking process of CaCl2, only Alg is crosslinked by calcium ions since MC cannot be crosslinked by divalent metal cations, defining and retaining the shape of the printed constructs. Thus the Alg/MC bioink combination showed high cell viability and shape fidelity. Meanwhile, eggwhite (EW) could also be applied to improve the cell response of Alg/MC bioink. 59 Further, Laponite was added into Alg-based bioink to enhance the printability and mechanical properties of printed structures for long-term cultivation. Large 3D macroporous structures (30 layers) with macropores in the lateral direction in the scaffolds were fabricated by the previously described bioink. 48 Liu et al. developed an albumin-rich alginate-based hydrogel ink, 60 which could only be used to fabricate structures with the assistance of in situ crosslinking agents. The authors tried to enhance the printability of the ink by temporarily adding gelatin. 61 This technique could fabricate large scaffolds, such as human ear models. The internal-enhancing strategy is a standard method widely used in extrusion-based bioprinting. However, the question of how to internally enhance a bioink efficiently while balancing printability and cell viability remains unanswered.
(4) Coaxial-assisted strategies use two or more bioinks extruded by coaxial nozzles to form a strong single strand. Common methods include the coaxial extrusion of a low-viscosity bioink (as the core) and a high-viscosity bioink or biomaterials ink (as the shell) to form a single core–shell strand. Moreover, the independent functions of the two different bioinks or biomaterial inks can simultaneously load drugs and fabricate functional TE constructs.36,49 Yeo et al. fabricated multilayered cell-laden structures using a core-shell nozzle and an aerosol crosslinking method. The cell-laden collagen bioinks functioned as the core, and alginate biomaterial ink as the shell to form cell-laden constructs. The cells showed higher viability in these constructs than pure alginate bioinks. 62 Using the same strategies, Liu et al. fabricated a 3D scalable islet delivery/implantation construct with good cell viability and structural stability. 49 Kilian et al. used the core-shell bioprinting strategy to load various differentiation factors into the cell-laden core-shell strands to construct osteochondral tissue engineering structures with excellent shape fidelity and cell viability. 50 However, the extruded core-shell strand usually has a large diameter, affecting the resolution of the resulting structures. The strand’s cell viability is also easily affected by shear stress. Consequently, each assisted bioprinting strategy has advantages and limitations for fabricating volumetric structures, and an optimal approach requires to be designed according to a research demand.
Multichannel, multi-material bioprinting strategy
Native tissues are highly organized, sophisticated, and heterogeneous structures comprising various components of extracellular matrix (ECM), functional living cells, and biomolecules. Hard tissues contain stiff mineralized matrices like bone.63–66 Although the various assisted bioprinting strategies mentioned in Section 3.1 construct volumetric structures, the soft bioinks are limited by the biofabrication window 38 and cannot easily mimic a suitable biochemical and biomechanical microenvironment of native tissue.
Multichannel multi-material bioprinting strategy displayed unique advantages to resolve the shortcomings mentioned above and even increase the throughput. 52 In this method, two or more printheads (or cartridges) with an array of nozzles were mounted on the multichannel bioprinter for loading and delivering various bioinks or biomaterials inks.52,67 The bioinks and biomaterial inks could be extruded via individual channels and deposited spatially according to predefined engineering constructs to form multi-material multi-cell composite volumetric scaffolds. Thus, multichannel bioprinting combined tough and robust cell-free biomaterial inks for mechanical support with soft cell-laden hydrogel bioinks. Using this technical strategy, Kang et al. developed a multi-nozzle printer called Integrated Tissue–organ printer (ITOP), 67 containing a multi-cartridge module that could print four different biomaterials, including two bioinks (See Figure 3(a)). Tissue constructs, such as ear cartilage (3.2 cm × 1.6 cm × 0.9 cm, with rabbit ear chondrocytes) and mandible bone (3.6 cm × 3.0 cm × 1.6 cm, with human amniotic fluid–derived stem cells), were successfully fabricated by multi-(bio)printing of stiff polycaprolactone (PCL) as the supporting material, and soft cell-laden hydrogel bioink, and Pluronic F-127 as the sacrificial component.
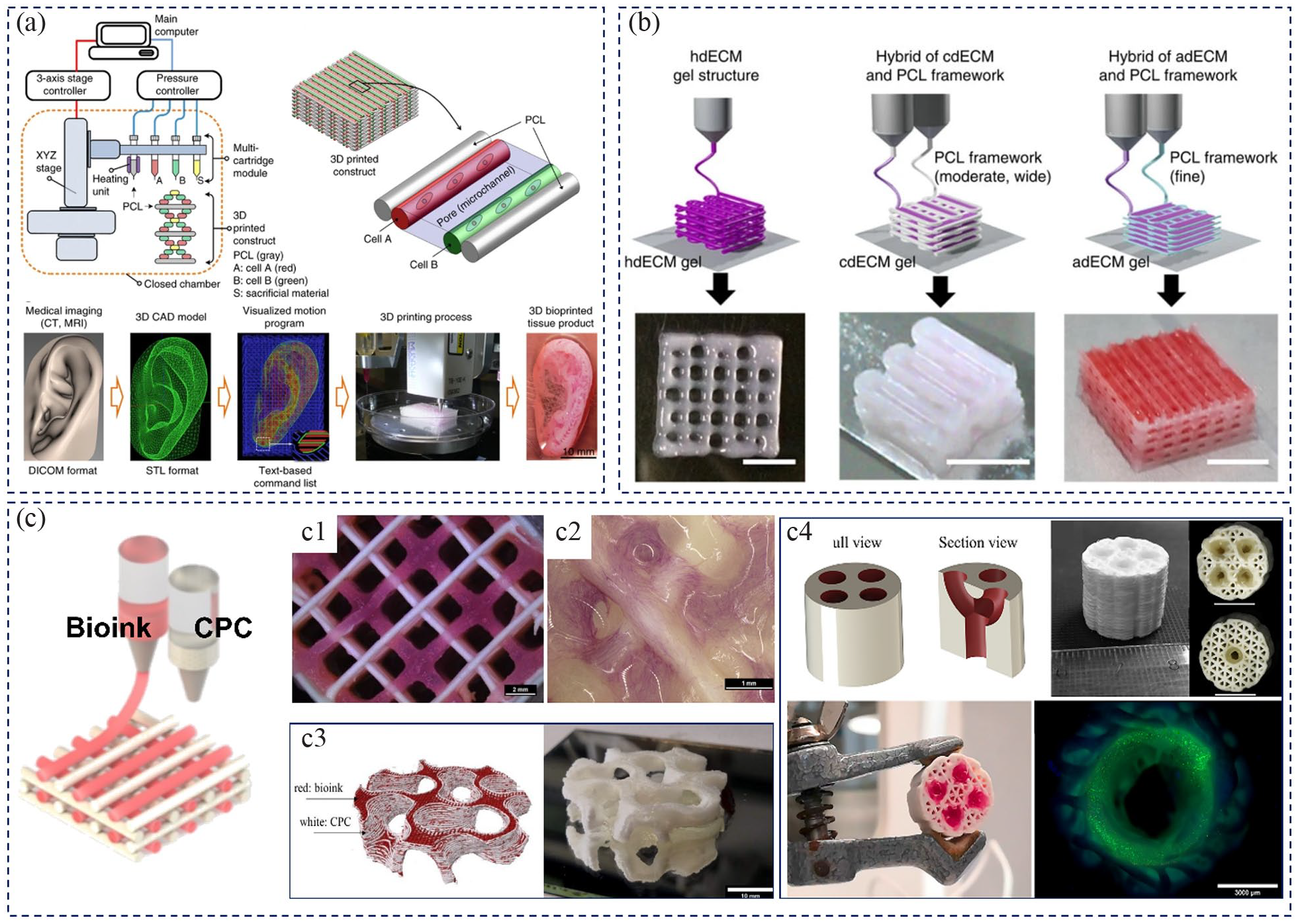
Multi-channel multi-material bioprinting strategy for the construction of living cell-laden volumetric structures: (a) Schematic illustration of ITOP system including four separate nozzles (two for the bioinks) and 3D (bio)printed basic pattern with multiple bioinks and the supporting PCL ink, as well as preparation process and bioprinted scaffold of target tissues of human ear; images reproduced with permission from Kang et al. 67 (b) The printing process of particular tissue constructs using soft dECM bioink combined with a robust PCL framework for the mechanical support (scale bar, 5 mm), which obviously exhibited the capability of this strategy for the fabrication of volumetric structures; images reproduced with permission from Pati et al. 68 (c–c4) Construction of functional biphasic volumetric bone substitute utilizing multichannel (bio)printing of mineralized CPC biomaterial ink and cell-laden plasma-based bioink: (c) schematic image of multi-(bio)printing biphasic scaffold and (c1) printed biphasic scaffold with open macroposity and (c2) ALP staining of cell-laden biphasic scaffold; construction of volumetric structure with complex geometries (c3) and complex prevascular structures (c4); images reproduced with permission from Ahlfeld et al.63,69
Following a similar strategy, Pati et al. improved the volumetric-formability of soft decellularized extracellular matrix (dECM)-based bioinks (hTMSCs) supported by PCL framework to construct large-volume scaffolds 68 (as shown in Figure 3(b)), such as adipose, cartilage, and heart tissue analogs.
To explore the clinical applications and construct a functional volumetric engineering scaffold, Ahlfeld et al. prepared cell-laden mineralized bone structures by utilizing multichannel nozzles of self-setting calcium phosphate cement (CPC) and cell-laden bioink 63 (See, Figure 3(c)). The biodegradable CPC had a bone-like composition of nanocrystalline hydroxyapatite. Mechanical strength was applied to construct biphasic bone structures with soft plasma-based bioink. 69 The open macropores were maintained in the fabricated large biphasic mineralized constructs (Figure 3(c1)) and contributed to the osteogenic differentiation of primary human osteoprogenitor cells (Figure 3(c2)). In addition, the combination of robust CPC and soft plasma-based bioink allowed the fabrication of more complex layer arrangements and advanced pore geometries like a gyroid structure (Figure 3(c3)). The complex perfusable prevascular structures in volumetric dimensions were also attained (Figure 3(c4)). Further, EW-enhanced bioink was combined with CPC to construct a partially mineralized volumetric bone tissue substitute.59,70 Thus, the multichannel multi-material bioprinting strategy was conducive to constructing cell-laden volumetric structures via the soft hydrogel bioinks supported by the strong and stiff cell-free biomaterial inks. The strategy could also be used to biofabricate functional TE substitutes, such as constructing appropriate ECM and vascularized channel networks.
Freeform bioprinting strategy
The strategies mentioned above (assisted bioprinting and multichannel multi-material extruded bioprinting strategies) significantly enhanced the complexity of the bioprinted volumetric construct and expanded the bioprintablility of bioink formulations. However, both strategies were based on the layer-by-layer printing process, making it difficult to achieve the complexity of the microstructures and 3D anisotropy as well as printing tissue mimetic soft hydrogels (elastic modulus below 100 kPa) or cells alone, 71 particularly for some soft tissues with overhanging structures. Therefore, another extrusion-based bioprinting strategy called freeform extrusion-based bioprinting was developed.72–75 This approach could overcome the abovementioned challenges by performing extrusion-based bioprinting within a support bath that could physically support the bioprinted structure.
Freeform extrusion-based bioprinting comprised two main parts: the bioinks for the target tissue scaffold and the supporting matrices for physical support for the target tissue scaffold formation. This technique has relatively open physicochemical demands for the bioinks and more strict requirements for the physicochemical properties of supporting matrices. For instance, the support material needed to behave solid-like to provide physical support. Yet, it had to become fluid-like under applied shear stress to allow the nozzle to move freely and self-recover immediately when the stress was removed to hold the printed structure in place.71,76 The fact that bioinks are extruded in supporting matrices minimizes the effect of gravity. Thus freeform fabrication can generate 3D complex and volumetric biological structures with 3D microvascular structures cooperation with fugitive inks, even suitable to low-viscosity bioinks or cell matrices. 75
Lee et al. developed a free-form reversible embedding of suspended hydrogels (FRESH) technique to direct-write hydrogels within a thermoreversible support bath composed of a gelatin microparticle slurry that provides support during printing (<25°C) and is subsequently melted away at 37°C to get engineered structures. This technique could engineer components of the human heart at various scales, from capillaries to the full organ. 73 Figure 4(a) shows the fabrication of a 3D human cardiac ventricle model with collagen bioink as the structural component with a high-density cell bioink. Further, a 3D multi-scaled perfusable and interconnected vascularized network channel, a tri-leaflet heart valve 3D model at adult human scale (diameter: 28 mm), and a neonatal-scale human heart with left and right ventricles and interior structures (37 mm × 55 mm) were fabricated by using FRESH, demonstrating the potential of this technique for constructing organ-scale volumetric tissue substitutes.

Freeform bioprinting strategy for fabrication of volumetric engineering constructs: (a) FRESH bioprinting approach, including schematic image of bioprinting human cardiac ventricle model and fabricated 3D ventricle structure, 3D multi-scaled perfusable and interconnected vascularized channel network, tri-leaflet heart valve 3D model at adult human scale and neonatal-scale human heart; images reproduced with permission from Lee et al. 73 (b) Schematic representation of the principle and process of freeform bioprinting within a developed fully transparent, cell-friendly, enzymatically/chemically degradable microparticulate support medium composed of alginate and xanthan gum, and bioprinted small-scale cellularized human heart with major blood vessels fabricated using two different bioinks; images reproduced with permission from Noor et al. 77
Following a similar strategy, Noor et al. 77 biofabricated cardiac patches and hearts using bioinks from patients’ own cells and tissues. Using an alginate-xanthan gum hybrid support media, they constructed major blood vessels within thick cardiac tissue using different bioinks (See Figure 4(b)). This work indicated the strong potential of the freeform bioprinting approach to construct personalized tissues and organs. Rapid progress has been made in this field since the development of freeform bioprinting techniques in 2015. 74 This technique opened a new avenue for modifying the physicochemical properties of scaffold bioinks. However, the high requirements of supporting materials limit its widespread application. Moreover, the precise control of longer needle in this strategy is also a challenge for biofabrication of volumetric construction. Nevertheless, the great potential of freeform bioprinting is clearly visible and is particularly viable for engineering human organ-scaled volumetric patient-specific tissue or organ substitutes.
Volumetric bioprinting strategy
Despite significant improvements made with these strategies, several limitations are still associated with lengthy extrusion-based layer-by-layer manufacturing methods, especially for the fabrication of volumetric constructs. The long printing process required to generate centimeter-scale constructs can impair scalability and clinical translation. 78 Printing time (which depends on the target structure size), printing resolution, printout composition, and fabrication technique are other critical parameters that may directly impact the fate of the incorporated cells. 28 Recently, vat-polymerization-based 3D bioprinting techniques (DLP and SLA) have been widely used in biofabrication.23,42 This technique exploits the versatile power of light to develop complex biomimetic cellular constructs within a broad range of size and resolution. 22 Based on these techniques and inspired by computed tomography (CT), a novel vat-based printing approach called volumetric printing (VP) was developed.79–82 This novel printing approach was also called tomographic volumetric additive manufacturing or computed axial lithography and enabled the fabrication of 3D geometries on a time scale of seconds. The term volumetric bioprinting (VBP) was used when the technique was applied in the biofabrication field. The principle of this method involved concurrent printing of all points within a defined 3D geometry by projecting a set of 2D images through a rotating tank containing a photosensitive resin from a different angle. The superposition of exposures from multiple rotational angles produced a 3D energy dose sufficient to solidify the material in the desired 3D geometry. The non-crosslinked photosensitive resin was washed away. The fabricated solid 3D structures could be harvested 80 (Figure 5(a)), and a 3D geometry could be formed in less than 1 min. With high printing speed, this technique is a powerful tool that could overcome the current limitations of extrusion-based bioprinting.

Volumetric bioprinting strategy for volumetric construct fabrication: (a) Graphical illustration of the computed axial lithography system (volumetric printing) and forming principle, the forming process of modeling structure and fabricated 3D geometry, scale bars: 10 mm; images reproduced with permission from Kelly et al. 80 (b) Overview of the volumetric bioprinting process with a cell-laden biocompatible resin in a rotating tank, and human auricle model printing process as well as printed hydrogel auricle structure; evaluation of the biocompatibility of such fabricate process and fabrication of cell-laden anatomical trabecular bone model with interconnected porous network (B1); and assessment of biofunctionality of bioprinted cells to synthesize new-tissue matrix though printing a meniscus-shaped implant with articular chondroprogenitor cells and relative results (B2); images reproduced with permission from Bernal et al. 79
For the application in biofabrication, an intriguing study was reported by Bernal et al., 79 demonstrating the feasibility of the volumetric printing technique in biofabrication of large living tissue constructs by applying cell-friendly hydrogel-based bioresins and a visible light laser-based printer. With this approach (Figure 5(b)), the complex, free-form structures could be generated within seconds from a volume of cell-laden hydrogels, such as the human auricle model (<23 s), which were difficult to produce through regular or conventional AM processes. The biocompatibility of these fabrication processes was demonstrated by volumetric bioprinting of cell-laden structures and even an anatomical trabecular bone model (cylindrical construct, 8.5 mm ` 9.3 mm). The high viability of cells (85%) incorporated in the structures was maintained after incubation for 7 days. Compared with other well-known bioprinting methods, there was increased metabolic activity, with no significant difference. Furthermore, vascular endothelial cells were seeded into the pore network of the constructs, leading to the formation of early angiogenic sprouts, typical of blood capillary network precursors (Figure 5(b1)). To assess the potential of bioprinted cells to synthesize a new-tissue matrix, a meniscus-shaped implant with encapsulated articular chondroprogenitor cells (1 × 107 cells/mL) was bioprinted and evaluated (Figure 5(b2)). The results revealed that cells could maintain high cell viability and increased metabolic activity over time. Neo-ECM was synthesized, resulting in a functional increase in the mechanical properties of the meniscal graft. Thus, this study demonstrated the potential application of volumetric bioprinting in tissue engineering and regenerative medicine for the biofabrication of living volumetric anatomical-shaped tissue or organ. VBP opened a new biofabrication window with far-reaching implications for future developments and applications. However, this technology also had limitations, such as low compositional complexity (including materials and cells) due to the process based on the selective curing of a single type, homogenous, pre-casted material. 28 While the native tissue and organ are extremely complex and contained different type of cells and various extracellular matrices. Therefore, how to address the multi-material and multicellular bioprinting and enhance the mechanical property of bioprinted structure are essential for its widespread applications.
In situ bioprinting strategy
For clinical applications, the fabricated 3D live constructs are either incubated in vitro for maturation before implantation or are 3D bioprinted in vitro and then directly implanted in vivo using surgical procedures.12,67,83,84 Besides the abovementioned methods, another novel and noteworthy approach, in situ bioprinting or in vivo bioprinting, was developed85–87 in recent years. This method directly printed constructs and transplanted them at the specific target site in the patient. Thus, the target constructs are located inside the patient’s body, serving as a living bioreactor.
Recently, a concept of intracorporeal or endoscopic 3D bioprinting was developed.85,86,88 The goal has been to develop tissue constructs using minimally invasive intravital bioprinting. Using this technology, Urciuolo et al. reported that the cell-laden photosensitive hydrogel structures were fabricated across and within the tissues of live mice. 85 As shown in Figure 6(a), the low-viscosity cell-laden hydrogels were injected into the desired organ sites and subsequently photo-crosslinked in vivo to form a 3D object by using near-infrared laser light according to the CAD/CAM system. Such intravital 3D bioprinting does not need and create by-products (e.g. additional supporting structures) and takes advantage of commonly available multiphoton microscopes to accurately position and orient the bioprinted structures into specific anatomical sites. This method fabricates complex structures inside live mice tissues and prepares volumetric constructs according to the defect size. The biocompatibility, cell-controllability engraftment, and tissue formation of intravital 3D bioprinting were evaluated by in vivo bioprinted dermis, skeletal muscle, and brain. Although exhilarating results were obtained using this strategy, there were several drawbacks, such as limited photosensitive hydrogels, 51 limited anatomical sites that can be exposed to a light source, 85 and appropriate cell and tissue-friendly light sources.
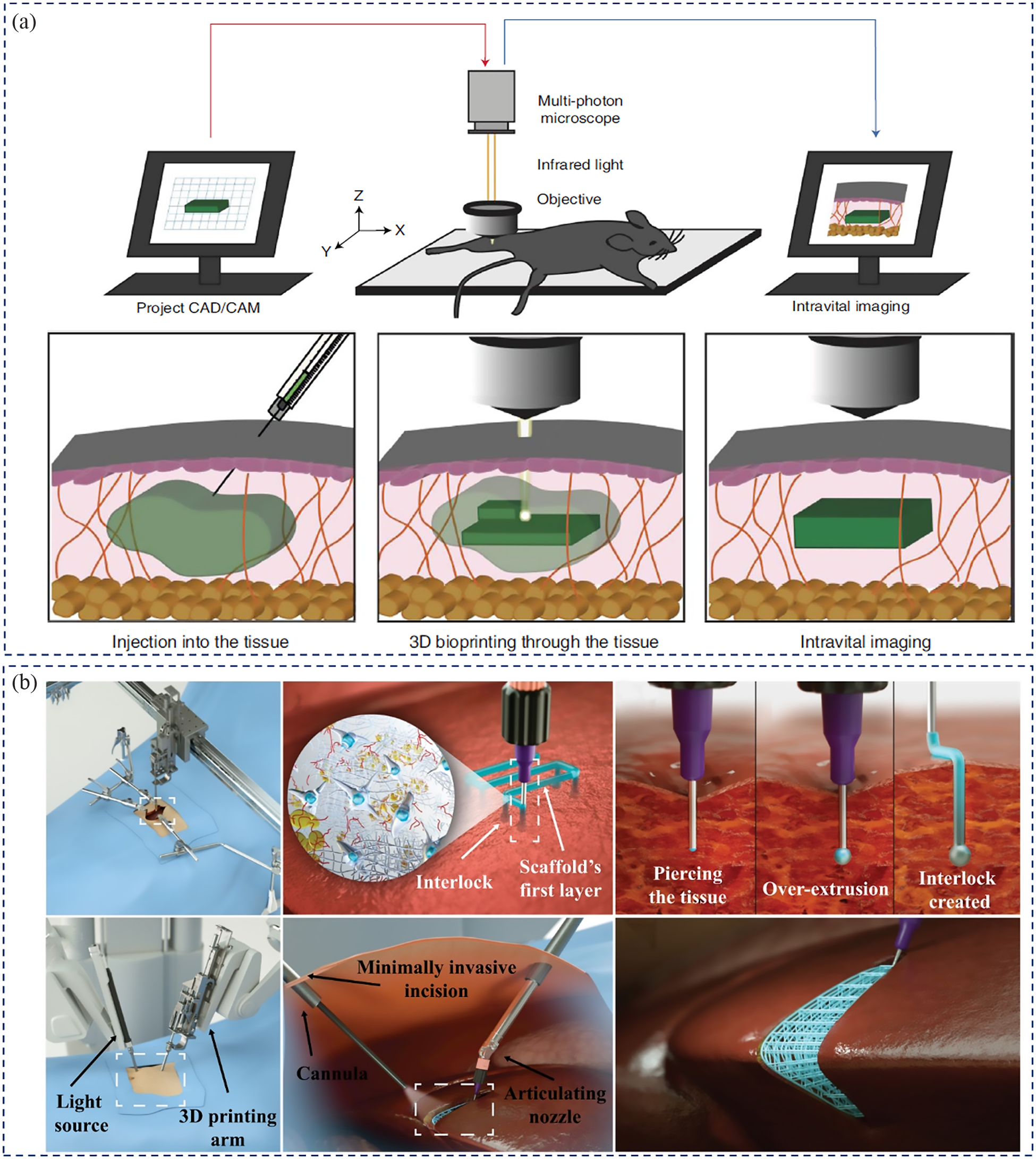
Minimally invasive in vivo bioprinting strategies: (a) Schematic illustration of intravital 3D (i3D) bioprinting setup and in vivo bioprinting processes, the i3D bioprinting required a multiphoton microscope equipped with a motorized x-y-z stage and a femtosecond near-infrared tightly-focused pulsed laser emission as well as CAD/CAM system; the fabricate process including injecting bioinks into target organs site of animal/human body, photo-crosslinking formation of 3D hydrogel objects, and intravital imaging for hydrogel identification and in vivo analysis; images reproduced with permission from Urciuolo et al. 85 (b) Schematic illustrating the concept of microextrusion-based direct-write 3D printing approach to print tissue structures directly in the patient body, developed bioinks was printed in target site via robotic-assisted minimally invasive DW 3D printing and using interlocking mechanism to improve the adhesion of scaffold with tissue inside the body, then in situ crosslinking of fabricated structures; a modeling application of filling a liver wedge resection; images reproduced with permission from Adib et al. 86
Adib et al. combined microextrusion-based direct-write (DW) 3D printing and visible light crosslinking system to fabricate 3D TE scaffolds inside a living patient in a minimally invasive manner. 86 Figure 6(b) illustrates the concept of this technique to 3D printing of a patient specific TE scaffold to fill a liver wedge resection defect. The DW 3D printing system was mounted on the operating room bed to 3D print patient-specific soft TE scaffolds in situ during surgery. The biomaterial inks or bioinks were bioprinted in the target site via a robotic-assisted minimally invasive 3D printing. Subsequently, the 3D-printed structure was crosslinked in situ to repair the tissue or organ. Thus, the advantages of this strategy include the repair or replacement of defective or malfunctioning tissue without tedious preliminary design and manufacturing processes, 28 long-term in vitro incubation, and the ability to construct volumetric structures with the support of surrounding tissues. However, the technique is also limited by the requirement to personalize the pre-prepared tissue to match the patient, both structurally and immunologically. 28 Because the bioprinting process and bioprinted structure were directly contacted with native tissues, and without post-fabrication and in vitro cultivation processes. Thus, how to design bioink with good operability and printability in vivo as well as good biocompatibility are essential in this strategy. Nevertheless, the in vivo bioprinting technology may be coupled with emerging optical-imaging-guided surgery or artificial intelligence surgical systems and applied in minimally invasive surgical techniques suitable for tissue repair and reconstruction.85,89,90
Clinical translation of bioprinted products
The ultimate goal of 3D bioprinting is to create living and functional tissue and organ constructs to repair or replace injured/necrotic tissues or organs. Thus the clinical translation of fabricated tissue substitutes is crucial and challenging. In orthopedics, for example, 3D printing can be applied to lesion and fracture repairs, arthroplasty implants, and custom casts. 91 However, most 3D (bio) printing clinical applications in humans have only involved non-living constructs designed to function as structural or space-filling prostheses.92,93 To the best of the authors’ knowledge, no bioprinted living tissues have been approved by FDA for clinical applications at the time of writing this manuscript.
Between 2010 and 2015, 80 approved additively manufactured medical devices received 510(k) clearance from the FDA, 94 by which the manufacturer “must demonstrate that the device is substantially equivalent to one legally in commercial distribution in the United States.” 95 These products were primarily used as implants (53% orthopedic-, 7% cranial/neuro-, 6% dental-implants) or surgical guides (34%). They did not yet contain a biological component (like mesenchymal stem cells), which would qualify them as living tissue. Due to the more intricate approval process, living tissues have not been approved yet. A 510(k) clearance is often insufficient when biologics are included, and a premarket approval or biologics license application is necessary. 94
Nevertheless, the implants were made of appropriate porous materials (72% Ti6Al4V, 2% CoCrMo, 5% CpTi, and 21% polymers) that could be further modified to increase the regenerative effect as a bone graft substitute. Some instances of increasing the regenerative effect of the scaffold are the addition of mesenchymal stem cells, 96 osteogenic differentiation-inducing RNAs, 97 growth factors, 98 and drug-loaded nanoparticles. 99 Witowski et al. 100 reviewed clinical trials utilizing 3D printing up to 2018 and found 92 clinical trials worldwide with N = 6252 participating patients. The number of clinical trials initiated has steadily increased from 1 (2012’) to more than 30 (2017’), reflecting the increasing momentum and maturity of 3D printing developments in the clinical field.
The two main reasons additively manufactured bone grafts are expected to prove superior in treating bone defects are the following: (1) They can be manufactured to fit the geometry of the bone defect precisely on-site 101 through automatically generated implant geometries based on computed tomography data of the patient. 102 (2) The implants can combine the beneficial properties of different materials manufactured in the same process, such as hydrogels with stem cells or growth factors for accelerated healing in combination with ceramic, metal, or polymer components for mechanical stability.91,103 Nevertheless, some challenges must be addressed for additive-manufactured bone grafts to be the clinical standard. On the one hand, the accuracy range of the models based on the CT scans must be improved with better segmentation methods, the current accuracy being 0.04–1.9 mm. 102 On the other hand, clinical standards for sterility and quality control must be achieved. Particularly achieving the desired sterility is challenging as the manufacturing process must occur under sterile conditions when cells are included. When cells are not included, using polymers as a scaffold substrate limits the useable sterilization processes.104,105
A positive side effect of 3D printers in the clinical setting is the ability to create inexpensive 3D models of the defect that help the clinician plan the surgery and explain it to the patient. Moreover, the capabilities of 3D bioprinting are at a stage where multiple biomaterials and cell types can be patterned into constructs approaching clinically relevant sizes and geometries.12,93 Nevertheless, there has been an increase in the transplantation of bioprinted living tissue in animals. The organ constructs, such as bioprinted bone,106–110 skin,111,112 and cartilage,113–115 were applied and evaluated. These bioprinted tissue substitutes were implanted into associated locations on animals to evaluate their biofunctionality, vascularization capability, and anastomosis with the host.
hAFSCs-laden volumetric 3D calvarial bone constructs in a circular shape (8 mm diameter × 1.2 mm thickness) were fabricated via a combination of soft bioinks and PCL/TCP reinforcing framework using the multichannel, multi-material bioprinting strategy. 67 The bioprinted living calvarial bone substitutes were implanted after in vitro incubation for 10 days into a rat calvarial bone defect for 5 months. The histology and immunostaining results demonstrated that many new vascular vessels grew into and throughout the implants to form vascularized bone tissue without necrosis, contributing to bone regeneration. Figure 7(a) shows the emergence of large blood vessels within the newly formed bone. This study demonstrated that combining different materials and cells via multichannel bioprinting holds great promise for generating the biological and mechanical properties essential for hard bone tissue, suggesting further potential for clinical applications.

(a) 3D bioprinting and transplantation of volumetric calvarial bone construct with PCL/TCP mixture and cell-laden hydrogel bioink: macro- and micromorphology of the printed calvarial bone construct, photographs after implantation (at day 0 and 5 months) and histological and immunohistological analysis; images reproduced with permission from Kang et al. 67 (b) In situ skin bioprinting concept: schematic images of skin bioprinting process (Ⅰ) and example of actual skin bioprinting process (Ⅱ); images reproduced with permission from Albanna et al. 112
Skin is the “first line of defense” and the largest tissue of the human body. It protects the body from external damage and, in the process, gets injured. Current repair strategies via 3D bioprinting skin substitutes include two main routes: transplantation of in vitro bioprinting skin construct with or without in vitro incubation and direct in situ (in vivo) bioprinting skin in the defect site.92,116 Albanna et al. reported a mobile skin bioprinting system based on in vivo (situ) bioprinting strategy combined with imaging technology for precise printing of dermal fibroblasts and epidermal keratinocytes directly into an injured site to form layered skin structure, leading to the rapid on-site treatment of extensive wounds 112 ( Figure 7(b)). In vivo experiments of in situ bioprinting cell-laden bioinks in murine and porcine full-thickness excisional wound models demonstrated accelerated wound repair and re-epithelialization. The regenerated tissue presented a dermal structure and composition similar to healthy skin, with extensive collagen deposition and mature vascular formation.
In recent times, many transplantations of bioprinted living tissue constructs in animals have been reported, leading to remarkable repair and healing effects of injured specific tissues.12,33,92,117,118 However, many limitations remain to be addressed before human clinical application. This exhibited that the solutions based on existing manufacturing volume structure and technical strategies still cannot meet the biochemical requirements of natural tissues. Moreover, these limitations also includes biomaterials and cell sources to prepare bioinks and vascularize volumetric tissues. 92 Most 3D bioprinted tissue substitutes are limited to small-volume structures, fewer cell types (one or two types only), and relatively simple structures with limited biofunctionality. Moreover, though this review discussed several technical solutions for fabricating volumetric structures and improving functionality, they are still far from meeting the biochemical requirements of native tissues.33,93,119 Cells are a key factor and a big challenge to the biofabrication of living tissue substitutes and successful transplantation in terms of cell sources and types, cell expansion, inducibility, or programmability. 92 Vascularization is also a major roadblock for fabricating engineered volumetric tissues. It influences the survival of implanted living tissue substitutes37,92,120,121 because, depending on the transplant site, the supply of oxygen and nutrients by diffusion kinetics after implantation is limited. 119
Nevertheless, significant progress has already been made in this area recently.60,61,122–124 Lee et al. demonstrated, as a proof of concept, the application of a human recombinant elastic bio-ink with which they could bioprint a vascularized cardiac construct that displayed the endothelium barrier function. 125 Therefore, constructing complex hierarchical vascularized networks in bioprinted tissue construct and establishing a connection with the host by surgical anastomosis or other technical solutions are issues that need to be addressed urgently. However, researchers have developed many new technologies and potential solutions in recent years. More details about these challenges, potential solutions, and other essential issues involved in the transplantation of 3D bioprinting can be found in the review article by Murphy et al. 92
Current challenges and future perspectives
The increasing demands on the biofabrication of human-scale volumetric structures, along with rapid development in 3D bioprinting, make the preparation of functional, transplantable, and complex tissues and organs indispensable. In the future, hybrid bioprinting strategies can be envisioned to meet the complexity of native tissue and organ structures and components. With various bioprinting strategies and novel bioinks, different functional tissue parts can be fabricated in advance, while entire and functional human-scale scaffolds can be constructed later. Among many strategies mentioned above, the multichannel multi-material bioprinting strategy, based on existing bioinks, shows great potential to form large-scale structures and to meet the complexity and heterogeneity of native tissues. In particular, the hybrid multichannel bioprinting of organic and inorganic inks is a promising method to form cell-laden constructs with gradients and high mechanical strength to mimic native hard tissues, as mentioned in Section 3.2. However, the long printing time and poor resolution of some irregular or complex overhanging structures fabricated through printing supporting materials need to be addressed in this strategy. The combination of multichannel multi-material bioprinting and freeform bioprinting, namely multichannel multi-material freeform bioprinting, has great potential to overcome these limitations, to achieve complex tissues construction.
In addition, one of the challenges faced by most existing extrusion-based multichannel multi-material bioprinting technologies is the long fabrication time when printing volumetric structures at clinically relevant sizes, which adversely affects cell viability in constructs. Volumetric bioprinting, an exciting approach for fast-printing speed, has great potential to fabricate volumetric structures within seconds. But it is still limited by low compositional complexity. Combining volumetric bioprinting with other bioprinting approaches to construct volumetric structures, which is called “volumetric bioprinting plus” models (VP+ biofabrication models), is a promising biofabrication strategy. In this way, the major part (or large part) of the whole tissue scaffold could be bioprinted with VP, and other parts (or small parts) can be fabricated with other bioprinting approaches. Such as the VP+ multichannel multi-material bioprinting models, which will solve the problem of low-compositional complexity of VP approach. Recently, Größbacher et al. reported a new strategy that combining volumetric bioprinting with melt electrowriting (MEW) to build geometrically complex objects, and successfully achieved the construction of multi-material and multi-cellular structures. 126 Beside the printing strategy, a smart bioprinting process is necessary via the addition of artificial intelligence (AI) or machine-learning approaches, such as computer vision, to predict or dynamically monitor and adjust to correct defects and errors, improving the printing quality and making the printing process more controllable and better visualized.
Ensuring sufficient vascularization of the bioprinted construct is necessary for the long-term viability of cells or newborn tissues in the constructs. The native-mimicking dynamic culturing system is essential to ensure the maturation and functionality of the bioprinted tissue substitutes. A bioreactor can not only maintain the viability of tissue constructs but also constantly monitor the culturing environment and physiological status, as well as specific stimulations (such as factors or microenvironment) for postprocessing tissue remodeling, maturation, and biofunction. However, developing a suitable bioreactor for each specific tissue type and mechanical stimulation is challenging due to the diversity and individuality of tissues and organs. The in situ bioprinting strategy, discussed in section 3.5, is an excellent alternative approach for transplanting bioprinted structures. The bioprinted structures are mounted in or on the specific defect and directly cultured in the patient’s dynamic natural “bioreactor,” avoiding complex postprocessing, long-term in vitro incubation, and contamination. Further advancements in situ bioprinting strategy can be coupled with emerging optical-imaging-guided surgery or artificial intelligence surgical systems (as discussed in section 3.5) with significant clinical applications, such as in vivo reconstruction and regeneration of tissues or organs immediately after injury or during surgery.
Conclusion
This review provides an overview of current developments in 3D bioprinting of tissue analogs and discusses their translational implications. The growing demand for tissue or organ replacement necessitates the development of engineered tissues or organs. When conventional methods fail to fabricate anatomically relevant functional tissues, 3D bioprinting becomes a powerful tool for engineering functional tissues and organs.
A literature survey reveals that most current research focuses on printing shape-mimicking tissue constructs, rather than replicating the functionalities of native tissues or organs in terms of the required structural, mechanical, biochemical, and biological features. Such features are a prerequisite for tissue repair and replacement. Advances in bioprinting and material development have led to the construction of high-resolution, complex, multi-material-based tissue constructs with precisely controlled architecture. In recent years, bioprinting research has thus focused on utilizing patients’ cells to print tissue constructs, with the aim of potentially reducing host tissue rejection. Another issue in bioprinted tissue constructs is vascularization, where small vascular networks and capillary-like channels must be generated before implantation.
Multiple studies have demonstrated the performance of printed implants in preclinical animal models, revealing their great potential for tissue regeneration. Despite significant advancements in 3D printing and notable developments in bioink formulation and pre- and post-printing processes, there are still limitations that need to be addressed, such as engineering complex shapes and human-scale functional tissue or organ substitutes. In conclusion, it is an exciting time to be involved in engineering bioprinted products as clinically viable substitutes for tissue regenerative medicine, with great challenges and expectations ahead.
Footnotes
Author’s contribution
All authors contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge funding support from the National Natural Science Foundation of China (grant no. 52175474 & 52275498), the China Scholarship Council (no. 202006890054, Suihong Liu), Natural Science Foundation of Sichuan Province, China (2022NSFSC1510), ICTS “NANBIOSIS,” in particular by the Drug Formulation Unit (U10) of the CIBER in Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN) at the University of the Basque Country (UPV/EHU) in Vitoria-Gasteiz, and the National Research Foundation of Korea (2018K1A4A3A01064257, 2021R1A5A2022318, RS-2023-00220408).
