Abstract
Osteoarthritis (OA) is a chronic degenerative osteoarthropathy. Although it has been revealed that a variety of factors can cause or aggravate the symptoms of OA, the pathogenic mechanisms of OA remain unknown. Reliable OA models that accurately reflect human OA disease are crucial for studies on the pathogenic mechanism of OA and therapeutic drug evaluation. This review first demonstrated the importance of OA models by briefly introducing the OA pathological features and the current limitations in the pathogenesis and treatment of OA. Then, it mainly discusses the development of different OA models, including animal and engineered models, highlighting their advantages and disadvantages from the perspective of pathogenesis and pathology analysis. In particular, the state-of-the-art engineered models and their potential were emphasized, as they may represent the future direction in the development of OA models. Finally, the challenges in obtaining reliable OA models are also discussed, and possible future directions are outlined to shed some light on this area.
Keywords
Graphical abstract

This review discusses the development of osteoarthritis (OA) models, including animal and engineered models. In particular, the state-of-the-art engineered models and their potential were emphasized, as they may represent the future of OA models.
Introduction
Osteoarthritis (OA) is a chronic degenerative osteoarthropathy characterized by chondrocyte apoptosis and an imbalance in extracellular matrix (ECM) synthesis and metabolism.1–3 The manifestations of OA predominantly consist of articular cartilage damage and degeneration, articular capsule hyperplasia and hypertrophy, and secondary accidental synovitis and bone remodeling.4–7 Microscopically, OA disease involves reduced proteoglycan content, deformed morphology of articular chondrocytes, increased inflammatory factors in the articular capsule and synovium, and overexpressed matrix metalloproteinases. 8 Studies have shown that the causes of OA mainly include aging, obesity, trauma, metabolic syndrome, lack of exercise, congenital joint abnormalities, and other related genetic factors, among which aging is deemed the largest risk.9–12 The aging of joint tissue leads to senescent chondrocytes, oxidative stress, mitochondrial dysfunction, and age-related changes in cell signal transduction and the biomechanical microenvironment.13–16 Figure 1 shows the major factors that may induce OA.

An overview of OA pathogenesis, treatment, phenotypes, and the importance of the OA model. (a) Structure and phenotype of the normal joint and schematic representation of hyaline cartilage morphology and structure. Reproduced from Thorp et al. 17 (b) Several pathogenic mechanisms contribute to the development of early-stage OA, such as injury, obesity and age, which can cause a significant increase in the expression of inflammatory factors. Reproduced from Hu et al. 18 (c) In addition to significant changes in the expression of signaling pathways associated with OA, late symptoms of OA gradually manifest. Reproduced from Hu et al. 18 (d) The ultimate symptom of OA, and current treatments for OA mainly include medical therapies and lifestyle changes. Reproduced from Lei et al. 19 (e) The phenotype of normal rat chondrocytes was stained with hematoxylin and eosin (H&E) and Safranin-O/Fast Green staining. Reproduced from Zhang et al. 20 (f) The phenotype of OA (monosodium iodoacetate (MIA)-induced OA) rat chondrocytes was stained with H&E and Safranin-O/Fast Green staining. Reproduced from Zhang et al. 20
As a heterogeneous disease, OA is widely believed to be the result of a combination of multiple risk factors, with early disease-related changes detected in cartilage and subchondral bone. 21 Additionally, OA can be caused by the improper adjustment of complex intricate biomechanical and biochemical interactions between various structures, which will destroy the normal homeostasis of joints. 22
The clinical treatment options for OA are largely confined to symptomatic treatments, which may merely relieve symptoms such as pain and delay disease progression.23,24 Medication is the main option for the early and medial stages of knee OA through intra-articular injection of corticosteroids or nonsteroidal anti-inflammatory drugs, 25 such as sodium hyaluronate (SH), 26 polydopamine (PDA), 27 platelet-rich plasma (PRP), 28 etc., or the use of systemic analgesic drugs. 29 Arthroscopic debridement is also an effective method for the early and medial stages of knee OA. 30 In the advanced stage of the disease, surgery is usually required to partially restore joint function, namely, joint replacement surgery. 31 Recent advances in OA treatment include stem cell therapy, antiaging approaches, and other targeted therapies.32–36 Some typical treatments for OA are also shown in Figure 1.
Although there are different ways to alleviate OA, it is still challenging to completely treat this disease. The reason for this situation is that the pathogenesis of OA has not been clarified. To treat OA, it is necessary to start with the pathological causes of OA and determine how it happens. Given the ambiguity of OA etiology and limited treatments, the development of reliable models for OA that accurately reflect the physiological and pathological characteristics of human OA is of great significance.
Animal models of OA
As a conventional paradigm for pathological and pharmacological studies, animal models play a dominant role in all aspects of OA research. 37 The purpose of animal models is to replicate the characteristics of human OA in animals, thus providing a living system to understand the disease mechanisms and evaluate potential treatment options.
Animal species for OA models
A variety of small animals and large animals have been used to develop OA models. Small animals are often used to generate disease models because of their effectiveness, low cost, ease of handling, and ease of genetic manipulation. However, due to their small size, their anatomical and tissue structures are much different from those of humans.38,39 In comparison, the anatomy of joints, cartilage morphology and biomechanical function of joints in large animals are closer to those in humans, thus providing more clinically relevant data. In fact, a series of disease-related experiments conducted with large animals may be more meaningful because they have a longer life span and enough time to slow disease progression and end-stage osteoarthritis. Table 1 summarizes the main experimental animal species used for OA models, as well as their advantages, disadvantages, and distance to the clinic.
Advantages and disadvantages of different animal species for OA models.

Generation of animal OA models
Animal OA models can be generated by spontaneous approaches and induction methods, as depicted in Figure 2. The common OA generation methods and their advantages and disadvantages are summarized in Table 2 and discussed below.

Animal OA models can be generated by spontaneous approaches and induction methods, the latter of which include surgical induction, chemical induction, mechanical induction, and genetic induction.
Common generation methods of animal OA models and their advantages and disadvantages.

Spontaneous animal OA models
Spontaneous models are the simplest approach to simulating OA in small animals such as mice. In spontaneous OA models, the natural aging process in the joints of the tested animals is the main cause of OA. In addition, spontaneous obesity, excessive exercise, decreased estrogen secretion and other related diseases are also common causes of spontaneous OA models. 64
OA can be spontaneously caused by age and natural environmental factors, and the development of OA in this situation was very similar to that of nontraumatic OA in humans. Theoretically, it can be regarded as the result of natural wear in the life process. 65 It is of great clinical value to study OA pathogenesis and evaluate therapeutic methods through these spontaneous animal OA models.
Spontaneous OA models have multiple advantages, such as convenient execution, no requirements for professional equipment and surgeons, and no uncertainties caused by the medical process.66,67 Although spontaneous models are widely used to study OA progression, such models often suffer from high uncertainty. The lower incidence of spontaneous models compared to other models and the greater variation in disease progression over time and environment require more animal models for statistical analysis to validate, yet often this is infeasible in some cases because of the increased costs. 68 Another issue lies in that skeletal development is often different between animals of different ages, even between different breeds of the same species. Moreover, the unpredictable time frame for OA generation and the uncontrollable severity of OA among different test animals are headaches. Additionally, accurate quantification of pain analysis in spontaneous approaches is challenging, as animals show fewer signs of pain than humans. 47 These limitations have greatly limited the application of spontaneous OA models.
Induced animal OA models
Animal OA models can be generated by various induction approaches, such as making the joints unstable, changing joint mechanics, locally changing cellular metabolism and promoting inflammatory processes. 69 The induced OA models can better represent the occurrence of secondary OA in humans, usually posttraumatic OA. 70 Induction approaches can be divided into surgical and nonsurgical approaches.
Surgical induction
Surgical induction methods for OA generation in animals mainly include anterior cruciate ligament transection (ACLT), ovariectomy (OVX), meniscus instability, collateral ligament transection, local cartilage injury, etc., 71 as shown in Figure 3(A). 72 OA was generated in rabbits through ACLT, which destroyed the cellular physiology and cartilage structure of rabbit cartilage, resulting in joint imbalance and ligament transection. Images of rat and rabbit cartilage after ACLT staining with H&E and Alcian Blue are shown in Figure 3(B). 73 Bilateral ovariectomy is a common surgical method for female animals to induce OA. 74 The mechanism is that ovariectomy may lead to decreased secretion of estrogen, which is a protective factor to prevent osteoporosis and osteoarthritis. 75 3D microcomputed tomography (micro-CT) revealed obvious bone destruction under articular cartilage. 76 Yoon et al. 77 induced OA by dissecting the right knee joint of 8-week-old C57BL/6 mice and destabilizing the meniscus and tested the full advantage of PUM1 for cartilage tissue expression in a DMM-induced OA model the next day using lentiviral-encoded pLenti-GIII-CMV-PUM1 or pLenti-GIII-CMV-NO for intra-articular injection. Compared with the control group without treatment of the meniscus ligament, OA symptoms had a significant tendency to ease.

Induced animal OA models. (A): Surgical induction. (a) Anterior cruciate ligament resection-induced OA; (b) collateral ligament transection; (c) meniscus instability, visible damage after induction; (d) OVX/DMM-induced OA in rats. Created with FigDraw. (B): Surgical induction. Images of rat and rabbit cartilage after ACLT staining with H&E and Alcian Blue. Reproduced from Kim et al. 73 (C): Chemical induction. Morphological changes and increased apoptosis of chondrocytes in the condyle induced by MIA injection. (a) TEM view of condylar chondrocytes; (b) TUNEL assay and H&E staining results. Reproduced from Wang et al. 78 (D): Genetic induction. Immunomarkers of dorsal root ganglion (DRG) and substance P (red) and pERK (purple) in the spinal cord 21 days after intraarticular injection of collagenase indicate that collagenase may activate neuronal nociceptors and injury pathways in the peripheral and central nervous system, leading to inflammation of joint injury. Reproduced from Ita et al. 79 (E): Mechanical induction. Graphical representation of the cyclic compression load tibia in the model. (a) Placed in a loading device when hind limbs estimate the position and loading direction; (b) applied the load of a single cycle diagram, according to keeping the size and peak load, load rate and intervening in the peak and baseline to keep time; (c) a graphic representation of five different loading schemes. Adapted with permission from Christiansen et al. 80
Systemic arthritis can be prevented by using surgical induction approaches, which have a high success rate in the generation of OA. Although small animals easily undergo surgery, surgical induction is irreversible in terms of damage to the animal, and the severity of the resulting OA cannot be accurately predicted, which can limit the reproducibility of the results. Large animals, whether macroscopically and microscopically, are far removed from human anatomy. Additionally, the high demands of animal experiments on the professionalism of the laboratory staff and the environment make the experiments very different in terms of reproducibility as well as standardization and make it difficult to perform in some laboratories.
Chemical induction
Intra-articular injection of toxic or proinflammatory chemical factors is one of the main approaches for the nonsurgical induction of OA. These chemical factors can either induce the degeneration of matrix compounds or inhibit the activity of chondrocytes, which serves as the trigger mechanism for the development of OA. 81
Injection of papain into joints can lead to osteoarticular degeneration. 82 Glyceraldehyde-3-phosphate dehydrogenase (GAPD) in chondrocytes is inhibited by MIA, which blocks the aerobic cellular glycolysis pathway and causes cell death. MIA leads to the rapid occurrence of arthropathy with obvious histological changes within a few days after MIA injection. 83 Serra and Soler 84 demonstrated that a 0.4 mg MIA dose could induce significant cartilage and synovial damage in mid-stage trials. Figure 3(C) shows that after MIA treatment, the condylar chondrocytes were changed from a polygonal morphology to a more shrunken one with vacuolar degeneration and increased apoptotic cells. 78 Intra-articular injection of collagenase is one of the common methods for OA induction; it leads to the decomposition of collagen fibers in cartilage and reduces the collagen matrix in tendons and ligaments, leading to rapid instability of joints and obvious cartilage lesions in a short period, thus gradually forming arthritis symptoms. 85 Collagenase may activate neuronal nociceptors and injury pathways in the peripheral and central nervous system, leading to inflammation of joint injury, as shown in Figure 3(D). 79
Dashnyam et al. 86 injected 20 μL of complete Freund’s adjuvant (CFA; 15 μg/mL) into the superior joint space to induce inflammation-associated OA in rats. Nanoceria (nCe) were injected into the synovial cavity at different concentrations after successful induction of inflammation, demonstrating the effectiveness of nCe in treating OA. However, the limitations of nCe are that it is dependent on specific redox reactions, and the therapeutic effect may vary greatly for different species. Further research is needed for future clinical translation.
Although moderate OA may self-heal without any treatment, the results of intra-articular injection of chemical factors are typically irreversible, which is inconsistent with the condition in the human body.57,87 The limitations of chemical induction models are that they have a different pathophysiology from posttraumatic OA, which means that chemical induction is not effective in examining the usual progression of the disease in humans. In general, chemical induction does not produce symptoms that mimic true OA as other models do, and the dynamic mechanical microenvironment of regular OA caused by mechanical load and wear in daily living activities is even less likely to be simulated by chemical induction. Instead, the approach is mainly used for observation and painological assessment and is able to intervene in pain or reduce/exacerbate inflammatory effects. 41
Mechanical induction
Posttraumatic OA can be triggered by applying an external mechanical load without skin rupture or joint capsule invasion. An intra-articular fracture of the tibial plateau is a traditional way to study the early changes in OA. The principle of this method is to damage the joint through external forces, thus inducing OA. 80 Periodic articular cartilage tibial compression is also a common noninvasive method for OA induction to study bone adaptability. This technique has also proven to be an effective method for studying mechanical loading after the degeneration of articular cartilage. 85 A graphical representation of a cyclic tibial compression loading model is shown in Figure 3(E). 85 In addition, anterior cruciate ligament fracture caused by tibial compression is also adopted in the scheme of inducing OA generation. 88
However, the disadvantage of mechanical induction is that it is not possible to define an appropriate mechanical stimulus, although it can to some extent cause structural damage, such as to the cartilage layer and the function of some tissues, for instance, the mitochondria, leading to the imbalance of superoxide and superoxide dismutase 2 (SOD2), thus resulting in cartilage degeneration. 89 Moreover, the molecular mechanisms that cause OA have not yet been fully explained, which limits the therapeutic strategies for these risk factors. 90
Genetic induction
Genetic modification approaches can produce osteoarthritis at specific sites in animal models by inserting or deleting specific genes when the genetic factors of OA are determined.91,92 Additionally, genetic modification is of great importance to explore the relationship between the genotype and phenotype of the disease. 93 Staines et al. 94 highlighted the genetic factors affecting articular chondrocytes and subchondral bone development and bone health utilizing an STR/ORT mouse model, which is particularly useful in studying OA development. Kang et al. 95 found that miR-204, a microRNA transcription factor that induces aging, was significantly upregulated in OA cartilage. Expression of miR-204 in ectopic articular tissue can trigger spontaneous cartilage loss and OA generation.
Animals with gene modification typically tend to be attacked by OA as a result of genetic manipulation, such as chondrocyte differentiation or apoptosis, cartilage matrix degradation, and subchondral bone metabolism. 96 Compared with the naturally occurring OA model, the location of lesions on the gene modification model is controllable, which can be its best advantage. However, the disadvantage is that the specific timing of OA onset cannot be predicted. In addition, the genetic induction method is expensive, and the operation is rather complicated, requiring dedicated reagents and equipment and skilled professionals.
Other induction approaches
In some cases, artificial distortion of environmental factors may induce OA. For example, Louer et al. 97 found that obesity aggravated OA after intra-articular fracture in mice fed a high-fat diet. In addition, deliberate high-intensity exercise, which gives rise to irreversible joint damage, is another important way to cause OA.
Summary of animal OA models
The advantages of animal models include rapid onset of action, a good ability to control disease severity, human-like anatomy and disease progression, and an ethically superior ability to test and track large numbers of subjects. 51 Unfortunately, none of the animal models used to construct OA can faithfully replicate the symptoms and pathologies of human OA due to the complexity and heterogeneity of OA and inherent species variation.
In general, the key differences in the potential impact of species-specific OA pathology are highly inconsistent, and the histological characteristics of each animal OA model also vary widely. The drugs and treatments involved in animal OA models are often not used in humans out of the consideration of the species relationship because of the lack of translational effects of the relevant models and the clinical results of the drugs in human subjects. Moreover, no matter what kind of animal models should comply with the Ethical Considerations and Reporting Guidelines (ECRG), it is impossible to achieve standardization and uniformity, only to refer to the prescribed guidelines as much as possible to achieve experimental details. 38 Considerations including cost, availability, housing, and length of the experiment need to be weighed against the outcome measures required to address the question being asked. Additionally, the potential for genetic modification is limited.13,98 Notably, large species generally require specialized laboratories and personnel to carry out high-level experiments, including experimental licenses and a high cost of ownership, which is why large animals are generally not used for routine experiments. Although animal models are still the most common in OA research, more rational animal models and appropriate generation approaches are desired for the development of more reliable in vivo OA models. 99 Additionally, standardized animal models are being pursued in this area.
Engineered OA models
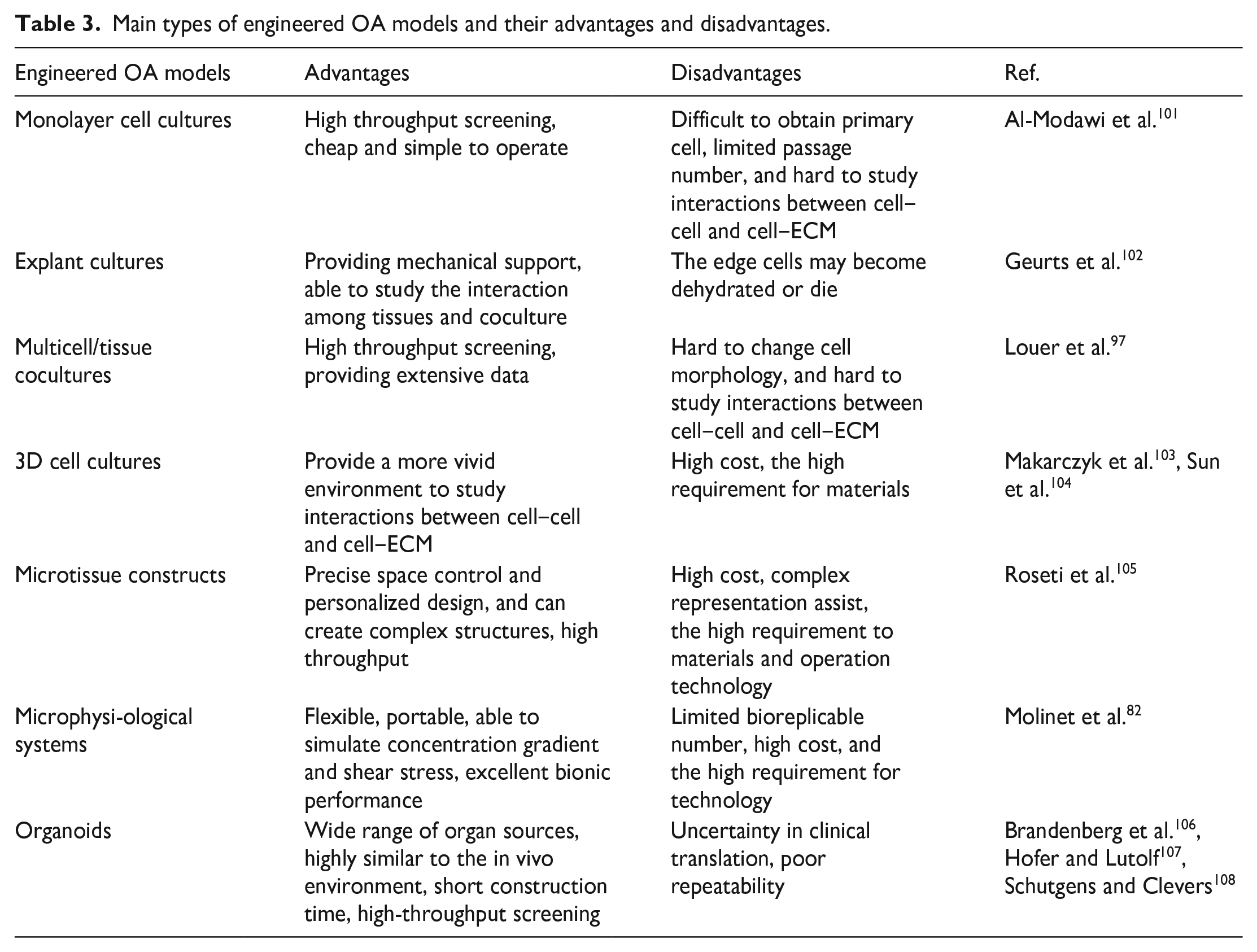
Apart from animal OA models, establishing OA models in vitro by engineering approaches is an emerging area in OA research. 100 Engineered models may represent future directions with great potential. Engineered OA models can be divided into several types: monolayer cell cultures, explant cultures, multicell/tissue cocultures, 3D cell cultures, microtissue constructs, microphysiological systems, and organoids, as depicted in Figure 4. Table 3 summarizes the advantages and disadvantages of these engineered models.
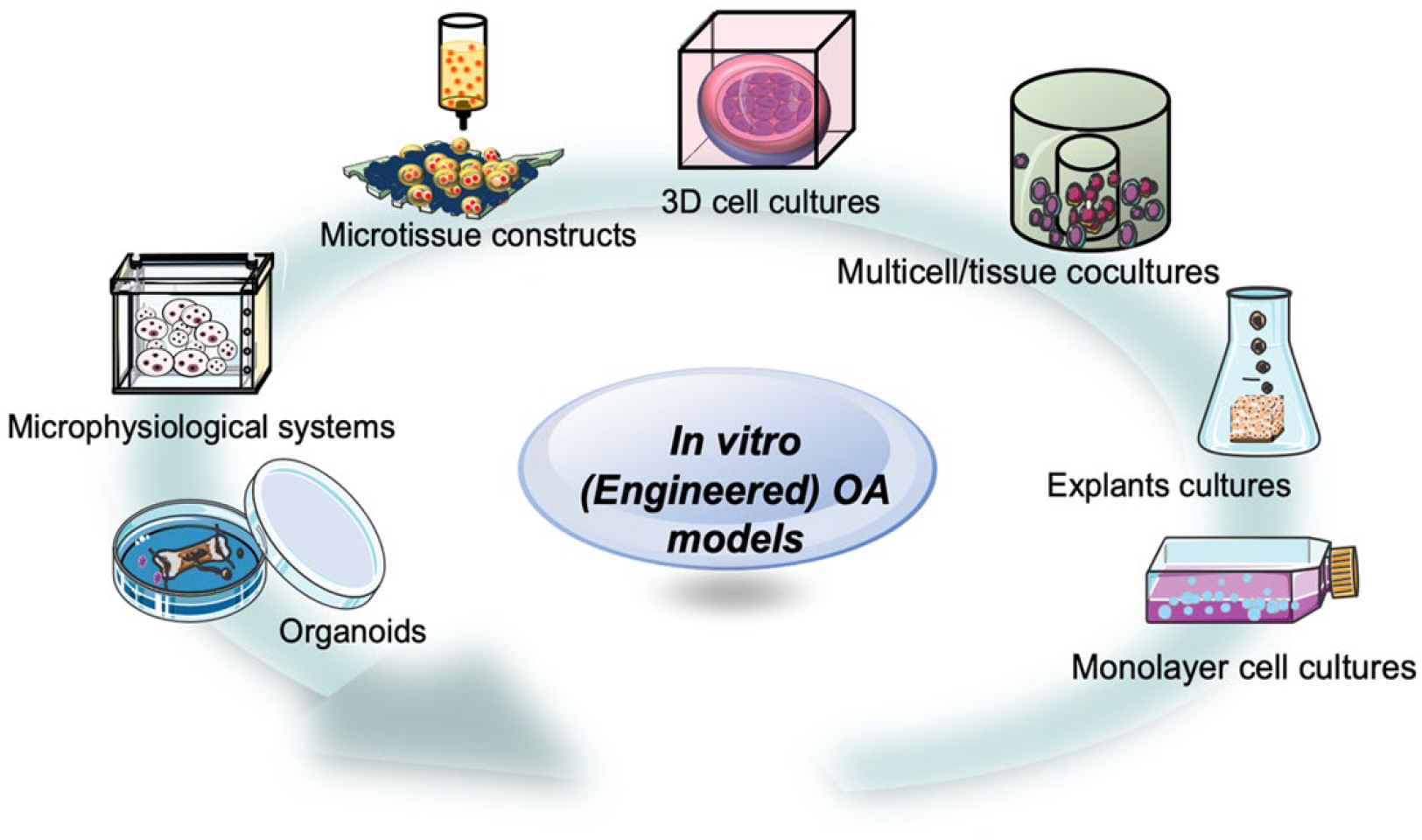
Schematic of the main types of engineered OA models. Engineered OA models have been developing from conventional and simple approaches such as monolayer cell cultures to more advanced and complicated systems such as organoids.
Main types of engineered OA models and their advantages and disadvantages.
Generation methods of engineered OA models
Monolayer cell cultures
The conventional and simplest engineered OA models are monolayer cell cultures of primary cells, which require various nutrients and necessary growth factors for cell development and proliferation. Monolayer cell cultures can be used as OA models to screen cartilage protective compounds, which aid in attenuating catabolic factors involved in articular cartilage degradation. 109 They are practical and economical tools in OA research, which makes them qualified for preliminary investigation of the underlying mechanisms of disease pathology and an effective assessment of specific compounds on cell phenotypes.
Yoon et al. 77 extracted mesenchymal stem cells (MSCs) from the knee joint of patients with OA and found that the cells are prone to oxidative stress and aging during in vitro expansion, limiting the regeneration ability of cartilage tissue. However, if the expression of PUM1 is retained during the expansion process, it can protect MSCs from senescence induced by H2O2, improve the potential for cartilage formation, and increase therapeutic effectiveness.
However, primary cells with limited passage numbers are difficult to obtain because their morphology may change during cell differentiation induction, and phenotypic cells are also likely to be polarized. Moreover, the OA models of monolayer cell cultures have limited potential in studying cell‒cell and cell‒ECM interactions and tissue load. 101
Explant cultures
Culturing explants usually requires the extraction of different human tibial cartilages with smooth or fibrous shapes. Because the tibial cartilage has not been stripped from the bones, it can remain in natural mechanical constraint for a long time.110,111 After a period of culture in vitro, methods for OA induction can be performed later on such explants.112,113 The construction and shape of the model barely changed under pressure several times, so it can represent the joint in the different parts and different periods of OA severity. Grenier et al. 114 constructed cartilage explants to simulate early OA injury. First, they used collagenase for cartilage preprocessing and then subjected them to repeated mechanical stress to simulate OA injury. The invention of this model provided new information on the mechanisms related to cartilage degeneration.
Explant culture models can be used to observe how inflammatory factors affect the knee joints, study the compression of overload, and provide data on mechanical load effects on the bone structure during disease development. However, one of the major challenges is how to avoid the environmental impact during bone tissue extraction. Cartilage cells at the resection margins have the risks of dehydration, deformation, bacterial infection, and even death. It should also be noted that explants in an artificial environment defy the study of mechanical loading or angiogenesis effects after surgical resection. 115
Multicell/tissue cocultures
As with monolayer cell cultures, cocultured models can be used to study multiple possibilities arising from experiments at the same time, and they are promising to provide extensive data on the pathological mechanism of OA.116,117 It is easy to apply cytokine and osmotic stimulation in cocultured models, such that these models have favorable prior conditions for OA-related induction.118,119 Furthermore, the coculture of the synovial membrane and chondrocytes is a common method, as the synovial membrane is the main source of osteoarticular media.120,121 Coculture of the synovium of OA patients with healthy cartilage explants can increase the expression of IL-1, IL-4, IL-7, IL-8, IL-10, IL-13 and osteoprotectin (OPG), a condition that is similar to the synovial fluid of OA, and reduce the generation of specific antigen-like genes in cartilage. 122 The most popular strategy to cause OA is by employing IL-1β. Designing carriers activated by IL-1β can reduce catabolic action, while IL-1β also increases reactive oxygen species through transcription factors, which ultimately causes cell apoptosis. 123 Araújo et al. 124 explored the cocultures of cartilage explants with synoviocytes and constructed an in vitro OA coculture model after stimulation with hydroxyapatite and IL-1β. The established OA model was then used to evaluate the pharmacological activity of Amentadione (YP). The drug could reduce the inflammatory/mineralizing conditions in the cocultures. Kato et al. 125 purified exosomes from the medium of human synovial fibroblasts (SFB) extracted from normal knee joints. SFB stimulated by exosomes or IL-1β was used to treat cartilage explants extracted from normal knee joints of mice. Detection of OA-related gene expression by real-time quantitative polymerase chain reaction (RT‒qPCR) showed that SFB stimulated with IL-1β significantly upregulated the release of human matrix metalloproteinase (MMP-13) and other polysaccharides in cartilage explants, which can induce OA morphology changes. Moreover, exosomes can transmit pathogenic signals among different cells in the OA model. 126 Shajib et al. 127 obtained engineered cartilage tissues formed from coculture of bone marrow-derived stromal cells (BMSCs) and expanded articular chondrocytes (AChs). The engineered tissues were then used to study the impact of enzyme treatment on cartilage tissue integration and matrix remodeling.
The coculture approaches allowed us to explore various factors affecting the progression of OA. However, it is worth noting that there are also limitations with cocultures, including cell morphology changes and difficulties in studying cell‒cell and cell-ECM interactions directly, which are similar to those with monolayer cultures.
3D cell cultures
3D cell cultures for OA models can be basically achieved through scaffold-dependent and scaffold-free approaches. At present, scaffold-dependent approaches for 3D cell cultures have been widely explored to develop OA models in vitro.
For instance, 100 pg/mL of IL-1β was administered to chondrocytes that were implanted in sodium alginate gel, and the expression of the aggregative proteoglycan gene was downregulated by 2–3 times when compared to the control group, which merely contained medium, while the transcription level of MMP-3 was increased by 12 times. Gene-6 induced by tumor necrosis factor-α (TNF-α 128 ) had a 7-fold increase in expression. 129 Murab et al. 130 investigated methods for the stabilization and localization of cytokines in scaffolds of sulfonated modified silk proteins. The proinflammatory cytokine IL-1β was then noncovalently solidified by the N-terminal end of TNF-α binding to sulfonated modified filaments, and human articular chondrocytes were stimulated to form an in vitro OA model Sun et al. 104 Human primary chondrocytes were grown for up to 21 days in a silk-protein porous scaffold before stimulating them with IL-1β, TNF-α, or macrophage medium (MCM); both cytokines and MCM upregulated MMP1, MMP3, MMP13, etc. This 3D cartilage tissue culture system aids in understanding the causes of illness.
A variety of factors in 3D scaffold-dependent culture, such as proteolytic enzymes, NO gas, hypoxic environments, and the application of mechanical stimuli, can lead to the degradation of cartilage tissues and induce OA models. Roncada et al. 131 achieved 3D culture of MSCs using 3D collagen and alginate scaffolds, which supported chondrogenic differentiation of MSCs and allowed deposition of collagen type II and aggrecan without the use of growth factors. It was further verified that chondrogenic differentiation and matrix deposition were attributed to the effect of stiffness. The stiffness and mechanical properties of the scaffolds play a crucial role in the formation and performance of cartilage tissues from MSCs, which suggests that the application of some specific mechanical stimuli may lead to the degradation of cartilage tissues and produce OA. However, a limitation of 3D scaffold-dependent culture is that it cannot fully replicate the biological properties of real ECM to support cartilage sculpting. 103
In scaffold-free cultures, anchorage-dependent cells must be loosely attached to form multicellular spheroids under suspension culture conditions. 132 3D spheroid cultures allow cells to proliferate in all directions and better simulate the internal environment, which provides a powerful but simple tool for the study of chondrogenesis and preliminary screening of OA drugs. 133 At present, scaffold-free methods for preparing 3D spheroids include microplate culture with ultralow adsorption surfaces, suspension drop culture, rotating culture, and magnetic suspension culture. Among these, 3D spheroids attached to microplates with ultralow adsorption surfaces are a simple and reducible substitute for microculture, which can better simulate the chemical and physical-mechanical environment of cells in cartilage tissues. 134
In general, 3D culture models can be used to observe and record the formation process of OA in a more tridimensional way and study cell‒cell and cell‒ECM interactions to a certain extent. 135 This approach allows cells to grow in a more biomimetic environment, which is also of certain research value for simulating the mechanical environment of joints in vivo.136,137 However, compared with 2D culture models, the deficiency is that the proliferation rate of cells in a 3D environment decreases, and the method of inducing OA formation needs to be further improved.
Microtissue constructs
Microtissue engineering has been a potent tool for creating 3D tissue models with properties resembling those of natural tissues. It may promote cell proliferation and differentiation by introducing microstructures. Modular bottom-up design and microenvironmental techniques have become hallmarks of OA microtissue construct models in recent years. Among different bottom-up approaches, the 3D printing technique is currently the most commonly applied in the development of in vitro OA models. 138 The construction of in vitro models of OA through 3D printing technology enables innovations in cell biology and material structure as well as modes of induction. 139 Moreover, by changing key settings, biological 3D printing technology may be utilized to alter the mechanical and chemical properties of the printed object, making it possible to create intricate in vitro bone tissue models. 103
Deng et al. 140 prepared Mn-TCP scaffolds by doping Mn2+ into β-TCP (β-tricalcium phosphate) using 3D printing technology. In the in vitro experiments, rabbit chondrocytes and mesenchymal stromal cells (rBMSCs) were implanted into the printed Mn-TCP scaffolds, and inflammatory cytokines (IL-1β) were introduced. RT‒qPCR results showed that the HIF (hypoxia-inducible factor) pathway was activated by Mn2+, with increased levels of HIF-1α and significantly lower expression of autophagy-related genes (MMP3 and MMP13 as well as all inflammatory mediators aggrecanase (ADAMTS5)) compared to the control, verifying that Mn2+ not only promotes chondrocyte proliferation and differentiation but is also able to preserve chondrocyte activity in the pathological environment of the OA scaffold constructed in vitro (Figure 5(A)). As a novel method for cartilage healing, 3D printing technology restored the structure of the cartilage while also causing the development of OA. 135 Gong et al. 141 constructed a cell-loaded bilayer scaffold (GelMA-IL-4+PCL-HA) in vitro by 3D printing technology. IL-1β and macrophage medium (M1CM) were added to mouse chondrocytes in culture to induce an inflammatory effect, and the induced mouse chondrocytes in this bilayer scaffold were inoculated to form an in vitro model of OA. The upper scaffold slowly released IL-4 during cell culture to reduce inflammation, and then this scaffold was implanted into rabbits, showing that this scaffold also promoted bone regeneration and cartilage defects (Figure 5(B)).
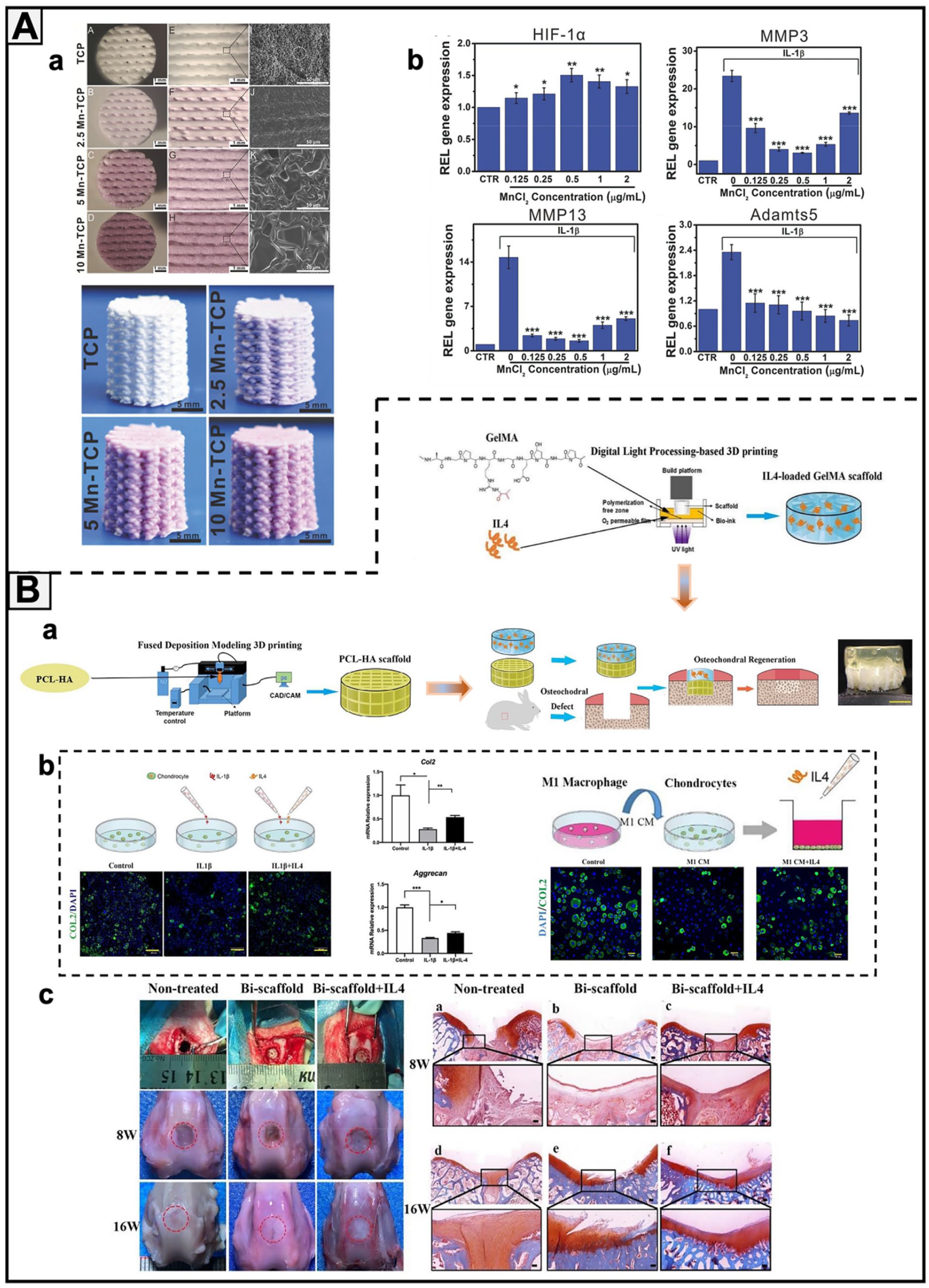
Some in vitro OA microtissue systems based on bioscaffold architecture. (A) Effect of 3D-printed Mn-TCP scaffolds with different concentrations on IL-1β-induced OA models. (a) 3D-printed surface morphology and microstructure and TCP scaffolds with different manganese contents; (b) the level of inflammation was significantly increased after treatment with IL-1β, and the addition of Mn was able to significantly decrease the inflammation level. Adapted with permission from Deng et al. 140 (B) Advantages of IL-4-loaded bilayer 3D-printed scaffolds in an IL-1β-induced inflammatory environment in vitro and in promoting rabbit osteochondral tissue regeneration in vivo. (a) Schematic diagram of how the stent structure was prepared (top layer: IL-4-loaded GelMA stent; bottom layer: PCL-HA stent; the top and bottom layers are connected by 3 μl GelMA gel after 10 s of UV curing); (b) during the culture of mouse chondrocytes, IL-1β and M1CM were added to induce an inflammatory state, and then the cells were inoculated into the scaffold. After 2 days of culture, the upper GelMA scaffold slowly released IL-4, and RT‒qPCR and fluorescent live-dead staining showed that IL-4 was able to protect cellular activity and had anti-inflammatory effects; (c) the bilayer scaffold was implanted into rabbit cartilage defects, and the efficiency of the bilayer scaffold for cartilage defect repair was assessed by contrasting macroscopic images of defects in three groups (untreated/spontaneous regeneration group, double scaffold group, and bilayer scaffold group) at 8 and 16 weeks postoperatively, as well as by staining with safranin-O. Adapted with permission from Gong et al. 141
3D bioprinting, as a promising approach to producing microtissue models, has been used to fabricate tissue-engineered bone and cartilage, with the advantages of creating gradient structures and properties. 142 Based on this, in vitro OA models can be induced by the addition of various proinflammatory stimuli, such as inflammatory cytokines and mechanical loads. Despite its high cost, 3D printing is a high-throughput technique with enormous potential for producing in vitro OA models.
Microphysiological systems (MPS)
In vitro simulation of the 3D structures and function of tissues and organs using advanced microfluidic technology, namely, organ-on-chip technology, has produced successful biological models for physiological and pathological studies. 143 Additionally, it is also an effective method to simulate OA in vitro and a crucial option for clinical OA drug screening. 144
Occhetta et al. 56 developed a microphysiological microchip system integrating a polyethylene glycol (PEG) hydrogel 3D microenvironment and mechanical actuation to simulate the creation of an in vitro on-chip cartilage (COC) model and OA model. Human chondrocytes (HAC) isolated from a donor were embedded in a PEG gel and injected into a microfluidic chip chamber. OA phenotypes were induced based on 3D chondrocyte culture through hyperphysiological compression. The OA model was then used to predict the use of anti-inflammatory and anti-metabolic medications, and the results were found to be compatible with preclinical findings already in existence. A microfluidic chip containing chondrocyte-laden hydrogels and enabling gradient compression was developed by Paggi et al. 145 The cell viability was evaluated under different compression stimulations, and it was found that hyperphysiological compression was more lethal to the cells, as shown in Figure 6(A).

Some samples of in vitro models of OA implemented on MPS. (A): A microfluidic device to study the effects of mechanical loading and vascularity on the progression of OA. (a) Mechanically driven chamber to simulate joint loading (top). Chondrocyte culture chamber representing cartilage (middle) media perfusion channel (bottom); (b) chondrocyte viability in response to normal and hyperphysiological mechanical stimulation: no stimulation (top), 800 mbar (healthy stimulation) (middle), and 1000 mbar (hyperphysiological stimulation) (bottom). Reproduced from Paggi et al. 145 (B): A drug delivery device for achieving OA treatment on a microfluidic device. (a) The design of OA models inspired by the superlubricated surface of ice that consists of a contiguous and ultrathin layer of bound water; (b) H2O2 was used as an inducer of oxidative stress to produce osteoarthritis disease and was treated by spray with 2-methylacryloxyethyl phosphate choline (MPC)-modified methacrylate anhydride-hyaluronic acid (HAMA) drug-delivery particles. Adapted with permission from Yang et al. 146 (C): Modeling OA on microfluidic devices. (a) The ideas for studying monocyte extravasation as a treatment for OA; (b) physical view of microfluidic organ-on-a-chip model. Reproduced from Mondadori et al. 147 (D): Implementation of an in vitro equine arthritis model on a microfluidic chip. (a) Microfluidic nutrient gradient-based in vitro equine chondrocyte culture model with staining characterization; (b) gene expression levels in inflammatory microtissues after 24 h and 1 week of incubation with 50 pg/mL TNF-α and IL-1β. Adapted with permission from Rosser et al. 148 (E): The “miniJoint”—OA modeling with a human mesenchymal stem cell-derived microphysiological system. (a) The miniJoint chip comprises engineered adipose tissues (AT), synovial-like fibrous tissues (SFT), osteochondral tissues to simulate fat tissue, synovium, and cartilage-bone complex in the native knee joint, and the timeline (up to day 63) of generating minijoint culture and modeling joint disease; (b) the inflamed joint model was created by challenging the SFT via the addition of IL-1β to the fibrogenic medium (FM). Reproduced from Li et al. 149 (F): An osteochondral tissue chip from iPSCs for OA modeling. (a) Schematic of the induction process. Following the formation of OC (osteochondral) or CH (chondral) after 28 days of differentiation, 1 ng/ml IL-1β was added into the top flow and perfused only onto the cartilage component; (b) assessment of IL-1β-treated OC or CH samples by Alcian Blue staining. Reproduced from Lin et al. 150 (G): Osteochondral constructs derived from human bone marrow stem cells (hBMSCs) induce osteoarthritis with a strong catabolic response induced by IL-1β, providing a high-throughput platform for testing potential disease-modifying anti-osteoarthritic drugs (DMOADs). Reproduced from Lin et al. 151
Microfluidics-based in vitro models of OA offer the possibility of building 3D cell aggregates, and various strategies have been developed to generate and culture microtissues in vitro, enabling a more systematic approach to inflammation induction and cell or tissue culture conditions. 152 By using H2O2 as an inducer of oxidative stress, Yang et al. 146 created an OA model in a microfluidic device and inhibited the inflammatory response with drug delivery by spraying. This work is made up of a contiguous incredibly thin layer of bound water (Figure 6(B)). The microfluidic microarray constructed by Mou and Jiang 152 used mononuclear macrophages as a mediator, with a particular focus on the interactions and mechanisms of action between synovial and articular cartilage and synovial fluid in the presence of inflammation, providing an effective entry point for the study of OA mechanisms, as shown in Figure 6(C).
Additionally, Rosser et al. 148 cultured equine chondrocytes encapsulated by fibrin hydrogels on microfluidic chips based on different nutrient gradients and exposed them to an inflammatory environment (50 pg/mL TNF-α and IL-1β) to establish an in vitro OA model. The levels of MMP13, ADAMTS5 and other inflammatory factors were significantly upregulated after 2 weeks of culture in the control group compared with the healthy group. Finally, 60 μg/mL tretinoin was used to treat microtissue inflammation, and significant experimental results were obtained, demonstrating the applicability of microfluidic 3D chondrocyte culture as a simple disease model for OA (Figure 6(D)). Lozito et al. 153 envisaged an in vitro 3D microsystem that was sized to fit a 96-well plate culture model, with separate compartments forming microspaces for synovial and bone components, the introduction of bioactive agents and cells, and tissue/media compartments. The aim of constructing an in vitro OA microphysiological system through extreme mechanical damage, exposure to an inflammatory factor environment, etc., is to screen for DMOAD drugs.
Li et al. 149 reported a microarticular system (miniJoint) that contained an engineered osteochondral complex, synovial fibroblastic tissue, and adipose tissue. This integrative microfluidic system allowed communication between different tissues, which still maintained their respective phenotypes. When exposed to IL-1β, the miniJoint exhibited physiologically relevant changes similar to OA inflammation in the human body. This microfluidic OA model may provide a powerful tool for joint pathology and novel therapeutic interventions (Figure 6(E)). 149
Lin et al. 151 built on this previous work by making hBMSC-derived constructs in situ and placing them in bioreactor culture to induce differentiation into cartilage for 4 weeks, while IL-1β was also added into one side of the culture medium that supplied the lumen. The addition of IL-1β leads to a strong catabolic response in the cartilage layer, which along one cartilage layer lumen affects and amplifies to some other lumen (e.g. osteogenic layer), leading to an increase in MMP13, for instance, and a specific expression of tissue inflammation levels (Figure 6(G)). This bioreactor will help to elucidate the role of various tissues in OA.
Lin et al. 150 triggered induced pluripotent stem cells (iPSCs) into mesenchymal progenitor cells (iMPCs) and then inoculated the iMPCs in a gelatin scaffold and placed them in a dual-flow bioreactor in their laboratory. The microphysiological osteochondral tissue microarray was characterized after 28 days of differentiation, and IL-1β was introduced into the side chamber supplying the cells with culture medium to generate the OA. The model was then tested in a system using NSAIDs to better investigate the pathogenesis of OA and the screening of DMOADs, as shown in Figure 6(F).
In comparison to other in vitro OA methods, microfluidic systems, which play a significant role in the research of OA simulation, exhibit greater advantages in the overall biomimetic properties of the biological joint, the experimental safety performance, and the physiological environment of the whole system for articular cartilage. 103 These benefits are mainly related to the characteristics of controllable fluid flow in organs-on-chips and the stable delivery of specific nutrients to cells or tissues, which enhance the differentiation, function, and long-term survival of chondrocytes.154,155 However, there are still limitations to the microphysiological system, including the amount of biological replication and the lack of appropriate biomechanical stimuli, which will require further research.128,156 For instance, the presence of air bubbles in microfluidic channels may harm cells, hinder the fabrication and control of chips, and make it difficult to completely remove them.149,157
Organoid
Organoids are becoming an emerging in vitro tissue engineering technology approach that is crucial in simulating the intricate biological operations of organs in vivo. The ability to imitate pathology at the organ level gives organoid cultures for disease modeling a clear advantage over conventional cell culture techniques, opening up new avenues for the testing and screening of investigational medications. 158 Bone organoids are 3D microstructures based on biomaterials that have been differentiated from stem or progenitor cells through appropriate induction measures to have certain bionic capabilities (including self-renewal and tissue differentiation). Several significant phases in the formation of bone organoids are depicted in Figure 7(A) and (D).159,160 An important application of organoids is the construction of bone disease models, which currently involve mainly osteoporosis models, OA models (artificially induced bone defect states mainly), bone tumor models, bone deformity models, and osteomyelitis models. 159 Herein, the creation and use of OA organoid models are the focus.

Organoid applications and implicated OA models. (A): A schematic of the biofabrication technique for bone organoids. Reproduced from Chen et al. 159 (B): Generating 3D-cultured organoids for preclinical modeling and treatment of OA, including 3D organoid culture, chondrogenic media and OA modeling. Reproduced from Sun et al. 161 (C) Bone-cartilage organoids as a model of joint inflammation and analysis of the agonistic and antagonistic effects of the A2A receptor. (a) Schematic representation of the process achieved by the inflammation program; (b) appearance of bone-cartilage organoids on live cell imaging at harvest; (c) expression of Collagen Type II (COL2) (green) and osteocalcin (OSC) (magenta) by immunofluorescence; (d and e) immunofluorescence staining shows expression of FOXO1 and FOXO3 in organoids; (f) expression of aggrecan (ACAN) and Sex-Determining Region Y Box 9 (SOX9) genes was detected by RT‒qPCR and normalized to organoids cultured in Chondro-Ost media only; (g) western blot detection of osteoclastogenesis. Adapted with permission from Abraham et al. 162 (D): The key applications of organoids include bioengineering, human biology studies, disease modeling, regenerative medicine and drug development. Reproduced from Lee and Son. 160 (E): An organ model for staphylococcal infection of human bone samples and investigation of the effect and efficacy of a microporous β-TCP ceramic loaded with hydrogels and clindamycin on infected human bone tissue over 28 days. Adapted with permission from Kuehling et al. 163
Sun et al. 161 constructed 3D organoids for preclinical modeling and treatment of OA (Figure 7(B)) based on self-assembled organoids of synovial mesenchymal stromal cells (SMSCs) in 3D culture mode and used miRNA and mRNA sequencing to phenotypically analyze them. The findings suggested that miR-138 might play a mediating role in the cartilage-specific effects on the development of OA and better chondrogenic properties of SMSC organoids, providing a new entry point for OA organoid modeling. It is known that the main symptoms of OA disease are concentrated in the skeletal and cartilaginous tissue layers; therefore, a self-assembling skeletal organoid would integrate the development of skeletal and cartilaginous tissue as well as an effective physiological model for disease prediction and development of therapeutic agents. A “minijoint” mixed human skeletal organoid was reported by Abraham et al. 162 (Figure 7(C)). The skeletal organoids were treated with IL-1β (5 ng/ml) for 24 h to induce an inflammatory model, and adenosine (A2A) receptor agonists were tested as therapeutic agents. The results showed that the levels of Forkhead Box O 1 (FOX01) and FOX03 were significantly upregulated (FOXO1 has been shown to antagonize the proinflammatory effects of IL-1β and reduce cartilage-degrading enzymes), thereby inhibiting the serious trend of the inflammatory response.162,164
Kuehling et al. 163 have proposed a microporous structure loaded with hydrogels (β-TCP loaded with hydrogels (alginate, alginate-di-aldehyde (ADA)-gelatin)) for use in a model of bone-infected organoid (samples were infected with S. aureus ATCC29213), and the targeted drug delivery treatment of the bone infected modules by porous hydrogels can effectively alleviate the symptoms of infection and is an excellent alternative to in vitro organoid model tests (Figure 7(E)). It is an excellent alternative to in vitro organoid models for testing and animal models.
Bone organoids show considerable promise for disease models that reveal biochemical, pathogenic, and therapeutic pathways, as well as to reproduce and improve the physiological functioning of cells and tissues. 165 When compared to previous in vitro models, organoid technology is more revolutionary in terms of complexity, spatial participation, and the biological microenvironment because it eliminates the moral dilemmas associated with animal models.166,167 It has always been difficult to build multiple organoid platforms, but the role of organoids in bridging the gap between preclinical and clinical trials is undeniable.
Challenges and future directions of engineered OA models
Compared to in vivo models, in vitro engineered OA models will be able to overcome more difficulties in the future and become an important mode for clinical translation, as shown in Figure 8. However, there are still many challenges with engineered OA models. The main limitation is that the current engineered models are not mature and only represent a prototype rather than the entire structure of the human bone and joint and cannot create a physiological microenvironment that is consistent with human OA. Engineered models are required to overcome the morbidity of tissues in specific situations, the need for large quantities of immune-acceptable cells to populate synthetic scaffolds, and the challenges faced under unnatural conditions due to the extensive expansion of cells in vitro, such as the lack of a reliable and reproducible source of cells and the loss of phenotype and gene expression of cells due to expansion. 168

Animal platforms and tissue engineering platforms for OA models. Animal models generally have limitations, such as large variation in species, ethical issues, low throughput, and long experimental cycles. In contrast, in vitro engineered OA models in the long run may solve these problems and exhibit a significant validity with advantages of low ethical risks, high throughput, low cost, more precise pathology, personalizability, etc.
Moreover, to construct a reliable OA model in vitro, it is necessary to consider the cells and their microenvironment, with the addition of one or more suitable induction methods. The complexity of the in vivo tissue allows cells and their interaction with the surrounding ECM, and the complex structure should be taken into account in the design of engineered OA models, corresponding to the design of in vitro ECM frameworks that allow cells to adhere, spread, grow, differentiate, mature and generate new ECM, similar to the in vivo microenvironment. Generally, it is quite difficult to simulate an absolute homeostatic microenvironment similar to that in vivo, which is a major challenge in engineered OA models. The most common types of ECM in engineered OA models are hydrogels and some protein-based scaffolds, where differences in pore size, structure and mechanical properties have a significant impact on the survival of cells. 169 The effects of autocrine and paracrine signaling are also difficult to recapitulate in vitro. In addition to ECM, the choice of cells is very important for in vitro engineered OA models, either from patients or animals. Primary MSCs, SFBs, macrophages and chondrocytes are the most commonly used cell types in engineered OA models. It is well known that in vitro cultures are generally maintained at 37°C and 5% CO2 to more closely resemble the in vivo growth environment and that appropriate physical or chemical stimulation is essential to produce OA and maintain cell performance. 170 In addition to better mimicking the in vivo microenvironment and the pathogenesis of OA, the advantages of high throughput, rapidity and no ethical issues of in vitro engineered models should be considered to screen for the most suitable drugs that intervene or treat OA for the purpose of in vitro repair. However, there may be some technical issues in the fabrication of some engineered OA models; for example, the microfluidic chip in the engineered model may be not suitable for mass production, mainly because of the toxicity of the chip manufacturing process and the high cost of microdetection. 171
Most recently, genome editing, single-cell RNA sequencing, and lineage tracing have been developed and used in engineered OA models, and these techniques greatly facilitate the discovery of new methods and drug candidates for OA treatment. 172 In future engineered OA models, the role and regulation of OA pathophysiological signaling pathways should be brought to the forefront. For example, focusing on the roles and regulation of pathological signaling pathways, such as the Wnt/β-catenin, NF-κB, focal adhesion, HIFs, TGF-β/ΒΜP and FGF signaling pathways, and key regulators AMPK, mTOR, and RUNX2 in the onset and development of OA. 173 It is also vital to construct the interfacial tissues of cartilage and bone in in engineered OA models. 142
Conclusions
OA models are crucial for studying the pathogenesis of OA and developing therapeutic interventions. Currently, animal models are still the first choice in OA research, and optimization measures are being explored to better simulate human OA, such as developing standardized animal models. However, in view of the limited variety of animal models, the impact of differences in disease pathology and species specificity, potential ethical issues and high cost, in vivo models will inevitably face challenges over the long term. In contrast, in vitro engineered models for OA have shown great potential for multiple applications in OA research. However, due to the complexity of the functional requirements of bone organs and tissues, there is still a long way to go for in vitro engineered models.
In conclusion, although there is currently no mature experimental model to study the full range of features that OA possesses, the fast-growing tissue engineering and availability of clinical data in the future will help develop and improve OA models, and the existence of a robust, physiologically relevant, reliable high-throughput system would radically change the way drugs are developed and tested before they are evaluated in clinical trials on OA patients.
Supplemental Material
sj-docx-1-tej-10.1177_20417314231172584 – Supplemental material for Osteoarthritis models: From animals to tissue engineering
Supplemental material, sj-docx-1-tej-10.1177_20417314231172584 for Osteoarthritis models: From animals to tissue engineering by Hongyuan Dou, Shuhan Wang, Jiawei Hu, Jian Song, Chao Zhang, Jiali Wang and Lin Xiao in Journal of Tissue Engineering
Footnotes
Author contributions
Hongyuan Dou: Data curation, visualization, methodology, writing – original draft, writing – review & editing. Shuhan Wang, Jiawei Hu: Data curation, visualization. Jian Song: Visualization, methodology. Chao Zhang, Jiali Wang: Methodology, writing – review & editing. Lin Xiao: Conceptualization, methodology, data curation, visualization, writing – review & editing, supervision, project administration. All authors approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the National Natural Science Foundation of China (52173151, 51803067), the Natural Science Foundation of Guangdong Province of China (2021A1515011084), the Fundamental Research Funds for the Central Universities (22qntd1302) and Shenzhen Outbound Postdoctoral Scientific Research Funding (SZBH202108) for their financial support. The authors are also thankful to Mr. Xiaohe Zhang for his participation in literature collection.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
