Abstract
Intracerebral hemorrhage (ICH) is a non-traumatic hemorrhage caused by the rupture of blood vessels in the brain parenchyma, with an acute mortality rate of 30%‒40%. Currently, available treatment options that include surgery are not promising, and new approaches are urgently needed. Nanotechnology offers new prospects in ICH because of its unique benefits. In this review, we summarize the applications of various nanomaterials in ICH. Nanomaterials not only enhance the therapeutic effects of drugs as delivery carriers but also contribute to several facets after ICH such as repressing detrimental neuroinflammation, resisting oxidative stress, reducing cell death, and improving functional deficits.
Introduction
Intracerebral hemorrhage (ICH) is caused by the spontaneous rupture of blood vessels in the brain, thereby leading to primary and secondary brain injury. Primary brain injury is a mass effect induced by hematoma formation and increased pressure on adjacent brain tissues. A series of metabolic reactions generated in response to the primary brain injury, such as hemolysis and thrombin activation, then lead to secondary brain injury. 1 Neuroinflammation and oxidative stress are involved in secondary brain injury after ICH,2 –4 and the extensive literature on these pathogenic mechanisms uncover new insights to counter the pathologic sequelae of ICH. At present, surgical or traditional treatments are still the main options,5,6 broad pre-clinical studies have been completed,7 –9 and numerous researches are pouring in, 10 – 12 however, there is no widely recognized and effective method to ameliorate secondary brain injury 13 and improve the prognosis of ICH. 14 For example, applying hyperosmolar agents and external ventricular drain placement to cope with the decreased alertness caused by mass effect of ICH may be appropriate.15,16 Recent clinical trial which used tissue plasminogen activator to resolve hemorrhagic products turned out to be unsatisfactory in prognosis. 17 Numerous clinical trials targeting correcting elevated blood pressure and reversing coagulopathy are still ongoing.18 –21
In the past few decades, nanotechnology has emerged as an important class in medical research, where it impacts molecular imaging, drug delivery, cancer therapy, and other aspects.22,23 Nanomedicine has attracted much attention as a safe and effective strategy because some materials possess special properties such as good biocompatibility, biodegradability, and low toxicity when they are modified into the nanoscale. Due to the multi-ligand valency, engineered nanomaterials offer other advantages such as long circulation time, crossing biological barriers, and enhanced targeting properties. 24 The different advantages of nanomaterials are highly dependent on their features which vary broadly from shape, size, and composition, to the surface charge, hydrophilicity, conductivity, and rigidity.25,26
Nanomaterials, mainly nanoparticles (NPs), have the potential to be applied in preclinical studies and clinical trials. They have exerted advantages in the studies of several neurological diseases such as Parkinson’s disease, Alzheimer’s disease, and ischemic stroke.27 –29 Although access to the brain is often restricted for many drugs due to blood-brain barrier (BBB) and its selective transport of drugs, 30 nanomedicines improve the bioavailability, pharmacokinetics, blood circulation time and biological distribution of drugs into the central nervous system (CNS), and achieve the purpose of micro, high efficiency, stability and targeting compared to free drugs.31,32 The rapid development of nanotechnology has made it possible to consider its prospects to improve the therapeutic efficacy of drugs in ICH.
Roles of nanomaterials in ICH treatment
Neuroprotection still represents a major therapeutic goal in ICH in order to prevent the progression of ICH-triggered damage in the brain parenchyma. 33 Unfortunately, most protective therapeutic approaches in animal research have been disappointing when they were translated into clinical trials, mainly because of the side effects of the drugs, low BBB permeability, or narrow therapeutic window.34 –36 The application of traditional medicines is often restricted by poor distribution and low selectivity. These problems may be solved by using nanotechnology, 37 which can conquer the main obstacles that hinder the implementation of neuroprotective approaches by transporting neurotherapeutic agents to the brain. 38 Multiple types of nanomaterials have been tested for delivering a variety of substances.38,39 Nanomaterials have been regarded as suitable carriers for overcoming pharmacokinetic limitations associated with conventional drug formulations. Nanotechnology has been mentioned as an emerging therapeutic strategy targeting ICH. 40 In this review, we summarize some nanomaterials which appear to exert influence on brain injury mechanisms of ICH including inhibiting brain cell death, resisting oxidative damage (Table 1), repressing neuroinflammation, and promoting brain tissue repair and functional recovery (Table 2), eventually improving neurological impairment and prognosis of ICH.
The strategies in targeted delivery of nanomedicines to inhibit cell death and resist oxidative stress after ICH.
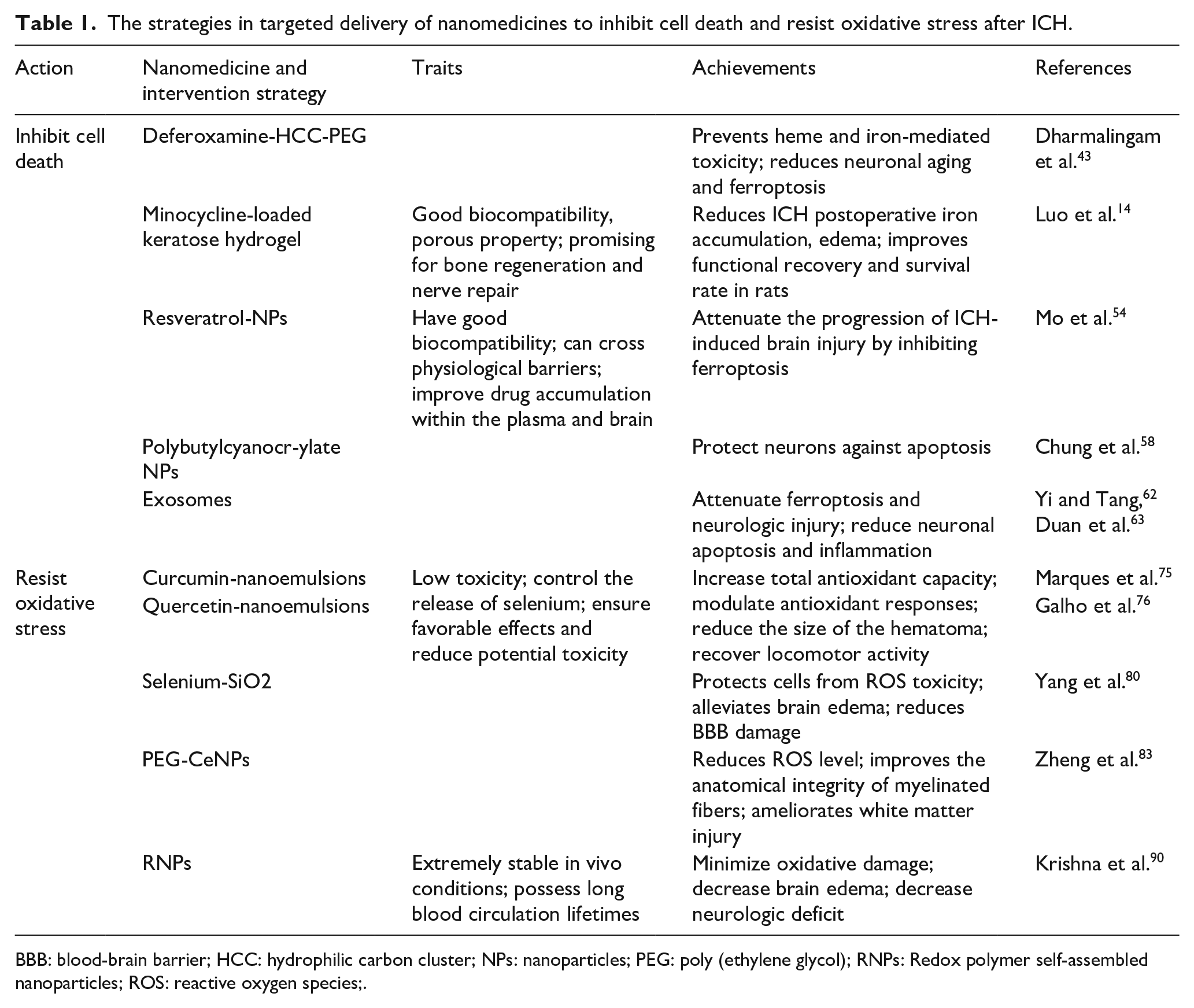
BBB: blood-brain barrier; HCC: hydrophilic carbon cluster; NPs: nanoparticles; PEG: poly (ethylene glycol); RNPs: Redox polymer self-assembled nanoparticles; ROS: reactive oxygen species;.
Strategies in targeted delivery of nanomedicines to repress inflammation and promote tissue repair and functional recovery after ICH.

Nanomaterials can improve the capacity of drugs to inhibit neuronal cell death
Drugs combined with nanomaterials can significantly reduce iron accumulation and ferroptosis
The blood components from hematoma after ICH including hemoglobin, heme, and free iron, have neurotoxic effects.41,42 However, the iron chelating agent deferoxamine (DEF) is dose-limited in human ICH trials due to its drawbacks of poor cellular absorption, toxicity, and short shelf life. A broad action antioxidant, poly (ethylene glycol)-conjugated hydrophilic carbon clusters (PEG-HCCs), was incapable of ameliorating the dual toxic effects of heme and iron and decreasing the susceptibility to ferroptosis. 43 The DEF-HCC-PEG, a synthesized, catalytic, multifunctional, and rapidly internalized carbon nanomaterial formed by PEG-HCC, covalently bonds to DEF. 43 The synthesized DEF-HCC-PEG was significantly more effective than PEG-HCCs, free DEF treatment alone, or combined but worked as a single drug, in reducing heme and iron-mediated toxicity, inhibiting neuronal aging and ferroptosis in culture. This combination strategy effectively protected neurons from the toxicity of ICH hemolysis (Figure 1). Using nanomaterials can greatly enhance the effect and decrease the dosage of drugs, and the optimal proportion of DEF and PEG-HCCs should be considered.

Model illustrating the effects of nanomedicine on the ferroptosis and apoptosis after ICH. Deferoxamine-HCC-PEG is a complex of the iron chelator deferoxamine and antioxidant PEG-HCC, which can effectively reduce heme/iron toxicity, and prevent senescence and ferroptosis. Minocycline-loaded keratose hydrogel can reduce iron accumulation, resveratrol-NPs and miR-19b-3p-enriched exosome can inhibit ferroptosis. miR-146a-5p-enriched exosome and cmv-neurotrophin-3-HRE-loaded polybutylcyanoacrylate NPs can reduce apoptosis following ICH.
Another iron chelator, minocycline, has been used to reduce ICH-induced brain edema, neuronal death, and neurological deficits.44,45 However, there were some adverse issues in clinical trials. 46 Keratose hydrogel, a kind of hydrogel formed from keratose, could delay hemoglobin-induced iron accumulation in rat primary neuronal culture owing to its adsorptive capacity, while minocycline hydrochloride-loaded keratose hydrogel displayed a stronger and more thorough cytoprotective effect than blank hydrogel (Figure 1). In vivo, the minocycline hydrochloride-loaded keratose hydrogel effectively reduced ICH postoperative iron accumulation and edema, and improved functional recovery and survival rate in rats compared to the systemic administration of minocycline. 14
Resveratrol, a widely used non-flavonoid polyphenol compound, 47 has neuroprotective roles in ICH.48 –51 However, its poor oral bioavailability and difficulty in crossing physiological barriers52,53 limit its clinical application. NPs, as a commonly used drug delivery matrix material with good biocompatibility, can transport across physiological barriers and improve resveratrol accumulation in plasma and brain. Resveratrol-NPs safely and effectively attenuated the progression of ICH-induced brain injury by inhibiting ferroptosis 54 (Figure 1). In this condition, it can be considered to combine other agents which target necrosis or necroptosis, with nanomaterials, to investigate if they can effectively ameliorate the death of neurons post-ICH.
Using nanomaterials can remarkably reduce the neuronal cells apoptosis
Polybutylcyanoacrylate NPs have shown potential as an appropriate non-viral system for gene delivery,55,56 and they are also efficacious vectors for delivering large molecules to the injured brain. 57 A delivery system comprising polybutylcyanoacrylate NPs of plasmid neurotrophin-3 containing hormone response element (HRE) with a cytomegalovirus (CMV) promoter was used to treat ICH rats. 58 The results showed that polybutylcyanoacrylate NPs could raise the delivery of cmv-neurotrophin-3-HRE across the BBB in vivo, therefore increasing the expression of neurotrophin-3, and protecting neurons against apoptosis after ICH in vivo (Figure 1).
Exosomes are nanosized 30–100 nm diameter membranous vesicles released from diverse cell types, and they transfer biomolecules from one site to another. They are thought to play important functions in various biological pathways including cell-to-cell communication, tumor progression, and cellular waste disposal. 59 A large collection of recent research papers has revealed that exosomes released from various stem cells can improve multiple diseases,60,61 including ICH. For example, exosomes derived from miR-19b-3p-overexpressing adipose-derived stem cells attenuated ICH-induced ferroptosis and neuronal injury. 62 MiR-146a-5p-enriched exosomes released from bone marrow mesenchymal stem cells elicited neuroprotection and functional improvement after ICH by reducing neuronal apoptosis and neuroinflammation associated with the suppression of microglial pro-inflammatory polarization 63 (Figure 1).
Nanomaterials can improve the ability of drugs to resist oxidative stress
Curcumin has been described to prevent or delay the onset of ischemic stroke and multiple CNS diseases.64 –70 Quercetin is known for its antioxidant activity and anti-inflammatory properties.71,72 However, they both have low bioavailability in their natural states.73,74 Nanoemulsions (NEs) are surfactant stabilized heterogeneous systems and their oil droplet sizes make them potential systems for improving drug delivery. 65 Studies have compared the possible therapeutic effect of these two free drugs versus their drug-loaded NEs in an ICH rat model.75,76 The results showed that the formulations of curcumin-NEs and quercetin-NEs increased total antioxidant capacity in ICH than them alone. Both drug-loaded NEs reduced the size of the hematoma, recovered locomotor activity, and attenuated weight loss caused by ICH, without obvious toxic effects. 71
Selenium is a cofactor of glutathione peroxidase and thioredoxin reductase, which are antioxidant enzymes that resist oxidative stress and maintain redox balance. 77 However, the use of selenium is restricted by the narrow gap between its beneficial and harmful effects. 78 The porous selenium-SiO2 nanocomposites with an average diameter of about 55 nm 79 could control the release of selenium, ensure favorable effects and reduce potential toxicity. 80 After intraperitoneal injection into ICH mice, the activity of glutathione peroxidase was increased, while the level of malonaldehyde was significantly decreased. By protecting cells from reactive oxygen species (ROS) toxicity, alleviating brain edema and reducing BBB damage, the porous selenium-SiO2 nanocomposites improved neurological functions of ICH mice.
Ceria NPs (CeNPs) are known to possess potent-free radical scavenging activity.81,82 One study found that PEG-CeNP, which was formed by CeNPs with a PEG coating, strongly reduced ROS levels in the brain tissue. It also modulated ROS-induced microglial polarization and astrocyte alteration improved the anatomical integrity of myelinated fibers and ameliorated white matter injury after ICH. 83
The nitroxide radical compound 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) is one of the strongest antioxidants that catalytically scavenge ROS.84,85 Under in vivo conditions, however, these low molecular weight nitroxide compounds have several problems, including nonspecific dispersion in normal tissues, preferential renal clearance and rapid reduction to the corresponding hydroxylamine form. Redox polymer self-assembled NPs (nitroxide radical-containing NPs [RNPs]) with diameters of approximately 40 nm were developed to solve these issues. 86 By contrast, RNPs are extremely stable in vivo and possess long blood circulation lifetimes.87,88 Nitroxide radicals in RNPs have catalytic ROS scavenging activity including for superoxide and hydroxyl radicals, 89 which is superior to that of superoxide dismutase (SOD). 90 Systemic RNP treatment at an early stage after ICH decreased levels of superoxide anion radicals and minimized ICH-induced oxidative damage to other molecules in addition to DNA. RNPs also decreased acute ICH-induced brain edema and neurologic deficit, probably by decreasing the hemorrhagic area.
Nanomaterials can improve the capacity of drugs to repress neuroinflammation
Statins are considered as neuroprotective agents that can reduce microglia activation and differentiation in many CNS diseases including ICH.91 –93 Rosuvastatin is a synthetic, highly effective second-generation statin with poor water-solubility and low oral bioavailability. Polymeric nanomicelles as self-assembled copolymers are good candidates for poorly water-soluble or hydrophobic drugs.94 –96 Rosuvastatin nanomicelles were prepared by cosolvent evaporation method using Poly (ethylene glycol)-poly (ε-caprolactone) (PEG-PCL) nanomicelles as nanocarrier 97 because of their good biocompatibility and nontoxicity.98 –100 The rosuvastatin nanomicelles employed in the treatment after ICH inhibited the expression of downstream inflammatory factors such as tumor necrosis factor-alpha (TNF-α) and interleukin-1beta (IL-1β), and promoted the polarization of microglia/macrophages toward a regulatory phenotype, effectively repressing neuroinflammation. 97
Injectable hydrogels are particularly suitable for ischemic and hemorrhagic stroke owing to their advantages of minimally invasive implantation.101,102 Hydrogels themselves can bind to specific cell-surface receptors of endogenous brain cells, inducing a variety of repair and anti-inflammatory cellular pathways. Gelatin is derived from denatured and partially degraded collagen. It has excellent biodegradability and biocompatibility, as well as adhesion to cells and lack of antigenicity, and can retain the cell adhesion motif of RGD. 103 There is an injectable hydrogel formed by Thiolated gelatin reacting with polyethylene glycol diacrylate. The in vivo experiment showed that the gelatin hydrogels interacted with different host cells, making it possible to attenuate postoperative neurological deficits, reduce neuronal loss, and repress the activation of astrocytes and microglia/macrophages. In addition, hydrogel injection reduced the release of inflammatory cytokines, IL-1β and TNF-α 104 (Figure 2).

Schematic presentation of the effect of the injectable gelatin hydrogel and rosuvastatin nanomicelles on neuroinflammation after ICH. The implanted hydrogels bind to integrins on microglia/macrophages via cell adhesion RGD peptide, and then promote the regulatory phenotype polarization of the microglia/macrophages. Rosuvastatin-loaded PEG-PCL can also promote regulatory polarization.
Using nanomaterials can significantly promote brain tissue repair and functional recovery
Rebuilding the damaged CNS is a momentous goal for studies in neuroscience. Typical scaffolds are unsuitable in the damaged brain owing to massive manipulation of overlying tissues during implantation.105,106 Moreover, the advances of exogenous stem cells are still limited by some practical and ethical hurdles.107 –110 In this light, injectable hydrogels are promising because they allow for a minimally invasive method that can eventually be compatible with current stereotactic procedures, as mentioned earlier.101,102 An injectable gelatin hydrogel containing epidermal growth factor (gelatin-EGF) as a therapy for ICH was evaluated. 111 This result indicated that growth factor-containing biomaterials could be safely introduced into an ICH cavity to rebuild tissue and potentially improve recovery in ICH rats. On this basis, the best time to inject the biomaterial and how long it will take to take effect after ICH still need exploration. Diverse recovery pathway and factors are involved in CNS recovery, 112 and given the good practices in brain ischemia, such as injectable hydrogels containing brain-derived neurotrophic factor was deposited into the cavity to promote repair and recovery,113 –115 nanomaterials containing different growth factors or neuroprotective agents could be further studied.
A traditional self-assembly peptide nanofibrous (SAPNS) called RADA16-I, is frequently used in tissue engineering. 116 In an ICH model that incorporated stereotactic minimally invasive hematoma aspiration to provide space for local delivery of nanomaterials, 116 SAPNS served as a biocompatible material in the hemorrhagic brain cavity. It replaced the hematoma, reduced acute brain injury and brain cavity formation, and improved sensorimotor functional recovery. 117 With this ICH model, it has been found that RADA16-I could reduce acute brain injury and decrease the inflammatory response. In addition, a modified synthesized neutral SAPNS, which structure is RADA16-RGD (Arg-Gly-Asp), could overcome the disadvantages of RADA16-I, including poor biocompatibility caused by its acidity, very fine functional recovery and even no nerve fibers to ICH mice. 117 Another self-assembly peptide nanofibrous scaffold, RADA16mix, reduced acute brain injury, decreased inflammatory response, and promoted functional recovery and nerve fiber growth. 118
Potential applications of nanomaterials
The volume of hematoma is one of the most important predictors of functional outcome in ICH patients. 119 However, the clinical trials of hemostatic measures have turned out to be unsatisfactory. For example, intravenous administration of an activated recombinant factor VII did not improve functional outcomes after ICH, 120 and platelet transfusion was detrimental in patients taking antiplatelet therapy before ICH. 121 Compared to cell-derived microparticles, such as thrombosomes and synthocytes, which are ineffective at reducing bleeding and increase the risk of adverse immunoreaction, 122 nano-engineered agents have efficiently improved hemostasis with fewer side effects. 123 These are promising to improve the progress of ICH. Various topical NPs have been developed,124,125 but only a few have been tested in ICH. In this case, polymeric NPs are applied to stabilize the structure of some clot’s components such as platelets or fibrin, and reduce bleeding time. Synthetic platelets, made of a poly (lactic-coglycolic acid)-poly-L-lysine block copolymer core with PEG arms terminated with RGD functionalities, 126 bind to activated platelets and favor platelet aggregation, thus reducing bleeding time in arterial injury models. Further optimized NPs, which is closer to endogenous platelets owing to deformability characteristics, 127 could better accumulate at the wound site and improve the hemostatic functions of natural platelets. A similar approach, intravenous administration of a hemostatic polymer (PolySTAT), enhanced fibrin crosslinking and prevented clot degradation because of multiple binding sites for fibrin, and therefore improving survival by reducing blood loss in the trauma model. 128 The efficacy of these treatments for limiting bleeding in ICH remains to be confirmed. 129
In recent years, stem cell therapy has been a promising method for ICH treatment, aiming to replace the damaged neurons and therefore improve the functional recovery and prognosis post-ICH. 130 A magnetic resonance imaging (MRI)-visible PAsp (DMA)-Lys-(CA)2 polymer-based nanoscale polymeric micelle, has been used to deliver small interfering RNA/antisense oligonucleotides (siRNA/ASO) against Pnky long noncoding RNA (lncRNA) into neural stem cells. This multifunctional nanomedicine can direct stem cell differentiation in vivo and in vitro, as well as track the stem cells after transplantation in vivo in ischemic stroke. 131 If MRI-visible nanomaterials could be applied in ICH, it will be more targeted and more controllable for nanomaterials to play their roles and deliver therapeutic agents. Apart from delivering therapeutic genes into stem cells to regulate neuronal differentiation as vehicles, nanomaterials can exert their effects on stem cells in several ways as well. Nanomaterials can protect stem cells from a series of pathological processes such as inflammation, 132 and deliver growth-related factors to provide microenvironments for stem cells to survive and proliferate then form new tissue in the brain.89,133 Furthermore, nano-sized exosomes released by stem cells can reduce neuronal death and improve neurological function on their own.62,63 It is promising to develop nanomaterials-based stem cell therapies for ICH.
Based on the benefits of good biocompatibility, good targeting, and well-controlled drug release, 134 nanomaterials not only hold great prospects in treating primary and secondary injuries, but also in improving the neurological recovery after ICH. Meanwhile, many crossing strategies have combined nanotechnology with multiple substances such as cell-penetrating peptides, receptor, shuttle peptide, and cells to transport the therapeutic drugs through the BBB for improved precision of drug delivery, prolonged half-life, great stability, and increased drug loads. 135 In this condition, it is worthwhile to consider which nanomaterial to choose, whether nanomaterials need to be modified, and how to combine nanomaterials and agents when applied to ICH. More importantly, attention needs to be paid to whether the application of nanomaterials improves prognosis.
The concerns of nanomaterials
Toxicological analyses are essential before formulations or drugs are applying in clinical studies. The good news is that according to the pre-clinical experiments in ICH models, nanomaterials have not been found to cause renal or hepatic toxicity.54,75 However, despite the many advantages and advances of nanotechnology to improve neuroprotection, potential side effects and the complex metabolic processes of nanomaterials may limit their applications in clinical practice. For example, NPs based on pure copper, silver, or aluminum, can impair endothelial cell function and increase BBB permeability in rodents. 136 These processes are regarded as adverse in ICH, 137 although they might promote drugs into the brain. Furthermore, NPs are more easily identified as pathogens by the host than small molecules, resulting in rapid clearance and response of the complement system. 138 In addition to immune reactions that could be triggered by contact with blood components, nanomedicines may damage organelles such as mitochondria, endoplasmic reticulum or lysosome of macrophages, leading to the release of excessive ROS and pro-inflammatory mediators. 139 Since the nucleus is also exposed, DNA may be damaged, so extensive monitoring for genotoxicity is required. All these reasons should be noted in the application of ICH treatment, therefore, further in-depth assessment of the risks associated with the use of nanomaterials is necessary.
Summary
ICH is an intractable disorder. The emergence of nanotechnology provides a promising option for treating ICH. Compared to free drugs, nanomaterials improve the capacity of agents to cross the BBB, prolong the half-life of drugs, reduce adverse effects, and enhance drug stability. Citations within this review highlight that nanomaterials can exert protective role after ICH. However, current intervention approaches based on nanomedicine are far from being clinical applications. Moreover, we should consider their side effects. The toxicity of nanomaterials should be thoroughly investigated before they are applied to patient treatment. Fortunately, the toxicity of nanomaterials has received increasing attention, and more measures have been developed in the field of nanomedicine140 –142 to support their eventual clinical translation. Furthermore, the application of nanomaterials should be based on clear drug targets and pathways.
Based on a series of conservative treatments, our attention has been turned to some innovative therapeutics targeting ICH. Among all, nanotechnology is undoubtedly a very efficient and effective method. More and more novel and useful nanomaterials are being developed to treat ICH and this has brought hope in improving the prognosis of this often-fatal form of stroke.
Footnotes
Author contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge operating grant support from National Key Research and Development Program of China (grant no: 2018YFC1312200), the National Natural Science Foundation of China (grants no: 82071331, 81870942, and 81520108011), and from the Canadian Institutes of Health Sciences (VWY).
