Abstract
Extracellular vesicles (EVs), including exosomes, carry the genetic packages of RNA, DNA, and proteins and are heavily involved in cell-cell communications and intracellular signalings. Therefore, EVs are spotlighted as therapeutic mediators for the treatment of injured and dysfunctional tissues as well as biomarkers for the detection of disease status and progress. Several key issues in EVs, including payload content and bioactivity, targeting and bio-imaging ability, and mass-production, need to be improved to enable effective therapeutics and clinical translation. For this, significant efforts have been made recently, including genetic modification, biomolecular and chemical treatment, application of physical/mechanical cues, and 3D cultures. Here we communicate those recent technological advances made mainly in the biogenesis process of EVs or at post-collection stages, which ultimately aimed to improve the therapeutic efficacy in tissue healing and disease curing and the possibility of clinical translation. This communication will help tissue engineers and biomaterial scientists design and produce EVs optimally for tissue regenerative therapeutics.
General properties, isolation, and utility of extracellular vesicles
Extracellular vesicles (EVs) are a collection of naturally derived particles enveloped by a phospholipid bilayer and considered a major cellular secretome, enabling cellular communications via transmission of macromolecules. 1 EVs are composed of multiple genetic or intracellular biomolecules such as nucleic acids (RNA, DNA), proteins, lipids, and metabolites. 2 Among the components of EVs, microRNA (miRNA) has gained a lot attention. It is a small, noncoding RNA (about 22 nucleotides) and is well known to regulate post-transcriptional gene expression by binding to the region of mRNA and its degradation, up- and down-regulating various biological activities of cells in proliferation, migration, differentiation, and disease progress. EVs are secreted from all types of cells such as osteoblasts, odontoblasts, chondrocytes, fibroblasts, endothelial cells, skeletal muscle cells, cardiomyocytes, keratinocytes, neurons, or other neuronal cells, hepatocytes, cancer cells, and immune cells. 3 Besides, EVs can be isolated from all kinds of tissues and a wide range of body fluids, including blood, plasma, breast milk, urine, saliva, synovial fluid, bile, amniotic fluid, semen, and ascites fluid.
EVs are currently categorized based on the vesicle biogenesis and the difference in size and composition; (i) “exosomes” (30−150 nm) when multivesicular bodies pre-formed by reverse budding of endosomal compartment fuse with the cytoplasmic membrane, (ii) “microvesicles” (150−1000 nm) when shedding or budding of the cytoplasmic membrane occur, and (iii) “apoptotic bodies” (500−2000 nm) when outward membrane blebbing in cells happen especially during apoptosis process in contrast with other EVs, which are produced by healthy cells as a part of regular membrane turnover and exocytosis. Among the three types of EVs, exosomes and microvesicles, without a strict distinction between them, have been collected together for applications in regenerative medicine due to the apoptotic characteristics of the apoptotic bodies.
Initially, EVs were believed to have the role of packaging and release of unwanted cellular materials like garbage bags. However, recent accumulating studies have highlighted other informative and therapeutic roles of EVs that are similar to conventional cytokines and growth factors, which spurred the utilization of EVs for therapeutic purposes of many diseases. 4 Furthermore, EVs are embedded in the extracellular matrix (ECM), which provides mechanistic insights into the mutual interactions of EVs and ECMs and the possible utility of EVs as an effective delivery tool through ECM to target cells.5,6
Significant efforts have thus been made to isolate EVs from various types of cells. In the isolation of EVs from cultured cells for regenerative medicine, it is recommended to use either a serum-free medium or EV-depleted serum-contained medium to minimize the effects of as-given EVs in commercially available serum. Thus, the isolation of EVs with high efficiency without contaminating (matrix) proteins and cell debris is necessary to ensure the accurate biological function of EVs of interest. There are many current and commonly used methods of EVs isolation, and their pros and cons were well-reviewed elsewhere 7 ; from the current widely used technique, ultracentrifuge-and-filtration that separates and concentrates EVs according to their density (1.13–1.19 g/mL) and then filtrate, to other improved techniques such as microfluidics, asymmetric flow field-flow fractionation, and nano-flow cytometry. Among the EVs isolation techniques, the ultracentrifuge-and-filtration is the most widely used that can collect EVs with relatively confined size, but it needs large sample volume and is time-consuming.8–10 Microfluidics method requires low sample volume and cost, but the isolated EVs amount per time is limited. On the other hand, the asymmetric flow field-flow fractionation technique can collect EVs in a gentle, rapid (<1 h) and highly reproducible way, but the particle loss and variation in the size due to sticking to the barrier membrane should be improved. 11 Lastly, the nano-flow cytometry method has shown the high fidelities in EVs sorting, but it has drawbacks such as swarm detection, detection of smaller vesicles than previously thought possible, and multiple sized EVs. 12 To overcome the disadvantages for clinical translation and therapeutics, multiple methods have been combined such as ultracentrifuge-combined asymmetric flow field-flow fractionation.7,13,14 Many studies have reported the impact of storage temperature and freezing/thawing cycles on the size and bioactivity of EVs and recommended −80°C as a storage temperature and minimal freezing/thawing for maintaining therapeutic and cargo potential.15–17
EVs have a huge impact on various biological and medical fields (as shown in Figure 1). EVs can inform the disease status and thus are new biomarkers of many diseases, including cancers. EVs may also be used to interpret the biological phenomena, to elucidate the unknown mechanisms of cell-cell and cell-ECM interactions. EVs are emerging therapeutics with their key packaged genetic molecules (e.g. miRNA). Especially as the delivery carriers for therapeutic purposes, EVs have innate merits compared with other synthetic polymers or viruses, such as their naturally-derived biocompatible bi-lipid layer structure and less possibility for invoking negative immune responses.18–22 The other abilities of EVs, such as enabling modifications with various ligands, penetrating blood–brain barrier, and cellular internalization, are beneficial for local or systemic delivery to target specific cell types. Thus, EVs have been spotlighted as versatile, tunable therapeutic tools for the healing and treatment of various traumatic injuries and diseases.23,24

Schematic showing the collection of EVs from various sources and their diverse applications for regenerative therapeutics. During or after biogenesis, various methods are used to tune/enhance the payload content, bioactivity, targeting ability, bio-imaging ability, and quantity.
In this context, recent efforts have been made to improve the preparation techniques of EVs, such as the genetic modification and specific drug/biomolecule treatment, during biogenesis process or at post-collection stages (as schemed in Figure 1). Furthermore, applying a biophysical and mechanical stimulus and providing 3D culture conditions with engineered biomaterials has been highlighted to improve the quantity and quality of EVs.4,25–28 Our communication here will help tissue engineers and biomaterial scientists design and generate EVs optimally for tissue regenerative therapeutics.
Key issues in the biogenesis of EVs
Several key issues are considered in EVs’ biogenesis: bioactivity (payload content), mass-production (quantity), targeting, and tracking. Firstly, EVs should contain bioactive components necessary for the therapeutic actions (“payload content/bioactivity” issue). However, native EVs have limited in their components, of which the content is mainly dependent on the donor cells and their microenvironment. Along with the quality of the contents, EVs should be mass-produced to achieve enough amount that allows clinically relevant treatment tools (“quantity/mass-production” issue). To date, one significant hurdle is the limited amount of therapeutic EVs per cell, requiring massive in vitro cell cultures to gather enough quantities of EVs. 29
Besides, EVs should interplay with target cells and deliver their bioactive contents to the cells (“targeting” issue). Native EVs have some tropism depending on the epitopes expressed by the donor cells or the culture conditions of the cells (e.g. growth, inflammation, or differentiation condition), although the mechanism of the process was not fully understood yet. However, the tropism of EVs may not be enough to target tissues or cells for therapeutic purposes. Further engineering the EVs would help specifically targeting tissues or cells and even intracellular components. In association with the targeting is the retention of EVs in localized sites; for this, EVs should internalize cells effectively to deliver the bioactive cargo molecules and reside for a long period to elicit biological efficacy. However, native EVs have a limited endolysosomal escape, albeit better than synthetic carriers, limiting the capacity to release their contents without degradation of the bioactive cargo. When EVs are possible to visualize, that is, to allow tracking in vivo, the biodistribution and targeting efficacy can be easily monitored. Native EVs are not detectable; thus modification and labeling of EV’s membrane without mitigating the innate bioactivity is needed. Fluorescence, luminescence, or radio-detector can visualize, monitor the delivery route, biodistribution, and targeting efficacy of EVs.
Over the last years, technologies to tackle the issues in EVs as mentioned above, that is, bioactivity (payload content), targeting and bio-imaging ability, and mass-production (quantity), have been significantly progressed. 28 Here we discuss the most recent technological advances possible by the control of donor cells during or at a post-collection of EVs biogenesis, which includes genomic insertion, recapitulating 2D and 3D culture conditions, and applying biophysical and mechanical cues (as depicted in Figure 2 and summarized in Table 1).

Schematic showing the recent technological efforts in solving some key issues in the biogenesis of EVs (bioactivity, targeting ability, mass production) utilizing various methods such as genomic insertion, recapitulating 2D and 3D culture conditions, and applying biophysical and mechanical cues.
Summary of emerging biogenesis technologies of extracellular vesicles for tissue regenerative therapeutics.

ESCRT: Endosomal sorting complex required for transport.
Cortactin: Actin cytoskeletal regulatory protein.
Payload content and bioactivity of EVs
Increasing the bioactivity of EVs by the modulated payload content has been a key issue in the biotechnology of EVs for their therapeutic uses. Studies have been focused on modulating the culture microenvironments of EVs-secreting donor cells, such as altering ingredients, oxidative stress, and physical cues. Specifically, EVs have been generated under different conditions, involving differentiation medium,30,31 hypoxia,32,33 3D culture, 34 mechanical force,35,63 and their combinations (e.g. 3D culture + mechanical force). 37 The key concept in the applications of those cues during the biogenesis of EVs is to allow cells to experience the conditions where the biochemical and biophysical conditions are mimicked or modulated to the native tissue microenvironments. Thus, the generated EVs can preserve the bioactivity of cells of interest.
One recent study by Wei et al. reported the effect of a specific differentiation medium on the biogenesis of EVs. EVs were collected at different stages (at day 5 and 11) during osteogenesis of mesenchymal stem cells (MSCs) in blood-derived hydrogels. The EVs secreted at an early stage of osteogenesis (at day 5) were shown to highly express the early osteogenic marker alkaline phosphatase (ALP). In contrast, those from a late-stage (at day 11) were rich in calcium phosphate crystals, implying the possibility of loading distinct cargo molecules within EVs by controlling the differentiation stage/period 30 (Figure 3(a)). In another study with human adipose-derived stem cells (ASCs), the endothelial differentiation medium generated the EVs with higher miRNA-31 levels compared to the normal growth medium, which ultimately contributed to the increased endothelial cell migration, tubule formation, and aortic ring outgrowth. 31 The approach of using a specific culture medium for the biogenesis of EVs is still in infancy, which yet can be potentially applicable to a variety of other situations where a specific payload of EVs is required to achieve the bioactivity of interest, such as for the lineage specification of stem cells, the polarization of immune cells, and rejuvenation of senescent cells.

Strategies to enhance the bioactivity of EVs. EV-secreting donor cells are modulated by culturing under various conditions: (a) EVs gathered from MSCs at different culture time points in an osteogenic medium to enrich either ALP (early gathering) or Ca/P (late gathering), (b) EVs obtained from stem cells under 3D spheroid culture condition to incorporate miRNAs with different profiles, and (c) EVs generated from periodontal ligament cells cultured under 3D gel matrix combined with mechanical stretch to enhance bone regeneration.
Not only the culture medium ingredients, but oxidative stress is a key microenvironmental regulator of cells. Furthermore, when primary cells are cultured in vitro, they experience higher oxidative stress than those in vivo native tissues. Modulated oxidative stress conditions are often used to tune the cellular behaviors, including the secretome profiles. A pioneering study by Gray et al. reported that the EVs collected from cardiac progenitor cells cultured under hypoxia (versus those under normoxia) could increase the tubule formation of cardiac endothelial cells, reduce the pro-fibrotic phenotype of cardiac fibroblasts, and improve the in vivo function of the infarcted heart. 32 When a hypoxia-conditioning was applied to MSCs, the EVs showed an increased vascular tubule formation in vitro without much difference in the concentration, size, and surface signatures compared to those with normoxia. 33
As noted, most studies on EVs have been carried out in 2D culture dishes, which cannot replicate the native tissue 3D environments. A variety of engineered biomaterials (scaffolds and hydrogels) can thus be promising candidates for the 3D cultivation of cells, allowing them to experience tissue mimic “3-dimensionality” and then adopt the right signals in the biogenesis of EVs. A recent study by Rocha et al. 34 applied 3D cultures using a microwell array in the biogenesis of EVs. When compared with the EVs from the 2D tissue culture plate, the EVs from the 3D microwell array showed higher numbers per cell, and the contents of EVs, including small RNA, micro RNA, and proteins, were significantly altered by the 3D spheroid cultures (Figure 3(b)). This study delivers a key idea that the 3D cultures enable cells to generate more EVs while modulating the quality of EVs. However, the mechanisms underlying the EVs’ biogenesis are yet to be elucidated. Given the 3D culture used in that study is the aggregated form of cells, the recapitulation of 3D conditions with various engineered ECMs such as scaffolds and hydrogels might be more accurate to mimic the in vivo tissues, which remains a further research area.
Along with the 3D cultures, externally applied mechanical forces are known to regulate diverse cell behaviors (e.g. development, homeostasis, and repair), 64 which is considered an effective way to alter the biogenesis process of EVs and ultimately to enhance the bioactivity. Recently, Najrana et al. 35 reported that the mechanical stretch applied to lung epithelial cells could regulate the expression of specific miRNA in EVs. Both cyclic (10%, 40 cycles/min) and continuous (5%) mechanical stretch could regulate the miRNA profile in EVs more significantly than the control (w/o stretch), implying the mechanical conditioning would be a possibly effective way of generating EVs that contain miRNAs for therapeutic purposes. In another similar study with MSCs, the cyclically stretched cells synthesized EVs that could inhibit osteoclast differentiation. 36 The event was demonstrated to relate with the attenuated RANKL-induced NF-κB signaling pathway, and the EVs could effectively rescue the in vivo osteoporosis caused by the unloading of the hindlimb, suggesting that the anti-osteoclastic role of endogenous MSCs in vivo might be due to the generation of stretch-induced therapeutic EVs. This study envisions mechanical conditioning as a potential strategy to collect therapeutic EVs from MSCs targeting diseases in skeletal systems.
Combinatory approaches of the above methods were further used to synergize the effects on the biogenesis of EVs. For example, Yu et al. 37 applied mechanical force while culturing cells under 3D gel conditions (Figure 3(c)). Periodontal ligament MSCs cultured in a 3D collagen hydrogel containing magnetic nanoparticles could be mechanically stretched under a magnetic field. The 3D mechanical stimulation significantly altered the miRNA content in EVs compared to the 3D culture only, and the EVs-treated cells showed enhanced proliferation, migration, and osteogenesis in vitro and bone formation in vivo in an alveolar bone defect model. Although studies on the combinatory treatment are still in infancy, the possible synergistic or additive effects of the biomechanical cues with the optimized 3D matrix and biochemical environment (medium and oxygen tension) are envisaged that can lead to the generation of EVs with enhanced bioactivity and therapeutic efficacy, which remains a further exciting area of research.
Quantity and mass production of EVs
Increasing the quantity (mass production) of EVs is considered a significant challenge for their in vivo application and clinical translation. The strategies for mass-production of EVs include modulating the components or secretary machinery proteins of EVs,38–40 increasing the intracellular Ca ions,41,42 adjusting biochemical cues such as extracellular DNA, liposomes, and proton concentration,43–45 and applying physicomechanical cues such as forces35,46–48 and other stimuli (e.g. electricity, thermal, photodynamic, radiative stress).49–53
One of the effective ways to increase the EVs amount is the modulation of key biogenesis machinery components. For example, the endosomal sorting complex required for transport (ESCRT) was modified using RNA interference (RNAi) to target 23 different components of the ESCRT machinery individually. Results revealed a key role of a few selected members (SCR, VPS4B, ALIX, HRS, STAM1, TSG101) of this family in either the secretion efficiency or the size and composition of secreted EVs. 38 In another study, when the expression of EVs constitutional proteins, that is, CD9, TSG101, and Alix in donor cells, was enhanced using lentivirus, the EVs amount and mean size were shown to increase. 39 These results supported the concept that the modulation or overexpression of EVs compositional proteins would be beneficial for mass production. The key component protein in secretory machinery was tuned to boost the EVs secretion after the intracellular biogenesis in a different approach. The overexpression of actin cytoskeletal regulatory protein, cortactin, could promote exosome secretion by controlling branched actin dynamics without altering exosome cargo content, demonstrating a useful alternative way of targeting secretion machinery in the post-cellular-biogenesis to enhance the EVs production. 40
An increase in intracellular Ca ions triggers the plasma membrane EV biogenesis, and the EV-production is dependent on the increased calcium mobilization and the activity of calpain, a calcium-dependent, non-lysosomal cysteine protease that modulates various cellular cascades, including EVs biogenesis. Thus, intracellular Ca ion enhancers (e.g. ionomycinor) were treated to donor cells during biogenesis.41,42 An acute elevation of intracellular calcium level stimulated an almost 5-fold increase in CD63+, CD9+, and ALIX+ EVs, general markers for EVs. Also, the EVs release was shown to depend on Munc13-4, an essential Ca ion-dependent synaptosomal-associated protein receptor that mediates vesicle fusion for the intracellular trafficking and exocytosis of intracellular granules such as secretory lysosomes and lytic granules. 41 Unlike using calcium activator drugs, one recent study introduced Ca ion-releasing biomaterials (e.g. calcium phosphate nanoparticles) in mouse macrophage-like RAW264.7 and human monocyte-like THP-1 cells, which led to a significant increase in EVs secretion (more than twice) (Figure 4(a)). 42 It was deduced that the calcium phosphate nanoparticles were internalized into cells and merged to acidic late-endosomes or lysosomes, resulting in membrane rupture and the release of Ca ions into the cytosol, which, however, warrants further investigation if the intracellular Ca delivery via nanoparticles would have the effect comparable to that though ion channels and would share the down-stream Ca signaling pathways.
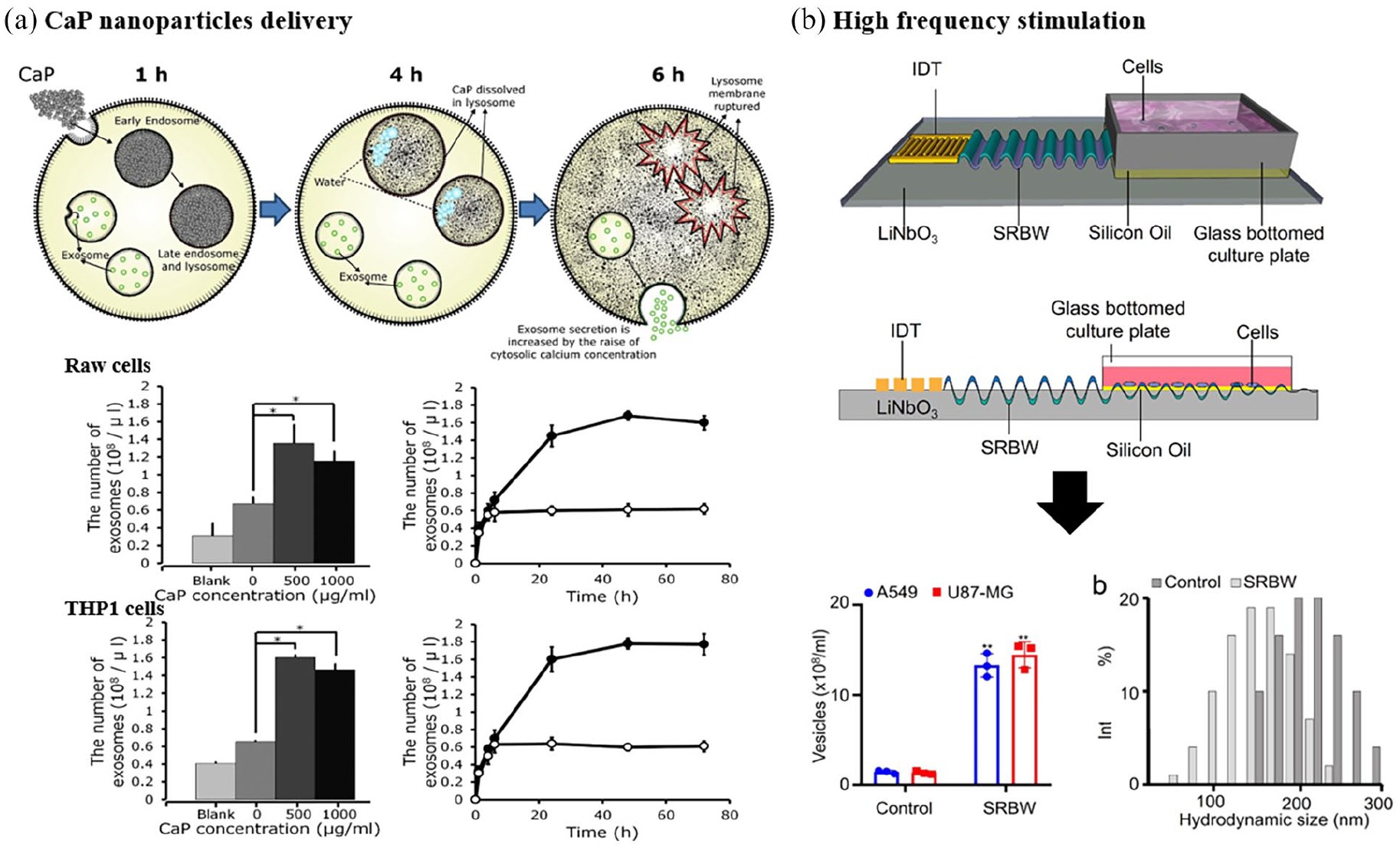
Strategies to increase the quantity of EVs secreted from cells: (a) application of calcium phosphate particles to mouse macrophage-like RAW264.7 and human monocyte-like THP-1 cells increased the EVs quantity more than twice and (b) high-frequency acoustic stretching to cells increased the EVs quantity by 15-fold.
Other exogenous biochemical cues, such as extracellular DNA, 43 liposomes (neutral or cationic liposomes), 44 or altered proton concentration, 45 were also used during EVs biogenesis to increase the secretion. The extracellular DNA, genomic bacterial, eukaryotic DNA, or synthetic CpG-ODNs are known to induce higher secretion of vesicles in even non-immunomodulatory mammalian cells. 43 Liposomes interact with the cell surface, also stimulating the generation of membrane vesicles, and among the liposomes, neutral, and cationic liposomes were shown to enhance the EVs secretion in a dose-dependent manner. 44 Low pH (mild acidic, pH 6.0) also increased EVs release compared with a buffered condition (pH 7.0) in melanoma cells. 45 Although it is not clear whether the pH-dependent EVs secretion is only for pH-sensitive cells such as cancer or immune cells or not, the positive roles of proton pump for EVs biogenesis and secretion might explain the pH-dependent EVs production phenomenon. 60
Along with the biochemical, molecular cues, the biophysical forces and matrix stimuli have recently been proved to enhance the EVs secretion. When cyclic (10%, 40 cycles/min) mechanical stretch was applied to lung epithelial cells, a 2-fold change of EVs production was noticed with respect to non-stretched cells. 35 The cyclic stretching (20%, ten cycles/min) of periodontal ligament stem cells also increased the EVs secretion by 30-fold, which could decrease the nuclear translocation of NF-κB and IL-1β production in LPS-inflamed macrophages. 46 A study by Yu et al. 47 suggested a possible mechanism underlying the EVs stimulation by the mechanical stretch. Stretched MSCs were shown to enhance the TNF-α production and its endocytosis, which is a key feedback event to promote the MSC function and EVs secretion. From an in vivo experiment in the hindlimb, the mechanical unloading was revealed to cause MSC functional decline and bone loss, suggesting a strong role for mechanical force in increasing the EVs secretion in vivo. Other mechanical forces have also been introduced to cells, such as acoustic vibration, to increase the EVs secretion (Figure 4(b)). 48 When a high-frequency acoustic vibration (10 MHz) was applied to cells using an electrical stimulating piezoelectric device, the EVs were produced significantly more (15 times) when compared to non-vibration conditions. The higher EVs production was reasoned to the increased influx of calcium ions. 48 The external mechanical stimuli might alter the cell membrane mechanics and the ion channels, and the intracellular mechanosensitive machineries that are involved in the EVs genesis,25,65–67 which yet needs future investigation.
Other biophysical cues such as electricity, thermal, photodynamic, and radiative stress were also applied to increase the EVs secretion.49–53 Thermal (40°C) and oxidative stress (50–100 µM of H2O2) induced a 20%–70% increase in overall EV production and a 50%–200% increase of functional NKG2D-bearing EVs in leukemia/lymphoma T and B Cells, which led to enhanced immunosuppression. 49 When the oxidative stress was maintained at a low level in a chamber (0.1%–1% O2), the EVs production was significantly increased to 50%–100% in a HIF-1α-dependent pathway. 50 Photodynamic therapy is one of the emerging regenerative treatments that use low-energy light and photosensitizing chemicals. When photodynamic treatment (1 h) with Foscan® photosensitizer was performed on cells or in vivo tissues, an approximately 400-fold increase in EVs number was detected compared to without treatment. 51 A high-energy X-ray (2, 4, 6, 8, and 10 Gy) irradiation was also shown to increase the EV production by 30%–100% in an intracellular ROS-dependent manner. 52 Recently, the cellular nanoporation method was also reported to enhance the EVs secretion. Compared to bulk electroporation and other exosome-production strategies (1% O2 hypoxia and 42°C thermal stress), the cellular nanoporation using a biochip made of 500 nm diameter of nanochannels could generate more exosomes (50-fold) as well as increased the designed cargo mRNA loading in EVs (103-fold). 53
There should be possible mechanotransduction signaling pathways underlying the events in EVs production related to the applied physico-mechanical stimuli. A recent high-throughput screening study revealed that human MSCs could be mechanically conditioned to enhance vascular regeneration in vivo via the mechanosensitive signaling pathways (YAP and Smad2/3) that can be possibly engaged in the EVs production. 68 From a metabolism point-of-view, mechanical cues have a potential impact on the synthesis of lipids, a major component of EVs, via activation of lipin-1 phosphatidic phosphatase, suggesting the implication of mechanical cues in EVs secretion through altered cellular metabolism. 69
Targeting and bio-imaging ability of EVs
Targeting cells with EVs allows the determination of their in vivo journey and biodistribution as well as enhances the therapeutic effects with reduced doses. Some recent studies on EVs have underscored this area of targeting by post-treatment of EV membrane54–56 or genetic modification.18,19,70,71 While targeting is important to localize EVs, the retention at the lesion sites for a certain (long) period can enhance the bio-imaging properties. Thus, many methods were developed to enhance the systemic biodistribution56,57 and cellular internalization.54,55,58,59 Detailed descriptions of the targeting strategies can also be referred to elsewhere. 72 Earlier studies have tagged the EVs lipid membrane with fluorophores, luminescence reporters, or radiotracers at post-collection for the visualization under microscopy, CT, PET-MRI, or SPECT, which has mainly been sourced from the technologies used in different nanoparticles. Regarding this, readers are guided to refer to other well-reviewed articles.27,28
On the other hand, the genetic modification of native components of EVs has been newly designed to express a reporter, and the process is relatively biocompatible without using toxic chemicals and has long-lasting imaging ability.61,62 For example, the cell membrane was engineered to express Gaussia luciferase reporter, which was fused to a biotin receptor domain 61 (Figure 5(a)). Thus, the EVs generated from the cells can be monitored both in vitro and in vivo under luminescent or fluorescent microscopy. Gaussia luciferase and metabolic biotinylation were combined to create a sensitive EV reporter (EV-GlucB) for multimodal imaging in vivo, and bioluminescence and fluorescence-mediated imaging on animals showed a predominant localization of intravenously injected EVs in the liver and spleen. In another report, a transgenic inducible GFP EV reporter mouse containing GFP-CD9, a major EV protein component, was designed for the in vivo imaging and tracking of EVs. 62 A transgenic mouse containing a CRE-recombinase inducible CAG promoter-driven GFP-CD9 was crossed to the nestin-Cre-ERT2 mouse. This highly selective promoter-driven CRE system was injected with tamoxifen to achieve EVs-labeling in astrocytes. The GFP-labeled EVs produced from astrocytes were shown to prove the immunomodulatory roles in vivo. Although the EVs imaging techniques by gene modification during biogenesis have some merits over the post-modification process, the low imaging sensitivity or absolute intensity remains a further issue to overcome.

Strategies to enhance the targeting and bio-imaging ability of EVs: (a) genetic modification of EVs by a design of bimodal (luciferase and click-fluorescence) reporter plasmid, and the in vivo imaging confirmed after IV injection and (b) the modification of EVs membrane component (Lamb2) by engineered plasmid (either multiple cell-adhesion peptide RGD or cell-penetrating poly-Arg, R11), showing the payload (siGFP) efficacy by a silenced EGFP in EGFP HeLa model cells.
After local and systemic administration (through intravenous route) of EVs to animal models or human clinical settings (i.e. Myocardial infarction, cancer, osteoarthritis, Parkinson disease, Alzheimer disease, rheumatoid arthritis, spinal cord injury, brain stroke, dental pulp inflammation, skin burn wound healing etc.) according to “clinicalTrials.gov,”73,74 the EVs are easily cleared out or trapped in non-target organs such as liver, spleen, and lungs with very short half-lives (usually a few to 10s of minutes). Thus, enhancing the biodistribution and retention of EVs at the lesion area is necessary to enhance the targeting ability and efficacy. Immobilization of polyethylene glycol, charged, or neutral liposome or magnetic nanoparticles is some of the representative examples.56,57 Furthermore, strategies to increase cellular entry and intracellular targeting of EVs have been made. For example, cell-penetrating peptides, cationic lipids, pH-sensitive peptides were used to increase the cellular uptake and endolysosomal escape. Indeed, approximately 60% of internalized EVs could co-localize with lysosomes after 48 h of contact. 58 The modification of EVs with cationic lipids and pH-sensitive fusogenic peptides enhanced the disruption of the endolysosomal membrane with the efficient cytosolic release of EVs. 59 Likewise, the arginine-rich cell-penetrating peptides could also induce active micropinocytosis and efficient cytosolic release of EVs.54,55 These methods used in EVs share the key ideas in common with other nanoparticle modification technologies that have long been developed for cellular entry and endosomal escape.
Furthermore, the conjugation of EVs membrane with RGD or specific cell target peptide was proven to be effective. 28 Apart from the conventional physical or chemical conjugation methods, generic modification has recently gained great interest. Either multiple cell-adhesion peptides (RGD), poly-Arg (R11), or cell-penetrating peptide was inserted into the N-terminus of the Lamp2b protein, which is abundantly expressed on EVs membrane, to facilitate the target-cell binding and intracellular uptake 18 (Figure 5(b)). Cells were transfected with EVs loading exterior cargo (siRNA) for silencing target model gene, and those expressing RGD and R11 (68.9% and 75.8%) significantly improved the knockdown efficiency by 11%–18% than the unmodified wild-type EVs. Specifically, when the siGNAS and siREST were delivered to stem cells via the modified EVs, the osteogenesis and neurogenesis of cells showed a 2–3-fold increase, respectively. Other similar studies on the EVs membrane modification by generic insertion used the C1C2 domain of milk fat globule-EGF factor 8 protein or the transmembrane domain of PDGFR.70,71
One recent intriguing study has focused on the ECM penetration ability of EVs via hyaluronan degradation to enhance the targeting ability. 19 In this study, PH20 hyaluronidase was inserted on the lipid raft at the EV membrane. The enzymatic ability of the PH20-inserted EVs to penetrating the hyaluronan-rich ECM was more than twice with respect to bare EVs, which led to significant enhancement in the antitumor effects in vitro and in vivo. As witnessed, the genetic manipulation of EVs membrane has shown some promise in enhancing the targeting ability, while the improvement of gene transfection efficiency and risk of viral delivery warrant further studies.
Concluding remarks
EVs are secreted from cells into microenvironments for cell-cell communications. Thus, one stream of studies has analyzed EVs to interpret the cell-cell interactions and to identify the status of several diseases like cancers. Accumulating evidence over the last years has disclosed that the EVs are diverse in size, payload content, and therapeutic efficacy. Therefore, the other stream of studies, gaining more interest in the regenerative medicine community, has focused on how to collect EVs with high quality and quantity for therapeutic purposes. As discussed, many efforts to tackle the major issues confronted in EVs biogenesis—how to control payload content, how to enhance bioactivity, how to provide targeting and bio-imaging ability, and how to generate in large scale for mass production—have been taken by various methods, either during biogenesis or at post-collection stages.
While earlier studies have mainly focused on the modulation of biochemical, molecular cues, such as culture medium ingredients and oxygen tension, more recent works started to utilize the biophysical and mechanical stimuli and 3D cultures with engineered biomaterials, which are considered to mimic the in vivo tissue conditions (e.g. endothelial cells under cyclic stretch, 3D spheroid cultures for tumor cells) where cells would favor the engineered environments to secrete therapeutically-relevant EVs. One recent study by Yang et al. 53 introduced a cellular nanoporation method where a focal and transient electrical stimulus was used to generate exosomes carrying transcribed mRNAs and targeting peptides. Compared to conventional bulk electroporation, the nanoporation stimulation could generate more exosomes (50-fold) and a significant increase in mRNA transcripts (103-fold), demonstrating the impacts of engineered stimulation of cells in the biogenesis of EVs.
Likewise, the tissue-mimic ex vivo culture systems might help to understand the roles of EVs and the related mechanisms happening in the in vivo tissues. One recent study by Lenzini et al. 75 has demonstrated the decisive role of ECM in the extracellular journey of EVs, underscoring that the design and recapitulation of the context-dependent tissue-mimic matrix are important for interpreting EVs transport in a specific disease or repair model. The interactive phenomena in the “EV-cell-matrix axis” are thus considered an important area to understand, which will help how to design and engineer the environments for EV-producing cells and ultimately to generate EVs more therapeutically relevant.
While some exemplar studies have shown the effective role of the biophysical and mechanical stimulation in the EVs biogenesis, the mechanisms underlying the events remain largely elusive. “Mechanobiology” would be the right language that can interpret the relationship of biophysical/mechanical stimulation and EVs biogenesis. External matrix and mechanical stimuli are well recognized to transmit toward intracellular and nuclear components through various mechanosensitive machineries (integrins, actin filaments, LINK molecules); including these, some recent highlights on the physical stimuli linked with ion channels and cell metabolism might help to elucidate the mechanistic events in the EVs biogenesis.
As witnessed, the potential and impact that EVs hold in the regenerative medicine area are high, and the technological advances in EVs’ biogenesis evolve very rapidly. The emerging technologies in EVs biogenesis to secure controlled payload content and bioactivity and enhance cellular/subcellular targeting and bio-imaging ability, together with the production on a large scale, are envisaged to speed up the clinical translations, as next-generation therapeutics for the treatment of diseased and injured tissues.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the National Research Foundation of Korea (NRF) (2020R1A2C1005867, 2018K1A4A3A01064257, 2019R1A6A1A11034536, and 2019R1C1C1002490). The present research was also supported by the research fund of Dankook University for the 2019 University Innovation Support Program.
