Abstract
Muscular dystrophies are a group of highly disabling disorders that share degenerative muscle weakness and wasting as common symptoms. To date, there is not an effective cure for these diseases. In the last years, bioengineered tissues have emerged as powerful tools for preclinical studies. In this review, we summarize the recent technological advances in skeletal muscle tissue engineering. We identify several ground-breaking techniques to fabricate in vitro bioartificial muscles. Accumulating evidence shows that scaffold-based tissue engineering provides topographical cues that enhance the viability and maturation of skeletal muscle. Functional bioartificial muscles have been developed using human myoblasts. These tissues accurately responded to electrical and biological stimulation. Moreover, advanced drug screening tools can be fabricated integrating these tissues in electrical stimulation platforms. However, more work introducing patient-derived cells and integrating these tissues in microdevices is needed to promote the clinical translation of bioengineered skeletal muscle as preclinical tools for muscular dystrophies.
Introduction
Muscular dystrophies (MD) are a group of genetically inherited muscle degenerative disorders characterized by muscle weakness and wasting. 1 The disorders differ in the age of onset, rate of progression, pattern of inheritance, and the type of muscles that are affected (Table 1). Depending on the type of muscular dystrophy, axial, limb, or facial muscles could be affected by a different degree of muscle degeneration and weakness. In specific MDs, other muscles such as cardiac and respiratory are also involved. Moreover, the pathology of some MD disorders has an impact on other organs and tissues as the brain, the skin, the testis, or the eyes. Very rare muscular dystrophies variants are continually identified. To date, more than 50 genes have been determined to be involved in more than 70 inherited muscular dystrophies. 2 These disorders are usually classified into nine main categories or types: myotonic, Duchenne, Becker, Limb-girdle, facioscapulohumeral, congenital, oculopharyngeal, distal, and Emery-Dreifuss. 3 Myotonic syndromes are the most common form in the adult population affecting 1 per 3000 people. 4 In contrast, Duchenne muscular dystrophy is the most prevalent in childhood and is found in roughly 1 per 5000 boys. 5 Although individually muscular dystrophies are considered rare diseases, the overall prevalence of combined muscular dystrophies was 16.14 per 100,000, 6 and therefore, they have a great impact on society.
Muscular dystrophies classification.
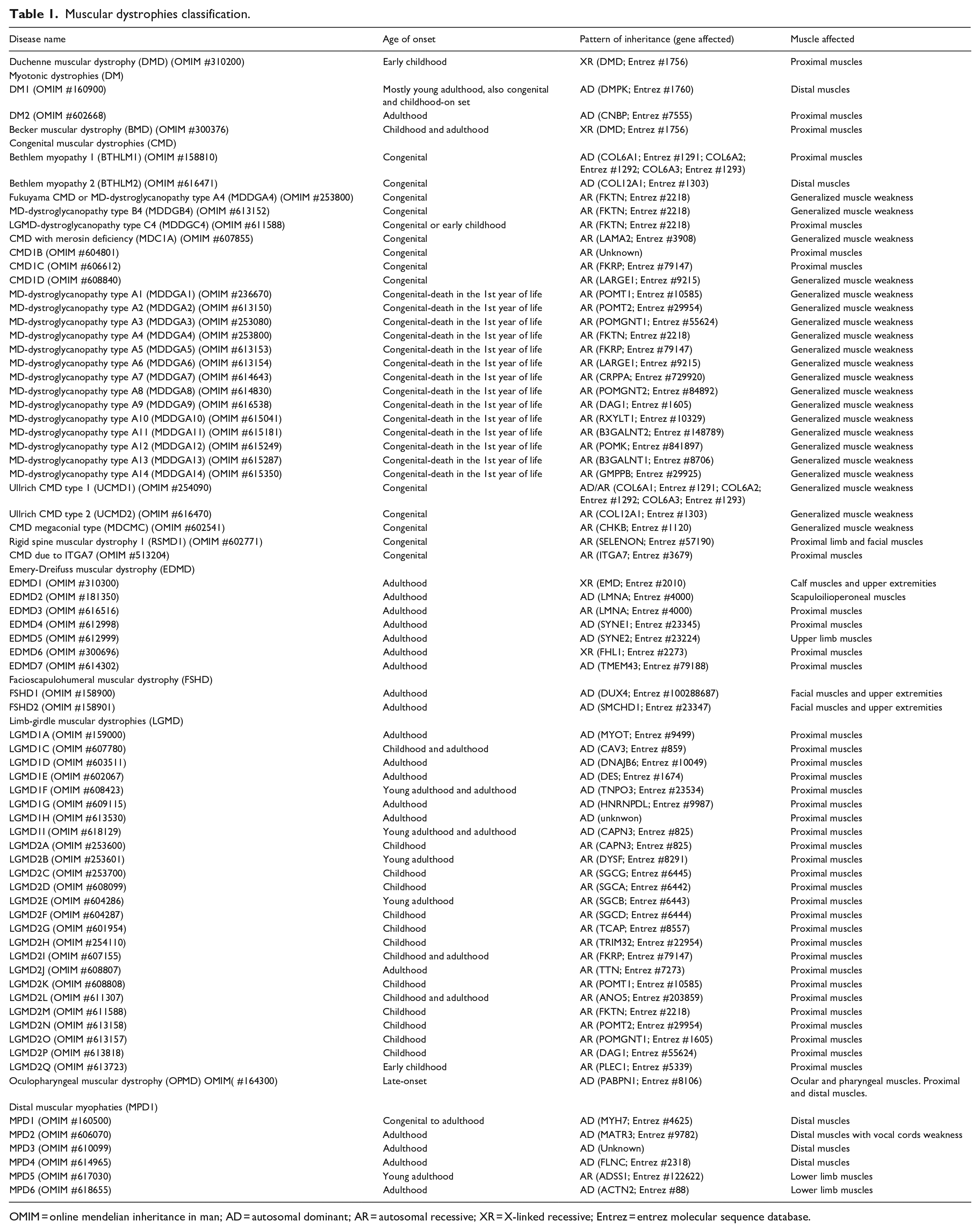
OMIM = online mendelian inheritance in man; AD = autosomal dominant; AR = autosomal recessive; XR = X-linked recessive; Entrez = entrez molecular sequence database.
Palliative treatments are availed for these diseases, although there is no cure for any muscular dystrophy to date. Remarkably, in the last decade, some drugs for specific muscular disorders have been developed, including small molecules and antisense oligonucleotides.7 –11 However, these treatments must demonstrate efficacy and safety in the clinical phases before being widely used in patients. The development of a new drug takes years, even decades, from preclinical studies to reach the market and it is a very costly process. 12 Drug development implies three main stages: discovery, preclinical studies, and clinical trials. During the preclinical stage, extensive studies with in vitro and animal models provide preliminary information on the efficacy, toxicity, pharmacokinetics, and safety of a drug candidate. However, the probability of success of a molecule after entering the clinical phases is only 10%. 13 This low success rate is mostly due to the high toxicity or low efficacy of the drug in humans. This suggests that current preclinical models should be improved to reduce costs and accelerate drug development time. Furthermore, the heterogeneity of muscular dystrophy manifestations anticipates that each patient would respond to treatments differently. Therefore, there is also a clinical need for personalized therapies to target these diseases effectively. Human bioengineered in vitro tissues are emerging as new tools for preclinical research.14,15 Specifically, bioengineered in vitro skeletal muscles would provide more efficient and predictive models to improve drug development for muscular dystrophies. Here, we discuss the recent advances in skeletal muscle tissue engineering, focusing on new human in vitro models for muscle analyses and their application in the study of muscular dystrophies. This review was designed with two main purposes: to bring the clinician community closer to the bioengineering technologies for skeletal muscle and to show a general overview of the complexity of muscular dystrophies to researchers working on skeletal muscles tissue engineering.
Skeletal muscle structure and myogenesis regulation
Skeletal muscle is the largest tissue in the body, accounting for almost 40% of body mass. 16 Skeletal muscle architecture is characterized by a very particular and well-described arrangement of muscle fibers, also called myofibers. Groups of myofibers form the fascicles, and bundles of fascicles make up the whole muscle tissue (Figure 1). Myofibers are formed by the fusion of myoblasts to produce multinucleated myotubes, which mature further in myofibers. 17 The dimensions of a single myofiber are approximately 100 µm in diameter and 1 cm in length, and its nuclei are located along the periphery of the whole fiber. The mature skeletal myofiber contains a highly organized cytoskeleton made up of aligned myofibrils, which are repeating consecutive sections of the functional unit of skeletal muscle, the sarcomere. 18 Sarcomere complex structure is composed of two main alternating sets of protein filaments: thin filaments (α-actin and associated proteins) and thick filaments (myosin and associated proteins), which run parallel to the muscle fiber axis. At a molecular level, the sarcomeric contraction consists of the movement of the myosin heads on actin filaments. Thus, the formation of correctly aligned myofibrils is fundamental for muscle function. So, to engineer skeletal muscle in vitro, its complex anatomy structure must be mimicked using biomaterial scaffolds and specific biofabrication techniques.

Structure of muscle anatomy. Epimysium covers each muscle. Muscle fibers are grouped forming bundles (fascicles). Each fasciculus is surrounded by perimysium while endomysium surrounds the myofibers. Each individual myofiber or myotube have a membrane (sarcolemma) and is composed of hundreds of myofibrils. Myofibrils are the functional contractile unit of the muscle and are surrounded by sarcoplasm.
Muscle development and regeneration are regulated by a hierarchy of myogenic regulatory factor (MRF) family of transcription factors 19 (Figure 2). Stem cells in the muscle are called satellite stem cells (SSCs) and are produced by activation of precursor cells expressing PAX3 and PAX7.20,21 The expression of MYF5 and MYOD1 in SSCs induces the myogenic program to produce myoblasts. Myogenesis process continues with the activation of MYOG and MEF2 and the downregulation of MYF5 and MYOD1. Activation of a second wave of MRFs (MYOG and MRF4) induces terminal differentiation of myoblasts into myotubes. Mature myotubes additionally express muscle-specific genes such as the contractile proteins of the muscle as myosin heavy chain (MHC), actin, titin, among others, and a reduction in expression of MYOG. 22

Regulation of myogenic differentiation by a hierarchy of transcription factors. Satellite cells expressing PAX7 derive from the PAX3/PAX7 expressing progenitor cells. Following activation of MYF5 expression in satellite myogenic cells, the myogenic program starts, and MYOD1 expression is activated. Activation of MYOG and MEF2, with downregulation of MYF5 and later MYOD1, marks the start of terminal differentiation into myofibers. Activation of MRF4 happens several days after the induction of differentiation, following a reduction in MYOG expression.
The skeletal muscle extracellular matrix
As mentioned above, the formation of aligned myofibers is an essential factor for skeletal muscle tissue engineering. Nonetheless, it is important to consider that in vivo, these fibers are embedded within a 3D scaffold, the skeletal muscle extracellular matrix (ECM), mainly composed of collagens, non-collagenous glycoproteins, proteoglycans, and elastin. 23 Besides being a scaffold for cell-matrix interactions, the skeletal muscle ECM plays a crucial role in skeletal muscle function. For instance, it allows a uniform distribution and transmission of force within muscle and from muscle to tendon. Furthermore, the ECM interactions are critical to maintaining the mechanical homeostasis within the muscle. 18
The skeletal muscle ECM is traditionally classified into three organized layers: endomysium, perimysium, and epimysium (Figure 1). The endomysium (also called basement membrane) surrounds individual myofibers. The perimysium is a thickened ECM that encapsulates the fascicles, and the epimysium is a dense layer that surrounds the whole muscle. However, as the knowledge about the ECM roles and complexity increases, it has been argued that a less simplistic ECM organization should be established.18,23,24
Among the skeletal muscle ECM proteins, collagens are the most abundant, glycoproteins are involved in tissue organization and cell-matrix interactions, and proteoglycans are involved in signaling and tissue regeneration. 18 The basement membrane consists mainly of collagen type IV and laminins, whereas collagen type I is found mostly in the peri- and epimysium. Together all types of collagens provide structural support and allow tissue mechanotransduction. 25 Briefly, fibrillar collagen type I provides tensile strength and rigidity, while collagen type III fibers form a loose mesh that gives elasticity to the inner layers of the intramuscular connective tissue. Moreover, collagen type IV integrates laminins and other proteins into a stable structure. In less abundance, collagen type VI is present in all ECM layers. 23 Interestingly, collagen type VI mutations cause disorders involving both muscle and connective tissue, such as congenital muscular dystrophies as Ullrich CMD type 1 (UCMD1) and Bethlem Myopathy type 1 (BTHLM1) (see Table 1). 26
Engineering topographical cues for skeletal muscle 2D culture
Skeletal muscle represents a complex and challenging tissue for in vitro generation through tissue engineering. Given the importance of the aligned structure of myofibers for their correct functionality, the main fabrication strategies are focused on providing geometric cues (Table 2). The most recent published approaches to culture myofibers on aligned structures can be classified into three main types: micropatterning, electrospinning, and bioprinting.
Novel bioengineering approaches to fabricate skeletal muscle models.

Micropatterning
The simplest models for generating skeletal muscle in vitro are based on the 2D culture of myoblasts seeded on micromolded substrates. RNAseq analyses of C2C12 murine myotubes on micropatterned gelatin hydrogels showed that cells display an increased ability to form aligned sarcomeres and increased contractile protein expression, demonstrating the effect of topographic cues in the maturation of muscle cells. 27 Another technique to generate 2D micropatterned scaffolds is microcontact printing. For example, Vajanthri and collaborators used the microchanneled flowed plasma process to generate different cell adhesive micropattern coated glass with 3-amino-Propyltriethoxysilane (APTES) and Octadecyltrichlorosilane (OTS). 28 Aligned myotubes grew onto stamped micropatterned regions. Micropatterned substrates also emerged as great potential substrates to align and differentiate myoblasts. 29 In this study, the authors generated a new platform by crumpling graphene uniaxially (Figure 3(a)). Micropattern dimensions were modulated by applying compressing strain. Culture of C2C12 mouse myoblast on this uniaxially crumpled graphene promoted the alignment and elongation at a single-cell level and enhanced differentiation and maturation of myotubes. Other graphene-based approaches used femtosecond laser ablation to generate a patterned substrate in polyacrylamide hydrogels. 30 Studying different pattern distances, the authors concluded that 50 µm of spacing produced the better alignment and maturation of myotubes. Myogenic differentiation could also be improved by applying electrical stimulation to these micropatterned cultures. An alternative approximation used poly(ethylene glycol) (PEG)-based microgrooved hydrogels to provide topographical and electrical stimuli to cells. 31 To enhance hydrogel electrical properties and cell attachment, poly(3,4-ethylenedioxythiophene) (PEDOT) and tripeptide Arg-Gly-Asp (RGD) were combined with the PEG-hydrogel.

Engineering topographical cues for the 2D culture of skeletal muscle. (a) Fabrication of an anisotropically crumpled graphene platform by releasing the elastomeric substrate on which a graphene film was attached. C2C12 cells were cultured on top of these substrates to differentiate and align C2C12 cells on crumpled graphene. Adapted from Kim et al. 29 . (b) Gelatin methacryloyl (GelMA) hydrogel micropatterns were 3D printed on a thermo-responsive polymer (poly(N-isopropylacrylamide), PNIPAm). Temperature control was used to promote directed collective C2C12 cell migration on the GelMA patterns to induce myotube formation and orientation. Adapted with permission from Du et al. 38 . Copyright 2019 American Chemical Society. (c) Schematic representation of the fabrication process of unpatterned and micropatterned GelMA fibers. Field emission-scanning electron microscope images showing surface structures of the GelMA fibers. Scale bars: 20 μm. Adapted with permission from Ebrahimi et al. 40 . (d) Characterization of endothelialized engineered murine muscle. Scanning electron microscopy images of randomly oriented or aligned scaffold nanofibers. Confocal microscopy images showing myosin heavy chain (MHC, green) and CD31 (magenta) staining in differentiated myotubes in randomly oriented or aligned scaffolds. Adapted from Nakayama et al. 41 . The images in panel (c) are not published under the terms of the CC-BY license of this article. For permission to reuse, please see Ebrahimi et al. 40 .
Electrospinning
One of the most common techniques for 2D cell guidance and tissue engineering is electrospinning. 32 Electrospinning technique has been successfully used to generate mature myotubes over microfibres fibrin bundles. 33 Mechanical strains were applied to enhance the myogenic differentiation of the immature myoblasts. In this study, several strain cycles were tested, and delayed strain onset improved or maintained myogenic outcomes. Electrospinning technique has also been used to co-culture C2C12 murine myoblasts and human umbilical vein endothelial cells (HUVEC). 34 To guide the cells and promote their fusion, the authors used Polyvinyl alcohol (PVA)-leached polycaprolactone (PCL) and collagen struts as mechanical supporters with topographical cues. HUVECs-laden alginate bioink was uniaxially electrospun on these supporters. C2C12 myoblasts seeded on this vascularized scaffold formed mature myofibers with striated myosin heavy chain (MHC) protein patterns. Recently, poly (vinylidene fluoride) (PVDF) was used as an electroactive biomaterial to enhance the myoblasts fusion and myogenic maturation due to its piezoelectric properties. 35 A piezoelectric material becomes electrically polarized upon a mechanical stimulation. 36 This propriety can be used to give an electrical signal to the cells. In this work, C2C12 cells were seeded on an electrospun aligned matrix of PVDF. Charged surfaces improved the fusion of muscle cells into differentiated myotubes.
Bioprinting
Similar to electrospinning, bioprinting and other 3D-based approaches have been widely used to generate highly aligned fibers.37 –42 E-field printing, a combination of e-field and 3D printing, was used to engineer PCL highly hierarchical scaffolds. 37 The growth of C2C12 myotubes on top of these scaffolds enhanced cell alignment and increased myogenic markers. Another technique to promote myotubes alignment is culturing them on bioprinted gelatin methacryloyl (GelMA) over a thermoresponsive poly(N-isopropylacrylamide) (PNIPAm) coated substrate. 38 After seeding cells, the temperature was lowered to detach cells from PNIPAm surfaces. With this approximation, directed collective cell migration was regulated (Figure 3(b)). Similarly, microfluidic spun GelMA fibers with have a well-defined surface morphology have been generated by extrusion through a microgrooved mold (Figure 3(c)).39,40 Topographical cues on micropatterned GelMA fibers promoted alignment of C2C12 myoblasts and myotube formation. The combination of topographical cues with agrin treatment further enhanced myotube maturation and functionality, as shown by improved contractility under electrical stimulation. 40 Extrusion of collagen fibers has also been used as geometrical cues to fabricate endothelialized and aligned skeletal muscle.41,42 Co-culture murine myoblasts with endothelial cells on nano-fibrillar collagen scaffold strips promotes the formation of highly organized myofibers and microvasculature (Figure 3(d)). Remarkably, the implantation of these scaffolds in injured mice muscle favored vascular regeneration as a promising treatment of volumetric muscle loss. 41 In summary, the results obtained by micropatterning, electrospinning, and bioprinting approaches confirm that the geometrical cues are fundamental for engineering mature myotubes in vitro.
3D Engineering for skeletal muscle culture
Bioengineering approaches in 2D models have been useful to study biomaterials and the importance of the topological cues for in vitro culture of skeletal muscle tissue. However, these techniques do not resemble the environment of the native skeletal muscle. For this reason, three-dimensional scaffolds have become the gold standard to generate skeletal muscle tissue. In order to mimic the extracellular environment and the native cellular morphology, the main bioengineering strategy is focused on the 3D encapsulation of muscular cell precursors in biocompatible materials. In the last years, 3D bioprinting,43 –54 hydrogel molding,55 –59 and microporous scaffolds60 –62 have been implemented to fabricate skeletal muscle tissues.
3D Bioprinting
Several biomaterials can be used as bioinks to print encapsulated myoblasts in various 3D structures to obtain aligned myotubes. Among these, GelMA and other methacrylated polymers are the most used biomaterials in bioprinting due to is photocrosslinkable properties. 3D bioprinting was used to fabricate tissue constructs using GelMA with alginate and C2C12 cells. 44 Interestingly, the metabolic activity of myotubes was improved by adding calcium peroxide, an oxygen-generating particle, to the bioink. Oxidized alginate has also been used in combination with gelatin as a bioink. The cells orientate with the proper nozzle size and extrusion pressure due to the shear stress during the bioprinting process. 47 The cells grow in the direction of printing, migrate to the hydrogel surface over time, and differentiate into aligned myotubes. 3D printed GelMA-alginate hydrogels with Plurionic F-127 as a sacrificial layer were used to induce a macroscale level of controlled cell alignment with angle variation. The variation in the grid pattern angles was designed to mimic the fibril orientation of native tissues, where angles of cell alignment vary across the different layers. In a similar approach, GelMA and collagen methacrylate were bioprinted and UV crosslinked to generate different 3D structures and obtain mature aligned myotubes. 48 To better control the cell alignment, cell aligners could be added to the bioinks. One way to achieve this was by printing collagen with gold nanowires and applying a field into this extruded biomaterial. These gold nanowires were aligned following the desired directionality by applying an electric field. The use of gold nanowires enhanced myoblast alignment inside the hydrogel by contact guidance. 49 Moreover, the addition of gold nanowires improves the electrical properties of the scaffolds. These works used bioinks based on natural polymers as gelatin or collagen due to their biocompatibility. However, these materials are degradable by mammalian cells, causing a loss of the hydrogel structure, limiting long-term cell cultures. To overcome this, materials that are non-degradable by mammalian cells have been combined with these natural polymers to improve the mechanical properties of the scaffolds. C2C12 cells were encapsulated in different combinations of GelMA-based composite bioinks. 50 A structure of hydrogel filaments was bioprinted with these bioinks (Figure 4(a)). The combination of GelMA with methacrylated carboxymethylcellulose (CMCMA) resulted in the most suitable properties for skeletal muscle tissue engineering. Remarkably, the GelMA-CMCMA composite biomaterial has been used for the long-term culture of C2C12 myotubes. The 3D structure of the hydrogel remained in time for 3 weeks, enhancing myotube maturation. 57 Alternative combinations of natural and synthetic biomaterials are suitable for bioprinting skeletal muscle cells. Aligned myoblast-laden hydrogels of PEG-fibrinogen have been produced using microfluidic-enhanced 3D bioprinting. 51 Moreover, Pluronic/alginate blends have been investigated as a model system for culture of C2C12 murine myoblast. 52 Fabricated constructs exhibited high cell viability, as well as a significantly improved expression of myogenic markers. To mimic the extracellular matrix, poly lactic-co-glycolic acid (PLGA) multilayered scaffolds were made with E-jet 3D printing. 53 By comparing different fibrilar gaps in the scaffolds, the authors concluded that 50 µm gaps enhance cell adhesion and proliferation. In all the previous works, the bioprinting design was based on lines or meshes to guide the alignment of myotubes. Alternatively, hydrogels can be printed to compact around post structures and generate skeletal muscle tissue.45,46,54 These kinds of platforms allow the force measurement of skeletal muscle after electrical stimulation. Skeletal muscle tissue contractions cause the bending of the pillar. This displacement is used as a proportional measurement of the muscle bundle forces.

Engineering strategies for the 3D culture of skeletal muscle tissue. (a) C2C12 myotubes formation in bioprinted or molded composite hydrogels. Top view images of the composite hydrogels after fabrication. Confocal microscopy showing F-actin in red, MHC in green, and nuclei in blue. Scale bar: 200 μm. Adapted with permission from Garcia-Lizarribar et al. 50 and authors unpublished results. (b) Multi-assay 3D printed poly(ethylene glycol) diacrylate (PEGDA) hydrogel platforms for casting fibrin hydrogel muscle bundles. Adapted with permission from Christensen et al. 59 . Copyright 2020 American Chemical Society. (c) Confocal microscopy images showing aminofluorescein-marked cryogels in green, cells marked with phalloidin in red and cell nuclei counterstained in blue with DAPI. Scale bars: 100 μm. Adapted from Velasco-Mallorqui et al. 61 . The images in panel (a) are not published under the terms of the CC-BY license of this article. For permission to reuse, please see Garcia-Lizarribar et al. 50 .
Micromolding
To generate aligned scaffolds, an alternative technique to bioprinting is hydrogel micromolding. Molds of polydimethylsiloxane (PDMS), a biocompatible transparent polymer, are the most used to transfer aligned micropatterns to the 3D hydrogels.63,64 Optimal stiffness of hydrogels for skeletal muscle bioengineering was determined by changing GelMA concentration and UV crosslinking time. Using molds with channels of different widths, the authors investigated the effect of geometrical confinement and hydrogel stiffness for C2C12 myotube culture. 55 The best results were obtained with the thinnest 0.5 mm channels and a low stiffness between 1 to 3 kPa. Murine skeletal muscle tissue has also been generated by micromolding of GelMA-CMCMA hydrogels.56,57 Here, C2C12 cells were encapsulated by photomold patterning of the hydrogel using a microgrooved PDMS stamp (Figure 4(a)). Interestingly, these tissues were implemented in cytokine sensing platforms to analyze responses to biological and electrical stimuli. Micromolding and micromilling techniques were combined with posts to develop a new organ-on-a-chip set up with a vessel embedded system. 58 To vascularize the engineered muscle bundle, C2C12 and HUVEC were co-cultured by using collagen and a sacrificial layer and supported by pillars. To mass-produce structures with posts or cantilevers, Christensen et al. described a stereolithographic method to 3D print poly(ethylene glycol) diacrylate (PEGDA) hydrogels with high precision and high accuracy (Figure 4(b)). 59 These PEGDA platforms with anchored cantilevers were used to cast fibrin hydrogel muscle bundles around these pillars.
Extracellular matrix derived materials obtained by decellularization (dECM) have emerged as novel natural hydrogels to engineer muscle tissue.65,66 These dECM-derived hydrogels contain growth factors, cytokines, proteoglycans, and structural adhesive proteins, which represent tissue-specific biochemical cues. 67 Murine skeletal muscle generated with dECM scaffolds present more mature myotubes than with collagen scaffolds. 65 Moreover, a significantly greater number of myofibers were observed when compared to collagen scaffolds after implantation of both engineered skeletal tissues in a rabbit tibialis anterior (TA) muscle defect model. Going a step further, methacrylation of dEMC allows bioprinting and photocrosslinking of the hydrogels, providing topological cues for skeletal muscle engineering. 66
Microporous scaffolds
An alternative fabrication method that allows the generation of millimeter range scaffolds is cryogelation. 60 Cryogels are microporous scaffolds with a pore range from a few micrometers up to hundreds of micrometers. 68 After freezing the polymer solution, ice crystals are formed. Once the cryogel is thawed, the ice crystals leave behind empty pores. The pore morphology can be modulated by applying different freezing directionalities.69,70 Highly aligned morphology is a necessary geometrical cue for skeletal muscle maturation. For this reason, anisotropic gelatin-cellulose cryogels were generated to engineer volumetric skeletal muscle using C2C12 myoblasts. 61 Anisotropic cryogels improve cell alignment, myotube fusion, and myogenic maturation (Figure 4(c)). Moreover, the addition of carbon nanotubes (CNTs) to the cryogel improves the electrical properties of the scaffolds, which enhances early myogenic maturation steps when electric pulse stimulation is applied. Interestingly, these gelatin-cellulose cryogels are easy to handle without affecting their shape. Therefore, this technique has strong possibilities for tissue engineering and organ-on-a-chip technologies.
Microporous scaffolds can also be used for skeletal muscle cell delivery by generating highly open porous microspheres (HOMPs). 62 These microspheres were fabricated of biocompatible PLGA by microfluidic droplet emulsion. These HOPMs with interconnected pores facilitated a high cell adhesion rate, continuous proliferation, and augmented myogenic differentiation of C2C12 after transplantation in mouse muscle.
Computational modeling
Despite the promising potential of all these approaches to generate 3D muscle tissues in vitro, there are still many technical limitations. To complement the lack of chemical or mechanical information of many experimental models, computational modeling has gained a relevant role. Using computational methodologies as agent-based model (ABM) for individual cell modeling, or finite element method (FEM) for cellular population density, it is possible to study the cell behavior inside scaffolds and between cells. As a clear example of this combination between technical and computational methods, Torri et al. 71 used the data from the previous work from Smith et al. 72 to analyze the muscle cell behavior in silico. They applied both modeling approaches with successful results achieving a good resemblance between their approximations in silico and the in vitro studies. These analyses point to the potential of computational modeling to predict uncertain variables and complement in vitro experiments.
Functional human skeletal muscle tissue models
In the last decade, skeletal muscle bioengineering techniques have been developed using mainly murine myoblasts as a cell model. These studies shed light on important features for skeletal muscle tissue engineering, such as topological cues, biomaterials, and biochemical factors. The next step in skeletal muscle bioengineering is incorporating human cells to obtain more relevant models for muscular dystrophies. Human skeletal muscle tissue models have been developed from primary and immortalized human myoblasts and human induced pluripotent stem cells (hiPSCs) [reviewed in 73 ]. It has recently been shown that human amniotic mesenchymal stem cells (hAMCs) can undergo myogenic differentiation. Moreover, hAMCs express key growth factors that promote endothelial cell proliferation and angiogenesis, representing a great advantage as a cell source for skeletal muscle tissue engineering. 74
In general, human myogenic precursor cells are encapsulated in hydrogel scaffolds that aim to mimic the 3D environment of native muscle tissues. The most common hydrogels are those of natural origin, especially collagen,75,76 fibrin,77 –82 and dECM. 83 The main fabrication strategies have been hydrogel molding75 –80 or 3D printing81 –83 (Table 2). In this way, cell alignment is achieved through passive tension, allowing long-term cell culture, and enhancing muscle maturation and function. The first 3D skeletal muscle tissues reported consisted of myoblasts encapsulation in a collagen I matrix that contracts around pillars.84,85 The contraction of the matrix promotes cell alignment in the direction of the anchoring points, producing long multinucleated myofibers. 75 Collagen I is one of the main components of the skeletal muscle extracellular matrix. 23 However, it has been observed that myogenic maturation and contractile force of the tissue can be compromised by the relatively high stiffness of the collagen hydrogel, especially for large macroscopic constructs.25,86 For this reason, in the last years, efforts have been directed to incorporate materials with better mechanical properties. Fibrin composite hydrogels have resulted in the most promising materials to generate functional skeletal muscle constructs due to their ability to be remodeled by cells and induce ECM synthesis. 87
Functional human skeletal muscle tissues or bioartificial muscles (BAMs) were developed for the first time in the Bursac lab using primary human myoblasts. By molding fibrin-Matrigel® hydrogels within PDMS molds inside a nylon frame, the authors created muscle 3D bundles 77 (Figure 5(a)). In this platform, active force measurements were performed in response to electrical and biological stimulation. In 2018, following the same biofabrication approach, the authors reported the generation of skeletal muscle tissues derived from direct reprogramming of hiPSCs. 78 Overexpression of PAX7 was induced to generate satellite cell-like cells. Remarkably, human muscle 3D bundles were kept in culture for up to 4 weeks, being the longest culture time reported to the date. Of note, muscle constructs presented a correct membrane localization of dystrophin and generated active twitch and tetanic contractions. Interestingly, after 4 weeks of culture, these tissues retained a pool of PAX7+ cells together with MYOG+ myotubes, mimicking the satellite-cells presence in native muscle. Although these models present different myotube maturation levels, it has been shown that electrical stimulation training enhances myofiber hypertrophy and metabolic flux. 79 Electrical stimulation training during 1 week of human myoblasts-derived muscle 3D bundles promoted an increase of myotube diameter by 40%. Using this electrical stimulation protocol, the authors measured the highest specific forces reported to date for an engineered human muscle (19.3 mN/mm2). A more complex human skeletal muscle model was achieved by co-culturing primary human myoblasts with human stem cells-derived motoneurons in a fibrin-based hydrogel. 80 Briefly, Fibrin-Geltrex® hydrogels were fabricated by molding with Velcro® anchors that acted as artificial tendons to apply uniaxial tension. The resulting bioengineered human skeletal muscle tissues were able to form neuromuscular junctions (NMJs) in a long-lasting culture for up to 3 weeks (Figure 5(b)). NMJs play a key role in several muscular dystrophies, 88 which could be modeled following this innovative approximation.

Human 3D engineered skeletal muscles. (a) Structure of hiPSC-derived muscle bundles anchored within a nylon frame. Scale bar: 5 mm. Representative longitudinal section of 2-week differentiated bundles showing aligned, cross-striated myotubes SAA = sarcomeric alpha-actinin, BTX = α-bungarotoxin labeling acetylcholine receptors (AChR) and DAPI counterstain myotube nuclei. Scale bar = 25 μm. Adapted from Rao et al. 78 . (b) Stitched phase contrast image of a representative 3D skeletal muscle-motor neuron (MN) co-culture. Neuromuscular tissue is outlined with a red dashed line in the left panel. The region outlined in the green box is magnified in the image to the immediate right. Red dashed lines in the right panel outline motoneuron clusters. Scale bars: 2 mm (left panel) and 200 mm (right panel). Adapted from Bakooshli et al. 80 . (c) Schematic representation of coaxial printing. Immunofluorescent image of a 3D printed muscle construct CD31 = cluster of differentiation 31 labeling endothelial cells, MHC = myosin heavy chain, and DAPI = stained nuclei. The images were taken from the center of the construct. Adapted with permission from Choi et al. 83 . The images in panel (c) are not published under the terms of the CC-BY license of this article. For permission to reuse, please see Choi et al. 83 .
3D bioprinting has also been applied to fabricate human skeletal muscle bundles.81,83 Cell-laden fibrin bioink was bioprinted using gelatin as a sacrificial material to generate organized multilayered muscle bundles supported by a PCL pillar structure. 81 Myofibers in these bioartificial muscles were densely packed and highly aligned. The generated constructs were studied in vivo, where they achieved 82% of functional recovery in a rodent model of tibialis anterior muscle defect at 8 weeks of post-implantation. Good integration with host vascular and neural networks was observed. Alternatively, dECM have been used as a bioink for 3D printing of human skeletal muscle bundles. 83 Primary human myoblasts were printed with dECM from porcine muscular tissues in granule-based reservoirs. Moreover, coaxial printing with endothelial cells, using porcine blood vessel-derived dECM bioink, has allowed the prevascularization of these muscle bundles (Figure 5(c)). Implantation of these prevascularized muscle bundles in an injured muscle rodent model resulted in the high viability of the cells without generating hypoxia and enhanced de novo muscle formation.
Bioartificial muscles have been recently tested as drug screening platforms. Human myoblasts encapsulated in fibrin hydrogels were introduced in a device with a stereotactic setup that allows controllable injection at BAMs. 82 While most of the biological stimulation assays in 2D and 3D in vitro models are performed by adding a compound to the culture media, this setup allows modeling intramuscular drug delivery in vitro. Using spectrophotometry and luminescence, the release of the injected compounds and their metabolites were measured over time. Although these models are useful for intramuscular drug injection studies, their relatively large size limits their potential as a drug screening platform. BAMs fabrication requires large numbers of cells, which can be costly and difficult to obtain. Current microfabrication technologies have allowed the miniaturization of these tissues to obtain high-throughput systems.46, 76, 89, 90 For example, a 96-well micro-muscle platform using primary human myoblasts. 76 Cells were encapsulated in a collagen-Matrigel® composite matrix around micropillars. The authors affirmed that with this approach, they could reduce the size, reagents, and cost by a factor of ≈25 compared to the state-of-the-art skeletal muscle bioengineering approaches mentioned above.77,78 However, the reported specific forces and protein expression levels obtained with this miniaturized system are representative of immature muscle in a fetal-like state. Hence, intensive efforts in training and maturation of the microBAMs are needed to develop a successful high-throughput screening platform.
Modeling muscular dystrophies using tissue engineering
The intrinsic heterogeneity of muscular dystrophies means that the future of effective treatments for patients lies in personalized medicine. To date, mutations that cause specific muscular dystrophies have been described in more than 50 genes (Table 1). As a consequence of this genetic heterogeneity, specific muscle types are affected with a variable degree of progression in the dystrophic syndromes. Moreover, in some muscular dystrophies, disease progression and severity depend on individual patients. Therefore, developing in vitro bioengineered tissues in the laboratory from patient-derived cells is necessary to study personalized therapies. To date, much effort has been put into developing better scaffolds or studying new biofabrication methods to develop skeletal muscle tissues in vitro. However, few works have introduced patient-derived cells, going a step further to create these personalized platforms. One of the first approximations was to culture myoblasts from Duchenne muscular dystrophy (DMD) patients on micropatterned polyacrylamide hydrogels functionalized with either laminin, fibronectin, or Matrigel®. 91 In this work, the authors functionalized the surface in parallel lines by micro-contact printing of these adhesion proteins. After 7 days of culture onto these hydrogels, the cells formed mature aligned myotubes with sarcomeric organization. Remarkably, myotubes cultured onto hydrogel with Matrigel® showed the highest level of expression of the muscle maturation markers as myosin heavy chain II and α-actinin. Moreover, DMD myotubes showed particular pathological hallmarks, such as the decreased expression of dystrophin, while the formation of sarcomeres remained unchanged. Although mature aligned myotubes could be obtained by culturing patient cells onto functionalized micropatterned polyacrylamide hydrogels, these 2D models do not represent the complexity of skeletal muscle tissues. In a recent study, human induced pluripotent stem cells (hiPSCs) were used to generate 3D engineered skeletal muscle tissues. 92 The authors cultivated human hiPSCs derived from Duchenne (DMD), limb-gridle, and other congenital muscular dystrophy patients encapsulated in fibrin hydrogels (Figure 6(a)). To mechanically stimulate myogenic differentiation, the hydrogels were cultured under tension. The resulting tissues showed mature myotube markers and recapitulated the pathogenic hallmarks of these muscular dystrophies. The authors investigate whether the 3D nature of the engineered skeletal muscle tissues would facilitate the detection of pathological hallmarks that are less evident in standard 2D cultures. To examine this, they generated artificial muscles from patient-derived hiPSC with muscular dystrophies caused by mutations in the LMNA gene. These diseases are also called laminopathies, and abnormalities in nuclear morphology are a key histological feature. LMNA mutant hiPSCs from patients with skeletal muscle laminopathies were used to create engineered skeletal muscle tissues, referred to by their mutation (i.e. LMNA L35P or LMNA R249W). Remarkably, 3D nuclear reconstruction of the engineered muscles highlighted features that were less prominent in traditional monolayer cultures. All mutant LMNA 3D engineered muscles showed a significant proportion of cells with nuclear aberrations (Figure 6(b)). These results demonstrate that these bioengineered skeletal muscle tissues from patient-derived cells are great tools to study the pathogenic pathways of muscular diseases and assay potential drugs. Nevertheless, the use of these platforms to test potential treatments for muscular dystrophies has not been reported yet. In 2009, Vandenburgh and colleagues developed an automated drug screening platform using contractile muscle tissue engineered from dystrophic myoblasts. 90 Primary myoblasts from the DMD mouse model (mdx mice) were encapsulated in a collagen-Matrigel® matrix and cast around two PDMS micro-pillars to engineer miniature bioartificial muscle (mBAMs). 46 These dystrophic mBAMs were electrically stimulated, and the force generation was measured. Then 31 drugs with potential anti-DMD effects were screened by measuring changes in force generation. Eleven drugs increased the dystrophic mBAMs tetanic force, similar to the response of DMD patients to many identical compounds. These results demonstrate the potential of this platform as a preclinical model. However, the use of mouse-derived cells is an important limitation of this approach since the heterogeneity of muscular dystrophy patients is not considered. Therefore, the integration of engineered skeletal muscle tissue from patient-derived cells in these automated drug screening platforms will bring powerful preclinical tools for these diseases.

Modeling muscular dystrophies using tissue engineering. (a) 3D artificial skeletal muscle constructs derived from healthy and dystrophic hPSCs. Immunofluorescence for myosin heavy chain (MyHC) on muscle constructs derived from hESCs and dystrophic hiPSCs (DMD, LGMD2D, and skeletal muscle LMNA) differentiated in 3D for 10 days. Nuclei are counterstained with Hoechst. Arrowheads: multinucleated myotubes. Scale bars: top 250 μm, bottom 25 μm. (b) Confocal (z stacks merge) immunofluorescence for DESMIN (myotubes), LAMIN A/C, and EMERIN (nuclear lamina) on hiPSC-derived (healthy and LMNA mutant) artificial muscles. Hoechst: nuclei. Scale bars: 15 μm. Adapted from Maffioletti et al. 92 .
Conclusions and perspectives
The latest advances in skeletal muscle tissue engineering have demonstrated the relevance of geometrical cues for the fabrication of in vitro muscle models. Aligned structures guide myotubes fusion and enhance the maturation of the myofibers.37,39 –42 Structured scaffolds can be obtained by bioprinting or molding biomaterials. Of note, the biomaterials for muscle engineering must provide mechanical support and allow nutrient diffusion through the scaffolds. Several studies conclude that electrical stimulation and mechanical tension favor the maturation of the tissues.35,61,79 Current studies to develop human functional bioartificial muscle use fibrin-based hydrogels. Fibrin composite matrices have tunable mechanical properties, can be remodeled by cells, and allow long-term cultures. 78 The biomaterials developed to date successfully provide geometrical and mechanical cues for engineered skeletal muscle. In the native muscle, the ECM not only acts as a scaffold but is essential for cell signaling. Accordingly, the future biomaterials for skeletal muscle tissue engineering should combine physical and biochemical properties that better mimic the complexity of native ECM. Extensive works have developed new fabrication techniques and biomaterials compositions to obtain functional skeletal muscle tissues. Nevertheless, high maturation levels of myofibers have not yet been reached. Thus, the bioartificial tissues have not accurately modeled functional adult muscles. Several approaches have recently attempted to improve the maturation of the in vitro muscles, for example, implementing electrical stimulation training protocols and co-cultures.79,80,83 Furthermore, incorporating endothelial cells to vascularize the tissues or innervation of the tissue with motoneurons are emerging strategies to obtain more complex skeletal muscle models. Innervation of muscle tissues with motoneurons is key for tissue maturation. Unfortunately, the protocols to obtain and co-culture motoneurons are complicated and not very well established. Hence, intensive research on co-culture techniques is one of the main future challenges. On the other hand, angiogenesis is still an important issue in tissue engineering. To date, full vascularization of engineered skeletal muscle tissue has not been achieved. Similar to motoneurons, it is essential to optimize protocols for co-culture. Besides, to mimic vascularized tissues, a specialized research field is focused on generating in vitro angiogenesis. The future perspective will be the confluence of both research areas to recreate the extensive network of blood capillaries in skeletal muscle.
Drug screening platforms exploiting new bioengineered skeletal muscle tissue models are promising tools to find treatments for muscular dystrophies. Interestingly, the integration of patient-derived cells to fabricate these bioartificial muscles could fill the gap in preclinical studies accelerating drug development. Moreover, patient-derived bioartificial muscles would allow testing personalized treatments in vitro, which are crucial due to the intrinsic heterogeneity of muscular dystrophy pathologies. Going a step further, these patient-derived bioartificial skeletal muscles can be integrated into microfluidic devices (organ-on-a-chip) with biosensors.56,93,94 These microfluidic chips permit the precise control of drug administration. In addition, muscle metabolism and disease-specific markers could be analyzed in real-time. Classically, the outcomes of these drug experiments are measured at time series or end-points. The challenge in this technological field is to integrate new sensing platforms that obtain data during the assays in a non-destructive manner. Altogether, the studies reviewed in this article show that tissue engineering technologies to develop these personalized drug screening platforms have a bright future perspective. Therefore, new efforts must point toward integrating patient-derived cells, biofabrication techniques, stimulation systems, and biosensors in personalized organ-on-a-chip preclinical platforms for muscular dystrophies.
Footnotes
Author contributions
JMF-C, XF-G, FV-M, and JR-A conceived this review. JMF-C coordinated the literature search and review design. JMF-C, XF-G, FV-M performed the literature search and wrote the paper. JMF-C generated the figures. JMF-C, XF-G, FV-M revised the manuscript and participated in the discussion with the input of JR-A.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge financial support from the European Research Council program under grants ERC-StG-DAMOC (714317), the Spanish Ministry of Economy and Competitiveness, through the “Severo Ochoa” Program for Centres of Excellence in R&D (SEV-2016-2019) and “Retos de investigación: proyectos I+D+i” (TEC2017-83716-C2-2-R), the CERCA Programme/Generalitat de Catalunya (2014-SGR-1460) and Fundación Bancaria “la Caixa”-Obra Social “la Caixa” (project IBEC-La Caixa Healthy Ageing) to JR-A. JMF-C was awarded with a postdoctoral fellowship (APOSTD/2017/088) from the Generalitat Valenciana, and XF-G was supported by a predoctoral fellowship (BES-2016-076681) from the Ministerio de Economia y Competitividad.
