Abstract
Three-dimensional engineered muscle tissues (EMTs) are transformative tools for modeling skeletal muscle physiology and pathology in vitro. Here, we perform a comprehensive comparison of EMTs derived from primary human myoblasts (hP-Myo) and hiPS-derived myoblasts (hiPS-Myo) to evaluate their structural, functional, and transcriptional characteristics. Contractile performance was quantified using a magnetic force-sensing platform, revealing that hP-Myo EMTs generate ~2 fold higher twitch forces and enhanced tetanic responses compared to hiPS-Myo EMTs. Tissue architecture and maturation were assessed and demonstrated significant larger myofiber diameters in hP-Myo EMTs. Transcriptomic profiling highlighted that hP-Myo EMTs maintain a mature skeletal muscle-like signature, marked by enriched pathways linked to sarcomere organization and fast-/slow-twitch fiber specification. To model metabolic dysfunction, hiPS-Myo EMTs were subjected to lipid overload, recapitulating hallmarks of intracellular lipid (IMCL) accumulation, including impaired contractility, blunted force-frequency responses, and dysregulated lipid metabolism genes.
Introduction
Skeletal muscle is the largest organ in the human body accounting for approximately 40% of the body’s total mass. Its unique structure and contractile function are crucial for movement, metabolism, and whole-body homeostasis. 1 Skeletal muscle research and drug development has traditionally relied on two-dimensional (2D) myotube cultures or poorly translatable animal models. Traditional 2D cell culture has provided fundamental insights into myogenic differentiation, muscle sarcomeric protein expression, and essential signaling pathways.2 –4 However, despite these productive uses 2D cell culture models are unable to produce contractile force, which is one of the most important functions of skeletal muscle. Additionally, 2D myotube models do not replicate the highly organized three-dimensional (3D) architecture, extracellular matrix (ECM) composition, and complex cell-cell interactions that are present in native muscle tissue.5,6
Over the past two decades, skeletal muscle tissue engineering has become a well-established approach to study contractile functions of skeletal muscle in vitro. 3D engineered muscle tissues (EMTs) derived from mouse or human myoblasts develop densely packed, aligned multinucleated myofibers that assemble functional sarcomeres, the basic units for synchronized contraction and force production. These structures produce quantifiable twitch and tetanic forces in vitro, mimicking key physiological properties of native muscle.7 –16 Previous studies show that incorporating controlled mechanical loading or electrical stimulation into EMTs can recapitulate the native biomechanical environment and improve muscle fiber size and force generation.17,18 Additionally, co-culturing with fibroblasts aids in the enhancement of ECM components and enhance muscle fiber viability. 19
Past human EMT studies have utilized myogenic cells, either derived from human induced pluripotent stem cells (hiPSCs) or human primary myoblasts and incorporated them into the 3D systems.14 –16,20 The benefit of hiPSCs is in their expansion capacity and genetic customizability enabling the production of large populations of myogenic precursors with a stable genetic background. Several previous studies demonstrated that hiPSC-derived myoblasts can be generated through directed differentiation or transcription factor-mediated programing via PAX7 or MYOD1 overexpression.13 –16 Although direct programing protocols reportedly yield purer, large-scale populations of myogenic cells, the resulting EMTs often show limited responsiveness to electrical stimulation and exhibit relatively low force production when comparing with primary myotube EMTs.15,21 Additionally, most studies have compared hiPSC-derived EMTs (hiPS-Myo) and human primary EMTs (hP-Myo) using different protocols or platforms, leaving uncertainties about intrinsic differences.12,13,21,22 One prior study assessed contractility in both hiPS-Myo and hP-Myo EMTs at a single timepoint (day 7). 16 Our work advances this paradigm by culturing hiPS-Myo and hP-Myo from multiple donors side by side in the same 3D platform with identical hydrogel composition, media, and force measurement conditions. While existing literature lacks longitudinal comparisons of hiPS-Myo and hP-Myo EMTs, our unified platform resolves this gap by measuring contractile force and bulk RNA sequencing profiles at day 7, 10, 14, and 21, enabling comprehensive evaluation of the 3D EMTs maturation and a direct comparison between hiPS-Myo and hP-Myo EMTs. By coupling four comprehensive functional assays, including twitch force, tetanic force measurement, force–frequency response and fatigue resistance, with global transcriptomic profiling, we characterized structure and functions of the EMTs and compared the contractile performance and RNA expression profiles of hiPS-Myo and hP-Myo from multiple donors at different culturing timepoints.
Studies using human primary myoblasts isolated directly from donor tissues, have demonstrated robust myotube formation, retaining relevant phenotypic features of the respective human donors, reflecting an advantage for modeling genetic myopathies and advanced personalized regenerative therapies. For example, EMTs generated from primary myoblasts of Pompe disease patients preserve the metabolic deficiencies of the donors, enabling disease recapitulation and drug testing. 23 However, when dealing with multifactorial, polygenic diseases like obesity-related metabolic dysfunction and age-related sarcopenia, the primary myoblasts isolated from biopsies face critical limitations. While these cells retain some donor-specific traits, such as genetic background or baseline metabolic profiles, their short lifespan in culture and lack of the native microenvironment underlying in vivo pathological conditions limit their capacity to accurately recapitulate the disease in vitro.24 –26 A hallmark of both obesity and sarcopenia in skeletal muscle is the abnormal accumulation of fat called myosteatosis.27,28 This ectopic fat is classified as intramyocellular lipids (IMCL) within muscle fibers and intramuscular adipose tissue (IMAT) between muscle bundles, both of which correlate strongly with insulin resistance, mitochondrial dysfunction, and reduced muscle mass and strength.28 –30 Here, we employed EMTs derived from hiPS-Myo to establish IMCL fat infiltration environment and examine how lipid accumulation influences both contractile function and metabolic activity.
In addition to IMCL modeling, in this study, we demonstrate that hP-Myo EMTs exhibit significantly greater twitch force, tetanus force, and fatigue resistance compared to hiPS-Myo EMTs, even after normalization to myofiber diameter. hiPS-Myo EMTs displayed a less mature phenotype, characterized by elevated expression of embryonic sarcomeric protein isoforms (e.g. MYH8 and MYH3) and markedly reduced expression of mature fast- or slow-twitch skeletal muscle markers (e.g. MYH1, MYH2, MYH7, TNNT1, TNNT3, TNNI1, TNNI2, MYL1 and MYL2) at the RNA level. Transcriptomic profiling further revealed that hP-Myo EMTs are enriched in pathways critical for myogenesis, contractile fiber assembly, and sarcomere organization (e.g. I-band and sarcoplasm components), underscoring their functional and structural maturity under identical culture conditions.
Materials and methods
Cell culture
iPS-derived muscle myoblasts were purchased from Curi Bio (MantaReady).
Primary human myoblasts isolated from adult donor muscle biopsies were purchased from Lonza (CC-2580), cultured and expanded according to vendor’s recommended protocol. The percentage of myoblasts in these cultures was verified by the myoblast specific marker CD56 (Novus Biologicals, AF2408) and fibroblast contamination was determined by studying cells positive for the fibroblast marker vimentin (Thermo Fisher, MA5-16409) using flow cytometry, and the myoblast population is 81.1% (Figure S1). P2 primary myoblasts were seeded on Matrigel (Corning, 356231) coated flasks (1:100) and cultured in Lonza’s skeletal muscle growth medium (SKGM-2, CC-3245). Expanded cells were cryopreserved in CS10 (Stem cell technologies, 100-1061). Primary myoblasts used in this manuscript were expanded until passage 5.
Primary normal human dermal fibroblasts that were used in EMTs were obtained from Lonza (CC-2511). They were cultured and expanded according to the vendor’s recommendations. Fibroblasts in this manuscript were incorporated into EMTs at passage 4.
2D differentiation of iPSC-derived myoblasts and primary human myoblasts
Cells were seeded at the density of 10,000/cm2 on collagen-I coated flasks/plates in Lonza skeletal muscle growth media (CC-3245). When the cells reached 70–80% confluence, media was exchanged to 2D differentiation medium (Curi bio, SKM-MED-IPS-D) and replaced every other day for day 7.
3D EMT generation
EMTs were generated using the Mantarray casting platform (Curi bio, Magnetometric Analyzer for engineered Tissue ARRAY) following the manufacturer’s guidelines. The diagram of the Mantarray plate and EMTs casting procedure were described previously. 14 First, expanded myoblasts were enzymatically detached following Lonza’s recommended subculture kit (CC-5034), collected from culture surfaces, and pelleted by centrifugation at 300g for 5 min. The workflow of EMT casting is shown in Figure S2. Briefly, approximately 675,000 myoblasts and 75,000 dermal fibroblasts were resuspended in 107 µL of high glucose DMEM (ThermoFisher), along with 30 µL of Matrigel, 10 µl of 50 mg/mL fibrinogen (Sigma-Aldrich), and 3 µL of 100 U/mL thrombin (Sigma-Aldrich), producing a total seeding volume of 150 µL at 750,000 cells per construct. The cell suspension was dispensed into the Mantarray 12X stiffness casting system and incubated at 37°C with 5% CO2 for 80 min to facilitate hydrogel polymerization. Addition of 1 mL prewarmed DMEM was added to each well to help release the hydrogel from the casting surface after incubation. The two-post array lid supporting the newly formed hydrogels was then transferred to a new 24-well plate containing Lonza’s skeletal muscle growth medium supplemented with 2 g/L aminocaproic acid (Sigma-Aldrich) for hiPS-Myo constructs or 5 g/L aminocaproic acid for hP-Myo constructs to prevent fibrinolysis.
Following overnight seeding, EMTs were switched to a low-serum (2%) medium (Curi Bio, SKM-MED-IPS-D) and differentiated for 10 days with medium replacements every 2–3 days. After differentiation, EMTs were transfer to a maintenance medium (Curi Bio, SKM-MED-IPS-M) for long term culture.
EMT contractility measurement
The contraction and force generation of the EMTs were stimulated using a commercially available stimulation lid (CuriBio, MA-STM-2) compatible with Mantarray system. EMTs were subjected to electrical stimulation at 10 V for 5 ms at 1 Hz for twitch contraction measurements and 50 Hz for tetanic response. Force-frequency protocol was used to assess the increasing electric frequencies from 1 to 50 Hz. Fatiguability protocol was used to assess the fatigue resistance of the EMTs by applying 20 repeated 50 Hz pacing within 3 min. Force production was calculated through the degree of post deflection upon electric excitation as previously described, 14 and the post-deflections were recorded using Mantarray’s Pulse Analysis software. This technique is non-invasive and individual EMTs could be tracked independently for functional changes over time. In this study, contractile functions were evaluated from day 7 through day 21 after tissue formation.
Treatment of 3D EMTs
For pharmacological treatment, EMTs were incubated with either 10 mM caffeine (Thermo Fisher, A10431) or 10 μM verapamil (Sigma-Aldrich, V4629-1G) in maturation medium (Curi Bio, SKM-MED-IPS-M) for 30 min at 37°C with 5% CO2 prior to functional assessment. After recording, EMTs were washed with PBS three times and fixed for other downstream experiments.
For IMCL induction, 3D EMTs were treated with lipid mixture 1 (Sigma-Aldrich, L0288) with 10% volume of culture medium for 72 h at 37°C with 5% CO2. After treatment, EMTs were washed three times with PBS and fresh maintenance media was added prior to functional assessment.
Immunohistochemistry and immunocytochemistry
EMTs were fixed in 4% paraformaldehyde (Thermo Scientific, J19943K2) overnight at 4°C and dehydrated through a series of ethanol washes from 70% EtOH to 100% EtOH at room temperature. Following dehydration, EMTs were immersed in 100% xylene (Sigma-Aldrich, 534056) for 30 min at room temperature and repeated three times. The EMTs were then transferred to a 1:1 mixture of xylene and paraffin wax for 30 min at 60°C. Next, they were placed in three separate baths of 100% paraffin wax, 30 min each at 60°C before being embedded in fresh wax.
After embedding, 7 µm sections were cut using a Leica RM 2135 rotary microtome and mounted onto microscope slides (Fisher Scientific) and air dried overnight at 37°C, dewaxed, and rehydrated in graded ethanol to water. Slides were stained with Hematoxylin (Thermo Fisher, 7211) and Eosin-Y (Thermo Fisher, 7111). Slides were imaged on Polaris (Akoya, Marlborough, MA) using whole slide brightfield scanning at 20×.
For immunohistochemical (IHC), fluorescent multiplexing was completed on the Leica Bond Rx automated Stainer (Leica Biosystems, Wetzaler, Germany). Tissues were baked, dewaxed and antigen retrieved using ph6. IHC was performed using Opal-TSA (Akoya, Marlborough, MA) staining with markers anti-MHC (Invitrogen, 14-6503-82) paired with Opal 520 (OP-001001; Akoya), anti-α-actinin (Invitrogen, MA1-22863), and anti-vimentin (Biocare, CM048A) both paired with Opal 690 (OP-001006, Akoya). Between each sequential antibody application, slides were incubated with Anti Ms & Rb HRP (ARH100, Akoya) secondary antibody, followed by Opal fluorophore labeling, stripped with heat induced epitope retrieval, leaving behind the Opal fluorescence signal. Slides were imaged on Phenoimager HT (Akoya, Marlborough, MA) using whole slide scanning at 20×. The antibody specific staining areas were quantified using HALO AI V3.6.4134 software area quantification fl v2.3.4 module (Indica Labs, Albuquerque, NM).
Whole mount immunofluorescence staining
While still suspended, EMTs were fixed with 4% paraformaldehyde (PFA, Sigma-Aldrich) for 30 min at room temperature, while lipid treated were fixed in 2% PFA for 15 min at room temperature. For immunofluorescence staining, EMTs were permeabilized in blocking buffer that contains 0.2% Triton X-100 (Sigma-Aldrich) +1% BSA in PBS for 1 h at room temperature on rocker. EMTs are detached from the posts and primary antibodies, anti-Myosin heavy chain (MF20, Invitrogen, 14-6503-82) or anti-sarcomeric alpha actinin (EA53, Invitrogen, MA1-22863) was diluted at 1:500 in blocking buffer and incubate with EMTs overnight at 4°C on rocker. The following day, EMTs were washed with PBS four times for 40 min total. Secondary antibody Donkey Anti-Mouse IgG [H + L] Alexa Fluor 647 (Invitrogen, A-31571) was diluted 1:1000 in blocking buffer and incubated for 2 h at room temperature in the dark on rocker. After washing four times with PBS for a total of 40 min, Alexa Fluor 594 Phalloidin (Invitrogen, A12381) or BODIPY 493/503 neutral lipid droplet dye (Invitrogen, D3922) diluted at 1:10,000 was added to EMTs with Hoechst nuclear stain (Thermo Fisher, 62249) diluted at 1:2000 for 30 min at room temperature.
For confocal microscopy, stained EMTs were placed in 24 well plate with glass bottom (Greiner, 662892) and imaged on Zeiss LSM 9880 Confocal with AiryScan (Carl Zeiss AG, Oberkochen, Germany).
AI-based analysis of muscle diameter measurement
All images were first rotated to align the myotubes perpendicular to the horizontal axis, ensuring a uniform orientation across the dataset. This was followed by the application of the Segment Anything Model 2 (SAM 2), 31 which was utilized to segment all dead cells present in the images. As illustrated in Figure S3, non-myotubes, which are cell aggregates, are visually distinguishable from live cells (elongated myotubes) based on their morphology. Subsequently, straight lines in the direction perpendicular to myotubes were drawn and spaced equidistantly at 202 micron across the image. Along these lines, peaks are detected, and the Full Width at Half Maximum (FWHM) are measured. During this process, any widths that overlapped with the segmented dead cells were discarded to ensure accuracy in our measurements.
The categorization of lipids is conducted based on their presence in either live cells or otherwise, utilizing the previously mentioned dead cell mask. Subsequently, the mean value of the lipid content is calculated specifically for the live cells.
Total RNA isolation
EMTs were washed with PBS twice and detached from posts. Monarch Total RNA Miniprep Kit (New England Biolabs, T2010S) was used according to manufacturer’s protocol. Tissue with 300 µL RNA protect solution was homogenized with 5 mm stainless steel beads in the TissueLyzer II (Qiagen) system at 30 Hz for 3 min. Proteinase K (New England Biolabs, P8107S) solution was added, and samples were incubated at 55°C for 5 min before continuing through manufacturers RNA isolation protocol.
Bulk RNA-sequencing
RNA samples were sent to Azenta Life Sciences for RNAseq. The strand-specific RNA sequencing library was prepared by using NEBNext Ultra II Directional RNA Library Prep Kit for Illumina following manufacturer’s instructions (NEB, Ipswich, MA, USA). The sequencing was performed using an Illumina NovaSeq instrument to generate approximately 30 million, 150 base pair, paired end reads. Reads were mapped against the Homo sapiens (human) hg38 /GRCh38 using the STAR aligner (v2.5.2b), 32 allowing for soft clipping of adapter sequences. Transcript annotation files from Ensembl version 86 were used for quantification, corresponding to GENCODE M11. Samples were quantified with the above annotations, using RSEM (v1.3.0) 33 and featureCount (v1.5.1). 34 Quality controls were implemented using FastQC (v0.11.5; Andrews S. 2010: Available online at: www.bioinformatics.babraham.ac.uk/projects/fastqc/.)
Gene sets and pathways enrichment analysis
To find differentially expressed genes in comparing different model systems and primary muscle, differential expression analysis was performed using Limma on voom-transformed data. 35 Differentially expressed genes were determined as having a fold change of ±2 and an adj. p-value <0.05. Differential pathways were identified using gene sets from MSigDB with the GAGE algorithm.36,37
Nanostring
Total RNA was isolated from EMTs using Monarch Total RNA Miniprep Kit (New England Biolabs, T2010S) as previously described. Groups from these experiments included fully differentiated and untreated 3D EMTs and lipid mixture 1 treated EMTs. The RNA from these experiments was measured by NanoDrop 8000 Spectrophotometer (Thermo Scientific) and normalized to 10 ng/µL with Molecular Biology Grade Water (Corning, 46-000-CM). Samples were processed on NanoString nCounter® Pro Analysis System in nCounter® XT Metabolic Pathways Panel according to manufacturer’s protocol. Gene expression was analyzed using the nCounter® advanced analysis software.
Data analysis
Data analysis was conducted using GraphPad Prism (La Jolla, CA). Pairwise comparisons were made with Student’s t-tests. For three-group comparisons, one-way ANOVA was used with Dunnett’s post-hoc test to assess treatment groups against the untreated group. Longitudinal datasets were examined via two-way ANOVA with repeated measures, and pairwise comparisons were evaluated within each timepoint. All results are expressed as mean ± SD, with individual data points illustrated in bar graphs. Statistical significance was defined as ns: no significance, *p < 0.05, ** p < 0.01, *** p < 0.001, and **** p < 0.0001 for all statistical tests.
Results
Functional characterization of EMTs on magnetic sensing system
A previous study using iPS-derived myoblasts in the magnetic sensing force measurement system (Mantarray) demonstrated contractility measurement and sustained contractile force from day 7 to day 14. 14 To ensure optimal tissue maturation, we used human primary myoblasts at passage 5 (hP-Myo) to construct EMTs on the same magnetic sensing force measurement device. They were first differentiated for 10 days and then kept in maintenance medium for an additional 4 days to day 14 (Figure 1(a)).
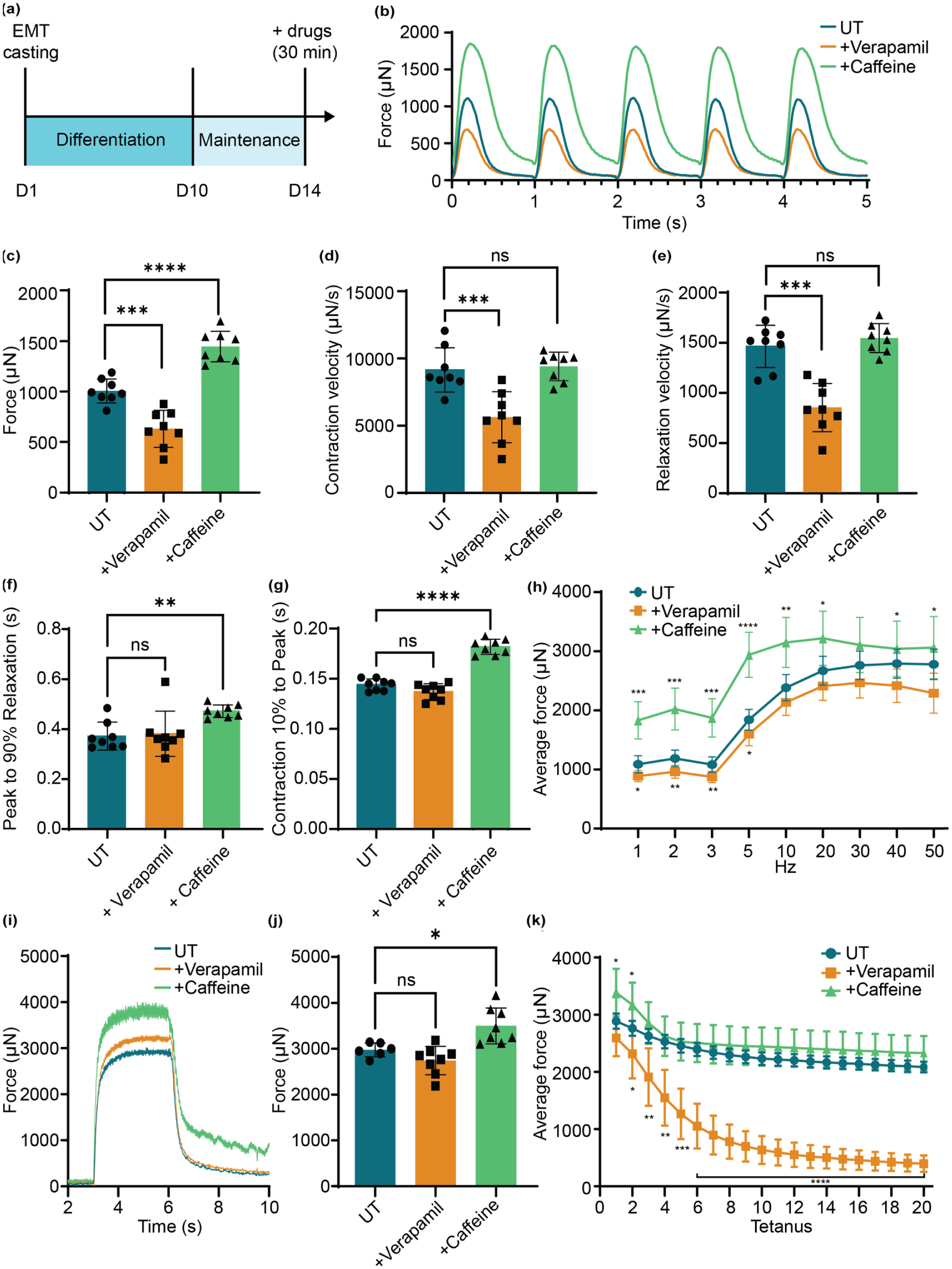
EMT contractility measurement and pharmacological modulation of contractility. (a) Illustration of EMT culture procedure. (b) Representative traces of five twitch contractions with and without drug treatment. Various EMT function parameters were quantified, including (c) average force comparison, (d) contraction velocity, (e) relaxation velocity, (f) active relaxation rate from peak contraction to 90% relaxation, and (g) active contraction rate from 10% contraction time to the peak (n = 8 per group). (h) Comparison of force-frequency response (1–50 Hz) of UT and drug treated EMTs. (n = 8 per group) (i) Representative tetanus contraction and (j) average force comparison (n = 8 per group). (k) Fatigue resistance stimulated by 20 tetanic contractions in UT and drug treated EMTs (n = 8 per group). ****p < 0.0001, ***p < 0.001, **p < 0.001, *p < 0.05 and not significant (ns) in drug treated groups versus UT. Data was reported as mean ± SD.
On day 14, EMTs were treated with the positive inotrope caffeine (10 mM) 38 or the calcium channel blocker verapamil (10 µM) 39 for 30 min before contractility measurement. When stimulated with 1 Hz electrical pacing, EMTs treated with caffeine exhibited significantly higher peak force and passive tension at baseline while verapamil treatment group showed declined force compared to the untreated (UT) control (Figure 1(b) and (c)). After subtraction of baseline tension, the average active force production for UT, caffeine treated, and verapamil treatment was 1005.78 ± 110.80, 1444.57 ± 142.37, and 630.87 ± 171.19 μN, respectively. In addition, verapamil treatment showed significantly decreased velocities of contraction and relaxation, but caffeine did not affect these velocities (Figure 1(d) and (e)). Instead, the contraction and relaxation time for the caffeine group were increased compared to our other conditions (Figure 1(f) and (g)).
Following twitch force measurements, EMTs were subjected to escalating stimulation frequencies from 1 to 50 Hz, which evoked tetanic contractions. Force outputs increased incrementally with frequency in all experimental groups. Among them, the caffeine group retain the highest active force in each frequency stimulation (Figure 1(h)). Maximum force during tetanic contractions (stimulated by single 50 Hz pacing) was increased by caffeine treatment but was unaffected by verapamil treatment (Figure 1(i) and (j)). When subjected to the fatiguability measurement (repeated tetanic contractions at 50 Hz, 20 cycles), the verapamil treated group showed much lower fatigue resistance and a progressive decline in maximal force output during repeated stimulation (Figure 1(k)), reflecting interrupted excitation-contraction coupling by verapamil administration.
Tissue architecture of the hP-Myo EMTs
EMTs were constructed by 90% human primary myoblasts with 10% human dermal fibroblasts to support myotube growth. To examine the internal architecture of the EMTs, EMTs were embedded in formalin-fixed paraffin (FFPE) and longitudinal sections were stained with hematoxylin and eosin (H&E) to visualize their internal structure (Figure 2(a) and (b)). Multinucleated myotubes are present within EMTs. Using the longitudinal and transverse sections, IHC was performed with a validated myosin heavy chain (MHC) antibody (Figure 2(c)–(f)), showing MHC expression in myofibers of the 3D structure. Dermal fibroblasts were detected by vimentin staining and the average area of vimentin positive / MHC positive stain is 4.61% (Figure S4).

Structure of hP-Myo EMT. (a and b) Representative H&E staining of a longitudinal section from FFPE tissue blocks. (c–f) Representative immunostaining of MHC on longitudinal and cross-sections of EMT. Representative whole-mount confocal images of (g) nuclei, (h) sarcomeric alpha-actinin (SAA), and (i) merged.
Due to dehydration during the FFPE process, myofiber organization was distorted. We therefore conducted whole-mount immunofluorescence imaging by confocal microscopy. EMTs were fixed with 4% PFA while attached to their posts and stained with MHC or sarcomeric α-actinin (SAA) antibodies. These whole-mount images revealed well-aligned, multinucleated, and striated fibers spanning the constructs (Figure 2(g)–(i)).
HP-Myo-based EMTs demonstrate enhanced contractile functions compared to hiPS-Myo-based EMTs
Human iPS-Myo-derived EMTs have demonstrated utility in modeling muscle biology and diseases, however, they persistently exhibit reduced contractile capacity compared to primary cell-derived systems.12,13,16 To directly compare functional performance, we generated EMTs using iPS-derived myoblasts and primary human myoblasts to assess their contractile force generations. Both hP-Myo and hiPS-Myo-based EMTs were cast using the same method, materials and the same number of human dermal fibroblasts, then differentiated for 10 days, followed by an additional 4-day maintenance period. Their contractile performance was measured and compared on day 14. hP-Myo EMTs have significantly greater muscle function and strength compared to hiPS-Myo EMTs, measured by twitch and tetanus stimulation respectively (Figure 3(a)–(d)). Notably, hP-Myo EMTs illustrated quicker wave summation in force-frequency response (10 Hz) and were more fatigue resistant in the tetanic stimulations (Figure 3(e) and (f)).

Muscle contractility comparison between hiPS-Myo and hP-Myo EMTs. Twitch contraction measurements for (a) average force, (b) contraction velocity, and (c) relaxation velocity was compared between hiPS-MYO and hP-Myo EMTs, and (d) averaged tetanic contraction for tetanic contraction (n = 4–5 per group). (e) Fatiguability was measured by 20 repeated tetanic stimulations, indicating declining maximal forces, and contraction amplitude (f) were normalized from the first tetanic contraction in the fatiguability experiment (n = 4–5 per group). (g) Quantified force-frequency response (n = 4–5 per group). (h) Myotube diameter distribution and average myotube diameters (i) from hiPS-Myo and hP-Myo (n = 4–5 EMT per group, ~300 myotubes measured for each EMT). Twitch force (j) was further normalized to their myotube diameters, as well as normalized contraction velocity (k), normalized relaxation velocity (l), and tetanic force (m). ****p < 0.0001, ***p < 0.001, **p < 0.001, *p < 0.05 and not significant (ns) in drug treated groups versus UT. Data was reported as mean ± SD.
To quantify contractile differences more precisely, we utilized AI-based analysis to identify and measure the diameter of myofibers inside the EMTs from the confocal images (Figure S2). Approximately 300 myotubes diameter were measured for each EMT, and the distribution of the diameter sizes shows a right-shift for hP-Myo EMTs (Figure 3(h)). The averaged diameters for hP-Myo and hiPS-Myo were 15.95 ± 0.39 and 13.56 ± 0.42 μm (n = 4) and hP-Myo diameter was significantly larger than hiPS-Myo EMT (Figure 3(h) and (i)). The cross-sectional areas (CSA) from both hiPS-Myo and hP-Myo EMTs were quantified from total MHC-stained regions, total nuclei count, and the percentage of nuclei count that overlayed with MHC stain to total nuclei count to account for potential differences in cell density between the two construct types. hiPS-Myo MHC + CSA and total nuclei count were not significantly smaller or fewer than hP-Myo, but the percentage of nuclei that overlapped with MHC staining to total nuclei numbers was significantly less than hP-Myo (Figure S5). We next normalized their generated forces to the averaged diameter. After this normalization, EMTs that originated from primary human myoblasts retained stronger contractile performance than those generated from iPS-derived myoblasts (Figure 3(j)–(m)).
RNA profiles of hiPS-Myo and hP-Myo EMTs
To elucidate the molecular profiles of EMTs derived from distinct cellular origins, we conducted bulk RNA-sequencing combined with systematic bioinformatic analyses. It allowed us to map dynamic shifts in global gene expression during differentiation and maturation of 3D muscle constructs.
Principal Component Analysis (PCA) revealed clustering patterns among samples. In this analysis, 2D and 3D models clustered separately, and hiPS-Myo and hP-Myo-derived samples clustered separately (Figure 4(a)). Undifferentiated 2D samples were collected prior to seeding for differentiation and undifferentiated 3D samples were constructed in 3D hydrogels first and then collected. Both 2D and 3D undifferentiation samples fell on the far left of the PC1 axis, while differentiated constructs clustered adjacent to human skeletal muscle reference samples. Notably, hiPS-Myo samples (2D and 3D) were shifted along the PC2 axis, distinctly separated from hP-Myo counterparts, highlighting transcriptional divergence between cell sources. Temporal analysis of genes associated with Gene Ontology Biological Process (GOBP): Regulation of skeletal muscle tissue development (GO:0048641) revealed divergent expression profiles between undifferentiated cultures (2D and 3D) and human muscle samples (Figure 4(b)). The stability of the 3D culture lengthens the differentiation phase (D7–D21), during this period, the gene signatures increasingly align with those observed in human muscle tissue. This demonstrates a more advanced maturation of our 3D constructs compared to 2D models, which experience a plateau in their culture stability at D7, preventing further differentiation.

Transcriptomic comparison of hiPS-Myo and hP-Myo in 2D and 3D cell models. (a) PCA plot based on the most abundant 700 genes from bulk RNA-sequencing. (b) Heatmap of genes from GO_Regulation of skeletal muscle development pathway in all groups. (c) GSEA of day 14 Hp-Myo versus day 14 hiPS-Myo (Padj < 0.05). Volcano plots highlighted the upregulated genes from the Hallmark Myogenesis pathway related to skeletal muscle master and stress regulators (d), structural/contractile machinery (e), and calcium handling mediators (f). (g) Heatmap of fiber type-specific genes from 3D cell models during culture at different timepoints and human skeletal muscle biopsies.
Consistent with our earlier functional data (Figure 3), hP-Myo EMT transcriptomes exhibited enriched pathways linked to myogenic maturation compared to hiPS-Myo EMTs. Gene Set Enrichment Analysis (GSEA) identified seven significantly altered biological processes (p < 0.05) in hP-Myo D14 EMTs, including Hallmark_Myogenesis (M5909) and contractile/sarcoplasmic pathways (Figures 4(c) and S7). Myogenesis-associated genes further outlined three functionally coherent subgroups: (1) master regulators and stress-response genes (Figure 4(d)), (2) structural/contractile machinery components (Figure 4(e)), and (3) calcium-handling mediators (Figure 4(f)).
Finally, fiber type-specific gene expression analysis revealed significant differences in mature fast/slow muscle isoform distributions between hiPS-Myo and hP-Myo EMTs. hiPS-Myo EMT predominantly expressed immature skeletal muscle markers, such as MYH3 and MYH8, in contrast to the more mature profile of hP-Myo EMTs (Figure 4(g)). These variations aligned well with their distinct contractile capabilities (Figure 3). Although numerous gene expression differences remained compared to human skeletal muscle biopsies, hP-Myo EMTs exhibited substantially higher levels of mature isoforms, including MYH2, MYH7, TNNT1, and TNNT2, indicating that hP-Myo constructs more closely approximate native human muscle gene expression, despite not fully replicating it. Importantly, the significantly lower expression of MYH1 in both hiPS-Myo and hP-Myo EMTs compared to human biopsies illustrates their incomplete maturation (Figure 4(g)), reflecting an intermediate rather than fully differentiated developmental state of engineered muscle tissues.
Exogenous FFA-induced IMCL phenotype attenuates the functionality of EMTs
Bulk RNA sequencing has provided a comprehensive transcriptional profile of EMTs. It also established a foundation for examining how such engineered tissues respond to metabolic challenges. We first compared the lipid related pathways from GSEA of hP-Myo and hiPS-Myo at D14, and there are not significant up- or down-regulated pathways (Fig. S8). Our goal was to induce IMCL, a pathological accumulation of lipid within skeletal muscle, within our EMTs to assess the resulting phenotypic changes related to force production and gene expression shifts associated with lipid accumulation. EMTs for lipid induction were produced from hiPS-Myo. The constructs were differentiated for 10 days and then supplemented with 10% total volume of lipid mixture 1 that contains seven types of saturated and non-saturated fatty acids (FA) for 72 h (Figure 5(a)). After treatment, contractile functions and structural alteration were evaluated to determine the impact of lipid exposure.
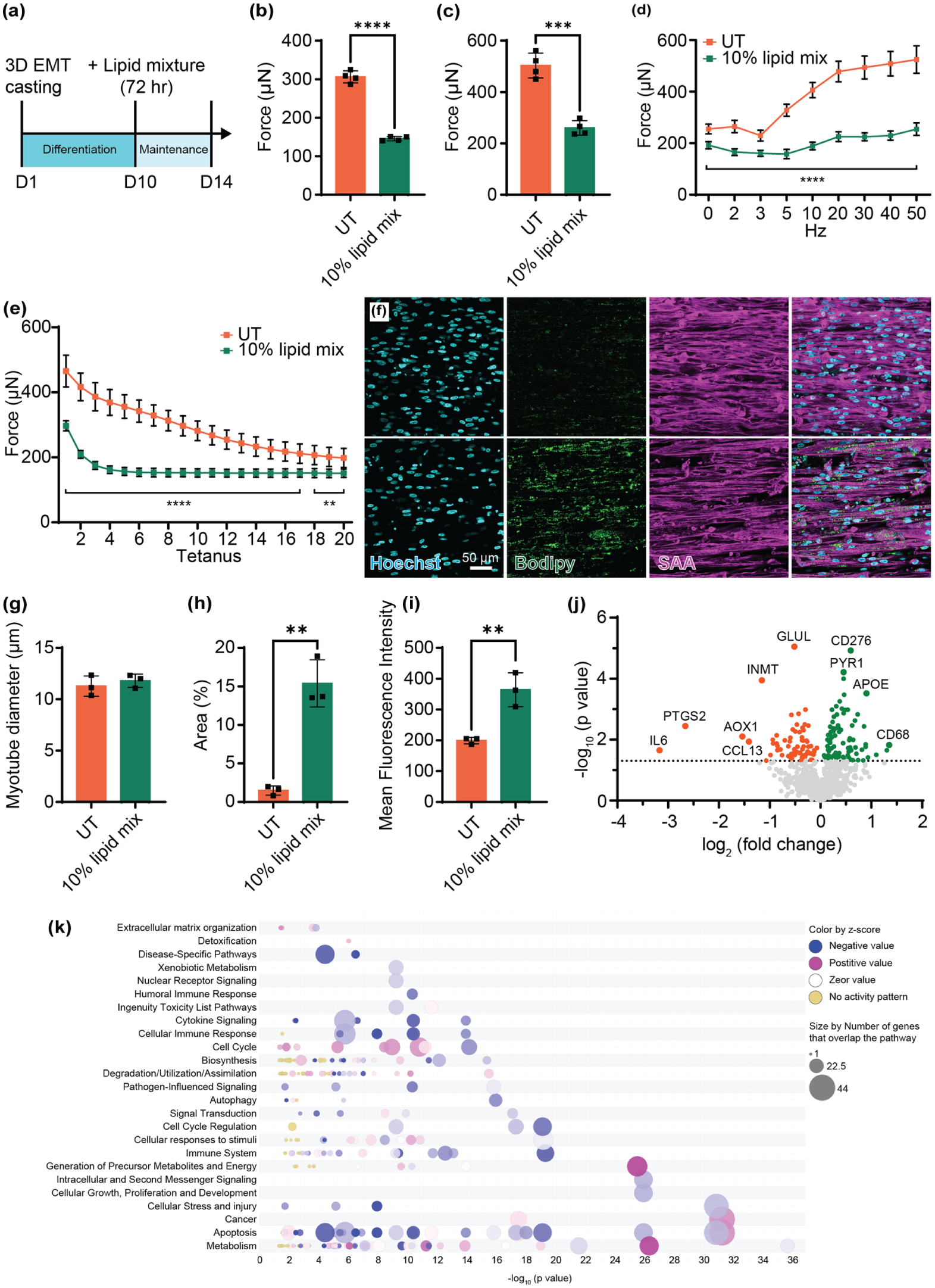
IMCL modeling in hiPS-Myo EMTs. (a) Timeline of EMT culture and IMCL induction. (b) Averaged twitch and (c) tetanus force was compared between UT and IMCL EMTs (n = 4 per group). (d) Force-frequency response and (e) fatigue resistance was measured and compared (n = 4 per group). (f) Representative whole-mount confocal images of lipid accumulation detected by BODIPY and muscle myofibers stained by SAA from UT (upper panel) and IMCL group (lower panel). Myotube diameter (g), lipid area of total myotubes (h), and mean fluorescence intensity of BODIPY (i) were compared (n = 3 per group). (j) Volcano plot of metabolic pathway related gene expression in IMCL EMT versus UT (p < 0.05). (k) Bubble chart of enriched pathways from the tested metabolic related genes. ****p < 0.0001, ***p < 0.001, **p < 0.001, *p < 0.05 and not significant (ns) in drug treated groups versus UT. Data was reported as mean ± SD.
Lipid-treated EMTs exhibited reduced twitch and tetanic force generation (Figure 5(b) and (c)). Their force-frequency response curves were significantly depressed across stimulation frequencies from the first stimulation at 1 Hz all the way to the final stimulation at 50 Hz, suggesting impaired metabolic capacity to sustain high-frequency contractile activity (Figure 5(d)). In addition, fatigability assessment showed a significant reduction in resistance to fatigue in EMTs exposed to lipids, implying that the accumulation of lipids negatively impacts both the strength and endurance of the muscle (Figure 5(e)).
To assess the structural changes in our EMTs, we used whole-mount confocal imaging to obtain detailed views of myofiber organization within the constructs. Analysis revealed an increase in lipid accumulation by measuring the lipid coverage area and lipid fluorescence intensity in myotubes (Figures 5(f)–(i) and S6). Despite decreased muscle function and increased lipid accumulation, myotube diameters were unaffected (Figure 5(g)).
In addition to imaging, RNA was extracted from the EMTs, and the expression of genes related to metabolic pathways was measured. Out of the 758 genes examined, our lipid-treated EMTs showed 68 down-regulated and 86 up-regulated genes compared to UT (Figure 5(j)). Canonical pathways were then identified and analyzed from the IPA (Ingenuity Pathway Analysis) libraries that were most relevant to measured metabolism genes from the metabolic panel. The top up-regulated canonical pathways included respiratory electron transport, oxidative phosphorylation, and complex 1 biogenesis, and the top down-regulated pathways included macroautophagy, signaling by VEGF, and mitochondrial biogenesis (Figure 5(k)).
Discussion
Our study investigated structural and functional differences of EMTs made from hP-Myo or hiPS-Myo in 3D. Our findings demonstrated that EMTs derived from primary myoblasts exhibit enhanced contractile function, gene signatures resembling human tissue and morphological features that point toward advanced muscle maturation. We also explored how the addition of lipids to 3D EMTs results in a metabolic dysfunction phenotype that mimics IMCL in skeletal muscle with impaired muscle functions in vitro and altered gene expression.
We first characterized the EMT contractile functions by measuring both twitch and tetanus contractions and the response to pharmacological treatments. Caffeine treatment significantly enhanced both twitch force and tetanus force (Figure 1(c) and (h)). However, unlike prior reports showing caffeine to improve muscle kinetics in vivo, the contraction and relaxation velocities remain unchanged in our model 40 (Figure 1(d) and (e)). This divergence may reflect microenvironmental or structural differences intrinsic to EMTs.
Caffeine enhances force production by binding ryanodine receptors (RyRs) on the sarcoplasmic reticulum (SR), triggering Ca²⁺ release into the cytosol. 41 Elevated intracellular Ca2+ unlocks actin-binding sites via troponin-tropomyosin in thin filaments and increases myofibrillar sensitivity to initiate force production. However, prolonged Ca²⁺ mobilization can impair SR Ca²⁺-ATPase (SERCA) activity, delaying Ca²⁺ reuptake and relaxation. 42 In a prior study examining the effect of caffeine dose on force and intracellular Ca2+, it was found that only a non-physiological dose (5 mM) could stimulate improvements in contraction of isolated mouse skeletal muscle fibers. 38 In our study we chose a 10 mM dose of caffeine as previously reported 43 and observed an increase in muscle contractility but slowed the rate of relaxation, indicating a decrease in the SR Ca2+ pump activity as shown previously. 42
Prior work in 3D EMTs have predominantly utilized either iPS-derived myoblasts or isolated primary myoblasts, and limited comparative data exists between hiPS-Myo and hP-Myo EMTs., In the current study, we cultured both EMT constructs in the same culture conditions and force measurement platform. We found that hP-Myo consistently produced higher twitch and tetanic forces even after accounting for myofiber diameter (Figure 3(h)–(l)). And the mean myotube diameters for hiPS-Myo and hP-Myo are 13.56 and 15.95 μm at day 14, respectively (Figure 3(i)). These values are similar to many earlier reports that Iuliano et al. found hiPSC-derived 3D-TESM fibers of ~17 μm diameter after 2 weeks culture, Rao et al. and Van der Wal both reported ~13 μm in diameter.13,16,44 In a different single-fiber microchannel system, Urciuolo et al. generated human myobundles ~10 mm in length and ~120 μm in the whole cross-sectional diameter, 45 illustrating how scaffold geometry drives construct size.
Our transcriptomic analysis revealed persistent elevation of embryonic myosin heavy chain isoforms (MYH3, MYH8) and markedly lower levels of adult MHC isoform genes (MYH1, MYH2, MYH7) in hiPS-Myo EMTs from day 7 to 21 of differentiation (Figure 4(g)). In contrast, hP-Myo constructs exhibited transcriptomic signatures closely aligned with native human skeletal muscle. This sustained fetal-like expression profile in hiPS-Myo tissues correlate with their reduced contractile force generation and impaired functional maturation. These findings align with previous results that hiPSC-derived muscle cells correspond to an earlier, fetal-like developmental stage compared to adult muscle cells. 46 Although the previous work by Rao et al. does provided foundational transcriptomic comparisons between hP-Myo and hiPS-derived skeletal muscle constructs, their study was limited using uncoupled datasets and lacked direct functional benchmarking between the two cell sources. Our study addresses this gap by integrating contractile force measurements and deep RNA sequencing within a unified experimental framework, enabling direct, time-resolved comparisons under identical conditions. This approach reveals not only the sustained fetal-like transcriptional profile of hiPS-Myo EMTs but also their reduced contractile performance and metabolic immaturity relative to hP-Myo EMTs. These insights extend prior observations and provide a more mechanistic understanding of the limitations and potential of hiPSC-derived muscle tissues for modeling disease and therapeutic development.
By comparison, van der Wal et al. reported similar embryonic (MYH3), neonatal (MYH8), and slow- twitch (MYH7) expression patterns in both cell types at day 7. 16 Notably, this early timepoint represents an immature state preceding full molecular and functional maturation observed in our extended timeline (day 21). Our data demonstrates that only hP-Myo EMTs progress to adult-like transcriptional programs, whereas hiPS-Myo tissues retain a developmentally arrested phenotype. Additionally, consistent with recent proteomic analyses by van der Wal et al., our bulk RNA-sequencing revealed significant enrichment of contractile fiber-associated genes in hP-Myo versus hiPS-Myo (Figure 4(c)). Van der Wal’s GO enrichment analysis identified “contractile fiber part” as the most upregulated category in primary EMTs during early differentiation (day 7), reflecting accelerated sarcomerogenesis. 16 Further analysis suggests that hP-Myo EMTs were enriched in pathways associated with oxidative metabolism and sarcomere organization, hallmarks of mature muscle function. In contrast, hiPS-Myo EMTs displayed a glycolysis-driven metabolic profile, consistent with their structural and functional immaturity. These findings suggest that metabolic and structural limitations of hiPS-Myo-derived EMTs.
In our study, the observed divergence in gene expression between our EMT models and native human tissue highlights the shortcomings of EMTs for phenocopying adult human skeletal muscle tissue. Muscle tissue in vivo comprises a complex milieu of cell types, including mature muscle fibers, satellite cells, skeletal muscle fibroblasts, endothelial cells, immune cells, and fibro-adipogenic progenitor cells, all of which interact in a highly coordinated manner to support muscle function and maturation. The absence of diversity in our models likely contribute to the observed limitation in achieving full structural and functional maturation, as evidence by the embryonic gene profile across our models. This limitation highlights the need for incorporating a broader range of relevant cell types to replicate the complex system of human muscle tissue and their dynamic properties more accurately. Another limitation is the absence of exercise-mimetic conditioning in our culture system. Chronic electrical pulse stimulation and/or cyclic mechanical loading are known to accelerate maturation of engineered muscle for enhancing myofiber size, sarcomeric organization, oxidative capacity, and specific force production. 47 Incorporating such stimulation protocols in future studies is likely to reduce the fetal-like transcriptomic signature observed here and bring EMT structure and function closer to native adult skeletal muscle.
IMCL accumulation in skeletal muscle is a hallmark of metabolic disorders like obesity and type 2 diabetes, driving insulin resistance and functional impairment.48,49 Traditionally, in vitro studies of “lipid-loaded” muscle have been conducted in 2D myotube cultures by treating cells with excess FFAs, particularly long-chain saturated fatty acids like palmitate. Numerous studies have shown that exposing myotubes to high palmitate for 16–72 h induce pathological changes, such as insulin signaling and blunted glucose uptake, increased oxidative stress, and activated muscle atrophy signaling. 50 To model metabolic dysfunction with quantitative functional results, we established an IMCL phenotype by adding exogenous lipid to hiPS-Myo EMTs. This allowed us to investigate whether ectopic fat infiltration induced by pathological concentrations of fatty acids might disrupt muscle function in ways that resemble clinically observed myosteatosis. 51 EMTs treated with lipid showed clear reductions in twitch and tetanic force, as well as flattened force-frequency profiles and diminished capacity to withstand repeated contractions (Figure 5(b)–(e)). Immunostaining confirmed accumulation of lipids within the myofibers (Figure 5(f)–(i)). Moreover, pathway analysis revealed upregulated respiratory electron transport and oxidative phosphorylation genes, suggesting FA oxidation Autophagy pathways were enhanced as well as CD68 indicating muscle injury caused by exposure to high concentrations of FAs 52 (Figure 5(j) and (k)).
These findings support the notion that excessive intramuscular fat can obstruct calcium handling, compromise mitochondrial homeostasis, and evoke inflammatory responses that all play a role in reduced contractile function. Our results share common ground with work by Turner et al., 53 who showed that physiological (200 μM) and pathophysiological (800 μM) concentrations of four types of fatty acids (oleic, palmitic, linoleic, and α-linolenic acids) induce lipid accumulation and impaired contractile performance in C2C12-based 3D engineered skeletal muscle, further highlighting the value of 3D-engineered systems in studying metabolic muscle disease. Our study extends it using human cell-derived EMTs treated with more diverse FFA mixture, including saturated, monosaturated, and polyunsaturated FAs, to mimic nutritional overload. Similarly, high-fat diet models in vivo show that increased IMCL content correlates with decreased specific force in oxidative muscles. 54
While our lipid overload treatment caused a significant decline in contractile function after 72 h, not all studies have reported functional deficits under similar lipid treatments. For instance, a recent study by Khodabukus et al. 55 exposed hiPSC-derived engineered muscles to a FFA combination of oleic, linoleic, and palmitic acid (100 μM each, 300 μM total) for 1 week and observed robust IMCL formation only in dysferlin-deficient (LGMD2B) EMT without any reduction in twitch or tetanic force in either healthy or LGMD2B EMTs. Different combination of FAs might play important roles for the different results. Our cocktail contained multiple saturated FAs (palmitate, stearate, myristate), and arachidonic acid in addition to oleate and linoleate, which may have exacerbated lipotoxic stress. Saturated long-chain FAs such as palmitate drive mitochondrial ROS and aberrant ER/SR Ca2+ release, depleting Ca2+ stores and impairing excitation-contraction coupling, while co-supplying unsaturated FAs, such as oleate, mitigates these effects by promoting esterification and partitioning of FAs into triglyceride droplets and blunting lipid stress-induced signaling.56,57 In Khodbukus et al., a three FAs treatment with a high unsaturated fraction may have favored neutral lipid storage and maintained contractile performance in healthy hiPSC-derived EMTs. By contrast, our broader mixture included more types of saturated FAs and also include arachidonic acid, a substrate for pro-inflammatory eicosanoids. Its inclusion may potentiate inflammatory signaling and alter membrane properties that impair contractility. 58 Our findings, alongside prior studies, highlight the need for systematic investigations into which fatty acid species or combinations are most culpable in driving IMCL-associated weakness.
The altered metabolic pathway-associated gene expression profile identified in our IMCL-loaded EMTs provides insight into potential therapeutic targets. Compared to healthy EMTs without excess FFA treatment, the most upregulated genes in IMCL EMTs are CD68, APOE, PYR1, and CD276, while the most downregulated are IL-6, PTGS2, CCL13, and AOX1 (Figure 5(i)). Specifically, APOE is a key apolipoprotein in lipid transport and is predominately synthesized by liver and macrophages to redistribute cholesterol and triglycerides. Data from previous publications indicates that, in various cell types, injury or lipid stress triggers increased APOE expression to facilitate lipid efflux. 59 Its induction in EMT likely reflects an adaptive response to excess intracellular fat, and higher APOE in skeletal muscle could alter local lipid handling and aid incorporation of FFAs into lipoproteins for clearance. IL-6 is the most downregulated in IMCL EMTs (Figure 5(i)). IL-6 is a pleiotropic cytokine with dual roles in metabolism and inflammation. 60 During exercise, muscle fibers actively secrete IL-6 as a myokine to increase lipolysis, fat oxidation, and insulin-stimulated glucose disposal. However, in obesity, chronically elevated IL-6 can lead to insulin resistance and low-grade inflammation along with other cytokines. 60 Moreover, a recent study revealed that lack of endogenous IL-6 leads to expansion of adipose tissue mass due to impaired FFAs mobilization in lean and obese men. 61 Reduced level of IL-6 in our IMCL model may represent impaired fat oxidation. Pharmacologically targeting IL-6 pathway in skeletal muscle could be complex, but drugs that mimic the muscle contraction/exercise response and activated downstream pathways, such as AMPK activator, irisin analogs, might be worth testing in the IMCL model. IMCL remodeling in EMTs provides a physiologically relevant platform for drug screening with contractile function as a quantitative readout, facilitating the identification of compounds capable of reversing lipid-induced muscle dysfunction.
In summary, our study provides a comprehensive comparative analysis of EMTs derived from hP-Myo and hiPS-Myo, elucidating critical differences in their structural, functional, and molecular maturation. Using a magnetic force-sensing platform, we demonstrated that hP-Myo EMTs generate superior contractile forces, exhibit larger myofiber diameters (quantified via AI-driven analysis), and display enhanced fatigue resistance compared to hiPS-Myo EMTs. Despite these differences, hiPS-Myo EMTs proved invaluable for modeling metabolic dysfunction: lipid overload induced IMCL features, including impaired contractility, blunted force-frequency responses, and dysregulation of lipid metabolism pathways, mirroring clinical hallmarks of ectopic fat accumulation in muscle. Our findings establish hP-Myo EMTs as robust platforms for studying physiological muscle function and highlight hiPS-Myo EMTs as tractable systems for interrogating complex metabolic diseases. By bridging the gap between primary and stem cell-derived models, this work advances the utility of engineered tissues for mechanistic studies, drug screening, and personalized therapeutic development.
Supplemental Material
sj-docx-1-tej-10.1177_20417314251382710 – Supplemental material for Human three-dimensional engineered muscle tissue characterization and intramyocellular lipid modeling
Supplemental material, sj-docx-1-tej-10.1177_20417314251382710 for Human three-dimensional engineered muscle tissue characterization and intramyocellular lipid modeling by Tianxin Cao, Eric Finnemore, Jon Hill, Shuo Wang, Chuanyu Wang, Heather Robinson, Jorge Villalona, Suzanne Segal and Curtis R. Warren in Journal of Tissue Engineering
Footnotes
Acknowledgements
Eric Finnemore thanks both Dr. Daniela M. Santos from Boehringer Ingelheim and Dr. Thomas Harris from University of Miami, Miller School of Medicine for their continued mentorship throughout his graduate studies. Tianxin Cao thank Dr. Husheng Qian from Boehringer Ingelheim for his guidance and support on histology. Tianxin Cao and Eric Finnemore thank Dr. Dongyue Xin from Boehringer Ingelheim for his support on data analysis. All authors extend their gratitude to their respective home departments of Boehringer Ingelheim for their support of these studies in service of discovering breakthrough therapies for patients in need.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Boehringer Ingelheim Pharmaceuticals, Ridgefield, CT, USA.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
