Abstract
Since myocardial infarction (MI) excessively damage the myocardium and blood vessels, the therapeutic approach for treating MI hearts should simultaneously target these two major components in the heart to achieve comprehensive cardiac repair. Here, we investigated a combinatory platform of ETV2 and Gata4, Mef2c and Tbx5 (GMT) transcription factors to develop a strategy that can rejuvenate both myocardium and vasculatures together in MI hearts. Previously ETV2 demonstrated significant effects on neovascularization and GMT was known to directly reprogram cardiac fibroblasts into cardiomyocytes under in vivo condition. Subsequently, intramyocardial delivery of a combination of retroviral GMT and adenoviral ETV2 particles into the rat MI hearts significantly increased viable myocardium area, capillary density compared to ETV2 or GMT only treated hearts, leading to improved heart function and reduced scar formation. These results demonstrate that this combinatorial gene therapy can be a promising approach to enhance the cardiac repair in MI hearts.
Introduction
Despite significant advances in modern medicine, the coronary heart disease (CHD) still remains a major cause of morbidity and mortality worldwide. Since the regenerative potential of adult cardiomyocyte (CM) in mammals is exceedingly limited, the massive loss of CMs following myocardial infarction (MI) caused by occlusion of main coronary arteries resulting in insufficient blood flow to the myocardium, has been regards as an irreversible damage.1–3 Although considerable efforts have been made to develop effective treatment strategies to treat this detrimental disease, currently available therapeutic options such as pharmacological and surgical interventions cannot reverse the pathophysiology associated with CHD, but only reduce symptoms and delay the progression of MI. 4 Therefore, revolutionary therapeutic solution that can be applied to heart failure patients have been urgently required.
Recently, gene therapy with transcription factors has come into the spotlight as a new way to overcome the shortcomings of conventional approaches involving classical gene therapy, since it can continuously deliver therapeutic proteins locally after single gene therapy and can potentially lead to reversal of pathophysiology associated with MI. 5 Along with this direction, several recent studies suggest the possibility that cardiac fibroblasts can be converted into the cells of the cardiovascular system, including CMs6–9 and endothelial cells (ECs)10,11 via overexpression of selected transcription factors termed as direct reprogramming. After screening multiple transcription factors known as key regulators for manipulating cardiac developmental, overexpression of three transcriptional factors such as Gata4, Mef2c, and Tbx5 referred as GMT efficiently converted cardiac fibroblasts into functional CMs, which exhibit representative CM phenotypes such as spontaneous contraction, calcium flux and organized sarcomere structure, under in vitro condition.12–15 Subsequently, local delivery of GMT into ischemic mouse myocardium in vivo by using retroviral platform resulted in a significant reduction in infarct/scar size, improvement in cardiac function along with clear evidences of successful conversion from cardiac fibroblasts to CMs in MI hearts. 16 These findings demonstrate that cardiac fibroblasts can be reprogrammed into CM-like cells using transcription factors in vivo for regenerative therapy purposes.
Increasing the number of vessels in ischemic region in the heart through vascular regeneration is another valid therapeutic strategy to promote cardiac repair. Recently, as a novel target for angiogenic gene therapy to promote the generation of new vessels, E-twenty six (ETS) Variant transcription factor 2 (ETV2) has been intensively investigated. It has been well delineated that ETV2 plays pivotal roles in embryonic vascular development and postpartum vascular regeneration.16–19 Indeed, expression of ETV2 especially in EC, is low during postnatal life in general, however its expression is substantially upregulated upon ischemic insult. It was evidenced that ETV2 deficient mice exhibited defective blood vessel development and ineffective vascular regeneration following ischemic injury. 20 Based on these results, it is believed that ETV2 is one of the critical genes responsible for new blood vessel regeneration in postnatal life. Subsequently, lentiviral delivery of ETV2 alone or together with other transcription factors into human dermal fibroblast could induce direct reprogramming into functional ECs that formed functional perfused vessels in vivo. 21 Furthermore, we reported that in vivo delivery of ETV2 into MI induced murine hearts led to a substantial improvement in vessel formation. 22 As underlying mechanism, we discovered that ETV2 promoted angiogenesis by directly regulating EC proliferation, migration, and vessel forming capability with enhanced expression of several angiogenic genes suggesting that ETV2 is a novel candidate for promoting angiogenesis in failing heart.
Since MI lead excessive damage to both cardiac muscles and vasculatures, which are two major components in the heart, therapeutic strategies for treating ischemic hearts should focus on repairing cardiac muscle and cardiac vessel together to achieve comprehensive cardiac repair. The principal behind cardiac gene therapy should adhere to the same principles as well. Accordingly, in this study, we investigated whether simultaneous delivery of GMT and ETV2 could rejuvenate both the myocardium and vasculatures concurrently in the ischemic heart. We hypothesize that in vivo deliveries of GMT and ETV2 would regenerate myocardium, and blood vessels, respectively, and simultaneous administration of both GMT and ETV2 will lead to comprehensive cardiac repair. Consequently, we demonstrate that the dual gene therapy applying GMT together with ETV2 lead to a significant improvement of cardiac function and improvement of vessels post MI. These results involve significant implications for cardiac gene therapy as an effective therapeutic mean for treating heart failure.
Materials and methods
Preparation of retroviral GMT and Adeno-associated virus ETV2
Retroviral vectors encoding mouse Gata4, Mef2c, Tbx5 in pMxs based vector was purchased from Addgene (pMx-puro-MGT; plasmid #111809). Retroviruses were generated as described 15 using pMXs retroviral vectors containing coding regions of Gata4, Mef2c, Tbx5. 10 ug of retroviral plasmid DNA was transfected using Fugene 6 (Roche) into Platinum E cells (Cell Biolabs) which were plated on a 10-cm tissue culture dish at a density of 3 × 106 cells per dish, 24 h prior to transfection. The spent medium was changed with 12 ml of fresh medium (DMEM supplemented with 10% FBS and antibiotics). After 36 h of transfection, viral medium was harvested, concentrated at 8000 g for 16 h at 4°C and filtered through a 0.45 μm cellulose filter. The viral supernatant was mixed with polybrene (Sigma) to a final concentration of approximately 1 × 1010 plaque-forming units (pfu)/ml. On the other hand, recombinant Adeno-associated virus (AAV) 9 vectors were produced as previously described. 22 Briefly, AAV vectors, rep2/cap9 packaging plasmids, and adenoviral helper plasmids were mixed with polyethylenimine and added to HEK293T cells (Thermo Scientific). At 72 h post transfection with pCSII-EF1α-ETV2-IRES-VENUS, pCAG-HIVgp and pCMVVSV-G-RSV-Rev (4:3:1), supernatant and cells were harvested separately for AAV9 preparation. Viruses in the supernatant were precipitated (mixed with 8.5% w/v PEG-6000 and 0.4 M NaCl for 2 h at 4°C), centrifuged at 7000 g for 10 min, and resuspended in a virus buffer (150 mM NaCl and 20 mM Tris, pH 8.0). Recovered AAV vectors were washed three times with PBS using Amicon 100K columns (EMD MilliporeViruses were diluted to various concentrations to test infection, and a concentration of approximately 2 × 1012 genome copies (GC)/ml was used for further experiments.
Rat myocardial infarction modeling and treatment
The animal studies were approved by The Catholic University of Korea Animal Care and Use Committee and IACUC and Department of Laboratory Animal (DOLA) in Catholic University of Korea, Songeui Campus. Fischer 344 rats (180–200 g, 8-week-old male, Koatec, Korea) were anesthetized with 2% inhaled isoflurane and intubated via the trachea with an 18G intravenous catheter. MI was induced by ligating left anterior descending (LAD) coronary artery using 7-0 prolene suture under mechanical ventilation with medical oxygen. Following experimental groups were made by injecting different types of virus particles into the myocardium. Experimental groups included (1) ETV2 only: AAV form of ETV2 (1 × 106 virus particle (vp)/m), (2) GMT only: retroviral form of GMT (1 × 106 vp/m), (3) ETV2/GMT combination (5 × 105 vp/ml of ETV2 + 5 × 105 vp/ml of GMT) and (4) control group (5 × 105 vp/ml of control AAV + 5 × 105 vp/ml of control retrovirus). After close the chest, the animals were recovered in cages and were maintained for further experiments.
Immunohistochemical analysis
Heart tissues were harvested and cryopreserved with OCT compounds (Shandon) and sectioned in 10 μm thickness. To perform immunofluorescent staining, frozen sections were permeabilized with 0.5% Triton X100 in PBS, blocked with 1% Tween 20 in PBS containing 1% bovine serum albumin (Sigma-Aldrich). Next, heart sections were incubated with primary antibodies including anti-cardiac troponin T (1:100 dilution; Sigma-Aldrich) and then incubated with secondary antibody (1:500 dilution; Alexa Fluor488; Invitrogen). The slides were mounted with a mounting solution containing 4’, 6-diamidino-2-phenylindole (DAPI) (Vector Laboratory Inc.) for further analysis.
TUNEL Assay
TUNEL staining was applied using a Click-iTTM Plus TUNEL assay kit (Invitrogen) according to the manufacturer’s instruction. Heart tissues were hydrated, fixed in 4% Paraformaldehyde (Chemcruz) and permeabilized with Proteinase K. Samples were then fixed again with 4% PFA and incubated with TUNEL reaction cocktail. Finally, samples were stained with a primary antibody specific for cardiac troponin T (1:100; mouse anti-cTnT; Thermofisher) and secondary antibody (1:500; Alexa 488 anti-mouse; Thermofisher) mounted with DAPI solution (Vector laboratory). Samples were imaged under a confocal microscope using a 20X objective. Five views were randomly selected within the infarcted zone from each section to assess the ratio of apoptotic CMs.
18F-FDG cardiac PET imaging
18F-FDG uptake in the infarcted myocardium was evaluated by using a small animal SPECT scanner (InveonTM; Siemens Preclinical Solutions, Malvern, PA). Animals were anesthetized with 2% isoflurane during the imaging process. All animals were given at an intravenous dose of 7.4–11.1 MBq of 18F-FDG. The 18F-FDG PET imaging was performed for 20 min at 1 h after injection. PET emission data were acquired with three spans and 79 ring differences through a 350–650 keV energy window and 3.43 ns timing windows. The acquired three-dimensional emission list-mode data were reconstructed for the PET image using the two-dimensional ordered subset expectation maximization (OSEM 2D) algorithm with four iterations. The PET images were visualized and analyzed using Siemens Inveon Research Workplace (IRW) software (Siemens Preclinical Solutions).
PET image analysis
The 18F-FDG cardiac PET image was processed by rotation and cropping, to be shown only heart area. The cardiac PET or SPECT images of the cardiac area (acquired from Inveon PET/SPECT scanner) were converted into DICOM format to read by other analysis software. The direction of cardiac PET or SPECT images of DICOM format were re-oriented to short-axis, horizontal long axis (HLA) and vertical long axis (VLA). Then, the cardiac PET or SPECT images generated as a polar map by using the Clinical QGS software (Cedars QGS 2008, Syngo, Siemens).
Determination of capillary density
Isolectin B4 (Vector Laboratory Inc.) conjugated with red fluorescence dye was intramyocardially injected into MI heart prior to harvest at 8-week post MI to visualize the capillaries. For measurement of capillary density, heart tissues were captured in 1 mm2 with 10× object lens under Nikon’s Eclipse Ni-E upright microscope (Nikon Instruments Inc). Two fields, border zone and infarct zone in the post-infarct left ventricle, were examined with a red fluorescent signal to quantify the presence of capillary using image analysis software, Image J. Capillary density from heart sections was calculated by a total number of capillaries per 1 mm2.
Echocardiography
The assessment of cardiac functions of infarcted hearts was performed with echocardiography as we previously described. 23 The rats were anesthetized with inhaled 2% isoflurane, and physiological data were recorded with a transthoracic echocardiography system equipped with a 15 MHz L15-7io linear transducer (Affniti 50G, Philips). The Echocardiograms were performed at 1, 2, 4, and 8 weeks after MI. The echocardiography operator was blinded to the group allocation during the experiment. Ejection fraction and fractional shortening were calculated using the following equation. 24
Evaluation of fibrosis
Infarct fibrosis was assessed using Masson’s trichrome staining kit (Sigma-Aldrich, HT-15) according to the manufacturer’s protocol. Three sections from different levels of each heart, from the level near the ligation site, the level far away from the ligation site and middle level, were stained with Masson’s trichrome for analysis of extends of fibrosis. To perform Masson’s trichrome staining, the samples were fixed in Bouin’s solution (Sigma-Aldrich) at 56°C for 1 h and then stained in hematoxylin for 5 min. The samples were stained with Scarlet for 3 min and treated with a mixture of phosphotungstic acid and phosphomolybdic acid for 5 min. Aniline (Sigma-Aldrich) was then used to stain the sample for 5 min and then treated with 1% acetic acid for 2 min. The stained samples were scanned with Epson Perfection V370 photo scanner (EPSON) to quantify both fibrotic-area and viable myocardium using ImageJ software.
Statistical analysis
All results are presented as mean ± standard error of mean (S.E.M). one-way ANOVA followed by Tukey’s or Bonferroni multiple comparisons post-test as indicated was used for statistical analysis, and significant differences are indicated by specific footnotes as indicated in the figure legends. Data analysis was performed with Prism 5.0 software (GraphPad, La Jolla, California).
Results
Treatment with GMT or ETV2/GMT induced cardiac muscle regeneration in MI hearts
We first evaluated whether overexpression of ETV2/GMT could regenerate the myocardium in the rat MI hearts induced by permanent ligation of the LAD artery. To achieve this, we intramyocardially injected control virus, adeno-associated virus (AAV)-ETV2 alone, retroviral-GMT alone or combination of ETV2 and GMT (ETV2/GMT) in the MI induced hearts and assessed the viable myocardium determined by measuring the area of cardiac troponin T positive (cTnT+) myocardium in the post-infarct left ventricle wall CMs at 8 weeks from MI induction. The result from histological analyses showed that area of cTnT+ myocardium in the ETV2/GMT treated MI hearts is higher than other hearts receiving GMT or ETV2 only (3.66 ± 0.68% in control, 8.19 ± 1.50% in ETV2 alone, 10.70 ± 2.03% in GMT alone and 18.35 ± 2.46% in ETV2/GMT, n = 3–6) (Figure 1(a) and (b)). No significant difference was detected between the hearts injected with ETV2 and GMT alone. In addition, the number of dead CMs in all experimental group was evaluated by TUNEL assay (Figure 1(c)). Fluorescent image analysis revealed that transduction of ETV2/GMT significantly reduced apoptotic cell death of CMs as evidenced by only a few TUNEL+ CMs were detected from the ETV2/GMT injected heart tissues compared to control, ETV2 or GMT alone injected MI hearts (13.00 ± 1.90% in control vs 7.31 ± 1.38% in ETV2 alone, 5.44 ± 1.17% in GMT alone and 1.34 ± 0.73% in ETV2/GMT, n = 3) (Figure 1(d)). Lastly, in order to more accurately quantify the infarct size of MI hearts in all experimental groups, we performed positron emission tomography (PET) using 2-[18F]-fluoro-2-deoxy-d-glucose (18F-FDG) with heart tissues collected at 8 weeks from induction of MI and injection of transcription factors (Figure 1(e)). The cardiac heat map illustrates that the tissues from the ETV2/GMT group show the smallest infarct size compared to control, ETV or GMT only groups (Figure 1(f)). These results suggest that the delivery of GMT or ETV2/GMT into rat MI heart have considerable effects on cardiac muscle regeneration.
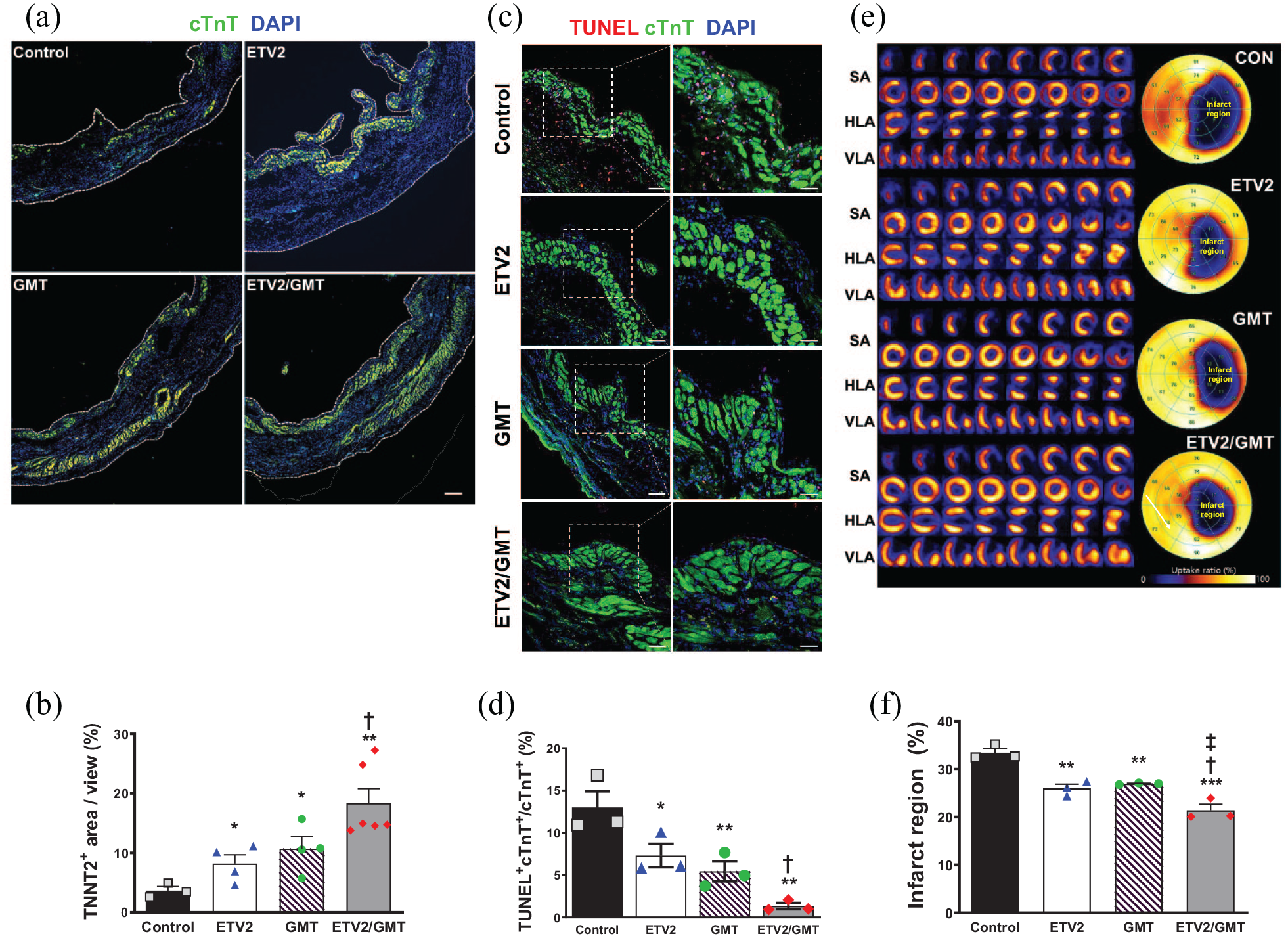
Treatment with GMT or ETV2/GMT induced cardiac muscle regeneration in MI hearts. (a) Representative immunostaining images from all experimental groups showing viable cTnT positive myocardium (green) in the LV of MI heart 8 weeks post MI and their quantification summary. Scale bar = 250 μm. (b) Quantitative analysis of cTnT positive CM density in the LV of MI heart. (c) Representative images of TUNEL assay using the heart tissues harvested from all experimental groups 8 weeks post MI and their quantification summary. Scale bar = 100 μm in left panels, 50 μm in right panels. (d) Percentage of cardiomyocytes double positive for TUNEL and cTnT in the LV of MI heart. (e) Representative left ventricular heat map of 18F-FDG PET illustrating the glucose uptake. Cardiac 18F-FDG PET image was reoriented to the short-axis (SA), the horizontal long-axis (HLA), and the vertical long axis (VLA). (f) Quantitative summary of 18F-FDG PET from all experimental groups.*p < 0.05, **p < 0.01, ***p < 0.005 versus control group; †p < 0.05 versus ETV2 group; ‡p < 0.05 versus GMT group, n = 3~6.
Overexpression of ETV2 or ETV2/GMT promotes vascular regeneration in MI heart
Since ETV2 has been reported as a key mediator of postnatal vascular regeneration, we investigated the effects of ETV2 or ETV2/GMT on blood vessel regeneration in ischemic hearts. To this end, we perfused isolectin B4 (IsB4) which conjugated with Red fluorophore, Rhodamine, the MI induced rats at 8 weeks prior to sacrifice to visualize functional cardiac vessels (Figure 2(a)). The fluorescent analysis showed that the number of capillaries in infarct area of ETV2 only injected hearts (59.27 ± 2.98/mm2, n = 3) and ETV2/GMT injected group (55.67 ± 2.28/mm2, n = 6) were significantly higher than that of control group (37.80 ± 2.04/mm2, n = 3) or GMT alone group (42.30 ± 2.08/mm2, n = 4) (Figure 2(b)). In addition, the number of capillaries in the border zone of ETV2 only (141.40 ± 8.32/mm2, n = 3) and ETV2/GMT injected hearts (105.83 ± 2.96/mm2, n = 6) were substantially higher than control group (68.33 ± 4.06/mm2, n = 3) and GMT alone group (78.05 ± 4.61/mm2, n = 4) (Figure 2(c)) suggesting ETV2 contribute for angiogenesis and has substantial effects on vascular regeneration in ischemic hearts.

Overexpression of ETV2 or ETV2/GMT promotes vascular regeneration in MI heart. (a) Representative fluorescent images of capillary density at 8 weeks post MI. Hearts were perfused with rhodamine conjugated Isolectin B4 to visualize vessels (Red) and nucleus was stained with DAPI (Blue). (b) Quantification summary of capillary density in border zone and (c) infarcted zone from all experimental groups. The number of capillaries was calculated from five randomly selected fields (1 mm2) in each heart. *p < 0.05, **p < 0.01, ***p < 0.005 versus control group; ‡p < 0.05, ‡‡p < 0.01, ‡‡‡p < 0.005 versus GMT group, n = 3~6. Scale bar = 50 μm.
ETV2/GMT combinatory treatment ameliorate cardiac dysfunction and scar formation in MI hearts
Lastly, to evaluate the therapeutic effects of overexpression of ETV2 or GMT or altogether in MI heart, we conducted serial echocardiography on a weekly basis to measure cardiac functions and remodeling of rat hearts in all experimental groups. Echocardiography results demonstrated that administration of the ETV2/GMT combination significantly improved cardiac function evidenced by ejection fraction (EF) (50.43% ± 4.33%, n = 6) and fractional shortening (FS) (22.53% ± 2.31%, n = 6) compared to control group (EF: 27.52% ± 0.55%; FS: 11.13% ± 0.22%, n = 6), ETV2 only group (EF: 42.38% ± 0.43%; FS: 18.57% ± 0.46%, n = 6) and GMT only group (EF: 40.05% ± 1.02%; FS: 17.05% ± 0.50%, n = 5) (Figure 3(a)–(d)). Both EF and FS, two functional parameters of cardiac function, in the ETV2 and GMT alone group were significantly greater compared to the control group. Of interests, while both EFs and FSs of the control, ETV2 only, and GMT only group were rapidly deteriorated after onset of MI and were maintained thereafter until 8 weeks, the cardiac function of the ETV2/GMT combination group was clearly augmented up to approximately 10% after MI induction (Figure 3(c) and (e)). Moreover, the parameters for cardiac remodeling such as left ventricular internal diastolic dimension (LVIDd) and left ventricular internal systolic dimension (LVISd) results suggested that the overall cardiac remodeling of the hearts in the mixture of ETV2/GMT injected group was significantly reduced compared to other experimental groups (Figure 3(f) and (g)). The results from Masson’s trichrome staining using the heart tissues harvested at 8 weeks post MI further revealed that the ETV2/GMT-injected group showed the most lowest scar formation compared to the control group (27.13 ± 2.21% in ETV2/GMT vs 46.46 ± 4.22% in control, 29.57 ± 3.95% in ETV2 and 37.14 ± 1.21% in GMT, n = 3–6) suggesting that combinatorial treatment of ETV2/GMT reduced adverse cardiac remodeling following MI (Figure 4(a)–(c)). Taken altogether, our results indicate that combinatory treatment with ETV2/GMT induced comprehensive cardiac repair in the ischemic hearts evidenced by improved cardiac function and reduced adverse cardiac remodeling possibly through simultaneous regeneration of both cardiac muscle and blood vessel.

ETV2/GMT combinatory treatment improved cardiac function in MI hearts. (a) Representative images of echocardiography from all experimental groups 8 weeks after interventions. IVS, interventricular septum; LVIDd, left ventricular internal Diastolic dimension; LVISd, left ventricular internal systolic dimension; LVPW, left ventricle posterior wall. (b–g) Summary of (b) Left ventricular ejection fraction (LVEF), (c) EF delta change, (d) Fractional shortening (FS), (e) FS delta change, (f) Left ventricular internal Diastolic dimension (LVIDd), and (g) Left ventricular internal systolic dimension (LVISd). *p < 0.05, **p < 0.01, ***p < 0.005 versus control group; †p < 0.05, ††p < 0.01 versus ETV2 group; ‡p < 0.05 versus GMT group, n = 3~6. CON, control group.
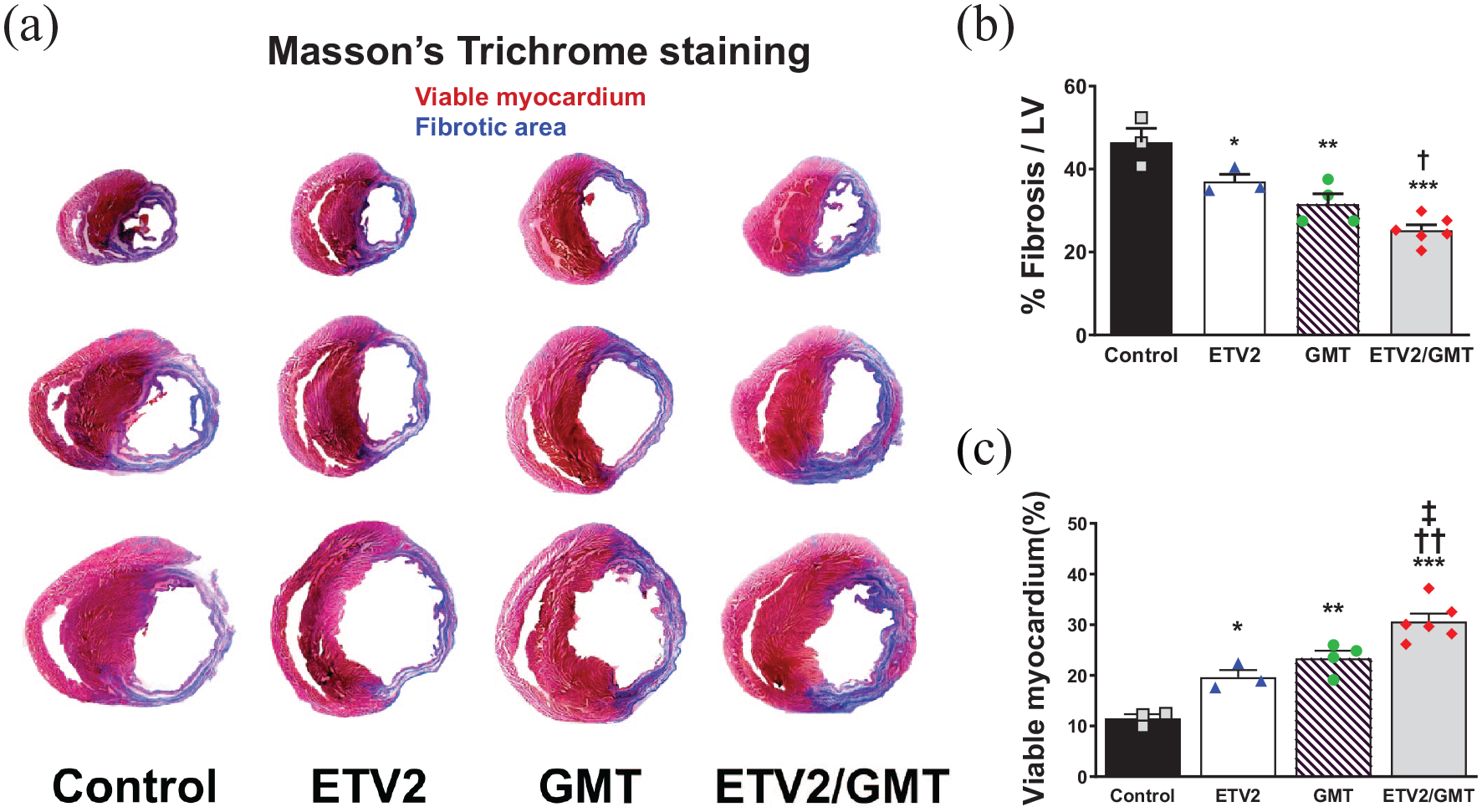
In vivo transduction of ETV2/GMT reduced scar formation in MI hearts. (a) Representative images of Masson’s trichrome staining showing the fibrotic composition (blue) and viable tissue (purple) in the LV of MI heart 8 weeks post MI. (b) Quantitation summary of the percentage of fibrosis from all experimental groups. (c) Quantitative analysis viable myocardium (red) in the LV of MI. *p < 0.05, **p < 0.01, ***p < 0.005 versus control group; †p < 0.05, ††p < 0.01 versus ETV2 group; ‡p < 0.05 versus GMT group, n = 3~6.
Discussion
A series of recent studies have reported a novel approach termed as direct reprogramming, that conversion or trans-differentiation of somatic cells into specific lineage cells such as CMs,5–9 ECs,10,11 neurons,25–27 or hepatocytes, 28 using lineage or cell type-specific transcription factors or miRNAs without dedifferentiation into a pluripotent state. This direct reprogramming approach has received enormous attention as the third-generation platform for regenerative medicine because it could reduce the time and cost for generating target cell, enable to avoid the potential risks associated with the use of human pluripotent stem cells or their derivatives including the teratoma formation from remaining undifferentiated stem cells. 29 In the present study, we sought to investigate whether a novel gene therapy strategy utilizing a combination of defined transcription factors, GMT and ETV2, could induce comprehensive cardiac repair through simultaneous regeneration of myocardium and blood vessels in ischemic hearts. Our results demonstrated that intramyocardial delivery of ETV2/GMT into infarcted heart synergistically promote the regeneration of cardiac muscle and blood vessels evidenced by the substantially greater area of viable myocardium and higher number of functional capillaries in the infarcted area compared to other GMT or ETV2 only treated hearts (Figure 5).

Schematic diagram of the underlying mechanism of in vivo combinatory gene therapy with GMT and ETV2 for comprehensive cardiac repair. Infarcted area (purple), regenerated cardiomyocytes (green), and blood vessels (yellow).
Indeed, there have been several previous studies that describe the therapeutic effects of GMT or ETV2 treatment in the ischemic hearts. Qian et al 16 previously demonstrated that local delivery of retroviral form of GMT successfully reprogrammed non-CMs to CMs which displayed well-organized sarcomere structures and functional properties of true CMs in MI induced hearts in vivo. Subsequently, transfection of GMT into the MI hearts promoted cardiac functions and reduced scar size compared to control hearts. By using several lineage-tracing experiments, authors verified that new CMs were not derived from cell fusion with resident CMs, but they were directly converted from resident CFs. Interestingly, authors claimed that efficiency for direct reprogramming under in vivo condition is substantially higher than in vitro conditions. In case of ETV2, we showed that in vivo transduction of ETV2 into the infarcted heart considerably increased the number of capillaries in the infarct zone of the MI hearts and substantially elevated heart function compared to control infarcted heart. 22 Compared with those previous reports, to the best of our knowledge, our study is the first to simultaneously examine the therapeutic effects of two distinct set of transcription factors, ETV2 and GMT, for inducing comprehensive cardiac repair.
One of the major findings of our study is that combinatory treatment with ETV2/GMT induced substantially higher cardiac function 8 weeks post MI even compared to GMT or ETV treated hearts, which still demonstrated significantly greater heart function over MI control hearts. Of interest, consistent with previous studies, we observed that the area of viable TNNT2 positive myocardium in the GMT only or ETV2/GMT co-treated hearts was significantly higher than ETV2 only or control virus treated hearts indicate the contribution of GMT in the regeneration of myocardium in the ischemic hearts. Conversely, the number of capillary in both the infarct zone and border area of MI heart receiving control vectors or GMT alone was substantially lower than ETV2 only or ETV2/GMT treated hearts suggesting that ETV2 play certain roles for vascular regeneration.22,30 Since the rapid vascular regeneration is critical for persisting the survival of injured myocardium, enhanced number of blood vessel by the delivery of ETV2 contributed at least to some extent to promote the regeneration of myocardium in the MI heart.
Despite our study showed several promising results, there are some limitations that should be considered in the follow-up studies. First, in addition to permanent ligation model that we used in this study, another heart injury model such as ischemia/reperfusion model should be considered to investigate therapeutic effects of ETV2/GMT since different injury model may generate distinct results. Also, non-viral delivery methods to overexpress gene(s) of interest in the MI hearts in vivo such as nanomaterials, hydrogels or CRISPR based approach should be developed to rule out the risk of the mutagenesis which can be happened by the viral delivery method.31–33
Conclusion
Collectively, we speculate that the fundamental reason that simultaneous treatment with ETV2/GMT in the MI heart exhibited considerably greater heart functions is probably due to the increased number of blood vessel from ETV2 treatment which further allowed to perfuse sufficiently to the myocardium regenerated by GMT treatment. This study may serve as a proof-of-concept approach that concurrent treatment of ETV2/GMT can be used as a sophisticated therapeutic tool to promote neovascularization and cardiac regeneration simultaneously and future studies are required to identify detailed molecular mechanisms underlying ETV2/GMT-based cardiac regeneration in failing hearts.
Footnotes
Acknowledgements
The authors would like to thank Dr. Hiroyuki Miyoshi at Keio University School of Medicine and Dr. Changwon Park at Louisiana State University for instrumental discussions and directions for this manuscript.
Authorship
Experimental conception and design, acquisition of data, analysis and interpretation of data, and manuscript drafting and revising: S.L., B.-W.P., and Y.J.L. Experimental conception and design, financial support, administrative support, manuscript drafting and revising, and final approval of the manuscript: K.B., and H.-J.P.
Data availability statement
The authors declare that the data supporting the findings of this study are available within the article and its supplemental information files. The reader may contact the corresponding authors with any request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Hong Kong Research Grants Council, Hong Kong (21100818 to K.B.) and the Bio & Medical Technology Development Program grant (NRF-2017M3A9B3061954 to H.J.P.) funded by the Ministry of Science & I.C.T, Republic of Korea.

