Abstract
The attachment of implants relies on bone and soft tissue biocompatibility. The aim of this article is to investigate the effect of fluoride-modified metallic titanium (Ti) surfaces (Ti-F) on proliferation and differentiation of human gingival fibroblasts. Human gingival fibroblast cells were exposed to hydrofluoric acid–modified Ti coins (Ti-F) for 1, 3, 7, 14 and 21 days, and untreated coins were used as controls. A five- to six-fold increase in the proliferation of human gingival fibroblasts on Ti-F compared to Ti surfaces was observed. Enhanced gene expression of interleukin-6 and osteoprotegerin was found at 7 days. Increased levels of sclerostin, interleukin-6 and osteoprotegerin in the media from human gingival fibroblasts cultured on Ti-F coins were found compared to controls. Our results confirm that hydrofluoric acid–modified surface may indirectly enhance the firm attachment of implant surface to junction epithelium, soft tissue epithelium, which would give protection for underlying osseous structures making osseointegration of the dental implant possible.
Introduction
Fluoride modification of titanium (Ti) is one surface modification that has been introduced with successful clinically results and significant improvements in the biological responses with increased bone-to-implant contact and functional attachment.1–5 According to Ellingsen, 6 fluoride-modified titanium implants give a firmer bone anchorage than the unmodified titanium implants. Fluoride on the titanium implant surface is suggested to have a stimulatory action on osteogenic cells and thus accelerate bone regeneration,7,8 and it has as well been reported to improve peri-implant tissue response.9–13
The effects of both fluoride surface modification of Ti and titanium dioxide (TiO2) on the biological response have been studied on murine osteoblast cells,14,15 human palatal mesenchymal cells, 9 human mesenchymal stem cells11,16,17 and human primary osteoblasts. 18 Fluoride-modified titanium surfaces have been found to stimulate both proliferation and differentiation of various cells in vitro; however, only increased proliferation of primary human osteoblasts was found in our previous study. 18
Soft tissue wound healing around dental implant is complex. The long-term success of an implant depends on the classical stages of wound repair, 19 and the establishment of soft tissue barrier to protect the underlying osseous structures makes osseointegration possible surrounding the implant body. 20 Connective tissue cells like gingival fibroblast is the soft tissue barrier that prevents bacterial penetration and thus gingival recession and bone resorption due to inflammation. 21 These cells have an important role in the development, function and regeneration of the tooth-support structures22,23 and production and maintenance of connective tissue matrix.23,24 Human gingival fibroblasts (HGFs) are also important for the implant success by inhibiting the epithelial down growth. 25
Much is known about the response from osteoblasts and bone to various surface modifications of titanium implants, but less is known about the soft tissue reactions. The surface modification by hydrofluoric acid (HF) has been reported to influence cell growth and adhesion of surrounding soft tissue to the implant surface. 13 In this in vitro study, we investigated whether fluoride-modified titanium implant surfaces had any effect on proliferation, differentiation and secretion of factors from primary HGFs.
Materials and methods
Metallic titanium
Coins (n = 120) of grade 2 titanium (Ti), with a diameter of 14.0 mm and a height of 1.0 mm, were grit-blasted TiO2 180–220 µm as previously described. 26 All the coins went through five washing steps as described elsewhere. 14 Half of the coins went through an additional surface modification procedure with 0.2 vol. % HF for 120 s, giving a fluoride-modified TiO2 surface (marked Ti-F), 14 whereas the rest were left untreated (marked Ti). All coins were sterilized by gamma-irradiation at 25 kGy with a Co-60 source (Institutt for Energi teknikk, Kjeller, Norway). The coins were placed in cell culture plates, y-sterilization and kept dry until culturing of the HGF started after 1 week. The coins have been fully characterized by profilometer, atomic force microscopy (AFM), X-ray photoelectron spectroscopy (XPS), contact angle and secondary ion mass spectrometry (SIMS) as described previously.14,18
Experimental design and analysis
Cell culture
Human gingival fibroblast-1 (HGF-1; American Type Culture Collection, Manassas, VA, USA) were cultured in 75 cm2 flask in Dulbecco’s Modified Eagle’s medium (DMEM, High Glucose, PAA Laboratories, Linz, Austria) containing 10% foetal bovine serum (FBS) supplemented with 100 U/mL penicillin and 100 µg/mL streptomycin (PAA Laboratories, Linz, Austria), defined as growth medium (GM), at 37°C in a humidified atmosphere with 10% CO2. The cell culture was changed three times per week until confluence. The cells were rinsed by phosphate-buffered saline (PBS; PAA Laboratories, Linz, Austria) and detached using a trypsin/EDTA (0.05%/0.02%) solution (PAA Laboratories, Linz, Austria) for 2 min. HGFs at 90% confluence were used at passage 6. The coins were placed in 24-well plates (Multidish, Nuncleon Surface, Nunc, Roskilde, Denmark), and the HGFs were seeded from a uniform solution in a dropwise manner onto each coin. The cell culture medium was changed twice a week throughout the experiment. At 1, 3, 7, 14 and 21 days, the cell culture medium was sampled and the cells were harvested. The cell culture medium was stored at −20°C until analysis. Cells were washed once with saline prior to cell lysis and messenger RNA (mRNA) isolation. The mRNA samples were stored at −80°C until analysis. Every experiment in this study was performed with three biological replicas.
Lactate dehydrogenase activity
The cytotoxicity of the fluoride surface-modified Ti coins (n = 18) was estimated based on the activity of the cytosolic enzyme lactate dehydrogenase (LDH) in the culture medium and compared to the activity from cells cultured on Ti coins (n = 18). The LDH activity from cells incubated with sodium fluoride (NaF: 1, 10 and 100 µg/mL; Fluka BioUltra, Sigma-Aldrich, St. Louis, USA) was compared to untreated cells. The LDH activity was determined in the medium collected after 1 and 3 days, according to the manufacturer’s instructions (Roche Diagnostics, Mannheim, Germany). In total, 50 µL of each sample was incubated with 50 µL of the kit reaction mixture for 30 min in the dark at room temperature. The absorbance of the samples was measured at 492 nm in a plate reader (BioTek El x 800, BioTek Instruments Inc., Winooski, VT, USA).
Effect of fluorine on cell proliferation
The proliferation rate of HGF was measured using [ 3 H]-thymidine incorporation on fluoride-modified and unmodified sample surfaces (n = 36). In addition, subconfluent cells on plastic were also incubated for 1 and 3 days with fresh cell culture medium with sodium fluoride (NaF: 1, 10 and 100 µg/mL) (Fluka BioUltra) as control. We want to measure the effect of the free bound fluoride from HF treatment with NaF on HGF in this study. The cells were pulsed with [ 3 H]-thymidine 12 h prior to harvest after a protocol described in a previous study. 18 The cells were counted for 3 min in a liquid scintillation counter (Packard 1900 TR, Packard Instruments, Meriden, CT, USA).
mRNA isolation
Cells were lysed in lysis/binding buffer (100 mM Tris–HCl, pH 8.0, 500 mM LiCl, 10 mM EDTA, pH 8.0, 0.5 mM dithiothreitol (DTT), and 1% sodium dodecyl sulphate (SDS)) and mRNA was isolated using magnetic beads [oligo (dT)25] as described by the manufacturer (Dynal AS, Oslo, Norway). Beads containing mRNA were resuspended in 10 mM Tris–HCl, pH 8.0, and stored at −80°C until use. One microgram of mRNA was used to synthesize complementary DNA (cDNA) using the Fermentas RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, Vilnius, Lithuania) for each of the samples.
Real-time polymerase chain reaction
The reverse-transcribed cDNA was subjected to real-time reverse transcription polymerase chain reaction (RT-PCR) using SsoAdvanced SYBR Green Supermix (Bio-Rad, Hercules, CA, USA) in the CFX384TM Real-Time System (Bio-Rad). cDNA samples (1 µL in a total reaction volume of 25 µL) were analysed for both the genes of interest and the reference gene (GAPDH). Gene name and primer sequences are listed in Table 1. The thermal profile was as follows: samples were denatured at 94°C for 5 min followed by 40 cycles of annealing at 60°C for 30 s, primer extension at 72°C for 30 s, and denaturing at 95°C for 30 s. Finally, one 3-min extension cycle completed the reaction sequence. Replicates (n = 4) actions were performed in duplicate in a 96-well plate. Cycle threshold (Ct) values were obtained graphically. Gene expression was normalized to GAPDH and presented as ΔCt values. Comparison of gene expression between untreated control samples and treated samples was derived from subtraction of control ΔCt values from treatment with equation (1)
ΔCt values to give a ΔΔCt value, and relative gene expression was calculated as 2_ΔΔ C(t) (2ΔΔCt) and normalized to GAPDH. The efficiency of each set of primers was always higher than 90%. The method to calculate the relative mRNA expression is by ΔΔCt. 27
Primer sequences used for real-time RT-PCR assays.

COL3A1: collagen type 3 alpha 1; MMP-1: matrix metalloproteinase-1; TGF-β1: transforming growth factor beta 1; ITGA2: integrin α2; ITGB3: integrin β3; SOST: sclerostin; TNFRSF11B: OPG; IL-6: interleukin 6; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; RANKL: receptor activator of nuclear factor kappa B ligand.
Levels of bone markers in culture medium
Prior to analysis, the samples were concentrated five times using Microsep Centrifugal tubes with 3 kDa cut-off (Pall Life Science, Ann Arbor, MI, USA). Multianalyte profiling was performed using the Luminex 200TM system (Luminex Corporation, Austin, TX, USA) and acquired fluorescence data were analysed by the 3.1 xPONENT software (Luminex).
The level of bone markers (interleukin (IL)-6, osteoprotegerin (OPG), osteocalcin (OC), leptin, osteopontin (OPN), sclerostin (SOST), tumour necrosis factor-α (TNF-α), fibroblast growth factor 23 (FGF-23)) was determined using Milliplex Map Human Bone Panel Magnetic Bead (HBNMAG-51K; Millipore, Billerica, MA, USA). RANKL was tested using a single plex kit (HRNKLMAG-51K-01; Millipore). All analyses were performed according to the manufacturers’ protocols.
Calcium analysis
Calcium content in the cell culture medium was determined by atomic absorption spectroscopy (AAS) (Analyst 400; PerkinElmer, Norwalk, CT, USA) after 1, 3, 7, 14 and 21 days of culture. Samples and standards were prepared according to Perkin-Elmer’s analytical methods for AAS. Prior to analysis, lanthanum chloride (VWR, Fontenay-sous-Bois, France) was added to the samples and standards to a final concentration of 0.5% (w/v) in order to counteract the negative effect of phosphorus on calcium sensitivity of the spectrophotometer. The absorbance was measured at 422.7 nm using an air–acetylene flame. Calcium content was calculated by WinLab32 AA Flame software (Perkin-Elmer) using a standard curve based on calcium carbonate (Sigma-Aldrich).
Calcium deposition in cells
Alizarin red staining was used to determine the presence of calcific deposition by the HGF cells. Cells on Ti and Ti-F coins were cultured in 24-well plates (n = 36, respectively) and incubated in GM and sampled after 1, 3, 7 and 14 days. The media were changed every third day and collected. The cells were washed with PBS three times and fixed in 95% ethanol for 30 min. The cells were then stained with 1% alizarin red S (pH 4.2) for 5 min and washed with distilled water, as described elsewhere. 28 The alizarin red deposition was extracted with cetylpyridinium chloride (Sigma-Aldrich) at room temperature and quantitated at 562 nm in ELx800 Absorbance Reader (BioTek instruments, Winooski, VT, USA).
Statistical analysis
The statistical analyses were conducted using SigmaPlot (V 13.0 for Windows, Systat Software Inc., Chicago, USA). Normality and equal variance tests were performed prior to the testing. The effect of fluoride-modified surface (Ti-F) was compared to the respective controls (Ti) at each time points. Data were presented as the mean ± SD when datasets were normally distributed. If datasets were not normalized, the median (Q2) and the interquartile range (IQR) were presented. Pairwise comparisons between groups and controls were made using analysis of t-test and comparison procedures with Tukey test. The significance levels were notified as followed in the figures and tables with probability of p < 0.05.
Results
LDH activity
No cytotoxic effect on fibroblasts was observed when the cells were seeded on the Ti-F compared to the Ti coins. Only the lowest concentration of NaF (1 µg/mL) in solution caused a significant higher LDH activity in the cell culture medium compared to untreated cells (Figure 1).

Lactate dehydrogenase (LDH) activity assay. LDH activity in culture medium from HGF (a) cultured on Ti coins with (Ti-F) and without (Ti) fluoride surface modification and (b) cultured on plastic incubated with NaF (1–100 µm/mL) (b). Values represent the mean ± SD. **p < 0.01 compared to control.
Fluoride modification of titanium enhances HGF proliferation
Ti-F induced a five- to six-fold increase in the proliferation of HGF compared to control (Ti) (p < 0.05) at 1 and 3 days. In comparison, the highest dosage of NaF (100 µg/mL) dramatically reduced the HGF proliferation after 1 and 3 days, whereas 10 and 1 µg/mL of NaF halved the proliferation rate compared to untreated cells at day 3 (Figure 2).

Cell proliferation measured by [ 3 H]-Thymidine corporation in HGF cultured on fluoride surface-modified (Ti-F) coins compared to Ti (a) and in cells cultured on plastic incubated with NaF (1, 10, and 100 µg/mL) (b). Data (counts per minute (CPM)) are presented as mean ± SD. **p < 0.01 compared to control.
Fluoride modification of titanium enhances gene expression (RT-PCR) of IL-6 and OPG
A significant increase in the expression of IL-6 and OPG at days 1 and 7, and 7, respectively (p < 0.05 for all) was found in cells cultured on surface modified with fluoride compared to controls. We found no significant alteration in the expression of genes related to differentiation of fibroblasts (RANKL, COL3A1, FN1, MMP-1, TGF- β1, SOST, Integrin α2 and β3) in HGF cultures on fluoride-modified Ti coins compared to controls (Figure 3).

Effect of Ti and Ti-F surfaces on the gene expression of (a) IL-6 and (b) OPG in HGF. *Significant difference between Ti-F and Ti in ΔCT/CT. *p < 0.05.
Fluoride enhances the secretion of SOST, IL-6 and OPG from HGF
The secretion of IL-6, OPG and SOST was higher in cells cultured on Ti-F compared to controls at all time points tested (Figure 4). No differences were, however, found in the protein secretion of OC, leptin, OPN, TNF- α and FGF-23 to the cell culture medium from HGF cultured on Ti-F compared to those cultured on unmodified Ti coins (data not shown).
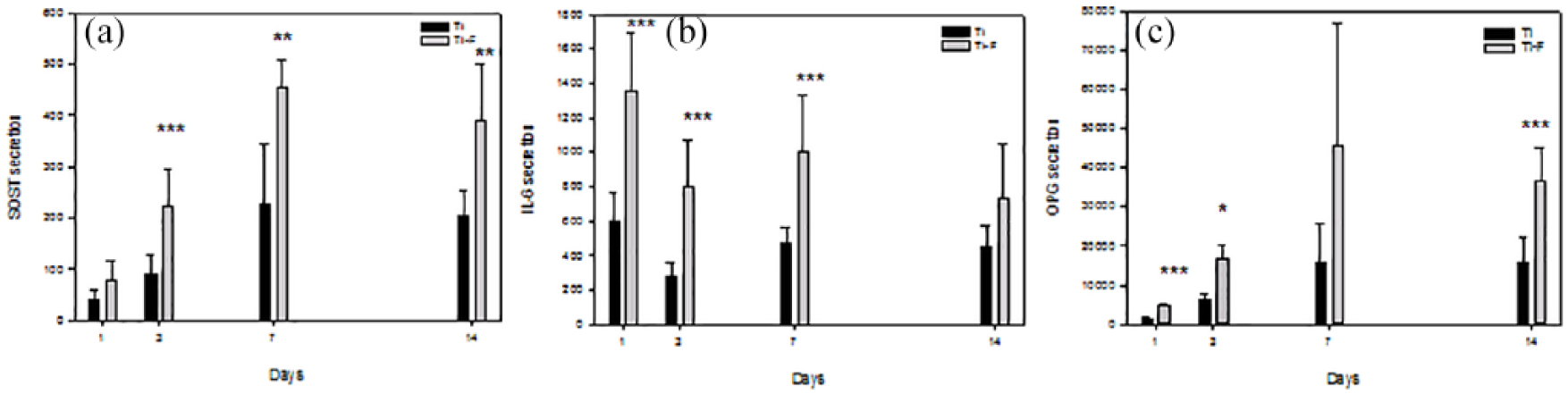
Secretion of (a) sclerostin (SOST), (b) IL-6, and (c) OPG to the cell culture media from HGF cultured on Ti-F surfaces compared to controls (Ti). *p < 0.05, **p < 0.01, ***p < 0.001.
No effect of fluoride modification on Ca2+ in cell culture medium of HGF
We did not find any statistically significant difference in the Ca2+ content in cell culture medium from cells cultured on coins with fluoride-modified surfaces compared to controls (Figure 5).

Total Ca2+ in cell culture media from human gingival fibroblast cultured on HF-modified Ti surfaces (Ti-F) compared to control (Ti). Values represent the mean ± SD.
Calcium deposition in cells
We found no significant differences in the Ca2+ deposition, measured with Alizarin red, on fluoride-modified surfaces compared to controls cultured on HGF (Figure 6).

Ca2+ deposition in cells on HF-modified Ti surfaces (Ti-F) compared to control (Ti). Values represent the mean ± SD.
Discussion
Surface-modified implants with HF are found to promote the early phase in osseointegration after the insertion of implants.1,29 Attachment of connective tissue to surface is one of many important determinants of implant success. 30 Frojd et al. 31 found that fibroblasts are directly involved with the inflammatory response, tissue remodelling, bone formation and resorption around dental implants.
In this study, we found a five- to six-fold increase in the proliferation of HGF cultured on surfaces modified with HF acid compared to controls. These results are homonymous with our previous study that showed increased cell proliferation on human primary osteoblasts cultured on the same surfaces. 18 Lamolle et al. 14 found a similar increase in growth, biocompatibility and no effect on differentiation of murine osteoblasts on machined titanium surface modified with HF.
Low levels of NaF in the cell culture medium induced a reduction of HGF cell proliferation. Our range of NaF concentrations was 1–100 µg/mL. Li et al. 32 used concentrations ranging from 1 to 5000 µM, equivalent to a range of 0.042–209.94 g/mL, and found significant increased cell proliferation of gingival periodontal ligament (PDL) by concentrations lower than 500 µM, whereas concentrations higher than 500 µM induced reduced cell proliferation and mineralization compared to controls. Li et al. explained this reduction in cell proliferation by reduced cell viability, measured by CCK-8 activity. We observed no effect of concentrations up to 100 µg/mL on the LDH activity in the cell culture medium. The samples in this in vitro study were titanium surfaces modified with 0.2% vol HF by immersion for 120 s. Immersion in HF more than 150 s could give cell death on murine osteoblast cells. 14 This surface modification with less than 1.2% fluoride concentration, described in previous study,18,33 confirmed no cytotoxic effect with no changes in the LDH activity.
In our study, we found significant increased cell proliferation, as well as the secretion and expression of IL-6, OPG and SOST in HGF on HF-modified Ti surfaces compared to untreated Ti surfaces. IL-6, an interleukin produced by several cells including gingival fibroblasts, 34 is assumed to regulate immune responses in inflamed tissues. As IL-6 presumably stimulates osteoclast formation, this factor might be of importance in the study of implant biocompatibility; 35 however, it has also been suggested that IL-6 may act directly on osteoclast formation and proliferation by suppressing osteoclast progenitor differentiation. 36
SOST has previously been found to be expressed in gingival fibroblasts and periodontal ligament cells and may play a role in periodontal regeneration. 37 In our study, we found a two to three times upregulated level of SOST in HGF cultured on HF-modified surfaces compared to untreated surfaces. According to Sutherland et al., SOST is bound to bone morphogenetic proteins (BMPs) and expressed in osteogenic cells for the anabolic bone formation. This product might modulate the activity of osteoblastic cells, 38 reduce the differ-entiation of osteoprogenitor cells 39 and enhance the osteoclastogenesis.
The combined enhanced levels in IL-6, OPG and SOST might promote the necessary osteoclast activation that is needed for the early inflammatory phase in bone healing and remodelling. 35 Cecchinato et al. 40 found a positive effect in peri-implant soft tissue response in dental implant with HF-modified surfaces and suggested that this might give a better aesthetic outcome for the implant. A soft tissue barrier with gingival attached to the abutment of the implant may improve the protection function 41 during the repair and regeneration processes of wound healing.42,43 Success rate depends on stable connections between implant surface and the surrounding tissue. 44 Our study showed that Ti-F induced an increase in both OPG mRNA expression and protein secretion from HGF compared to control; OPG has an inhibitory effect on osteoclast activation and bone resorption 45 and may therefore have an impact on bone density.45–47 The biological influence of changes in OPG expression and secretion from HGF on bone remodelling and osseointegration is dependent on the levels of RANKL. We were unable to reliably detect and quantify the levels of RANKL expression and secretion from HGF cultured on Ti and Ti-F coins.
Conclusion
HF surface modification enhanced the proliferation of HGF, whereas aqueous fluoride had the opposite effect. Our results confirm that HF-modified surface may indirectly enhance the firm attachment of implant surface to junction epithelium, soft tissue epithelium, which would give protection for underlying osseous structures making osseointegration of the dental implant possible.
Footnotes
Acknowledgements
The authors would like to acknowledge Aina-Mari Lian, from Faculty of Dentistry, University of Oslo, for valued contribution.
Declaration of conflicting interests
M.H.P., H.J.H., A.R., and J.E.E report no conflict of interest. J.E.R is a member of the COST Action CA16119 CellFit.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by Research Council of Norway (FRINATEK grant number 231530).

