Abstract
Strategies to replace retinal photoreceptors lost to damage or disease rely upon the migration of replacement cells transplanted into sub-retinal spaces. A significant obstacle to the advancement of cell transplantation for retinal repair is the limited migration of transplanted cells into host retina. In this work, we examine the adhesion and displacement responses of retinal progenitor cells on extracellular matrix substrates found in retina as well as widely used in the design and preparation of transplantable scaffolds. The data illustrate that retinal progenitor cells exhibit unique adhesive and displacement dynamics in response to poly-
Introduction
Specialized biomaterials1–3 have been used in conjunction with extracellular matrix (ECM) proteins to introduce a variety of replacement cell types4–7 into damaged and/or injured retina8,9 via transplantation therapy. The microenvironments within such transplantable biomaterials provide structural and chemical cues that influence nearly all aspects of the cells that populate them, including de/differentiation, 10 migration, 11 intercellular connectivity, 12 and long-term survival. 13 Reported advancements 14 in the survival and integration of transplanted photoreceptor progenitors have inspired dramatic innovation in contemporary transplantation strategies, including elastomeric grafts 15 and protein-coated, electrospun fibers, 16 whose properties mimic in vivo cues affecting cell fate, behavior, and response in host retina.17,18
A significant, but largely understudied, group of physical cues guiding biomaterial design for retinal transplantation are those which support and/or influence the migratory behavior of transplantable cells. This is surprising because the migration of transplanted cells is fundamental to cellular and synaptic integration within host retina. As illustrated in Figure 1, cells transplanted into the sub-retinal space must exit the biomaterial and navigate the complex retinal architecture via the outer nuclear layer in order to connect with neuronal targets of the host.8,19 Few studies have explored how properties of biomaterials affect outward cell migration into host retinal laminae. Such migration processes are complex because they include movement of individual cells as well as the collective migration of neuroclusters. Collective behaviors of transplanted cells are particularly important because donor cells are explicitly derived to mimic earlier developmental states, where cells largely interact as neuroclusters.20,21 The mechanisms of collective cell adhesion and migration are also acutely affected by factors such as population density and interstitial interactions, 20 both of which are routinely altered in order to increase the integration of transplanted cells.22,23
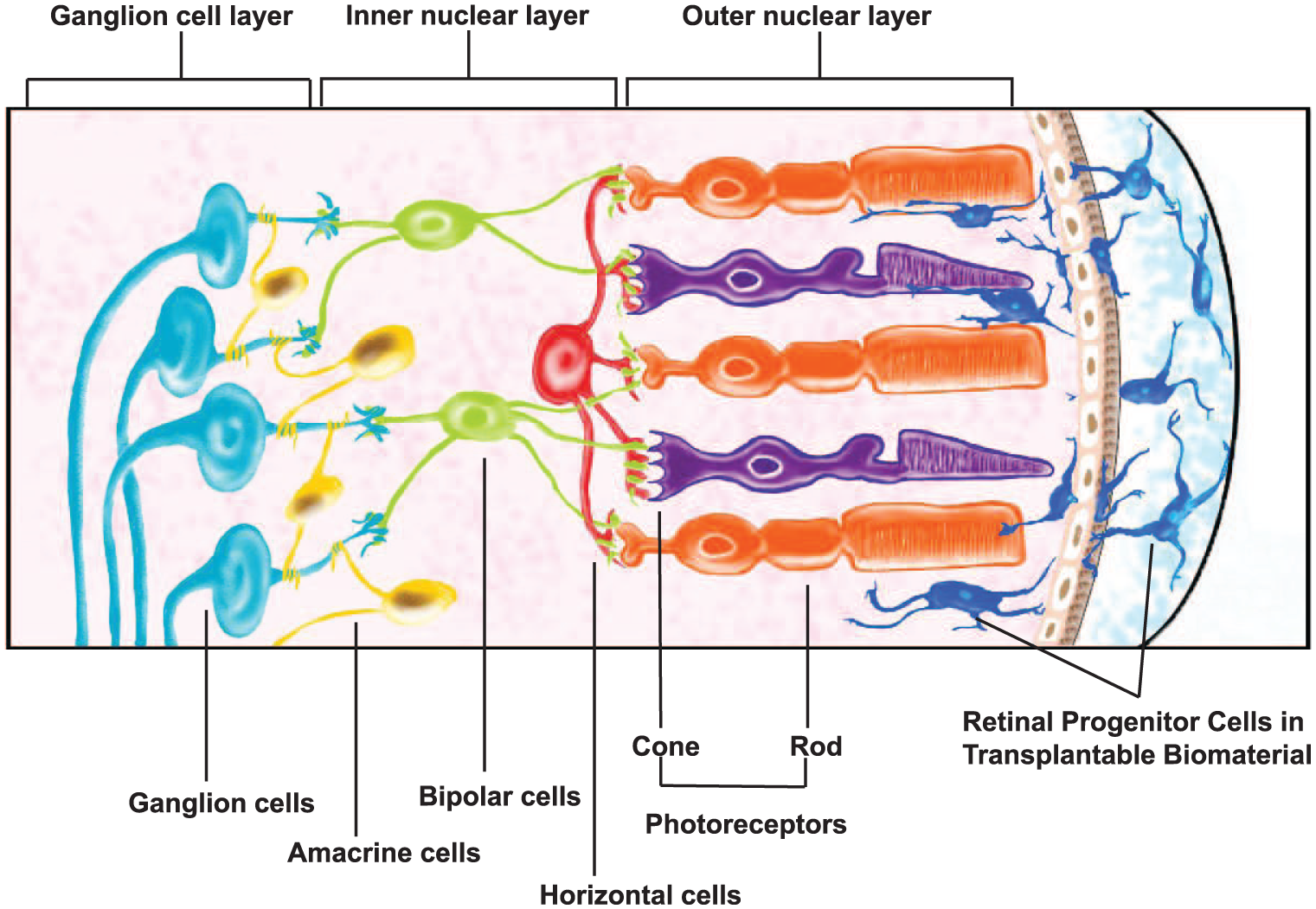
Cell-seeded biomaterials in photoreceptor transplantation. Retinal progenitor cells (RPCs) are embedded within biomaterials and transplanted into the sub-retinal space in the eye posterior. Viable cells exit the biomaterial and navigate into and within the host outer nuclear layer to achieve synaptic integration with neuronal targets.
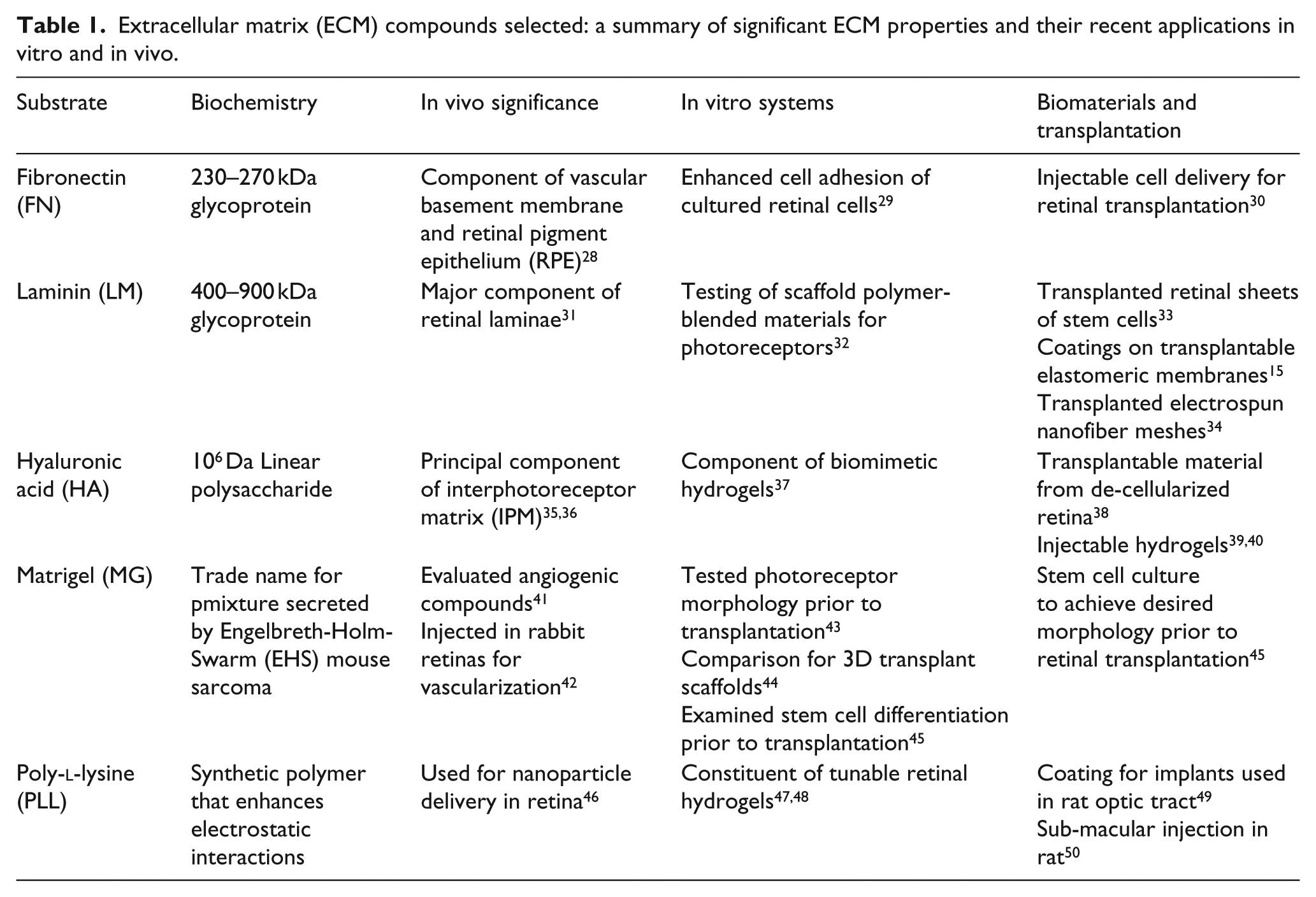
Our group has previously evaluated the collective11,24 and individual25–27 migration of retinal progenitor cells (RPCs) and photoreceptor precursor cells (PPCs) toward ligand gradients derived from light-damaged retina. In this article, we quantify the clustering, adhesion, and displacement of RPCs in response to four ECM substrates present in developing and mature retina, and/or used extensively in the development and testing of transplantable biomaterials as described in Table 1: fibronectin (FN),28–30 laminin (LM),15,31–34 hyaluronic acid (HA),35–40 and Matrigel (MG).41–45,51 RPC behavior upon these substrates was also compared against poly-
Extracellular matrix (ECM) compounds selected: a summary of significant ECM properties and their recent applications in vitro and in vivo.
Methods and materials
Cell isolation and culture
Mouse RPCs were isolated from dissociated retinas of post-natal (P4) C57BL/6J mice, homozygous for expression of enhanced green fluorescent protein under control of the beta actin promoter (EGFP+/+) (gift from Dr Young, Harvard University
52
). RPCs were expanded and maintained at 37°C in a 5% CO2 incubator in neurobasal complete medium (Invitrogen-Gibco, Rockville, MD) containing 2 mM
ECM substrates
Mouse laminin (LM, Sigma-Aldrich, St. Louis, MO) and human fibronectin (FN, Thermo fischer, Waltham, MA) were dissolved and diluted to 10 μg/mL and 100 μg/mL, respectively, in phosphate-buffered saline (PBS). Growth factor-reduced MG (BD Biosciences, Franklin Lakes, NJ) was diluted to 100 μg/mL in PBS. HA (Alfa Aesar, Ward Hill, MA) was added to PBS at 30 mg/mL with thorough sonication and allowed to dissolve for 24 h at room temperature. PLL 53 was used as a control substrate. Wells were coated with a 15 ng/mL solution in PBS at 5°C overnight and left to air dry in a laminar flow hood for 1 h prior to study. Surfaces of 12-well culture plates (VWR International, Bridgeport, NJ) were functionalized with a 1 mL volume of LM, FN, or MG per well and 0.5 mL of HA for 1 h. All wells were washed afterward with 1 mL of PBS. Substrates were functionalized at room temperature (25°C) for all solutions except LM, which was performed on ice as per vendor protocols. All wells were used on the day of surface functionalization.
Morphology and adhesion density of individual RPCs
RPCs were plated at (1) low cell density of 104 cells/well; (2) moderate, but sub-confluent, density of 105 cells/well; and (3) high cell density of 106 cells/well that approached confluence in 12 well plates (~1 mL/well). The size and morphology of individually adhered cells were evaluated using the cell shape index, CSI, defined in equation (1) as a dimensionless parameter that quantitatively represents cell asymmetry54–56

where AS represents the surface area of individually adhered cells and P represents the cell perimeter. Values of CSI range from 1.0 for an idealized circular shape to 0.0 for cells that exhibit a perfectly linear elongation, as shown in the schematic of Figure 2. In this study, individual cells (i.e., not part of a neurocluster) were defined as those whose contact with neighboring cells was limited to either (1) extended, continuous interfacial contact with a single cell along the plasma membrane (e.g., daughter cells following mitosis) or (2) discrete point contacts via processes or extensions with one or more other cells. In addition, the average cell density of individually adhered cells was quantitatively represented by the cell adhesion density, πADH, defined in equation (2)

where ASC denotes the area of individually adhered cells within a substrate region of interest, i, and ASA, i denotes the surface area of that region of interest.
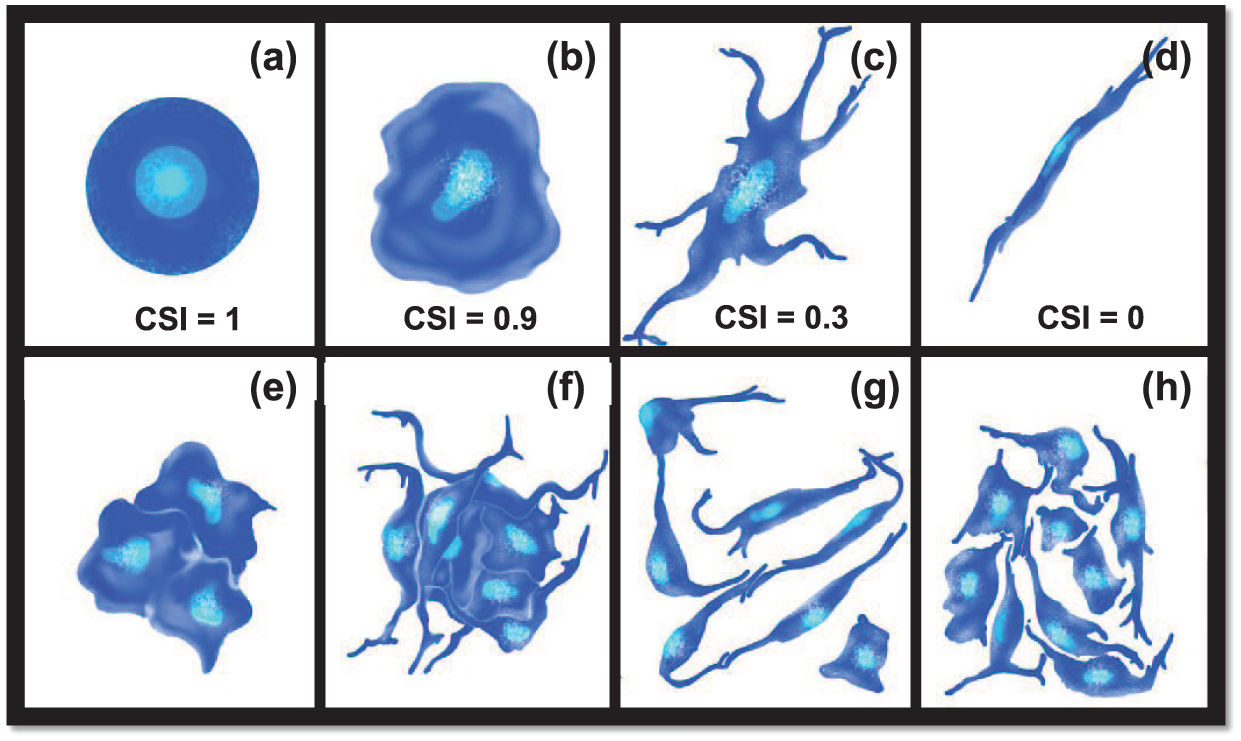
Morphology of adhered cells. Schematic representation of different cell morphologies with corresponding values of cell shape index (CSI): (a) idealized circular cell (CSI = 1.0), (b) typical rounded cell (CSI = 0.9), (c) typical elongated cell with process extensions (CSI = 0.3), and (d) idealized linearly elongated cell (CSI = 0). Cell clusters were defined as (e, f) groups of three or more cells in direct and continuous interfacial contact along common plasma membranes. (g, h) Cells in direct contact with neighbors along only one interface or extension were not evaluated as clusters.
Mean size and adhesion ratio of retinal neuroclusters
Retinal neuroclusters were defined as groups of three or more cells with continuous and extended interfacial contact along their plasma membranes,
24
as described per Figure 2. The mean size of each neurocluster,

where ANC is the projected surface area of adhered neuroclusters within a substrate region and ASC represents the total surface area of singly adhered cells. In this way, RADH denotes the percentage of total cell-adhered surfaces that contain neuroclusters.
Expression of adhesion receptors
Expression levels of four genes encoding adhesion receptors were measured using quantitative polymerase chain reaction (qPCR) for integrin α3, integrin α7, integrin β3, and the adhesion molecule CD44 with primers shown in Table 2. Primer specificity was verified using Basic Local Alignment Search Tool (BLAST), which confirmed the selected forward and reverse primers listed. RNA was isolated from cells using Trizol (Sigma-Aldrich, St. Louis, MO) and measured photometrically. First-strand complementary DNA (cDNA) synthesis was performed using random hexamers followed by amplification with specific primers on a Rotor Gene 6000 thermal cycler (Qiagen, Inc., Germantown, MD) as per manufacturer instructions. The following amplification conditions were used: 95°C denaturation for 10 min, followed by 40 cycles of 95°C for 15 s and 60°C for 1 min, followed by a hold at 4°C. Raw data were analyzed with Software version 2.2.3 (Qiagen Inc.) to determine the cycle threshold (CT) setting for assigning baseline and threshold CT determination. Relative expression (RE) of the sample gene was calculated using the conventional ΔΔCT method.57–59
Gene regulation examined via quantitative polymerase chain reaction (qPCR): a listing of the genes encoding cell and surface adhesion molecules studied, alongside primer sequence, size in base pairs (bp), and accession number.

Adhesion and displacement within microfluidic environments
A previously described microfluidic system from our group, the µLane, 56 was used in this study to evaluate RPC adhesion and displacement upon different ECM protein substrates. As shown in Figure 3, µLane systems comprised channels of 150-µm diameter and 12-mm length, as fabricated via conventional lithography, PDMS (poly(di)methyl-siloxane) micromolding, and glass bonding. The µLane inner surfaces were functionalized with LM, FN, HA, or MG at the concentrations described earlier. Solutions of RPCs at three different seeding densities of 104, 105, or 106 cells/mL were then inserted into the µLane interstitial spaces. Cells within the µLane were imaged every 24 h for 3 days using phase-contrast microscopy.

The µLane system. (a) Projection images of µLane construction, comprising two layers of PDMS polymer ((1), (2): poly-dimethyl siloxane) ozone-bonded upon a (3) glass coverslip. The µLane systems has a channel length of l = 1.2 cm, reservoir volume of V = 0.5 mL, and PDMS thickness of th = 0.75 cm. (b) Image of closed µLane system with highlighted microchannel. Scale = 1 mm.
Cell imaging and analysis
Phase-contrast images of adhered RPCs and neuroclusters were evaluated using a 20× objective lens (Nikon TE2500 microscope, Morrell Instruments, NY). Images from well plates were gathered from five non-overlapping fields via a conventional checkerboard pattern, 55 while images from µLane systems were captured from five channel and reservoir areas. Size and morphology of individual RPC and cell aggregates were quantitated using ImageJ shareware (NIH, Bethesda, MD) as described previously by our group. 24
Statistical analysis
Differences among adherent cell groups were evaluated using one-way analysis of variance (ANOVA) and post hoc test (Tukey). A one-way ANOVA test at the 95% confidence interval assessed statistical significance across biomaterial substrates and cell seeding densities, while the post hoc test (Tukey) determined the disparity among different groups. Calculated p-values < 0.05 were considered statistically significant and denoted by an asterisk. The numbers of experiments performed for each test were at least 10 per condition.
Results
Adhesion of RPCs
Adhered RPCs were evaluated 2 days post-seeding upon substrate surfaces of PLL, FN, LM, HA, and MG, as shown in Figure 4. RPCs adhered to all substrates in different combinations of single cells and neuroclusters (defined in Figure 2), each with varying morphology and mean size. No significant differences in viable cell numbers were observed among RPCs adhered to substrates, individually or within neuroclusters, as measured via MTT assays and conventional DAPI staining (data not shown).

Aggregation and adhesion of retinal progenitor cells (RPCs). Images of RPCs at a cell density of 105 cells/mL adhered as individual cells and neuroclusters upon surfaces of: (a) poly-
Retinal progenitor morphology (CSI) and adhesion density (ΠADH)
Cells that did not adhere as part of neuroclusters exhibited varying cell morphology and density with biomaterial substrate. Changes in cell morphology were measured via the CSI shown in Figure 5(a). As seen, only morphology changes measured upon LM were statistically significant (p < 0.05), as RPCs became increasingly elongated with higher seeding density. RPCs upon LM demonstrated the only statistically significant elongation, as average values of CSI upon LM decreased dramatically from CSI = 0.67 at low seeding density (104 cells/mL) to CSI = 0.33 for high seeding density (106 cells/mL). There was no statistical significance in CSI values among and across the other biomaterial groups (p > 0.5).

Metrics of individual cell adhesion. The (a) average cell shape index (CSI) of retinal progenitor cells (RPCs) adhered as individual cells upon poly-
In contrast, the density of cells adhered as individuals rather than in clusters, ΠADH, varied depending on both the biomaterial substrate and the initial number of cells plated. Changes in ΠADH were statistically significant across cell seeding density for LM, HA, and MG, as per Figure 5(b). Average ΠADH values were greatest upon LM and HA at moderate seeding density (105/mL), but steadily increased upon MG. By contrast, increases in ΠADH were most pronounced upon PLL and FN, where at high seeding density (106/mL) the largest ΠADH value was measured upon PLL, and a complete RPC monolayer was observed upon FN. Mean values of calculated parameters are summarized in Table 3.
Summary of parameters measured for adhered retinal progenitor cells (RPCs).

Average values of individual cell shape index (CSI) and single cell adhesion density (ΠADH) summarized alongside values of mean cluster size,
Size
and extent (RADH) of RPC neuroclusters
Neuroclusters in Figure 4 exhibited a range of mean sizes,
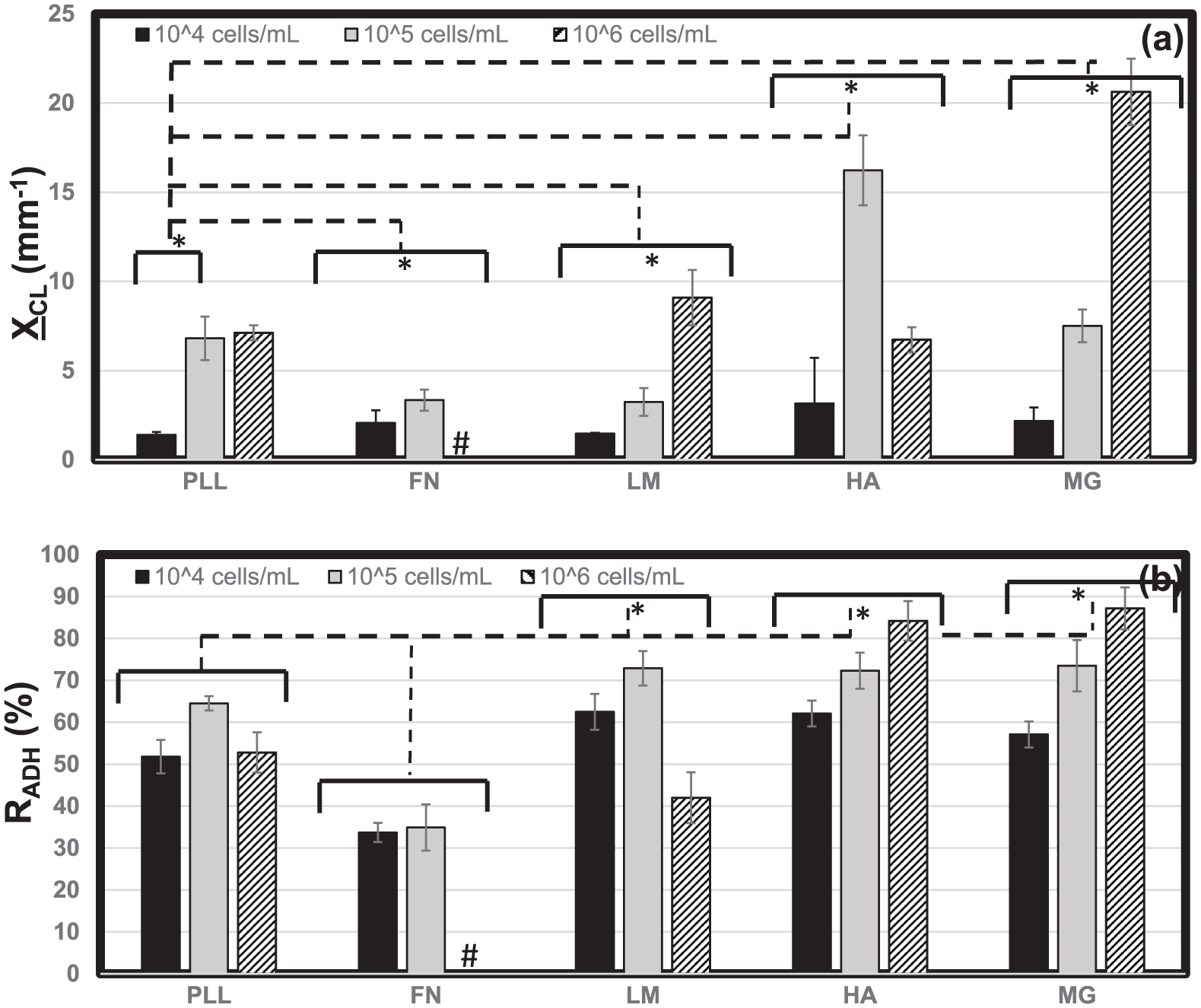
Metrics of adhered neuroclusters. The projected surface area of adhered retinal neuroclusters was measured to determine (a) mean cluster size, XCL, upon poly-
The percentage of cell-occupied area containing neuroclusters (vs individually adhered cells) also varied with both biomaterial substrate and cell seeding density, as measured by the adhesion ratio, RADH, in Figure 6(b). Values of average RADH increased with cell seeding density upon FN, HA, and MG and decreased with seeding density upon PLL and LM. The highest values of RADH were measured upon both HA and MG at the highest seeding densities (106/mL), where 85% of adhered surface areas contained neuroclusters. As previously noted, RPCs formed a complete monolayer on FN at high seeding density rather than discrete neuroclusters. Conversely, the lowest adhesion ratio of RADH = 31% was measured upon FN at low cell seeding density (104/mL), where less than a third of cells adhered as part of neuroclusters. Furthermore, RPCs within adherent neuroclusters exhibited similar morphologies upon all biomaterials, with an average CSI = 0.82 ± 0.4 that was significantly higher (indicative of more rounded cells) than that measured for any individually adhered cell group (Figure 5(a)). Mean values of calculated parameters are summarized in Table 3.
Expression of adhesion receptors
The observed changes in the adhesive behavior of RPCs were evaluated in relation to expression of a panel of cell surface adhesion molecules known to serve as receptors for one or more of the ECM materials used as substrates in this study: integrin α3, integrin α7, integrin β3, and CD44. As noted in Table 4, integrin α360–65 and integrin α766–69 bind to FN and LM, CD4439,75–78 is the receptor for HA, and integrin β370–74 is expressed by cells of the retinal pigment epithelium (RPE) and highly implicated in retinal angiogenesis. We observed that RPCs seeded onto substrates identified as ligands expressed somewhat higher levels of most receptors than when seeded onto PLL. Figure 7 shows that integrin β3 was up-regulated by FN and LM, but down-regulated by HA and MG, and CD44 was down-regulated by FN and LM, but up-regulated by HA and MG as per the ΔΔCT method.
Summary of adhesion molecules examined alongside their applications to neural cells in the retina.

LM: laminin; FN: fibronectin; RPC: retinal progenitor cell; RPE: retinal pigment epithelium; HA: hyaluronic acid.

Differences in gene expression of adhered RPCs. Fold change in regulation of the genes ITGA3, ITGA7, ITGB3, and CD44 that encode (a) integrin α3, integrin α7, integrin β3, and CD44 upon adhesion with fibronectin (FN), laminin (LM), hyaluronic acid (HA), and Matrigel (MG). (b) Schematic representation of up- and down-regulation of encoding genes as measured via the ΔΔCT method.
Attachment and displacement of RPCs
To evaluate RPC adhesion and displacement within the microscale of sub-retinal spaces, cell suspensions were seeded into µLane devices previously functionalized with FN, LM, HA, or MG. RPCs were seen to adhere as neuroclusters, individual cells, or combinations of the two, depending on the substrate used. As shown in Figure 8, RPCs did not aggregate into neuroclusters within µLane devices functionalized with FN or MG, but rather adhered solely as individual cells. By contrast, RPCs were observed to adhere within µLane systems functionalized with LM and HA as both single cells and neuroclusters. As summarized in Table 5, neuroclusters within µLane systems functionalized with LM and HA exhibited net displacements of 18–26 µm (i.e., two diameters of individually adhered cells), while that of individually adhered RPC within LM- and HA-treated µLane substrates was 3–6 µm, or less than one cell diameter. Individual RPCs adhered upon FN- and MG-treated devices underwent net displacements of 4–9 µm.

Displacement of adhered retinal progenitor cells (RPCs) within the µLane microfluidic system. Images describing attachment of RPC cells and/or neuroclusters within (a) µLane systems whose inner surfaces were functionalized with (b) fibronectin (FN), (c) laminin (LM), (d) hyaluronic acid (HA), and (e) Matrigel (MG). Scale bar = 75 µm.
Retinal progenitor adhesion and displacement within µLane system.

Discussion
This study examined the ability of biomaterial substrates used in transplantable biomaterials to alter RPC adhesion, clustering, and displacement. The balance between cell adhesion and motility is a critical consideration in the design of retinal scaffolds, as insufficient adhesion can result in cell death79–81 while overly strong adhesion may adversely affect RPC morphology and differentiation or prevent cell migration needed to re-establish functional connectivity.82–84 Moreover, since RPCs migrate as both clusters and individual cells during retinogenesis, 85 it is also important to know the extent to which retinal scaffolds can mimic this behavior.
This study defined two parameters, adhesion ratio, RADH, and adhesion density, ΠADH, which quantitatively represented the ratio of individually adhered cells to adhered neuroclusters and the portion of total substrate area covered by cells, respectively. Results illustrate that both parameters differed depending on both ECM substrate and seeding density used. Of note, increasing the number of RPCs seeded on a substrate generally resulted in increased numbers of single cells (Figure 5) but did not necessarily yield proportionately larger neuroclusters. Rather, larger seeding densities produced (1) larger numbers of moderately sized neuroclusters upon HA and LM, (2) elicited no change in neurocluster size or adhesion upon PLL, and (3) produced larger numbers of larger-sized neuroclusters upon MG (Figure 6). Interestingly, on most substrates, formation of neuroclusters, even at the highest density examined, resulted in substantial areas of open space not covered by either clusters or single cells. This suggests that the interactions between cells and substrate were notably weaker than those between cells. In contrast, cells plated on FN at high density formed a complete monolayer, indicating that cell–substrate interaction was predominant. The implications of these differences for RPC adhesion and migration, either as single cells or clusters, have yet to be established, although the ability to characterize clustering behavior, quantitatively, will facilitate this process.
Expression of adhesion receptors as assessed by polymerase chain reaction (PCR) was altered when RPCs were cultured on their respective ligands (Figure 7). However, the substrate-dependent differences in adhesion receptor expression were modest. Consequently, while the receptors examined are likely to contribute to changes in cell shape, formation of neuroclusters, or the displacement of either individual cells or clusters, we could not establish a clear relationship between those processes and receptor expression levels. That expression of adhesion receptors, such as integrins, can be modulated in response to the amount of available ligand (substrate) in ECM has been well established. 86 However, differences in gene expression may underestimate changes in the number and organization of adhesion receptors on cell surfaces. In addition, averaging techniques like PCR may not be sensitive enough to detect subtle expression changes in mixed populations such as RPCs present both as clustered and individually adhered cells. Furthermore, it has been reported that cell adhesion even to single-component extracellular matrices, such as LM or FN, may involve multiple integrin family members 87 while CD44 is capable of binding both HA and collagen.75,88 Hence, adhesion to the various ECM substrates in this study may have activated more than one receptor, including some outside the limited group whose expression was examined. Future study will examine expression of molecules able to mediate cell–cell interactions that aid formation and stabilization of cell clusters, for example, cadherins.89–91
The use of the µLane system to evaluate the microscale displacement of mixed (individual and clustered) RPCs (Figure 2) within microfluidic environments represents a novel application of technology little used in retinal cell replacement studies despite widely reported usefulness in a variety of cell-based systems87,92 (Figure 3). Our system readily enables quantitative study of cell migration over sub-retinal distances <50 µm as well as distances >1000 µm for chemotaxis driven by extrinsic fields, as shown previously by our group.11,26,27 Our studies demonstrated that RPCs formed neuroclusters and underwent displacement both as individual cells and as clusters within the interstitial spaces of the µLane functionalized with LM or HA, although FN- and MG-treated substrates exhibited single-cell adhesion, only (Figure 8). Why RPCs did not form neuroclusters on FN and MG in the µLane system, as they did in bulk culture, is so far unclear, but suggests differences in substrate composition or organization between the microfluidic and bulk microenvironments that warrant further investigation. In any case, the absence of clustering suggests a predominance of cell–matrix over cell–cell interactions (Table 3), as previously noted.
Finally, the displacements observed by RPCs as both single cells and clusters on LM and HA were clearly measurable and distinct. We also note that those displacements were observed in the absence of applied stimuli, indicating that chemotactic or chemokinetic responses of RPCs to chemical and physical signals are readily amenable to study. Why the observed displacement of neuroclusters was greater than that of single cells is not known (Table 3). While quantitative study of collective migration is beyond the scope of this article, the size and composition of neuroclusters is known to affect migration modalities20,93,94 which may directly influence cell distances traveled. Furthermore, different ECM substrates may preferentially regulate cell adhesion molecules that maintain the cohesion of motile clusters 93 and whose interactions may outweigh the cell–matrix adhesion studied here. In any event, our results suggest that, among the substrates examined, LM may be preferred for retinal cell transplantation. LM is present in mature and developing retina, RPCs express cell surface LM receptors, and LM mediates adhesion both as single cells and as neuroclusters. These features suggest that LM matrices may enable tuning to achieve both collective and single cell migration with desirable displacement distances. Future study will benefit from using known physical and chemical cues of retinal injury and/or neurodegeneration to directly evaluate RPC migratory behavior into injured and/or damaged laminae.
Conclusion
Composites of ECM substrates and biomaterials have evolved to support increasing numbers of cells, extend cell survival, and promote cell adhesion as strategies to improve outcomes of transplantation therapy. This study evaluated the abilities of selected substrates to regulate RPC adhesion, shape, and cell displacement as individual cells and as aggregates to design and/or enhance transplantable biomaterials. Results illustrate that combinations of clustered and individual cells on different substrates led to different behaviors that may influence the ability of transplanted, or replacement, cells to migrate effectively and achieve connectivity with host cells in retinal laminae. These findings suggest that by characterizing and/or manipulating the migratory behaviors of replacement cells, transplantation strategies can be enriched to improve cell integration.
Footnotes
Acknowledgements
The authors thank Jason Mighty, Onyekwere Onwumere, and Stephanie Zhang for their contributions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institutes of Health (R21 EY026752, SC3 GM113782) and the National Science Foundation (CBET 0939511).
