Abstract
The purpose of this study was to compare, by means of in vitro cultivation technique, five marketed brands of wound covers used in the treatment of burns and other skin defects (Biobrane®, Suprathel®, Veloderm®, Xe-Derma®, and Xenoderm®) for their ability to stimulate the keratinocyte growth, stratification, and differentiation. In three independent experiments, human keratinocytes were grown on the tested covers in organotypic cultures by the 3T3 feeder layer technique. Vertical paraffin sections of the wound covers with keratinocytes were processed using hematoxylin–eosin staining and immunostaining for involucrin. Keratinocyte populations on the dressings were assessed for (1) number of keratinocyte strata (primary variable), (2) quantitative growth, (3) thickness of the keratinocyte layer, and (4) cell differentiation. The Xe-Derma wound cover provided the best support to keratinocyte proliferation and stratification, with the number of keratinocyte strata significantly (p < 0.05) higher in comparison to all products studied, except Xenoderm. However, in contrast to Xe-Derma, Xenoderm did not significantly differ from the other dressings. The results of this in vitro study show that the brands based on porcine dermal matrix possess the strongest effect on keratinocyte proliferation and stratification. The distinctive position of Xe-Derma may be related to its composition, where natural dermal fibers form a smooth surface, similar to the basement membrane. Furthermore, the results indicate that in vitro evaluation of effects on epithelial growth may accelerate the development of new bio-engineering-based wound covers.
Introduction
The limited amount of donor sites for skin autotransplantation is a constant problem in the therapy of extensively burned patients. While full-thickness burns are resurfaced with split-thickness skin autografts, deep dermal burns are usually covered with biological or synthetic covers (dressings). Wound covers work as temporary substitutes. If the wound does not heal spontaneously, they have to be replaced with the patient’s own skin.
The ideal wound cover should be elastic, be optimally adhesive, be nontoxic, prevent external infection, and copy the surface of underlying structures. In addition, it should, like native extracellular matrix (ECM), support growth of both dermal fibroblasts and overlying keratinocytes, leading to skin restoration without contraction. 1 The success of healing is highly dependent on the properties of the wound cover, especially its ability to support keratinocyte proliferation and differentiation. 1 Only a few sophisticated biomaterials allow sufficient cell adhesion, which is the prerequisite of cell proliferation. 2 A host of modern wound covers have recently become available and are commonly used in clinical practice. However, the properties and applicability of the wound covers differ significantly, being primarily influenced by the materials used and the manufacturing process of the dressing material. Acellular wound covers (i.e. free of living cells) may be produced from (1) biological materials, (2) synthetic (man-made) materials, or (3) composite materials (containing two or more components either biological or synthetic). 3 Composite and synthetic materials allow better control over the composition of the dressing and the production process; yet, there is a higher risk of cytotoxicity or bioincompatibility. 4 On the other hand, the biologic materials are likely to be more similar to native ECM, allowing more natural new dermis to be formed.
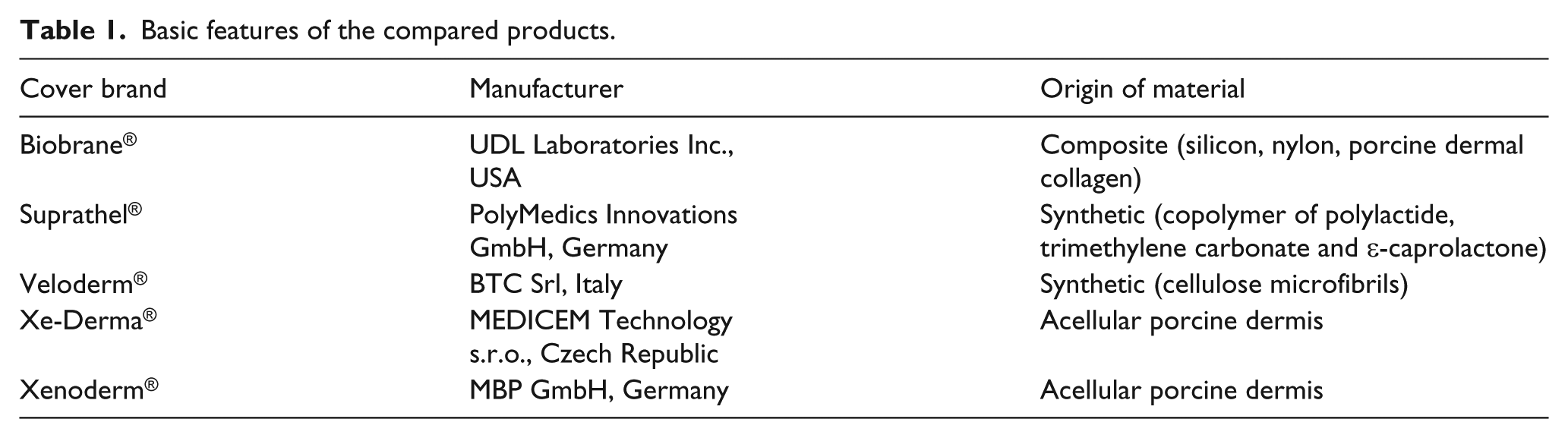
The aim of our study was to evaluate whether the material composition and manufacturing procedure may influence the formation of neoepidermis, which is one of the crucial factors of the wound healing. Therefore, we have compared five marketed biological or biosynthetic wound covers (Table 1) of biologic (Xe-Derma®, Xenoderm®), biosynthetic (Veloderm®), synthetic (Suprathel®), or composite (Biobrane®) origin concerning their ability to stimulate the keratinocyte growth, stratification, and differentiation in vitro. The common clinical feature of all compared dressings is either spontaneous detachment without the need for changing the cover (Biobrane, Suprathel, Xe-Derma) or smooth painless removal (Veloderm, Xenoderm), both preventing further traumatization once applied. The common biomechanical feature of all these compared dressings is the possibility to grow cells on their surface.
Basic features of the compared products.
The comparison of growth and stratification was based on histological evaluation of the structure of epidermis (keratinocyte layers) grown on the cover surface at the medium–air interface (organotypic culture system). 5 In addition, we assessed the expression and distribution of involucrin—a keratinocyte differentiation marker, typically expressed in the terminally differentiated cells in the granular and horny layers of normal epidermis.
Materials and methods
Design of the study
This was an exploratory, comparative study, employing blinding procedures during both the cultivation process and evaluation of images. To avoid conclusions based on accidental factors, the study was performed as a series of three successive independent experimental waves, each obeying the same procedures. The study obtained the approval of the Ethics Committee of the 3rd Faculty of Medicine, Charles University, Prague.
Keratinocyte cultivation
Human primary keratinocytes were obtained from redundant skin of one 24-year-old male donor undergoing plastic surgery. The samples of wound dressings sized 3 cm 2 were placed into 60-mm tissue culture dishes filled with a standard culture medium (Hank’s Minimum Essential Medium,H-MEM, enriched with non-essential amino acids, 0.12 g/L sodium pyruvate, 1 g/L NaHCO3, 10% bovine serum) 6 and kept prior to cultivation overnight at 37°C. Keratinocytes were cultured by the 3T3 feeder layer technique.6–9 Briefly, lethally irradiated NIH-3T3 cells, used as a feeder layer, were seeded to the tissue culture flask at a concentration of 2.5 × 104 cm−2. The keratinocytes were grown on the 3T3 feeder cells in the keratinocyte medium (standard culture medium enriched with 2% fetal bovine serum, hydrocortisone 0.5 µg/mL, cholera toxin 10−10 M and epidermal growth factor (EGF) 5 ng/mL (all Sigma–Aldrich, USA), and insulin 0.12 U/mL (Novo Nordisk, Denmark)) to subconfluence. Keratinocytes in the second passage were seeded at a concentration of 5 × 104 cells/cm2 on the wound dressings containing 3T3 cells and cultured at 37°C in 3.5% CO2 atmosphere. After reaching confluency, the wound dressing was lifted to the air–medium interface on a stainless steel grid covered with two layers of sterile gauze and subsequently cultured for the next 7 days.6,9 Critical manipulations with the cell cultures (addition of feeder cells, lifting of the dressings with cell layers to the air–liquid interface, medium changing) were performed by laboratory staff who did not know the identity of the products.
Histology
Specimens of wound covers with cultured keratinocytes were fixed in 4% buffered formaldehyde and processed by routine histological procedure. Paraffin sections were mounted on glass histological slides and used for hematoxylin–eosin staining and immunostaining for involucrin (Novocastra, UK).9,10
Assessment of keratinocyte layer characteristics
Narrative evaluation of images based on the most representative set
On the basis of global impression, the most representative image out of all three experimental waves was selected for each of the wound covers. A narrative description of the respective images was then provided, integrating both hematoxylin–eosin staining and immunostaining for involucrin. This assessment was focused on evaluation of the number of cell layers and the degree of cell differentiation.
2. Assessment of quantitative variables using the “best” image set
To support the validity of the evaluations, all quantitative assessments were performed separately for each of the three experimental waves and subsequently averaged. First, for each experiment and dressing, the “best” hematoxylin–eosin-stained image (showing the thickest continuous keratinocyte layer) was selected. All images were printed using the same scale. Each of the selected images was then split to nine adjacent columns (Figure 1). Within each column, three variables were assessed: (1) the number of keratinocyte strata, as a measure of the stratification level; (2) the number of keratinocyte nuclei, as a measure of cell proliferation; and (3) the thickness of the keratinocyte layer, as a measure of the overall cell mass of neoepidermis. The latter two assessments were performed separately by two independent raters and then averaged.

Splitting the enlarged picture of a paraffin section stained by hematoxylin and eosin to nine equivalent columns for assessment of keratinocyte layer parameters. The columns allowed easy quantification of keratinocyte strata, number of keratinocyte nuclei, and thickness of the keratinocyte layer. The sum over all columns yielded a value valid for the whole section.
Statistical evaluation
The data of the primary variable, the number of strata, were submitted to statistical evaluation mutually comparing data of all pairs of covers. To prevent the effect of simultaneous statistical inference, the Bonferroni adjustment of p values was applied.
Results
A narrative description of the representative images
The cultivation on Biobrane generated no more than one stratum of keratinocytes (Figure 2). The sections were extremely fragile, preventing cohesion of the cell layer. The immunohistochemical staining was not successful.

Keratinocytes cultured in organotypic culture system on different wound dressings. Keratinocytes culture was kept submerged for 1 week, then lifted to the air–medium interface and cultured for another week. For each dressing, a representative picture was selected. Left: Hematoxylin and eosin staining. Right: Involucrin immunostaining. A reference picture of normal skin was added (bar: 50 µm).
Suprathel allowed formation of one to three layers of keratinocytes, present in the form of cuboidal cells in the basal layer, and mixture of cells and cell exudate in the second and third layers. Staining for involucrin showed that all cells were terminally differentiated (positive for involucrin, inv+).
Veloderm allowed formation of one to three layers of keratinocytes. Cells in the basal layer were negative for involucrin (inv−). Wherever the second or second and third layers were formed, the respective cells were terminally differentiated (inv+).
The keratinocytes cultured on Xe-Derma lead to a multilayer structure with up to 12 strata, differentiated in the epidermis-like manner. Similarly as in the normal skin, the lower one to three layers were negative for involucrin (inv−), while the suprabasal layers were positive (inv+).
Keratinocytes grown on Xenoderm formed three to six layers with epidermis-like stratification, with some keratinocyte “nests” in the dermal matrix, originating from the migrating keratinocytes. Staining for involucrin showed rather early terminal cell differentiation.
Results of quantification of the “best” images
Tables 2–5 show the results of quantification, averaging all three waves of the experiment.
Results of evaluation of images for the count of keratinocyte strata.

Statistical evaluation of differences in number of keratinocyte strata.

T-tests with Bonferroni adjustment for multiple testing.
Results of evaluation of images for the keratinocyte layer thickness.

The Biobrane® specimen data were not included due to the dressing layer fragility and too small a keratinocyte population.
Results of evaluation of images for the count of keratinocyte nuclei.

The Biobrane® specimen data were not included due to the dressing layer fragility and too small a keratinocyte population.
The Biobrane specimens of Wave 3 did not allow reliable evaluation; hence, some data on Biobrane are only tentative and were not entered in the tables. The keratinocytes hardly formed one stratum, which is 79% less than the mean number of strata calculated for all covers and experiments. The count of the keratinocyte nuclei per column was about 0.3, which is 97% less than the mean of all covers. Furthermore, the thickness of the keratinocyte layer was 10.4 µm, which is 64% less than the mean thickness calculated from the values of all substitutes.
The counting of keratinocyte strata of Suprathel and Veloderm showed the average number of 2.1 and 2.2, respectively (36% and 33% less than the mean of all covers). The counts of nuclei per image column on Suprathel and Veloderm samples were 6.3 and 6.2, respectively (38% and 39% less than the mean of all covers). The thickness of the keratinocyte layer was 12.6 and 13.9 µm, respectively, which is 56% and 55% less than the mean thickness calculated from the values of all dressings.
On average, cultivation on Xe-Derma resulted in 7.3 keratinocyte strata (126% above the mean of all covers) and reached the number of 18.1 keratinocyte nuclei per image column (78% above the mean of all covers). The thickness of the keratinocyte layer was 59.4 µm, which exceeds the mean thickness calculated from the values of all dressings by 106%.
Application of Xenoderm led to development of the average number of 3.9 keratinocyte strata (21% above the mean of all wound covers) and 10.1 keratinocyte nuclei per image column (1% less than the mean of all dressings). The thickness of the keratinocyte layer was 30.5 µm, which exceeds the mean thickness calculated from the values of all dressings by 6%.
Statistical evaluation of the primary variable, number of strata (Table 2), revealed that Xe-Derma formed significantly (p < 0.05) more strata than any other cover, except Xenoderm. However, in contrast to Xe-Derma, Xenoderm did not show any statistically significant difference from any cover studied.
As a convenient measure of each dressing’s relative position, the results of the respective dressings in terms of a percent difference from the average of all dressings in the experiment were calculated; the results are presented in Figure 3.

Results of the quantified variables, expressed as the percentage of difference between the value obtained for the respective covers and the mean value over all covers.
Discussion
An essential prerequisite for the proliferation of skin cells is cell attachment. 2 It is achieved by the presence of collagen 11 or other material showing similar adhesiveness. 5 Stratification and sufficient differentiation appear only on materials with biological activity, for example, acellular dermis 5 or a dermal equivalent—collagen populated with fibroblasts.12,13
The most widely used skin wound covers—based on hydrogels or paraffin gauze—protect the wound but do not stimulate the epithelization. In this study, five innovative products, declaring stimulating effect on wound epithelization, were compared for their ability to support growth and stratification of keratinocytes in 3D cultures at the air–medium interface.
The results showed marked differences among the evaluated wound covers in supporting the keratinocyte growth, stratification, and differentiation in vitro. These differences are likely to be related to the design of the dressing material. We have demonstrated that while synthetic and semi-synthetic dressings allowed growth of keratinocytes in a monolayer, biological dressings, Xe-Derma and Xenoderm, induced a multilayer growth, mimicking stratification of normal skin.
The composite dressing Biobrane is a semipermeable silicone film with a nylon fabric, chemically binding reconstituted porcine dermal collagen. Since it results in formation of only a fragile single keratinocyte stratum, its efficacy in supporting keratinocyte proliferation when applied to a wounded skin area is rather modest with a slow onset of action.
The synthetic dressing Suprathel is a microporous lactocapromer terpolymer matrix. Clinically, it is distinct by strong adhesive power with a marked hemostatic effect. Although it shows better in vitro results than Biobrane in terms of the number of strata, a modest level of stratification indicates rather slow stimulation of the biological activity in a clinical situation.
Veloderm is a polymeric wound cover made of cellulose microfibrils. The properties in the applied experimental setting are similar to Suprathel. However, the staining with involucrin indicates lower keratinocyte differentiation. This feature may indicate longer persistence of proliferation potential.
The remaining two dressings, Xe-Derma and Xenoderm, are both derived from acellular porcine dermis; they are based on natural collagen fibers and other components of normal dermis. The presence of collagen fibers does not solely provide a mechanical support, but the collagen/cell interactions stimulate production of a number of cytokines and growth factors, leading to enhanced cell proliferation and migration. 11 This is probably the reason why they show better stratification in comparison to the synthetic wound covers.
From the clinical point of view, it is important that the new keratinocyte population is not damaged by changing the cover, since it peels off spontaneously during the recovery process. However, while sharing the origin from acellular porcine dermis, Xe-Derma and Xenoderm are prepared by means of different manufacturing procedures. Xenoderm is lyophilized, which results in disruption of the basement membrane, loosening of collagen fibers, and, consequently, rather high fragility. This brings about the occasional cell migration inside the dressing structure that we have observed. The resulting cell islands show terminal differentiation. On the other hand, Xe-Derma mimics the basement membrane by having a flat, solid, confluent surface, integrating fragments of the basement membrane. 10 This feature allows optimal adherence and easy surface locomotion of the cells, which—together with cell/xenomatrix interactions—induce a high level of metabolic activity. We assume that it is the reason why the number of strata observed in neoepidermis grown on Xe-Derma is double and better mimicking the structure of normal skin in comparison to Xenoderm (Figure 2).
The best cover of a burn wound is an auto- or allograft. A wound cover should have the properties as close as possible to normal human skin to provide optimal stimuli for wound healing. It was demonstrated that human and porcine acellular dermal matrices show a similar structure and are biocompatible. 14 Our previous clinical results show that Xe-Derma, more than hydrogel covers, vaseline gauze, or antibacterial silver sulfadiazine cream, accelerates the recovery of burns, without the need for dressing changing.9,15 Moreover, Xe-Derma combined with suspension of fresh (non-cultured) autologous keratinocytes and other skin cells enables repeated harvest of skin from donor sites for autotransplantation up to five harvests in 2-week intervals from the same location (in preparation).
Conclusion
The Xe-Derma wound cover provided the best support to keratinocyte proliferation and stratification in vitro compared to four other wound covers of different matrix structure (Xenoderm, Veloderm, Suprathel, and Biobrane). Statistical evaluation using t-tests with Bonferroni adjustment revealed no statistically significant difference as to the number of keratinocyte strata between Xe-Derma and Xenoderm. However, unlike Xenoderm, Xe-Derma produced significantly (p < 0.05) more strata in comparison to Veloderm, Suprathel, and Biobrane.
Furthermore, our study shows that there is a sufficient test-to-test reliability in using the applied in vitro model, opening the possibility of studying possible parallelism between in vitro results and clinical healing effect and, consequently, accelerating the development of future bio-engineering products.
Footnotes
Disclosure
Eva Matoušková is an associated scientist of the MEDICEM Institute.
Declaration of conflicting interests
Ondrej Mestak declares no conflict.
Funding
This work was supported by the Ministry of Health of the Czech Republic (grant no. NT 11392-6).
