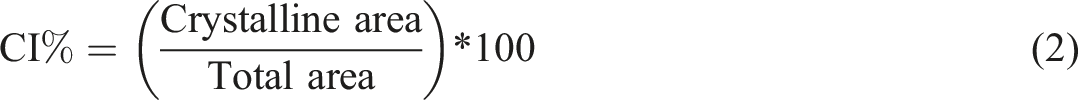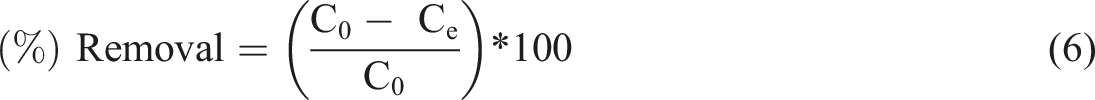Abstract
The discharge of dyes, including methylene blue (MB), contributes to environmental pollution. Adsorption is one of the most widely used techniques for removing colorants from water, with hydrogels being the materials that have attracted the most attention due to their excellent adsorption capacity. In this work, hydrogels composed of cellulose/pectin/starch were synthesized to remove MB dissolved in water. For the characterization of the hydrogel, Fourier-Transform Infrared, Thermogravimetry/Differential Thermal Analysis, XRD, and Scanning Electron Microscopy techniques were applied. At pH 9, the maximum swelling capacity (326.1%) of the hydrogel and the maximum MB removal percentage (37.05%) were obtained. The Weber-Morris diffusion kinetic model indicated that other adsorption mechanisms exist in addition to the diffusion process of MB through the hydrogel pores. Temkin's model better described the adsorption process, therefore an interaction between the adsorbate and the adsorbent surface is assumed, while the MB adsorption kinetics at pH 5 followed a pseudo-first-order model; and at pH 7 and pH 9, they followed a pseudo-second-order model.
Introduction
The textile industry is one of the main sources of pollution in the world. In addition to using a high consumption of water and energy, it releases a large number of hazardous effluents. 1 This industry represents ∼75% of the global dyestuff market and involves around 10,000 different dyes used for printing and/or coloring multiple types of fabrics. However, most of the textile industries are located mainly in developing countries, where effluent discharge standards are not respected, resulting in huge environmental pollution. 2 For example, it is estimated that 2%–20% of textile dyes are directly discharged as aqueous effluents, representing approximately 5000-10,000 tons of dyes per year. 3
One of the most used dyes in the textile industry to dye wool, cotton, and silk is methylene blue (MB). Methylene blue (Figure 1(a)) is a cationic and primary thiazine dye, which was first synthesized in 1800 by Heinrich Caro. Methylene blue is an odorless, dark green powder that when dissolved in water produces a stable blue solution at room temperature. However, it causes various risks to human health, such as respiratory distress, blindness, and abdominal, digestive, and mental disorders.4,5 Schematic representation of the structure of (a) methylene blue and (b) cellulose.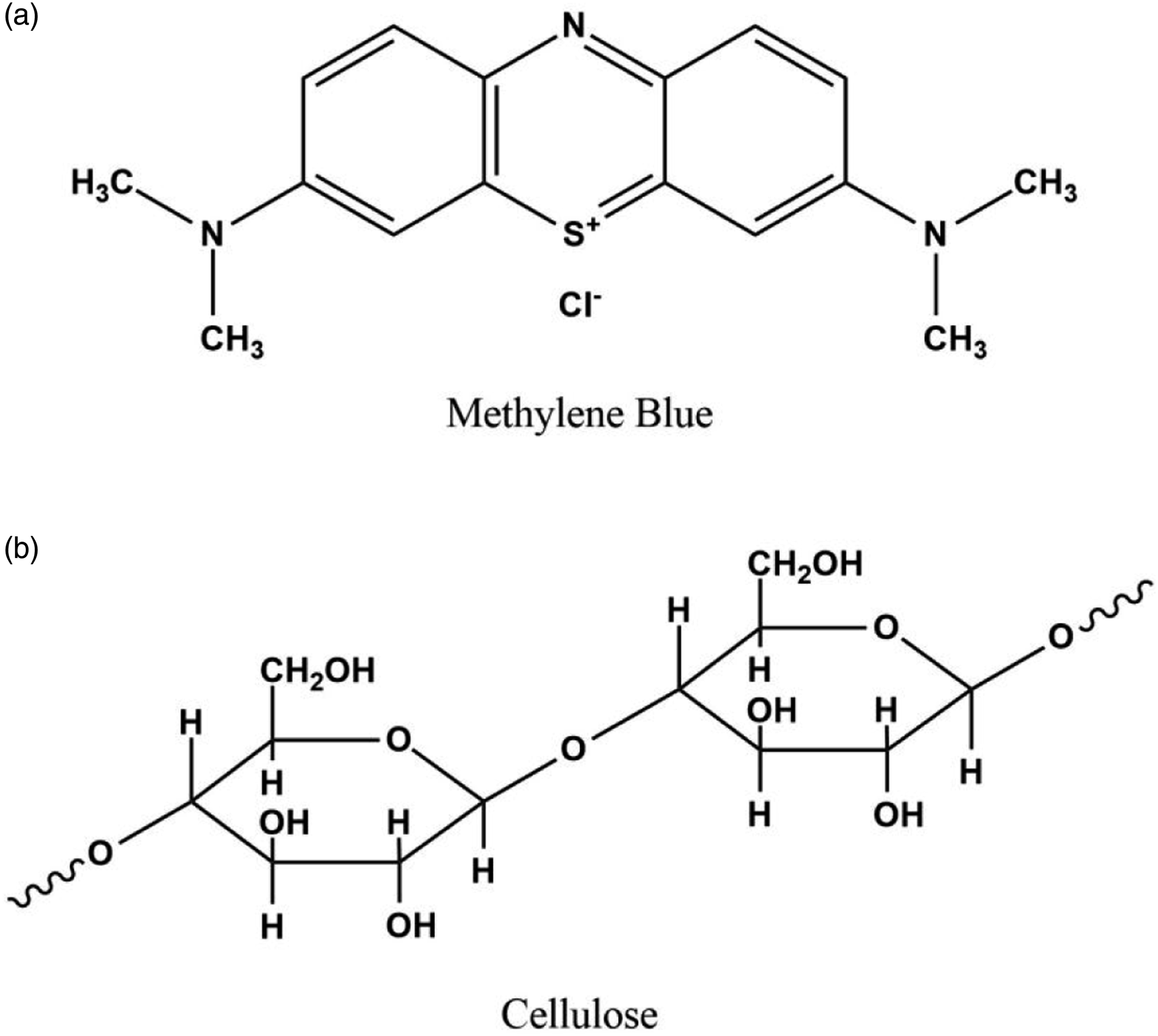
It is estimated that water-related diseases claim the lives of more than 5 million people a year, which is why in recent decades various water purification techniques have been used, including adsorption, filtration of membrane, electrochemical oxidation processes, and biological methods.6,7 Among the many dye removal techniques, adsorption is the technique that gives the best results and can be used to remove different types of dyes. 8
Adsorption is a non-reactive mass transfer process, by which a solid surface (adsorbent) is concentrated by a substance (adsorbate) initially present in a gaseous or liquid environment. 9 In this process there are two sorption mechanisms: physical adsorption (physisorption) and chemical adsorption (chemisorption). In physisorption, the adsorbate binds to the surface of the adsorbent due to weak forces such as electrostatic attraction and Van der Waals forces. Whereas, in chemisorption, the adsorbate is attached to the surface of the adsorbent due to strong covalent bonds. However, under desirable conditions, both mechanisms can occur at the same time. 10
The advantages of this technique are its simplicity of design, initial cost, insensitivity to toxic contaminants, ease of operation, non-production of harmful substances, 11 reuse of adsorbents and high efficiency. 12 However, its performance is influenced by physico-chemical factors (e.g. pH, temperature and pressure), the chemical structure of the contaminant, the presence of ions in the aqueous solution, and the properties of the adsorbent (pore size, molecular weight, molecular structure, molecular size, and polarity). 13
Compared with other adsorbents, hydrogels (HG) have excellent adsorption capacity. Hydrogels are porous adsorbent materials made up of 3D networks of hydrophilic polymers, 14 composed of numerous hydrophilic functional groups (e.g., -OH, -COOH, -NH2, -SO3H, -CONH2, etc.) that allow the adsorption and retention of a large volume of water (hydrogels swell to more than a thousand times their original size when they absorb water),15,16 and provide multiple adsorption sites for the removal of pollutants. 17
Conventionally, hydrogels are made from synthetic polymers; however, due to the environmental pollution caused by the accumulation of conventional solid waste, current research is focused on the development of new biodegradable materials made from natural polymers (polysaccharides).18,19 Natural polymers, such as polysaccharides, have properties that make them widely used in the development of new materials for applications in various areas, such as healthcare, agriculture, and environmental protection. 20
Cellulose is one of the most widely used polysaccharides in the preparation of hydrogels due to its natural abundance. It is a linear polymer composed of repetitive β-(1→4)-linked
Zinc chloride (ZnCl2) is an inorganic molten salt highly used for the dissolution and hydrolysis of cellulose.
23
The dissolution capacity of cellulose with ZnCl2 depends strongly on the amount of water present in the form of hydrates, ZnCl2.nH2O. e.g. Zinc chloride trihydrate (ZnCl2.3H2O) dissolves cellulose, but zinc chloride dihydrate (ZnCl2.2H2O) and tetrahydrate (ZnCl2.4H2O) only swell the cellulose, forming a fine distribution without dissolving it.
24
The hydrolysis of cellulose is carried out through a coordination between the glycosidic oxygen of cellulose with zinc, which acts as a Lewis acid allowing the breaking of the glycosidic bond. Subsequently, the coordinated water molecules of the hydrated ZnCl2 participate as a nucleophile, obtaining
Due to the presence of a large number of hydroxyl groups in its structure, cellulose has a big potential in the preparation of superabsorbent hydrogels. 27 Cellulose-based hydrogels are biodegradable, biocompatible, have high water retention, low cost, and good mechanical properties; allowing them to be applied in tissue engineering, controllable delivery systems, blood purification, sensors, agriculture, and water purification. 28
In recent years, research has focused on improving the properties of hydrogels. These improvements can be obtained from different types of interactions, such as (i) electrostatic interaction (cation-polymer), (ii) hydrophobic interaction (polymer-polymer), and (iii) covalent cross-linking. 16 For example, cellulose/chitosan hydrogels demonstrated superior swelling properties with possible application in antimicrobial packaging, 29 and the percentage of swelling for cellulose/gelatin hydrogels increased from 4516% to 4933% with increasing gelatin weight percentage. 30 Additionally, these composite hydrogels can be used to remove dyes from water. e.g. cellulose/sodium carboxymethylcellulose hydrogel, 31 cellulose/sodium alginate/iron hydroxide hydrogel, 32 and cellulose/guar gum/biochar hydrogel 33 had a maximum MB adsorption capacity of 362.3 mg g−1, 105.93 mg g−1 and 598.28 mg g−1, respectively. Furthermore, the cellulose-chitosan hydrogel removed 95.88% of MB at pH 9. 34
Due to not finding research on cellulose/pectin/starch composite hydrogels. This research focuses on the preparation of these composite hydrogels, the study of swelling at different pH and temperature values, and their application in the removal of MB dissolved in water.
Materials and methods
Materials
Microcrystalline cellulose, pectin, starch and MB were purchased from Sigma. Ethanol absolute, zinc chloride and calcium chloride dihydrate from Scharlau. Commercial chemicals were of analytical grade.
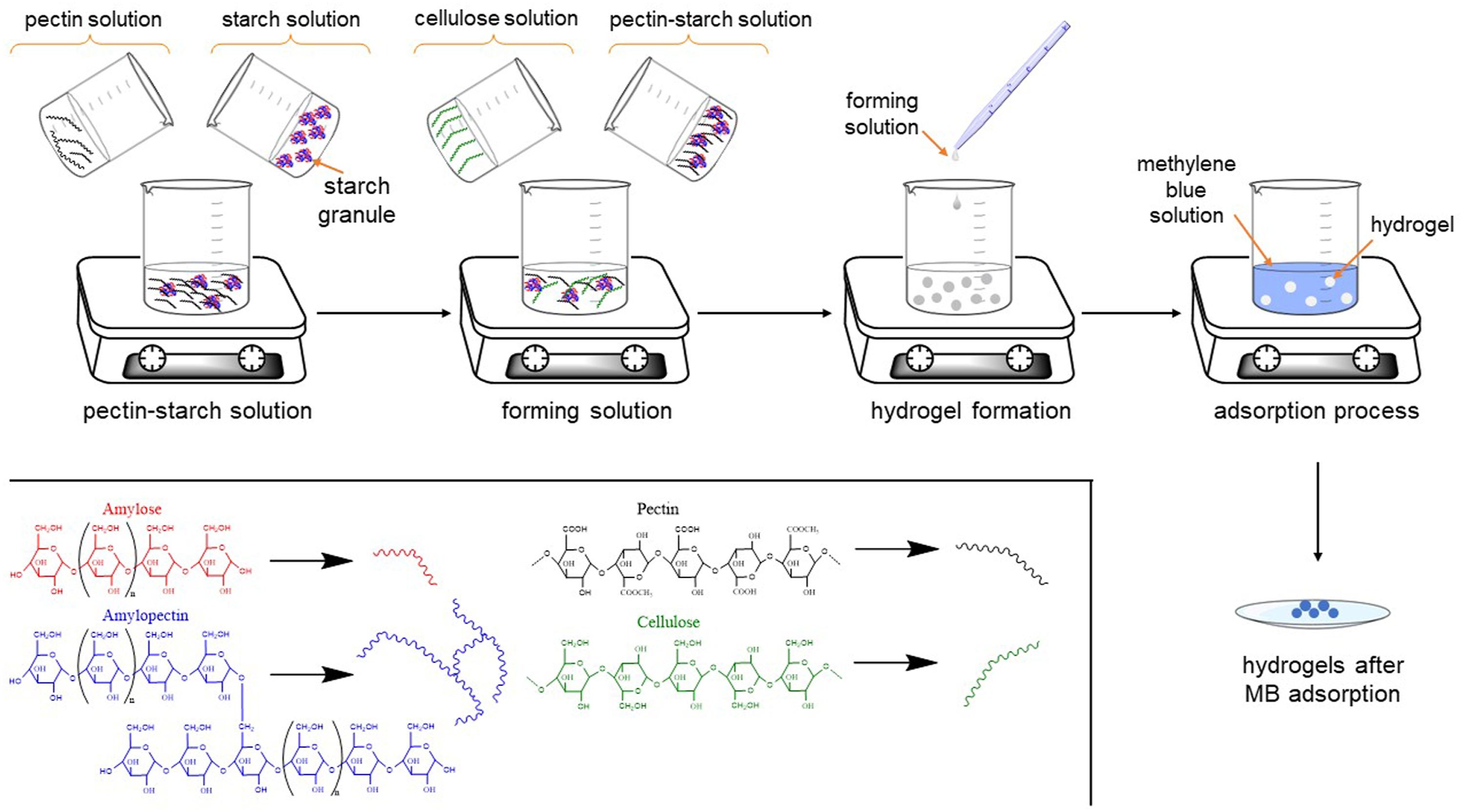
Preparation of composite hydrogels
Based on the methodologies developed by Chen et al., 35 Nie et al., 36 and Dafe et al. 37 The composite hydrogels were prepared using the following procedure: Firstly, 0.4 of cellulose was added to 10 g of ZnCl2 and stirred for 6 h at 90°C until dissolved. In addition, pectin solution at 3.0% w v−1 and starch solution at 2.5% w v−1 were prepared separately and then mixed for 1 h at 70°C. Subsequently, the pectin-starch solution was mixed with the cellulose solution and stirred at 70°C for 1 h. Finally, the resulting mixture was added dropwise into crosslinking solution (500 mL of 70% w v−1 CaCl2) with constant slow stirring at room temperature and left for 2 h. The obtained hydrogels were washed at room temperature with ultrapure water, and then with ethanol three times.
Attenuated total reflectance fourier-transform infrared spectroscopy
Fourier-Transform Infrared (FT-IR) measurements were recorded on Nicolet iS10 spectrophotometer (Thermo Scientific, Madison, WI, USA), and the wavenumbers ranged from 600 to 4000 cm−1.
XRD analysis
The XRD diffractograms were obtained by D2 Phaser (Brüker, Germany) equipment with a scattering angle from 5° to 80° at a scanning speed of 0.05°/s. The crystallinity index (CI%) of cellulose was determined by the Segal method
38
(equation (1)), and of the hydrogel was determined using the relationship between the crystalline area by the total area
39
(equation (2)):
Thermogravimetric analysis (TG/DTG)
The thermogravimetric curves were gathered on a Simultaneous Thermal Analyzer (STA) 6000 (PerkinElmer, Waltham, MA, USA) using 5.0 ± 0.1 mg of sample in an N2 atmosphere (flow rate at 20 mL min−1) with a temperature range of 38 °C–595°C (heating rate of 10°C min−1).
Scanning electron microscopy Analysis
Scanning electron microscopy (SEM) images of composite hydrogels (before and after MB removal) were studied using an Inspect S50 microscope (FEI, Hillsboro, OR, USA). Prior to analysis, all samples were gold plated in a 11,430E-AX high vacuum metallizer (SPI Supplies, West Chester, PA, USA).
Swelling studies
Swelling values of the hydrogels in response to pH (5, 7, and 9) and temperature (9°C and 25°C) were measured in triplicate via the gravimetric method. In brief, 0.1 g of the dried hydrogel was immersed in 25 mL of distilled water and withdrawn at defined time intervals (15, 30, 60, 90, 120, and 240 min). The hydrogels were then dried with filter paper and weighed. The pH of the solutions was adjusted by 0.1 M HCl and 0.1 M NaOH. The swelling value (S), expressed in g H2O g−1 hydrogel,
40
was determined from the following equation:
Methylene blue adsorption studies
The removal of MB was carried out according to the methodology developed by Herrera et al.
39
0.1 g of composite hydrogel in 50 mL of 3.1 x 10−6 mol L−1 MB dye solution was added at room temperature. Six-time intervals (15, 30, 60, 90, 120 and 240 min) and three pH levels (5, 7, and 9) were used. The absorbance values (
Isotherm models studies
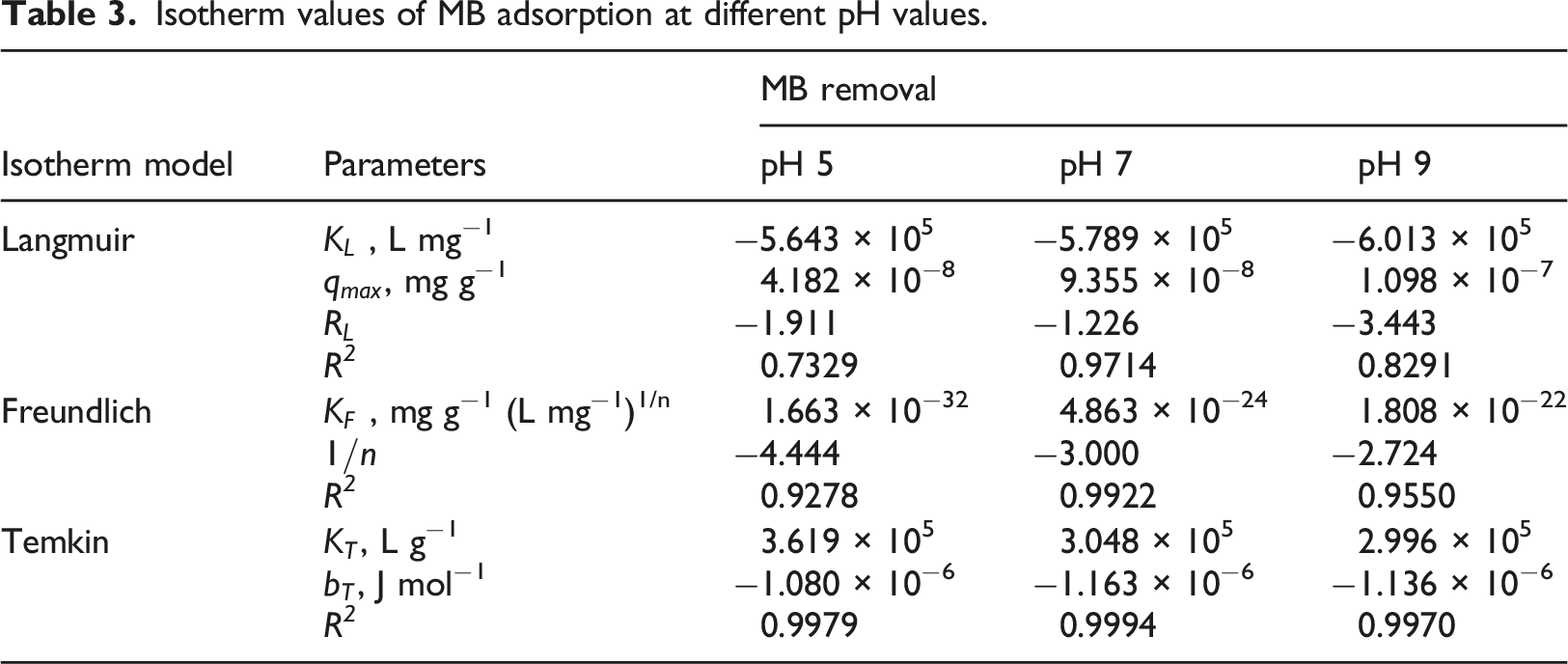
The adsorption isotherms describe the interactive behavior of the amount of adsorbate adsorbed on the surface of the adsorbent and the residual amount in the solution, until reaching a dynamic equilibrium at a constant and defined temperature.
42
To characterize the MB adsorption process, the Langmuir (equation (7)), Freundlich (equation (8)), and Temkin (equation (9)) adsorption isotherm models were used. In addition, the Langmuir isotherm’s value (equation (10)), denoted as
Kinetics models studies
To study the time dependence of the adsorption progress, the adsorption dynamics were investigated. Kinetic models can indicate whether chemical or physical adsorption is dominant in the adsorption process. To determine the relationship between the adsorption capacity and contact time, the kinetic models of pseudo-first-order (equation (11)), pseudo-second-order (equation (12)), and Elovich (equation (13)) were used;
44
Diffusion models studies
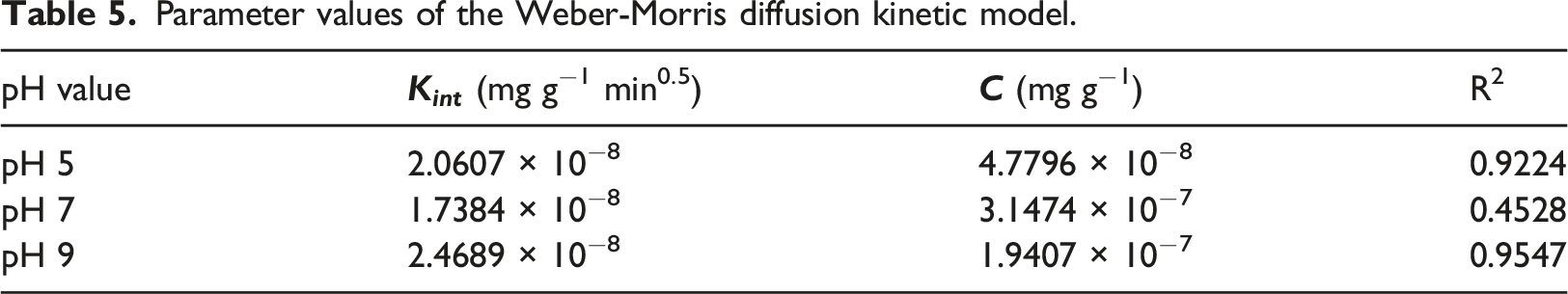
Diffusion models are based on a three-step adsorption process: (1) diffusion through the liquid film surrounding the adsorbent; (2) diffusion within the pores and/or along the pore walls of the adsorbent; and (3) adsorption and desorption between the adsorbate and the active sites of the adsorbent.
43
The Weber-Morris model (equation (14)) describes the mechanism of diffusion and transfer of adsorbate through internal pores. Solute adsorption is proportional to
Results and discussion
Characterization and analysis of the composite hydrogels
Photos of composite hydrogels
The appearance of the hydrogels was photographed as shown in Figure 2. The hydrogels obtained are solid white hydrated spheres (Figure 2(a)) that after the drying process became harder and brown in color (Figure 2(b)); however, when they were submerged in water, they returned to their initial appearance due to their ability to retain water. Photos of the hydrogels (a) initial stage and (b) after the drying process.
Morphology of hydrogel
The morphology of the hydrogels was obtained by scanning electron microscopy (SEM). Figure 3 shows the SEM image of the hydrogel previously dried for 72 h at 30°C. From this image it is confirmed that the hydrogel has a rough surface (Figure 3(b)), which according to the Wenzel's model would act as a wettability amplifying effect, allowing the surface to be highly hydrophilic.
46
Scanning electron microscopy images of surface morphology of dry composite hydrogel at (a) 500 µm and (b) 100 µm.
Fourier-Transform Infrared and XRD analysis
Fourier-Transform Infrared was used to investigate the structural interactions of the polymer matrices in the composite hydrogels (Figure 4). The hydrogel exhibited characteristic peaks for cellulose at 3320 cm−1 (OH vibration bonds), 1429 cm−1 (CH2 scissoring in C6), 1313 cm−1 (CH2 wagging), 1159 cm−1 (glycosidic bonds C-O-C), 1109 cm−1 (C-O vibrational bonds), and 1050 cm−1 (vibration C-O-C of the pyranose ring).47,48 However, it presented an additional peak at 1620 cm−1 which is related to the stretching of the carboxylate ion present in the pectin polymer matrix,
49
and an increase in intensity at 1429 cm−1 which suggests a reorganization of the cellulose segments present in the hydrogel.
50
In addition, the peak close to 1370 cm−1 characteristic of the cellulose-water interaction and the C-O-H bending51,52 was not evidenced, but rather the appearance of a peak at 1004 cm−1 product of the O-H expansion mode,
53
suggesting a rearrangement and possible interaction between the hydrophilic groups of the polymer matrices. Fourier-Transform Infrared spectrum of cellulose and hydrogel.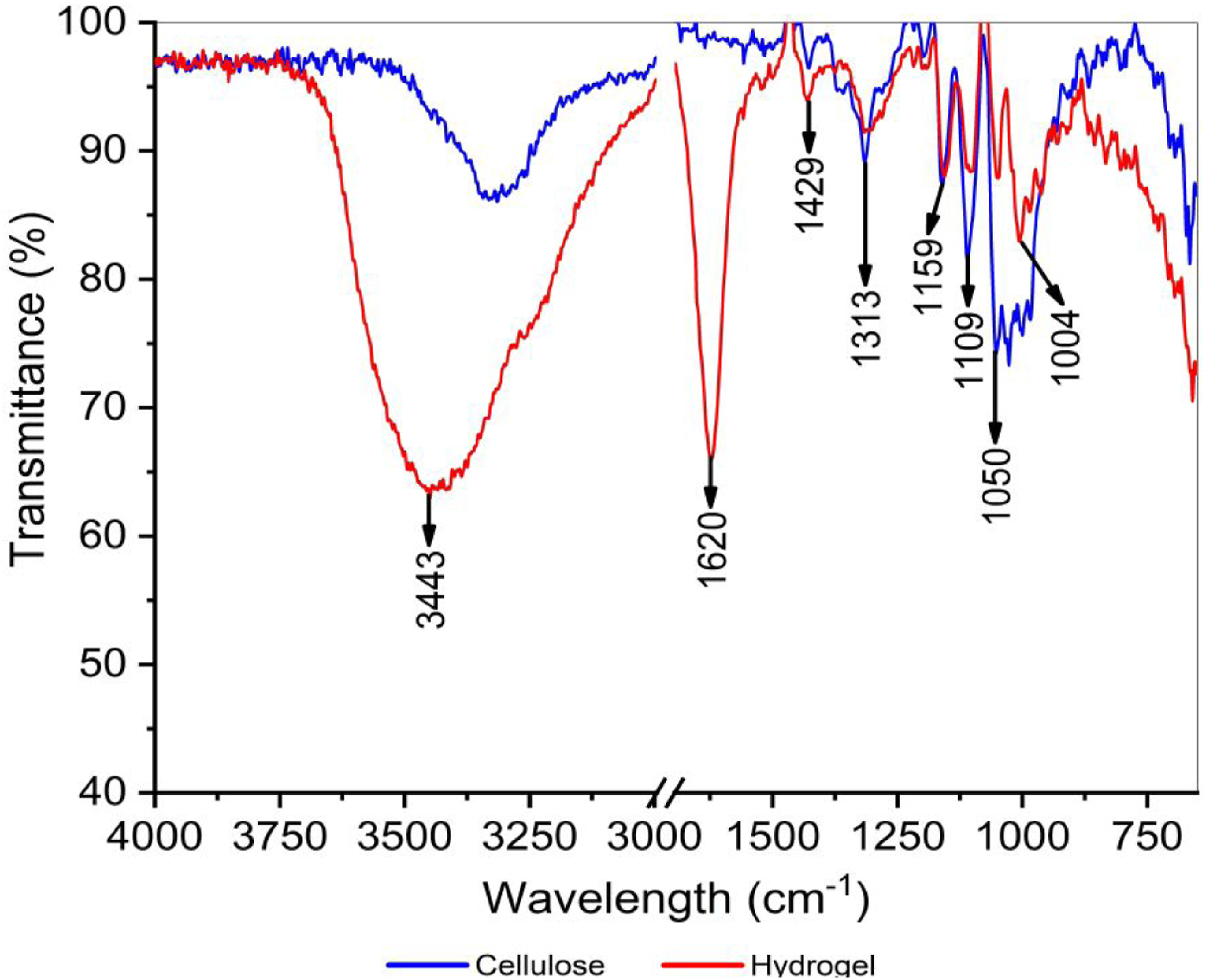
Figure 5(a) shows the XRD diffractogram of the cellulose, which presented a crystallinity index of 79.79%, and peaks close to 15°, 16°, 22.5° and 34.5° corresponding to the crystalline patterns (101), (10 XRD diffractograms of (a) cellulose and (b) hydrogel.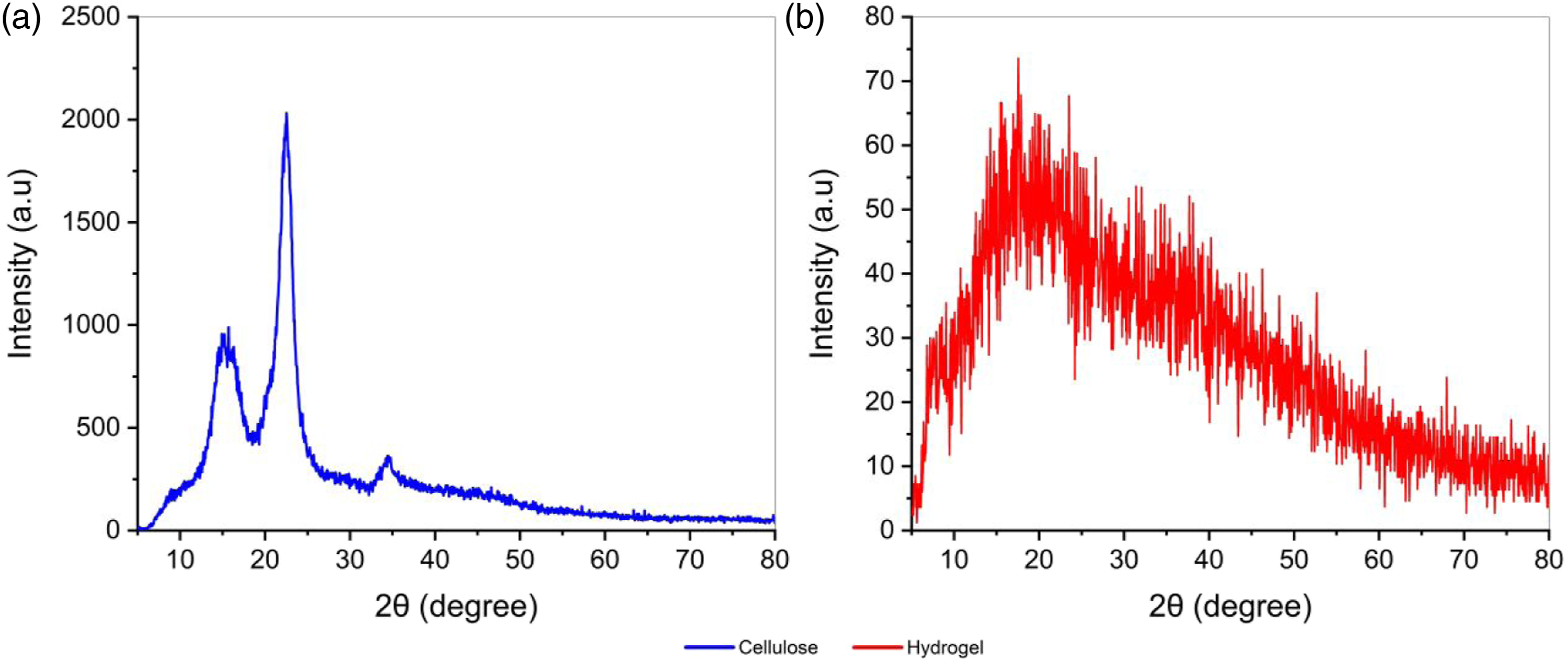
Thermogravimetric analysis
The study of the thermal decomposition was determined by means of the thermal analyzes of TG (thermogravimetry) and DTG (differential thermal analysis). Figure 6(a) shows the thermal events of the cellulose, which presented a small mass loss between 38–226°C attributed to the process of dehydration (eliminating absorbed water) and a more pronounced degradation event between 226–400°C. The sharp DTG peak centered at 333°C was due to cellulose degradation processes caused by dehydration, depolymerization, and decomposition processes of glycosyl units, which can produce CO, CO2, H2O, and charred residue.55,56 Thermogravimetry/Differential Thermal Analysis curves of (a) cellulose and (b) hydrogel.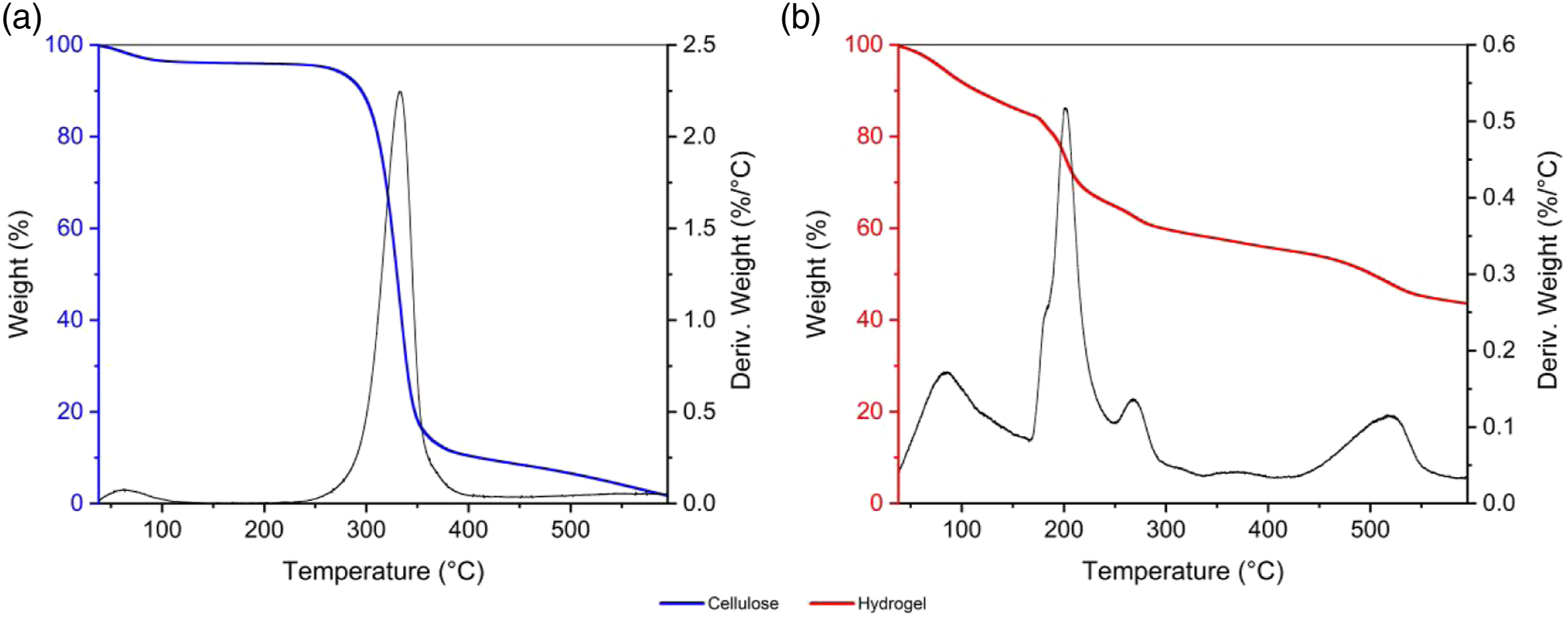
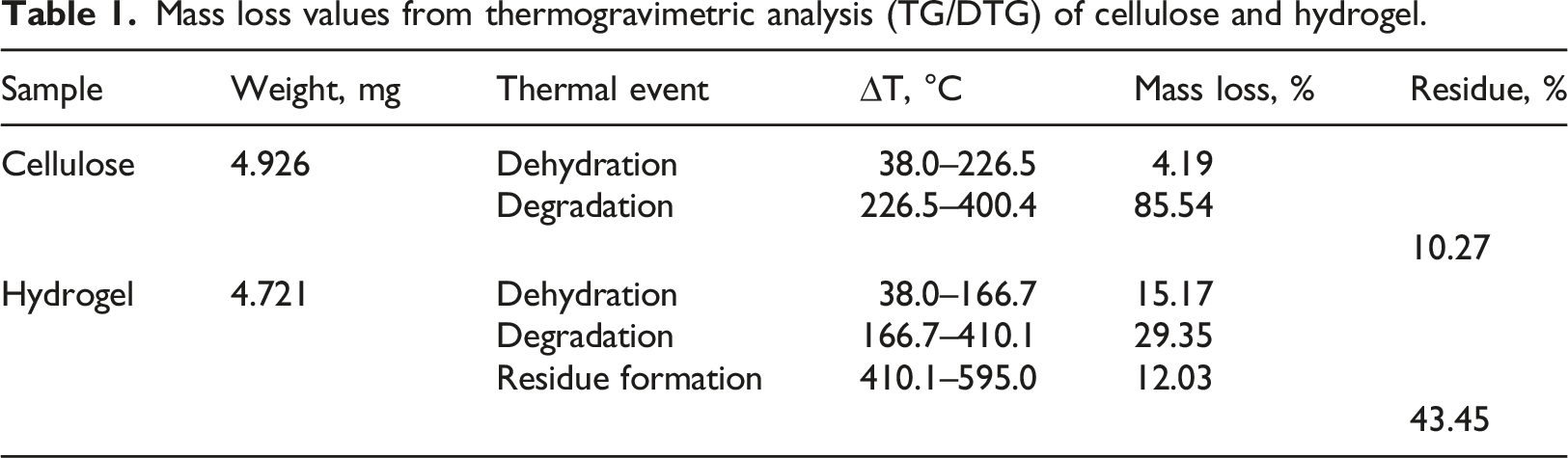
Mass loss values from thermogravimetric analysis (TG/DTG) of cellulose and hydrogel.
Swelling capacity
The swelling behavior of the hydrogels at three pH levels of the solution and at different temperatures is shown in Figures 7(a) and (b). At 9°C, the swelling value was 0.268 g g−1 (26.8%, pH 5), 0.281 g g−1 (28.1%, pH 7), and 0.298 g g−1 (29.8%, pH 9). While at 25°C a swelling value was 1.480 g g−1 (148.0%, pH 5), 2.215 g g−1 (221.5%, pH 7), and 3.261 g g−1 (326.1%, pH 9). This is due to high temperature increasing the mobility of the polymer chains, destroying the hydrogen bonds present in the hydrogel,
58
and generating an expansion in the polymer matrices. On the other hand, at low temperatures, a lower swelling capacity was observed as a consequence of a limited movement between the polymeric chains and with it a lower diffusion of water in the internal structure of the hydrogel.
59
Swelling value (S) for hydrogels at (a) 9°C and (b) 25°C.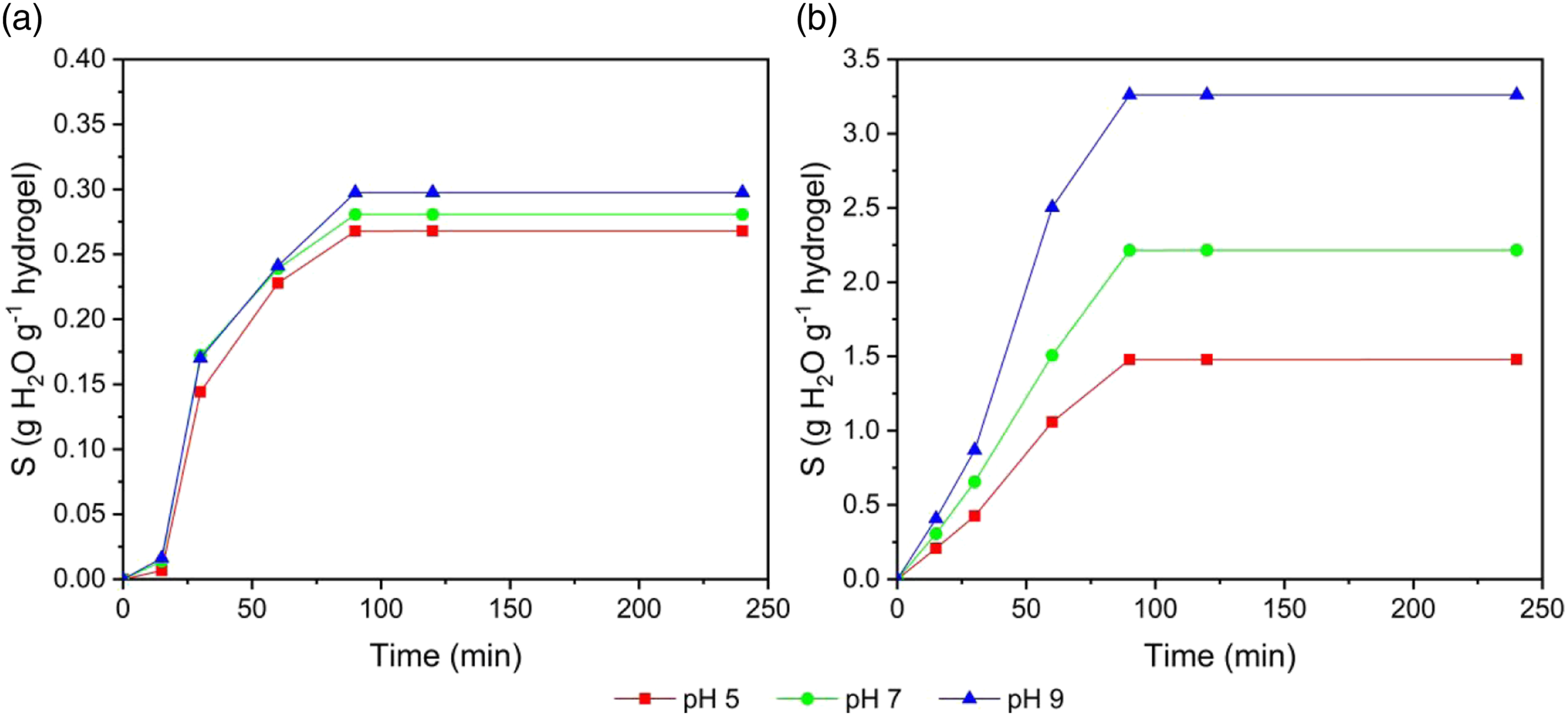
Figure 8(a) shows a higher swelling capacity at alkaline pH (pH 9) in the solution compared to the test performed at acid pH (pH 5). According to the literature, at lower pH levels, the amount of H+ ions increase, so an additional physical cross-linking would be formed, causing the hydrogel network to contract and decrease its swelling capacity; while, at higher pH levels, H+ ions decrease and OH− ions increase
60
generating negatively charged sites that promote expansion and displacement of polymeric matrices, increasing the pore size of the network and improving the swelling capacity of the hydrogel.
61
However, these hydrogels presented swelling values at 25°C well below those of hydrogels based on cellulose derivatives reported in the literature: 550% and 725%.62,63 Therefore, it is presumed that the presence of zinc, from ZnCl2 used in the dissolution of cellulose, produced cellulose-zinc and starch-zinc complexes.
22
While with pectin a cross-linking mechanism described by the egg-box model occurred.
64
For this reason, it is presumed that the presence of zinc and various polymer matrices could produce strong bonds that would prevent further expansion of the polymer chains in the hydrogel, reducing its swelling capacity. Bar charts of (a) swelling ratio of hydrogels and (b) Methylene blue removal percentages.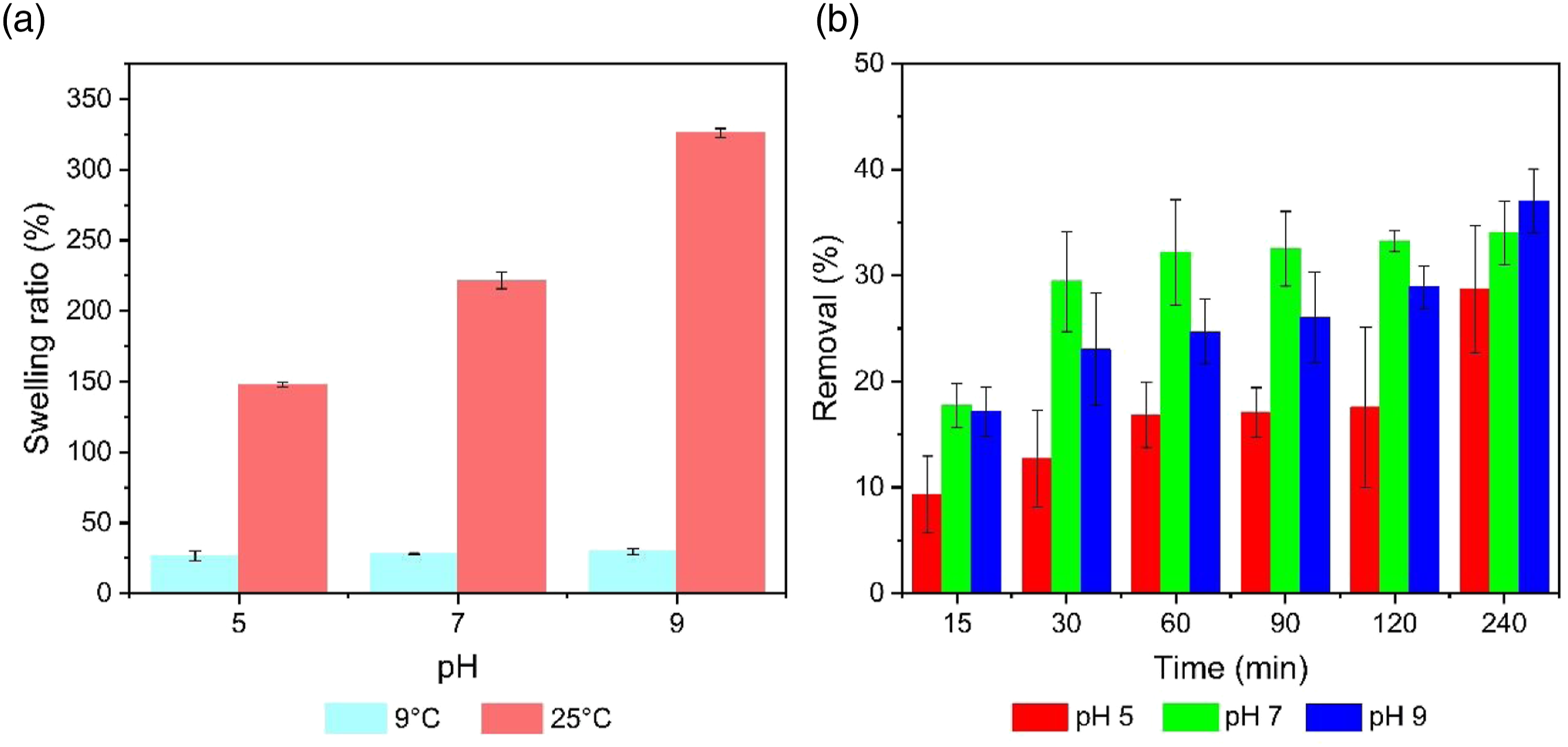
Methylene blue adsorption experiments
Fourier-Transform Infrared and SEM analysis after MB removal
Figure 9(a) shows the peak at 1620 cm−1 in the FTIR spectrum corresponding to the stretching of the carboxylate ion of the hydrogel before and after the MB adsorption process. According to Wang et al.
65
under alkaline conditions, carboxyl groups in their ionized form (-COO-) could be attached to MB molecules through electrostatic interaction, thus achieving efficient adsorption. However, under acidic conditions, carboxyl groups (-COOH) would be formed, so the adsorption of MB would not be favorable. Fourier-Transform Infrared spectrum before and after MB adsorption (a) and Scanning electron microscopy images of surface morphology of hydrogel after MB adsorption at pH 5 (b), pH 7 (c), and pH 9 (d) at 100 µm. Note. MB: Methylene blue.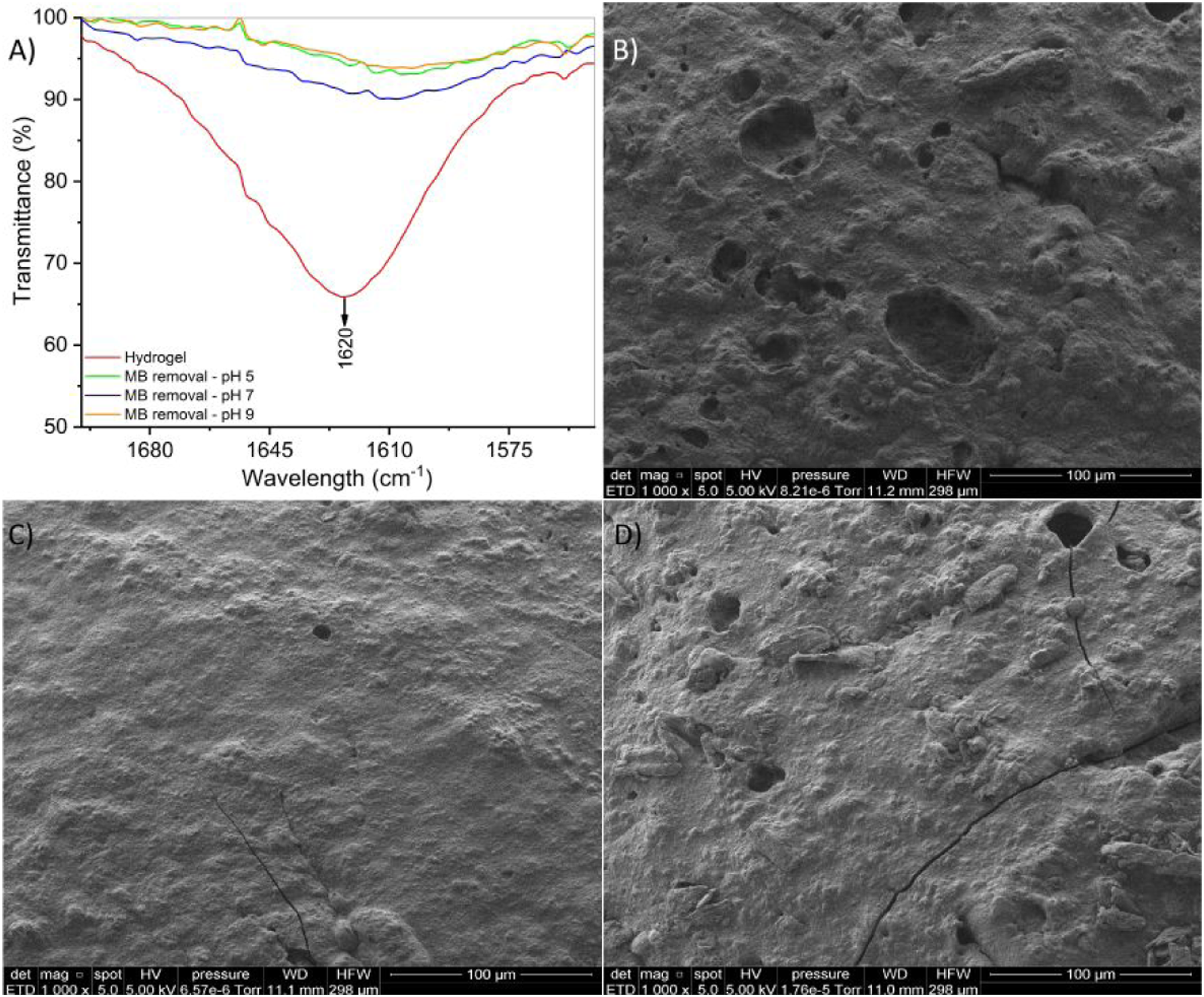
The surface morphology of the hydrogels after MB adsorption was obtained by SEM. At neutral pH, the surface of the hydrogel became smoother and more homogeneous (Figure 9(c)) compared to the surface of the hydrogels after the adsorption process at acid (Figure 9(b)) and alkaline (Figure 9(d)) pH, which presented higher porosity and heterogeneity.
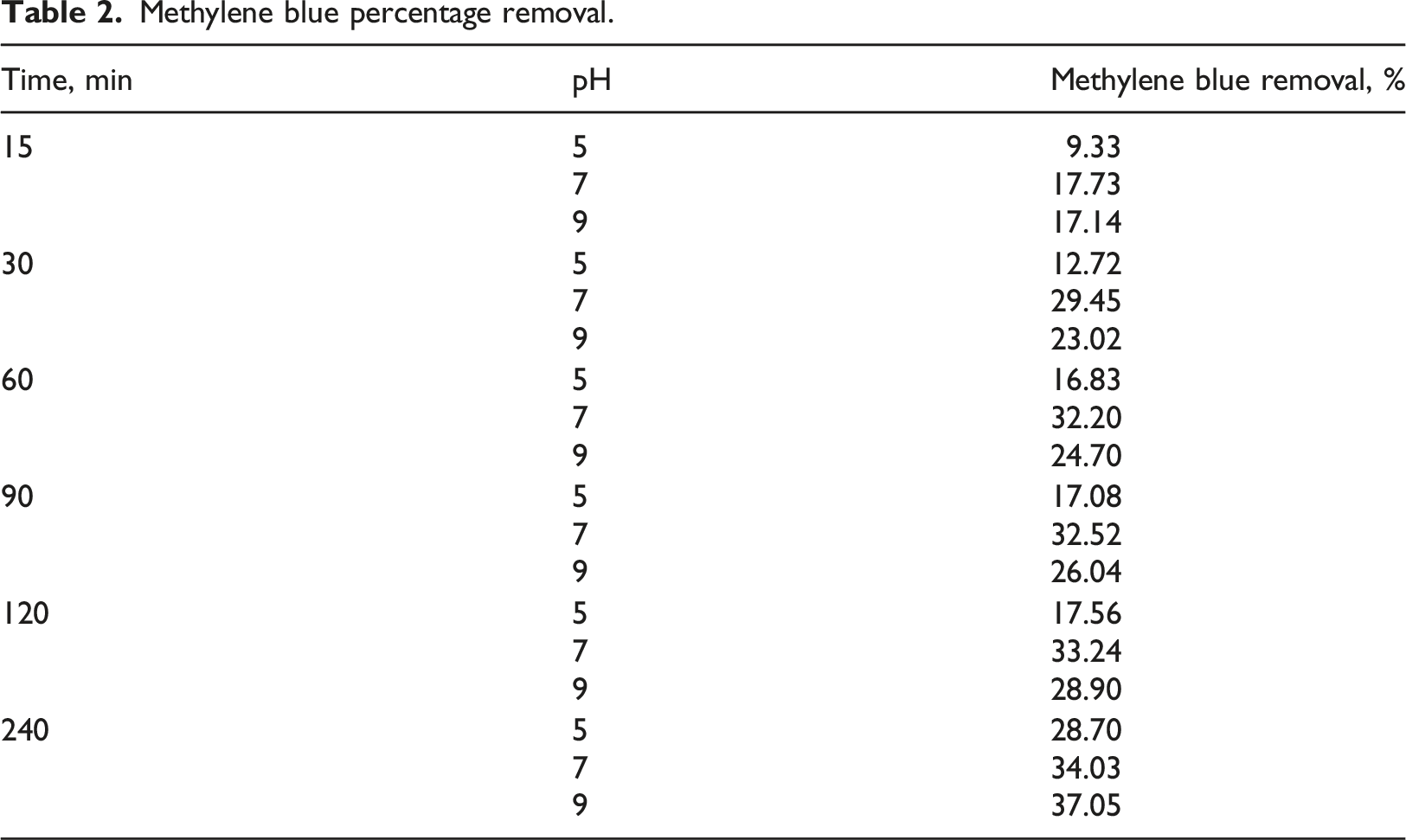
Methylene blue percentage removal
Methylene blue percentage removal.
Adsorption isotherm models
Isotherm values of MB adsorption at different pH values.
Kinetic adsorption models
Kinetic values of MB adsorption at different pH values.

Diffusion models
Parameter values of the Weber-Morris diffusion kinetic model.
Conclusions
A composite hydrogel based on cellulose with pectin and starch was used for MB removal. Structure, morphology, swelling and MB removal ability of hydrogels were investigated by different techniques. SEM analysis revealed the rough surface of the hydrogel, which became smoother and homogeneous after the MB removal test at neutral pH (pH 7). Alkaline and acidic pH values did not induce any changes on the hydrogel surface. At pH 9, due to the interaction of carboxylate groups with MB molecules represented by the pseudo-second-order kinetic model, the highest MB adsorption (37.05%) was obtained. Moreover, the increase of OH− ions at pH 9 and 25°C allowed obtaining the maximum swelling capacity of the hydrogels (326.1%). However, higher values were not obtained because the presence of ions could have formed complexes with the polysaccharides and/or cross-linking mechanisms. These hydrogels are not good adsorbents under the conditions used. In general, the physicochemical properties of a hydrogel may be influenced and limited by the presence of different polysaccharides.
Supplemental Material
Supplemental Material - Preparation of cellulose-based composite hydrogels for the removal of methylene blue
Supplemental Material for Preparation of cellulose-based composite hydrogels for the removal of methylene blue by Lizbeth Aymara, Nora Gabriela Herrera, Hélmer Helí Lezama and Nelson Adrián Villacrés in Polymers from Renewable Resources
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Federico Villarreal National University through the “Basic and Applied Research Projects for Knowledge Communities 2021 - CANON Funds” program, project grant R. N° 9078-2021-UNFV.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.