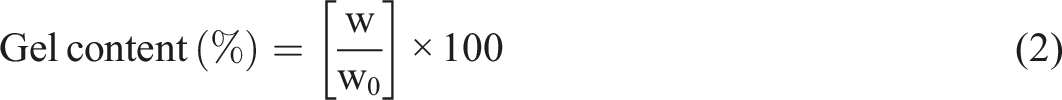Abstract
The coating industry may comply with new safety rules by using more environmentally friendly and harmless alternative materials. The present study intends to investigate the characteristic of agricultural waste material-based bio-polyol derived polyurethane wood protective coating for building and construction applications. This study covers the synthesis and characterization of cardanol based bio-polyol through dual epoxidation of phenolic -OH and terminal unsaturation to generate monoglycidyl ether of cardanol followed by ring opening reaction. The resultant bio-polyol is used for formulation of polyurethanes using aliphatic (Desmodur N75) and aromatic (Desmodur L75) isocyanates. The formulated polyurethanes employed for sustainable wood protective coating as well as architectural and multitudinous applications with enhanced antibacterial activity. The resultant wood protective coating was characterized by attenuated total reflectance-fourier transform infrared spectroscopy (ATR-FTIR), mass spectroscopy, 1H NMR, thermogravimetric analysis (TGA), surface morphology, mechanical and antibacterial properties.
Highlights
Cardanol from CNSO is used as a feed stock for the synthesis of a novel bio-polyol with high hydroxyl value. The cardanol derived bio-polyol based polyurethanes giving vary good thermal, mechanical and chemical resistant properties due to a greater number of hydroxyl groups. The polyurethanes formulated using aliphatic and aromatic isocyanates gives enhanced antibacterial properties. The resultant polyurethane coating gives long-lasting and durable wood coating for various construction applications.
Introduction
With the help of various functional polymeric materials, our daily life becomes more convenient and comfortable. Most of the polymers are derived from petrochemical based materials which causes global warming by increasing greenhouse gas emission which become a gradual worldwide environmental crisis along with the depletion of fossil fuels. Therefore, the exploration of resources for the development of different functional polymeric materials is necessary for reduction of the global warming and preventing the consumption of fossil fuels. For environmental concerns like sustainability, cost-affectivity, and abundantly availability, use of bio-based materials from renewable resources such as plant and non-food materials become a wide area for research for academician and industries.1–3
The use of fossil fuel-based petrochemical feedstocks for synthesizing the necessary polymers can be reduced by replacing it with non-edible oil-based feedstocks. In order to decrease dependency on petrochemicals, it is necessary to find an alternate naturally occurring non-edible oil based materials for the synthesis of polymers. 4 In addition to effectively addressing the issue of pollution and reducing synthetic dependability of polymers on petroleum resources, the use of biodegradable polymers and their composites can also help to reduce the release of greenhouse gases and advance the high-value utilization of biomass resources, which is consistent with the objective of sustainable development and the foundational principle of the circular economy. Finding and extracting natural, non-edible oils that may be used to make polymeric materials becomes essential to reduce the dependency on petrochemicals. 5 Plant fibers are an ideal replacement for polymer-based reinforcing materials owing to they are inexpensive, renewable, and benign to the environment. 6 Global concern is growing associated to ecological issues, such as waste management and carbon emissions. To combat rising waste materials contents, effective waste management is required, encompassing monitoring, separation, and recycling. 7 Petrochemical based feedstock was replaced by oligomeric plasticizer with high efficiency from renewable bio monomers glycerol and adipic acid. 8 P. Zarrintaj et al. 9 reported the biopolymers implemented in tissue engineering. With the introduction of new technologies and techniques, recycling technology has gradually improved in recent years.10,11
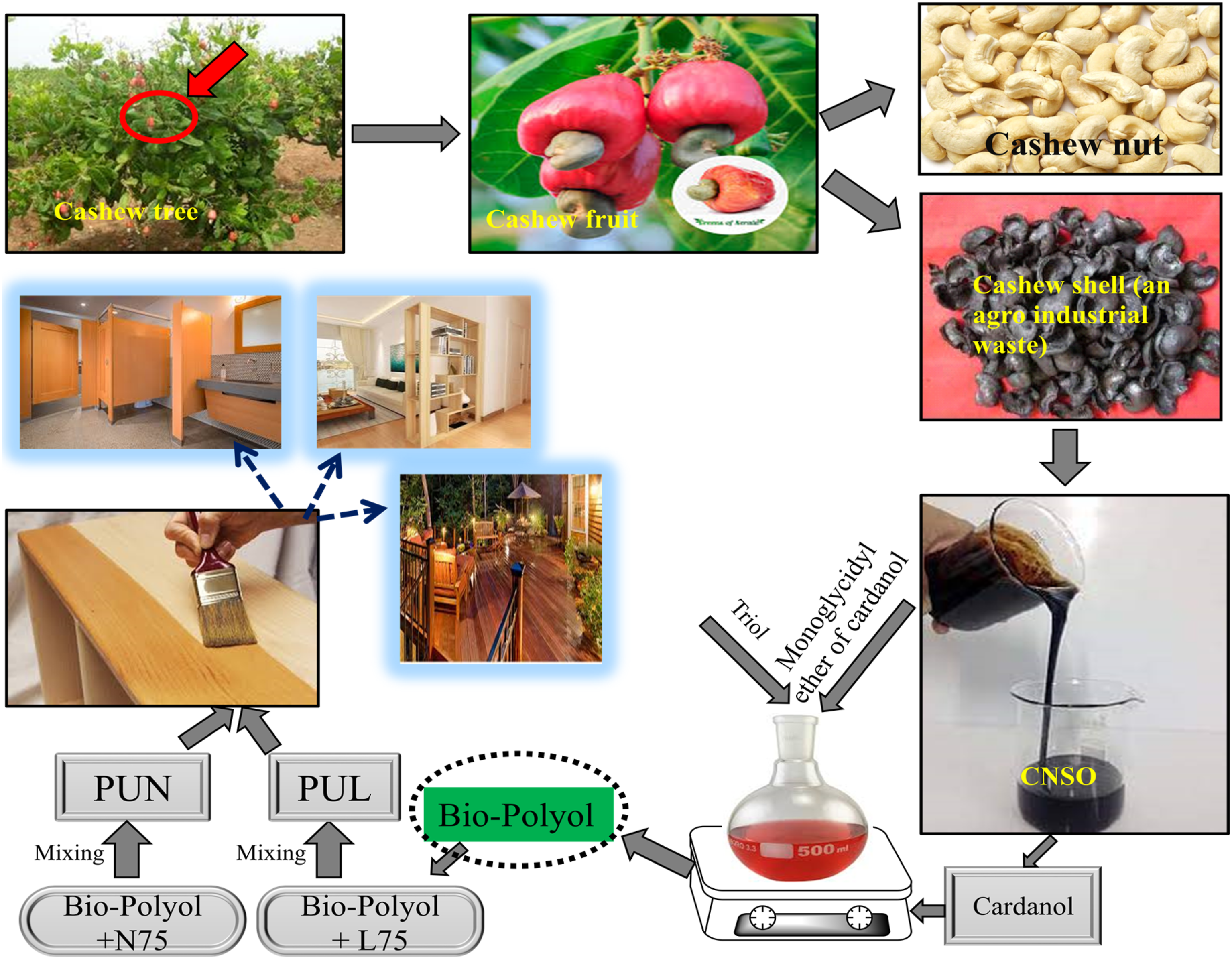
Vegetable oil-derived polymeric material has commendable chemical and physical properties. 12 Plant oil is one of the important renewable resources for the chemical industry. 13 Cashew nut shell oil (CNSO) is an agricultural waste and natural oil extracted from the non-food cashew plant (Anacardium).14–16 In the year 2002, the worldwide export of CNSO is approximately 1900 metric tonnes with the United States amounting to 737 metric tonnes. Technical CNSO comprises cardol, 2-methyl cardol and cardanol (62%–63%) whereas natural CNSO mostly contains anacardic acid (64%–65%). 17 Cashew nut shell oil (CNSO) can be used as a possible replacement for petroleum-based feedstock because of its sustainability, abundantly available reactive functionalities, and cost-effectiveness.18,19 Among the four components of CNSO, cardanol has become a great research area in recent years and have been widely used in adhesives, coatings, and plasticizers. Cardanol cannot dissolve in water but is easily dissolved in an organic solvent such as chloroform, ether, and petroleum ether. 20 Cardanol has long alkyl chain at meta position of phenolic ring which allows cardanol derivatives facile processing and considerable solubility in organic solvents. 21
Wood is widely used construction material; it must be meticulously protected in order to accomplish its full potential. Indeed, the food and agriculture organization (FAO) recognized using wood in building as an essential tool in the battle against climate change. Wood must be used more frequently in construction because steel and concrete are less environmentally friendly materials which stores enormous quantities of carbon dioxide. Good methods of construction can stop the deterioration of wood, through preventing the prolonged exposure of wood to some sources of degradation. 22 Over the past 5 years, coatings have received the greatest attention for wood surface preservation. Wood is easily accessible in our environment, in contrast to steel, whose production requires a drawn-out and complicated process. Additionally, wooden homes are cheap to maintain, which results in long-term cost savings. Wooden constructions reduce humidity, promoting easier breathing. 22
Polyurethanes can be formulated using two components; polyol and isocyanates. Polyols derived from non-renewable petrochemical based materials are known as synthetic polyols and synthesized from environmentally friendly and renewable resources like vegetable oils are known as natural/bio-polyols. Natural/bio-polyols can be synthesize from through three various pathways, epoxidation, hydroxylation, and purification. 23 On the bases of hydroxyl value of the polyol, it finds application in the development of polyurethane based coating, adhesives, rigid and flexible foams etc. Polyurethane has almost touched every area of material engineering, from shoes to aeronautical coatings because of its versatile properties. The main advantage of polyurethane has been its durability, toughness, and admirable chemical resistance 24 and may be used for fibers, foams, adhesives, thermoplastic elastomers, coatings, and sealants. 25 Currently, most thermosetting polyurethanes are still prepared from petrochemical-based polyols by reacting with an isocyanates. 26 Some hazardous petrochemical derivatives are being replaced with safer and more ecologically friendly ones based on biomass as part of new greener approaches in synthesizing polyols. 27 Polyurethanes prepared from non-renewable petroleum resources based polyols offers numerous benefits but have a low rate of biodegradation and cause an enormous risk to the environment. 26 T.W. Pechar et al. 28 developed network using soy based polyurethane with various diisocyanates and their mixtures with petroleum based polyol. The three countries USA, China, and Russia supplies the majority of the polyols to polyurethane industries in the other countries. The worldwide supply chain has recently been impacted by the COVID-19 outbreak. A prohibition on international boxing matches and import-export commerce were just two of the measures implemented to stop the COVID-19 virus from spreading globally. Such a restriction on the import and export of raw resources has a significant impact on the economy. 29 The petrochemical crisis will ultimately result from the earth crust declining oil reserves. Therefore, it is now more important than ever to concentrate on creating the innovative materials that are ecological and can be formed locally. It presents an opportunity for researchers and businesses to generate bio-based polyols and stop their reliance on petroleum-based polyols. Thus, developing bio-based polyols is meaningful for sustainability in the PU industry. 30
The novelty of the present invention is that many researchers have synthesized polyol from cardanol, but no one has attached a hydroxyl group to the phenolic hydroxyl group or a lengthy alkyl chain to the meta position of cardanol. The amount of urethane linkages is directly impacted by the number of reactive hydroxyl groups (OH) that are present on the polyol, which in turn has a significant effect on the final physical characteristic of polyurethanes. The current study deal with the investigation of novel eco-friendly, long-lasting, and cost-effective agro-industrial waste cardanol derived bio-polyol based polyurethane. The synthesized cardanol based bio-polyol consists of more numbers -OH groups. The amount of reactive hydroxyl groups (-OH) on the bio-polyol directly impacts the number of urethane linkage. Synthesized bio-polyol was used for preparation of the two different polyurethanes, which employed as a wood protective coating for construction applications. These polyurethanes demonstrate good chemical, mechanical and antibacterial property. Additionally, the synthesized PU may also utilize for various industrial applications like in textile industry, corrosion-resistant coating, paper coating, as a phase-change material, in food packaging etc.
Experiment
Materials
Cardanol was purchased from a Cardatech limited Valsad, Gujarat, India. Hexamethylene diisocyanate biuret (Desmodur N75, manufactured by Covestro) was accepted as a benefaction, from Atul Ltd, Valsad, Gujarat, India. Aromatic polyisocyanate based on toluene diisocyanate (Desmodur L75, manufactured by Covestro) was received as a kind gift from Grand Polycoats Company Pvt. Ltd. Padra. Sodium hydroxide (96%), H2O2 (30% w/v), formic acid (98%) was purchased from Finar, Gujarat, India. Epichlorohydrin (98%), anhydrous ZnCl2 (96%) were purchased from Sigma Aldrich, Gujarat, India. Sodium acetate, acetic anhydride and ethanol were purchased from local vendor.
Methods
All the synthesized compounds were characterized by using different techniques. Physicochemical properties like appearance, physical state, specific gravity, refractive index, hydroxyl value, molecular weight, and –OH functionality, various spectroscopic techniques; ATR-FTIR, 1H NMR, Mass spectroscopy, mechanical properties like pencil hardness, dry film thickness (DFT), cross cut adhesion (%), gloss, dry to touch time (min), gel time (min), water absorption (%), gel fraction (%) were characterized by following methods.
Physicochemical properties of bio-polyol
The physicochemical characteristics provide information about the composition, procedure, and focus of some specific application. Physicochemical properties like appearance, physical state, specific gravity, refractive index, hydroxyl value, molecular weight, and –OH functionality were measured for the synthesized compound.
Specific gravity
Specific gravity provides a measurement of density compared to the density of water. Specific gravity was measured by specific gravity bottle at the department of chemistry, Sardar Vallabhbhai National Institute of Technology (SVNIT), Surat.
Refractive index
Refractive index was carried out for the optical properties. Refractive index was done by Abbemat 300 Refractometer Anton Paar at the department of Chemistry, SVNIT, Surat. Refractive index was done as per ASTM D1747-09. 31
Hydroxyl value
The concentration of reactive hydroxyl (OH) groups, which directly affect functionality, is reflected by the hydroxyl value, which is crucial in the manufacture of polyols. For many different applications, such as coatings, adhesives, and foams, customizing the hydroxyl value is essential. Hydroxyl value was measured at the department of chemistry SVNIT, Surat. The pyridine-acetic anhydride method was used to assess the hydroxyl value as per ASTM-1957-86. 32
Spectroscopic analysis of bio-polyol
Various spectroscopic methods were used for the structural analysis of synthesized compounds.
Attenuated total reflectance-fourier transform infrared spectroscopy
ATR-FTIR characterization of synthesized compounds carried out in JASCO/6600 instrument, at department of chemistry, SVNIT, Surat.
Mass spectroscopy
Mass spectroscopy was carried out in the presence of CDCl3 at MNIT Jaipur.
Nuclear magnetic resonance (1H-NMR)
Nuclear magnetic resonance (1H-NMR) carried out through ECS 400 MHz instrument in the presence of CDCl3 at MNIT, Jaipur.
Mechanical properties of PU coated wood panels
Mechanical properties like pencil hardness, dry film thickness (DFT), cross cut adhesion (%), gloss, were carried out at ISTAR, Vallabh Vidyanagar, Gujarat, India. Dry to touch time (min), gel time (min), water absorption (%), gel fraction (%) were measured at the chemistry department, SVNIT, Surat.
Pencil hardness
Pencil hardness testing was done by Mitsubishi pencil. Pencil hardness was done by using ASTM D3363. 33
Dry film thickness
Dry film thickness (DFT) was measured by positector-200 (prob-3).
Cross-cut adhesion
Cross-cut adhesion was done by accordance with ASTM D3359-02. 34
Gloss
Gloss was carried out with the picoolosi-503. The gloss of the coating was observed using a digital gloss meter at 20o, 60o, 85o angle.
Water absorption
In accordance with ASTM D570, the water swelling property of film was evaluated by submerging it in water for 48 h at room temperature to examine moisture resistanceof the film. After drying, the film was weighed once again. Water absorption (WA) was determined using equation (1).
Gel content
The gel present in the cured film served as an indicator for the degree of curing. After being immersed in acetone for 48 h, the cured film was completely dry. The weight change was used to compute the gel content (% by mass).
Thermo gravimetric analysis
TGA analysis was carried out at the department of chemistry, SVNIT, Surat. TGA was carried out under N2 atmosphere with flow rate of 100 mL/min and a constant heating rate of 10°C/min utilizing instrument SDT Q 600 V20.9 Build 20, Module DSC-TGA standard equipment. The testing was carried out in an aluminium pan at temperatures ranging from 25°C to 600°C.
Thermal conductivity
Thermal conductivity of polyurethane coating was examined by C-THERM thermal conductor at department of physics, SVNIT, SURAT.
Chemical resistance performance of PU coated wood panels
Chemical resistance test like acid, alkali and salt and solvent resistance test was carried out at the department of chemistry, SVNIT, Surat.
Acid, alkali, salt and solvent resistance test
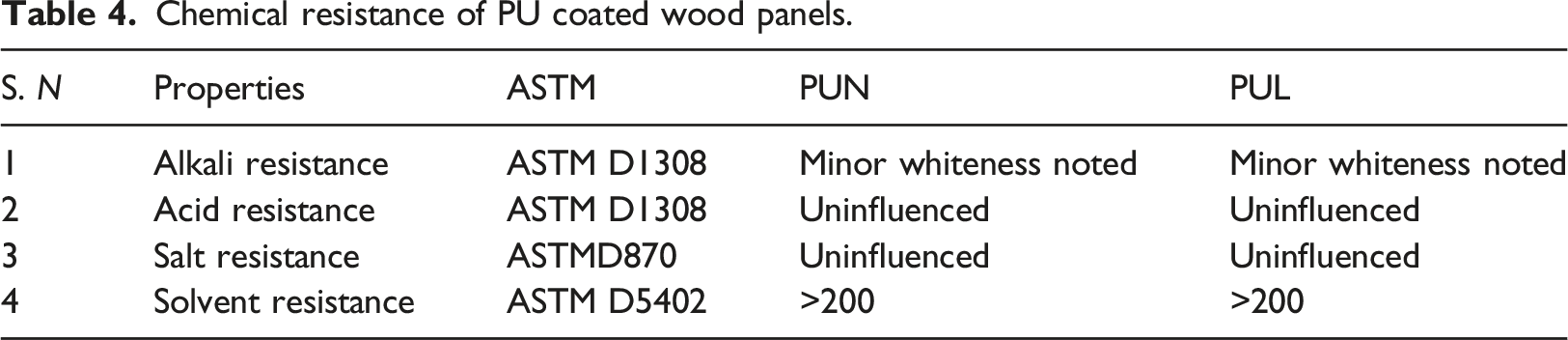
Acid and alkali resistance test were used to the evaluation of chemical resistance, according to ASTM D1308 35 and ASTMD870, 36 respectively. Coated wood panels were immersed in a 5% aqueous solution of HCl, and NaOH. After 72 h, changes in the appearance of the coating were studied. Coated wood panels were immersed in salt water. Changes in the appearance of the coating were studied after 72 h.
Solvent resistance test was carried out using methyl ethyl ketone (MEK). MEK double rub test was employed to determine the ability of PU based wood coating solvent resistance. The film was removed from the wood panels by rubbing them with a delicate cloth dampened with the solvent. The number of rubbings necessary to remove the film from the coated wood panels was recorded as the solvent resistance capability of the coated panels. MEK double rub test was done according to the ASTM D5402. 37
Beverage (tea, coffee, and soft drinks) stain resistance test
The stain resistance of the coatings was evaluated using a spot test. The beverage resistance was performed in accordance with ASTM D1308. 35
Cigarette ignition resistance
Cigarette ignition test of PU coated wood panels was done at the department of chemistry, SVNIT, Surat. A burning cigarette was placed on the surface of the coating for an appropriate time to assess the coated wood durability against cigarette ignition, and any destruction or changes in the PU coated wood panels appearance was observed.
Surface morphology
Surface morphology was used for the surface characteristics including smoothness, roughness, texture, patterns, and irregularities. Surface morphology was done at mechanical department, SVNIT, Surat.
Antibacterial properties
Antibacterial property of the prepared polyurethanes was measured by zone of inhibition using disk diffusion method against microbial strains known as Gram-positive Stayphylococcus aureus, Stayphylococcus pyogenes, Streptococcus pneumonia, and Gram-negative bacteriaPseudomonas aeruginosa, Escherichia coli. 38
Synthesis of bio-polyol
In the present work bio-polyol was synthesized in the following three different steps. (1) Synthesis of mono glycidyl ether from cardanol (2) Synthesis of triol from monoglycidyl ether (3) Synthesis of bio-polyol from triol
The detail synthetic process carried out as per follows.
Synthesis of monoglycidyl ether from cardanol
In the present study, monoglycidyl ether was synthesized from cardanol. Epichlorohydrin and cardanol were reacted in the presence of alkaline conditions to yield epoxide. 500 mL round bottom flask (RBF) with a mechanical stirrer, thermometer, and dropping funnel was used for the reaction synthesis. Cardanol, 119.4 g (0.4 mol) was heat up to 95°C. Followed that, anhydrous ZnCl2, 0.12 g (0.1%) was added. Theoretical quantity of epichlorohydrin, 37.3 mL (0.48 mol) was added drop by drop by keeping temperature constant. The reaction was extended for 2-3 h. The stoichiometric amount of NaOH (19.3 g/100 mL water) was added drop by drop. The reaction temperature was increased to 100°C, and it was maintained for 2-3 h. The product was separated and washed with excess water to remove the byproduct sodium chloride and other unreacted reactants. It was then dried using anhydrous sodium sulfate. 39 The isolated yield was 87%. Monoglycidyl ether from cardanol was synthesized according to scheme 1.
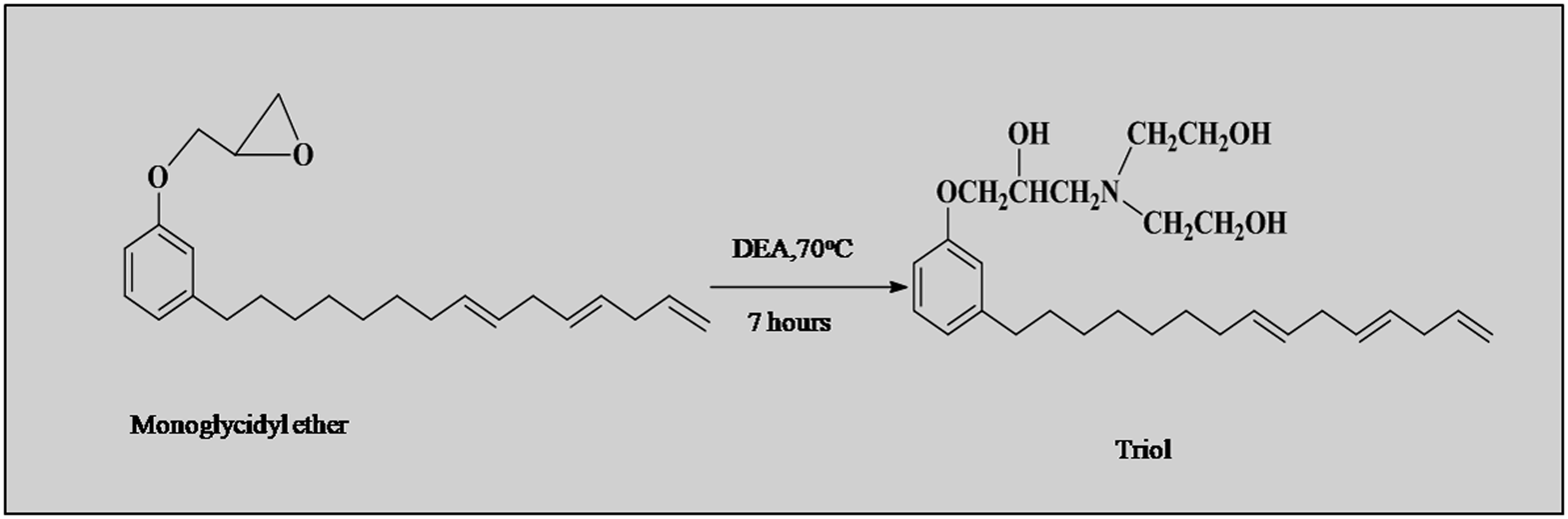
Synthesis of triol from monoglycidyl ether
The triol was synthesized by reacting monoglycidyl ether with diethanolamine at 70°C in the presence of ethanol. In a typical procedure, monoglycidyl ether, 10 g(0.028 mol) was combined with an equal weight of ethanol and a molar excess of diethanolamine 3.49 g (0.033 mol) in a 250-mlRBF equipped with a mechanical stirrer, thermometer and reflux condenser. Ethanol was removed from the product after 7 h using a rotary evaporator. The product was separated and rinsed with water-ethanol mixture (1:1) before being washed with water to remove excess diethanolamine. It was dried with anhydrous sodium sulfate. 39 The isolated yield was 83%. Triol was synthesized from monoglycidyl ether of cardanol as illustrates in scheme 2.
Synthesis of bio-polyol from triol
The bio-polyol of cardanol from triol was synthesized using two steps (1) oxidation, and (2) saponification. Epoxide primarily formed as intermediates which react with excess organic acid to produce the hydroxyl formoxy ester. During the oxidation, the hydroxyl formoxy ester formed, and then the product was directly subjected to hydrolysis. In the presence of sodium acetate, hydroxyl formoxy ester is converted to a bio-polyol (Scheme 3). Synthesis of monoglycidyl ether from cardanol. Synthesis of triol from monoglycidyl ether.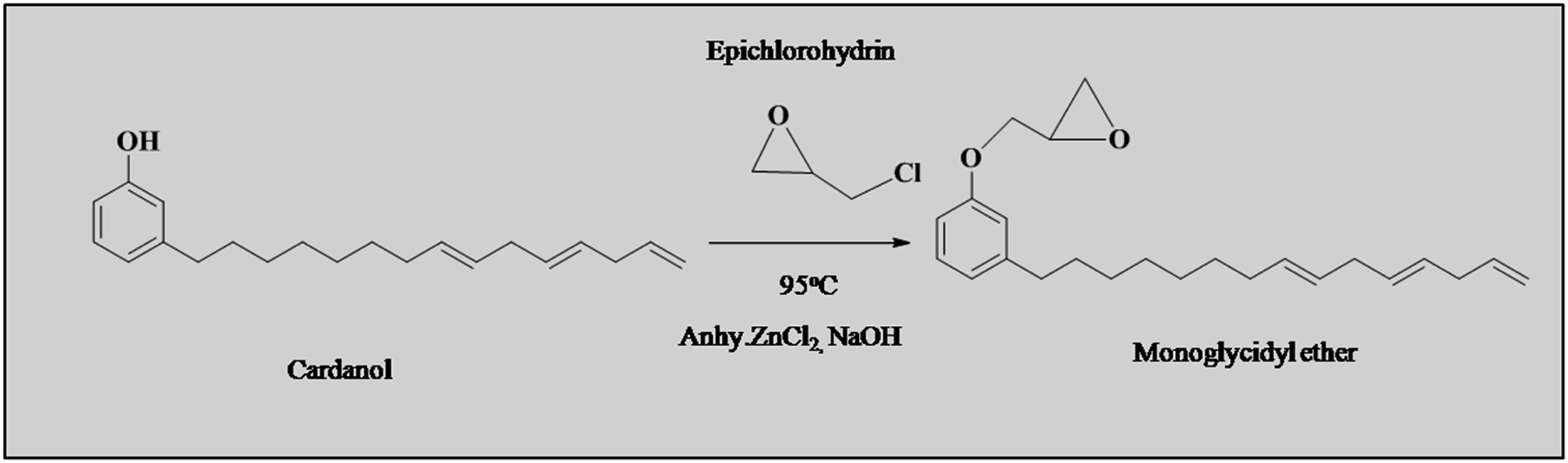
For the cardanol-based polyol, studies on per acid oxidation of triol have not been published. One of the most used oxidation processes in the fine chemical sector is the per acid oxidation of olefins. These oxidants are produced in situ when carboxylic acid reacts with aqueous hydrogen peroxide. High active oxygen content, low cost, high oxidation potential and the creation of just water as the reaction by-product are all advantages of utilizing hydrogen peroxide as an oxidant. There was no attempt was made to separate the intermediate (Scheme 3). Synthesis of bio-polyol from triol.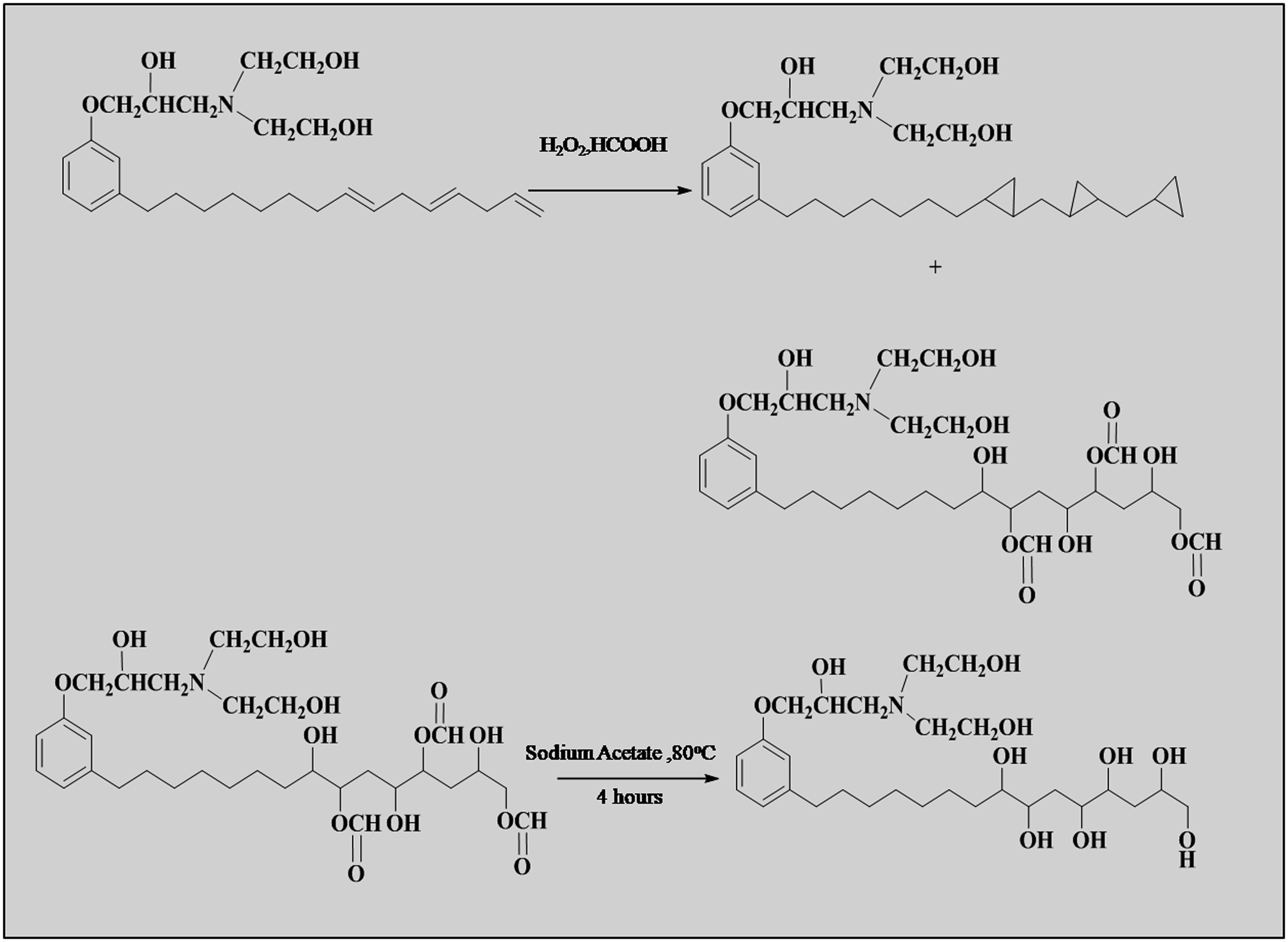
In the present work bio-polyol was synthesized from triol, hydrogen peroxide and formic acid. In a glass vessel, formic acid, 7.82 g (0.17 mol) and triol, 79 g (0.17 mol) were combined and cooled to 0°C. The H2O2 38.56 g (0.34 mol) was added drop by drop with stirring to keeping constant temperature. The reaction mass was stirred for 24 h at of 0°C.
Saponification was carried out using a hydroxyl formoxy ester with 10% sodium acetate solution. The reaction was carried out at 75 °C–80 °C for 4 h. The product was washed and dried on anhydrous sodium sulfate after being neutralized. 40 The isolated yield was 80%.
Figure 4(d) and (e) shows FTIR spectra of oxidized triol (hydroxyl formoxy ester), before and after hydrolysis (bio-polyol). Unsaponified product exhibits ester carbonyl absorbance emerging with free acid at 1712 cm−1.The half-ester group will transform into the hydroxyl group during saponification. Thin layer chromatography (TLC) was used monitor progress of the reaction. The product was purified by using silica gel (60-120) chromatography. FTIR spectra of (a) cardanol (b)monoglycidyl ether (c) triol (d) hydroxyl formoxy ester (e) bio-polyol.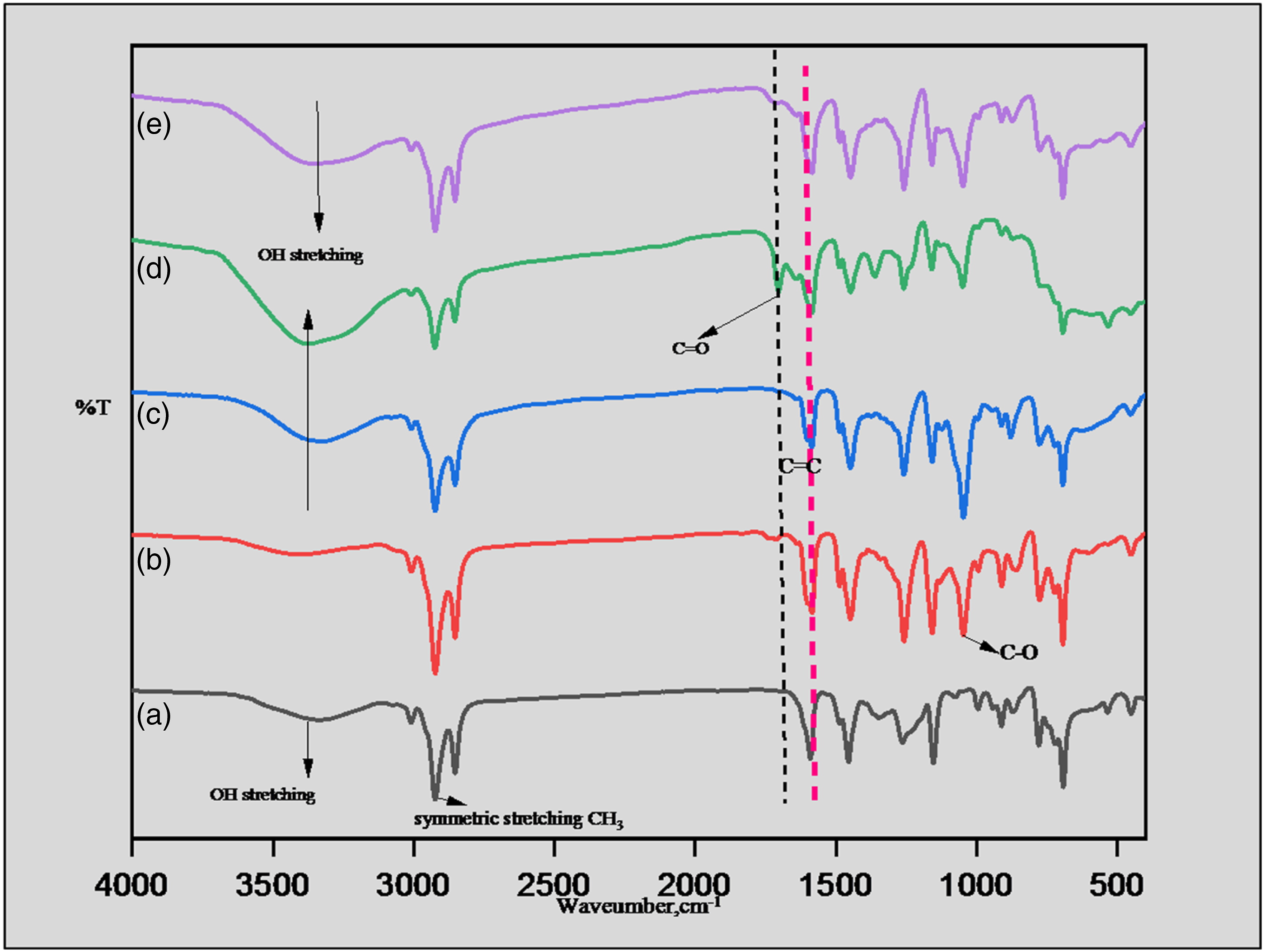
Formulation of polyurethanes
In the present study polyurethanes were formulated from synthesized bio-polyol and aliphatic/aromatic isocyanates for wood protective coating. Polyurethane formulation was done by mixing bio-polyol with aliphatic isocyanate (Desmodur N75)/aromatic isocyanate (Desmodur L75) in 1:1 ratio. The mixture was allowed stand at ambient temperature until it reached to pourable viscosity. The polyurethane formulated from bio-polyol and Desmodur N75 was denoted wereas PUN. The polyurethane formulated from bio-polyol and Desmodur L75was denoted as PUL.
Sample preparation
Treating a wood surface for coating entails several of procedures for effective adhesion, durability, and an optimal finish. To provide the smooth surface of wood panels, 80 and 150 grit sandpaper were applied. Followed that, surfaces of the 4 × 6 inch wood panels were treated with a sanding sealer. After achieving the pourable viscosity of formulated polyurethanes, the mixture was brushed onto the prepared wood panels, and under the visual examination, the coating was allowed to cure at ambient temperature.
Results and discussion
This study addressed the development and implementation of a cardanol derived polyurethanes-based wood protective coating. As a protective coating for wood surfaces, the synthesized bio-polyol based polyurethanes showed good results. The coating established an incredibly durable and malleable layer. If the formulation of the coating continues to be improved and its mechanical and chemical properties are examined, the industry may be able to utilize it systematically.
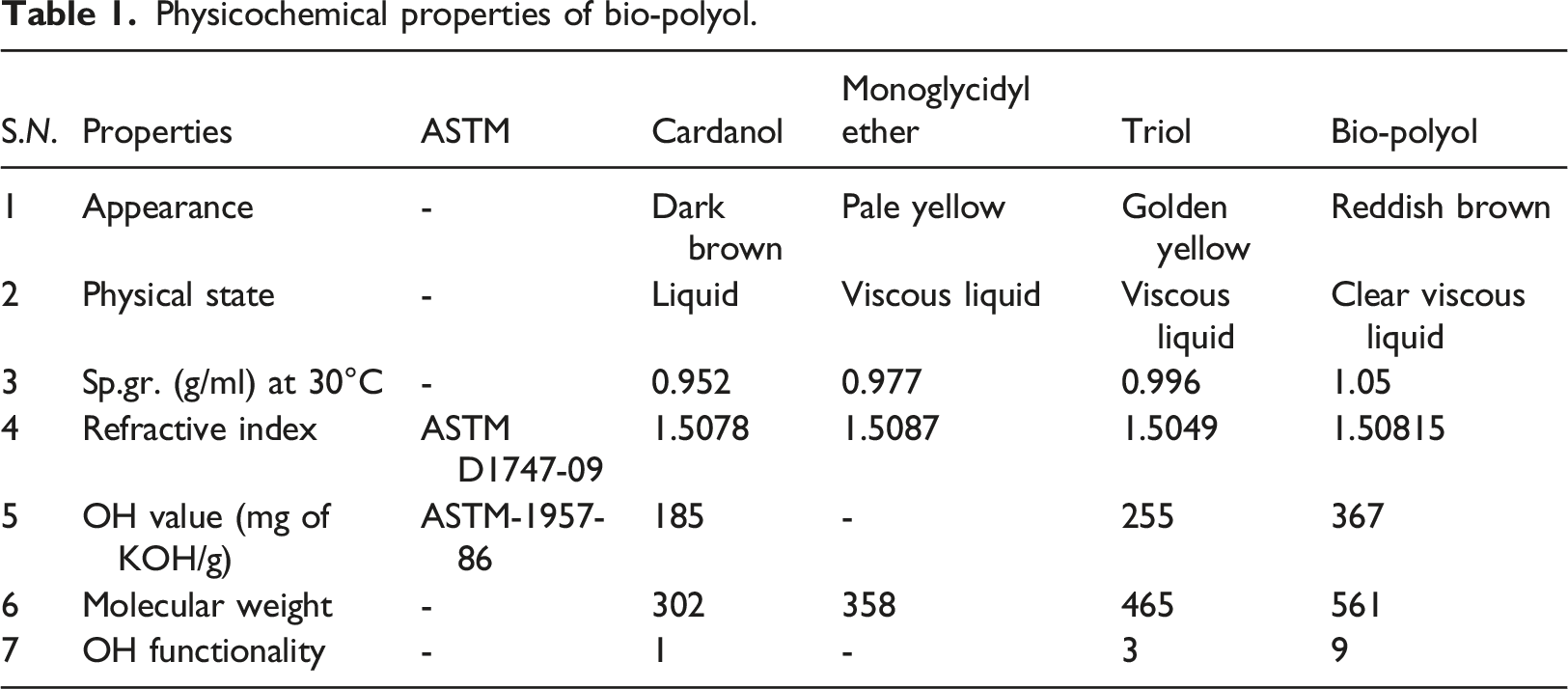
Physicochemical properties
Physicochemical properties of bio-polyol.
Spectroscopic analysis of bio-polyol
ATR-FTIR characterization
The FTIR spectrum of cardanol depicted in Figure 4(a), the broad band at 3361 cm−1 due to the -OH stretching. The spectrum at 2851 cm−1and 2923 cm−1 was because of stretching of -CH2 and -CH3, correspondingly. The band at 1352 cm−1 and 1421 cm−1 were of -CH2 and –CH3 bending vibrations. The peak at 1567 cm−1 is due to vibration of the aromatic C=C linkages. Monoglycidyl ether of cardanol was confirmed by FTIR spectrum Figure 4(b), the characteristic absorption bands at 1045 cm−1, indicating the formation of the phenolic ether linkage. Triol Figure 4(c) was confirmed by reappearing hydroxyl peak and C-N stretching at 1223 cm−1. Hydroxyl formoxy ester Figure 4(d) and bio-polyol Figure 4(e) was assigned by the carbonyl peak at 1712 cm−1.
Mass spectroscopy
Mass spectroscopy of synthesized cardanol based bio-polyol was carried out in the CDCl3solvent.In mass spectral studies (Figure 5), the high intensity peak for cardanol based bio-polyol has been used to confirm the molecular formula. Bio-polyol consist calculated molecular weight is 561 and the high intensity peak at m/z 561 represents peak with an intensity of about 100% of the compound. The MS spectrum shows intense peaks at m/z 560 (M-1) and 562 (M + 2) which is corresponding to bio-polyol. Mass spectra of bio-polyol.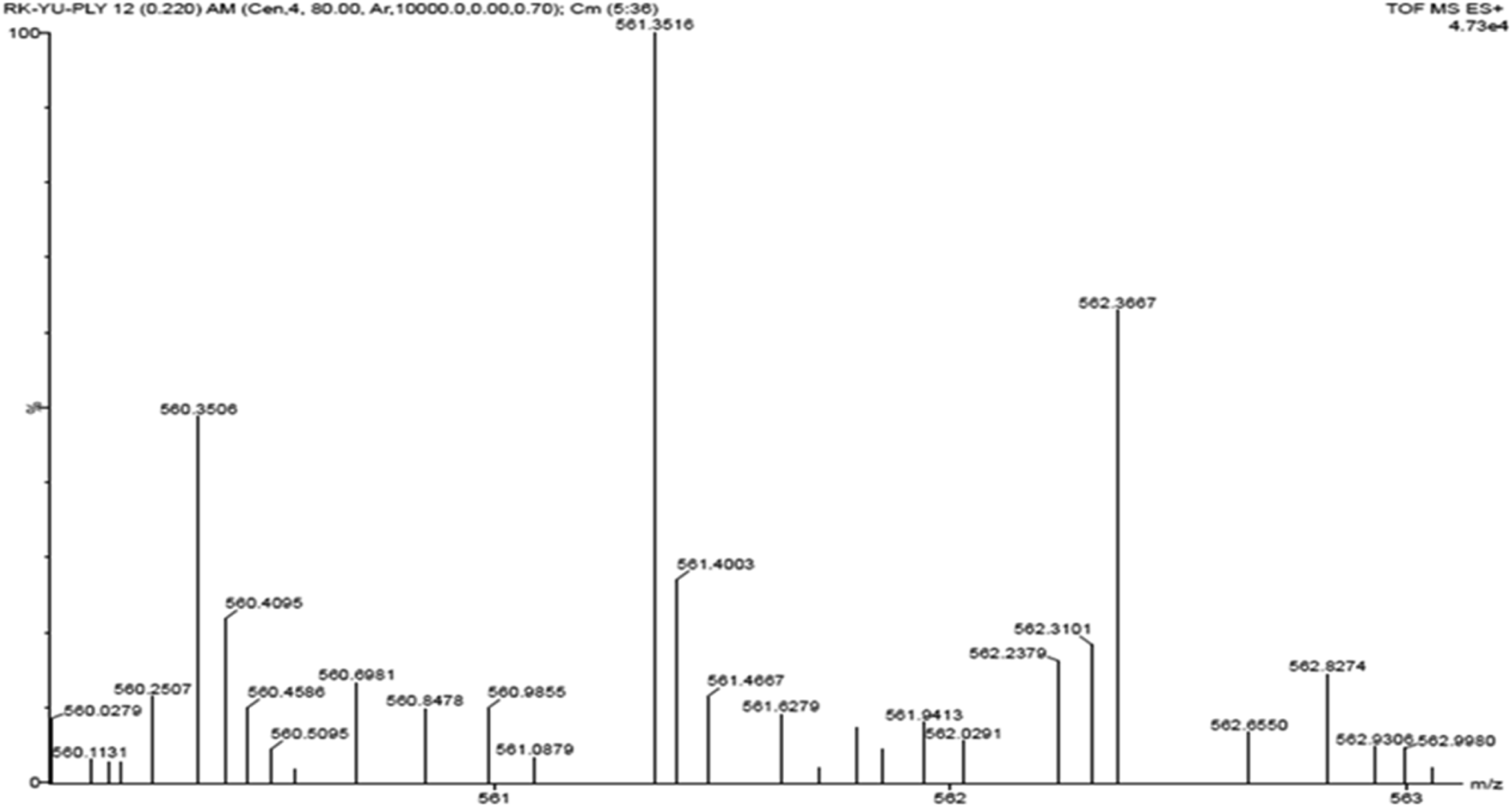
Nuclear magnetic resonance (1H-NMR)
The 1H –NMR spectroscopy shows the spectra of hydroxyl (-OH &H α to hydroxyl) in the range δ 3.28-5.5 ppm Figure 6. Aromatic proton shows spectra at δ 6.58-7.11 ppm.0.80-2.77 ppm range shows the characteristic for –CH2 (methylene of alkyl chain). Compound shows peak around δ 8.05 ppm for aldehyde proton but, herein no peak observed around this range which identifies the conformation of bio-polyol.
41
1H NMR spectra of bio-polyol.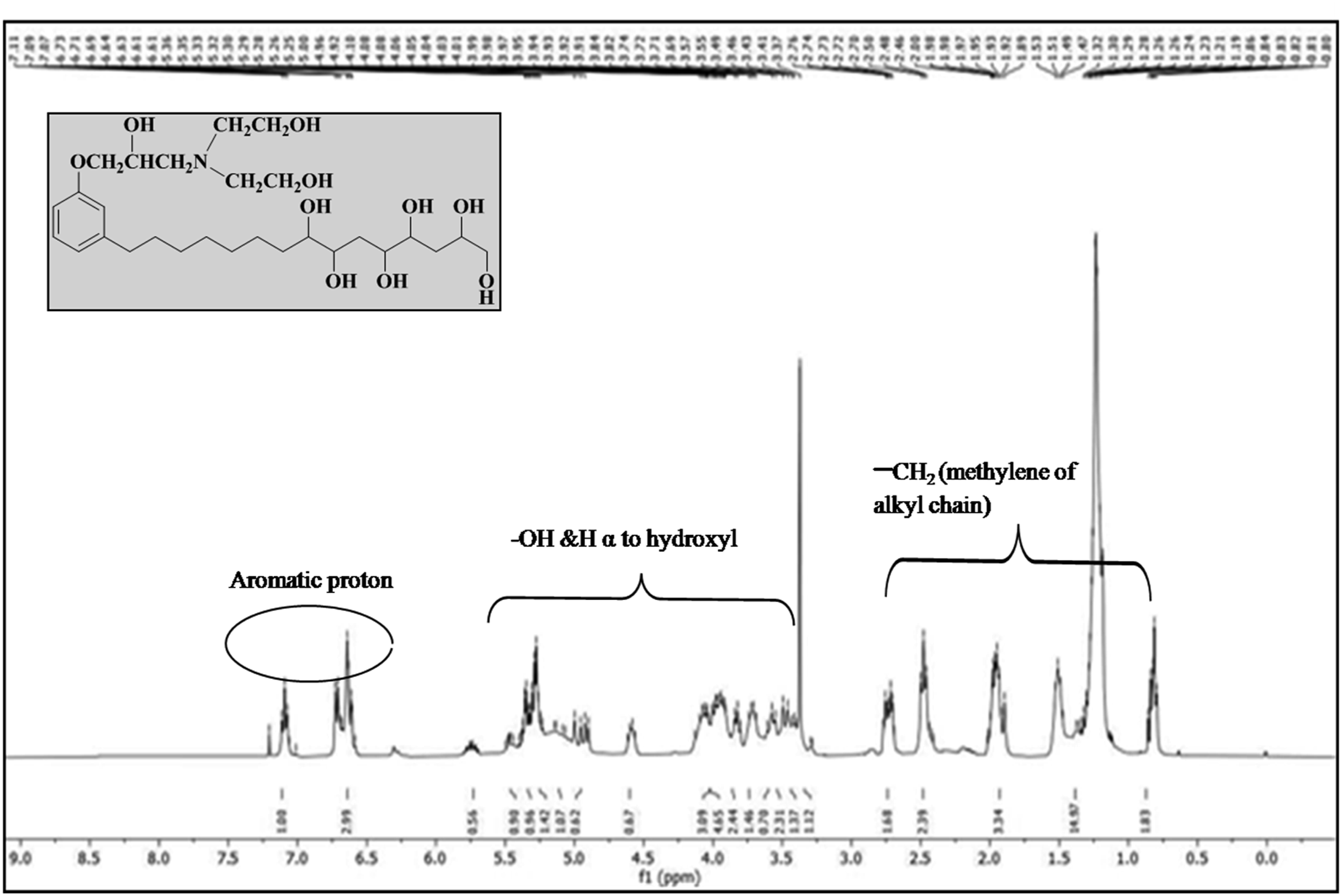
ATR-FTIR characterization of PU coated wood panels
Figure 7 depicts the FTIR spectra of PUN Figure 7(a) and PUL Figure 7(b). The presence of amide carbonyl was confirmed by the occurrence at 1687 cm−1 and 1679 cm−1 owing to the –C=O stretching (urethane linkage) in PUN and PUL respectively. The spectrum observed around up to 3000 cm−1 in PUN and PUL due to –NH stretching. Spectra observed at 1260 cm−1, 1254 cm−1 due to the N-H bending in PUN and PUL respectively.42,43 ATR-FTIR spectra of (a) PUL (b) PUN.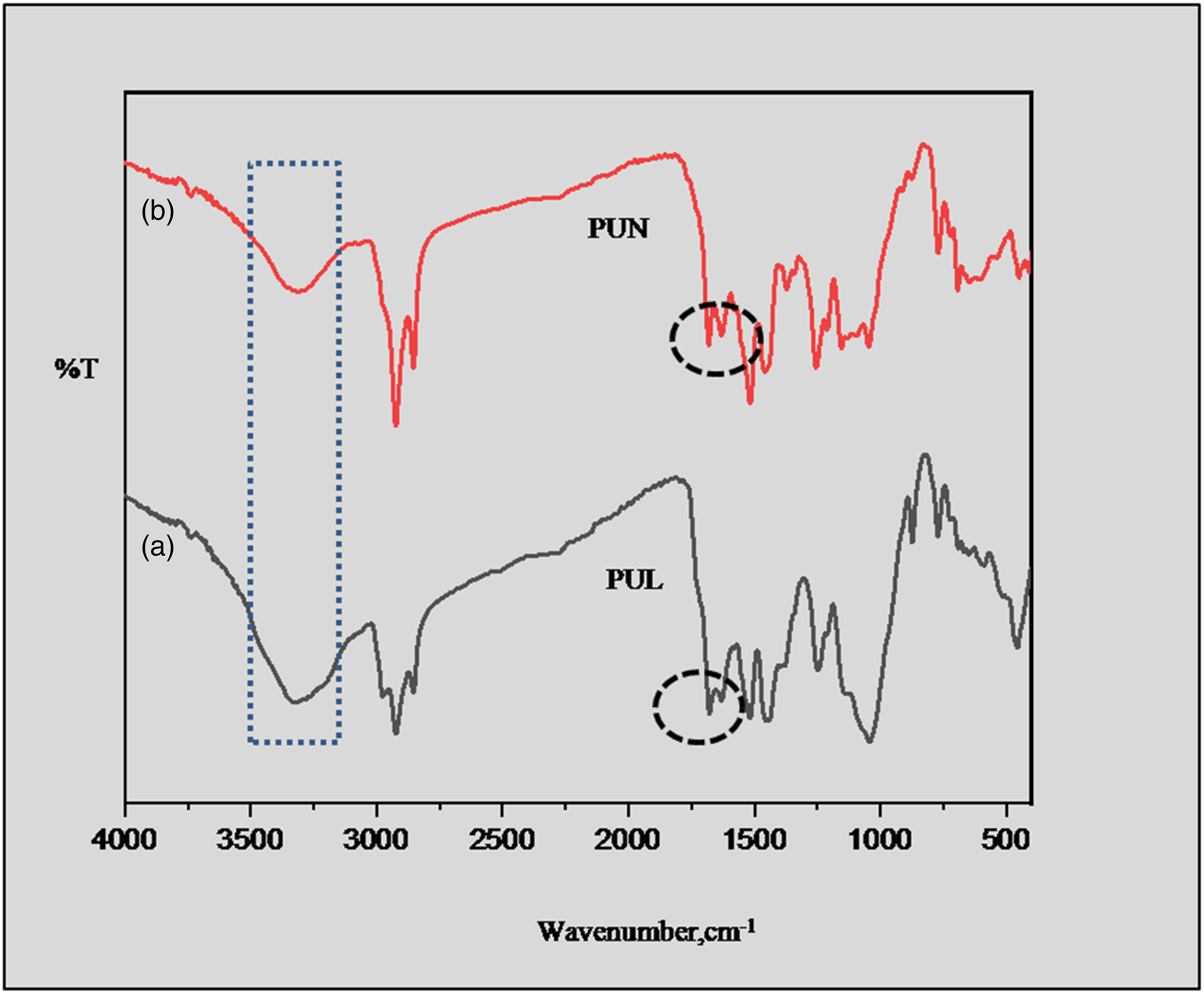
Performance properties of PU coated wood panels
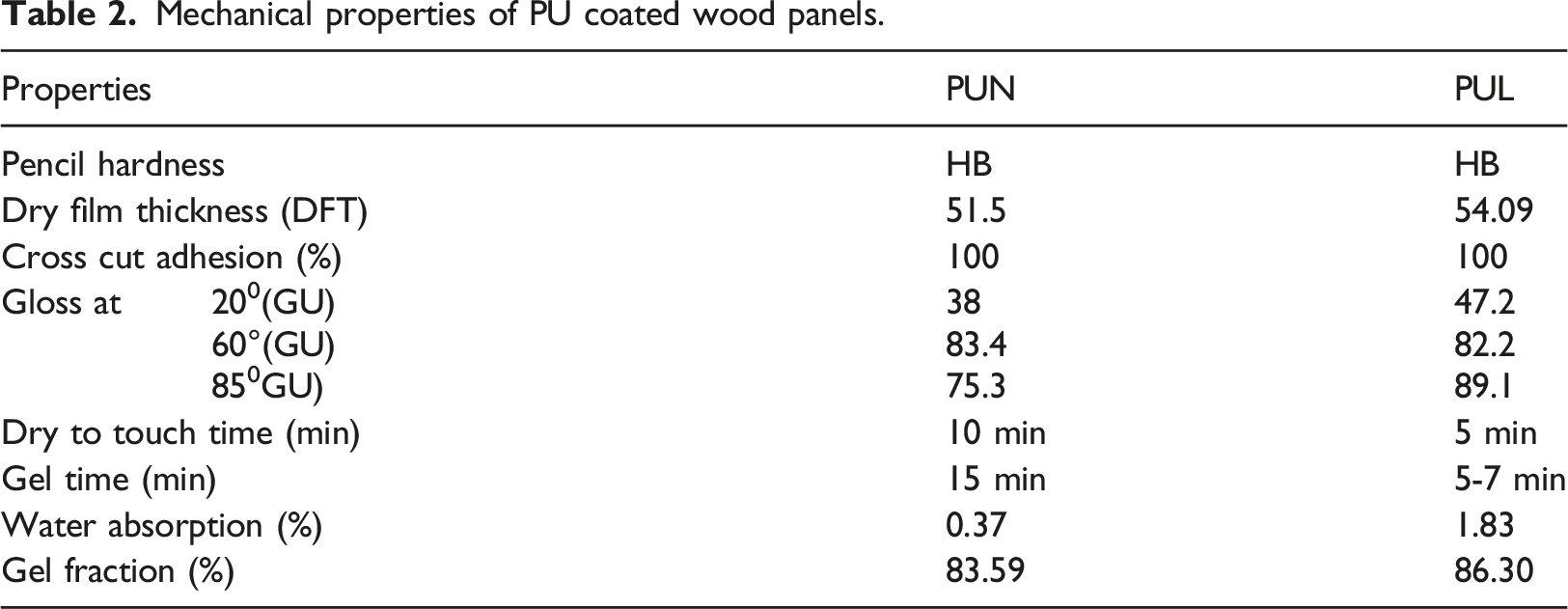
Mechanical properties
Mechanical properties of PU coated wood panels.
Thermogravimetric analysis
TGA is the most widely used method for determining thermal stability of polymer. PUN and PUL showed pattern of thermal behavior with two thermal degradation steps.
The initial phase of degradation was detected at temperatures in the range of 201°C–285°C for PUL and 202°C–350°C for PUN. Figure 8. This might be because the urethane bond (–NHCOO–) cleavage occurred at these temperatures, resulting in a weight loss of 37% for PUL and 23% for PUN. The second stage of deterioration was found at temperatures ranging from 285 to 480°C and 345 to 490°C, with a weight loss of 55% and 46% for PUN and PUL, respectively. This weight loss might be caused by the degradation of the amide and hydrocarbon chains. Degradation of polyurethanes at higher temperatures demonstrates greater thermal stability of the polymer; this may be attributed to bio-polyols going through thermal cross linking at high temperatures.44–48 (a) TGA and (b) DTG graph of PU coated wood panels.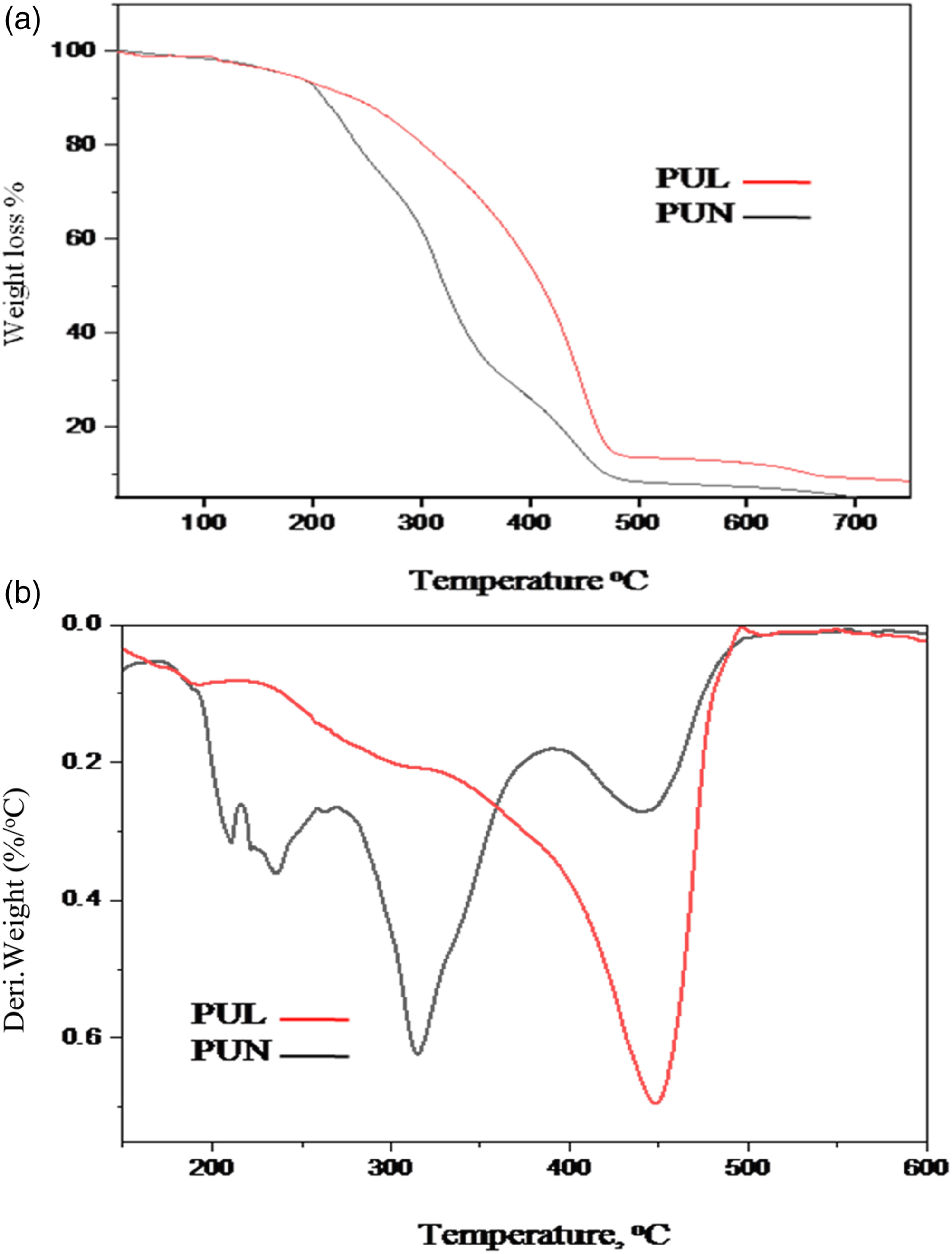
Thermal conductivity
Thermal conductivity is a measurement of materials ability of heat transfer. According to previous research that the effective thermal conductivity of the polyurethane varied between 0.020 W/m K and 0.030 W/m K
23
This study investigated the thermal conductivity 0.01966 W/m K for PUN and 0.03066 W/m K for PUL. The thermal conductivity of the manufactured polyurethanes has almost identical with other polyurethanes prepared from vegetable oils. From the thermal conductivity measurement both polyurethanes demonstrate good insulating properties. Figure 9 revealed thermal conductivity study of PU coated wood sample. Table 3 Thermal conductivity analysis of PU coated wood sample. Thermal conductivity of PU coated wood panels.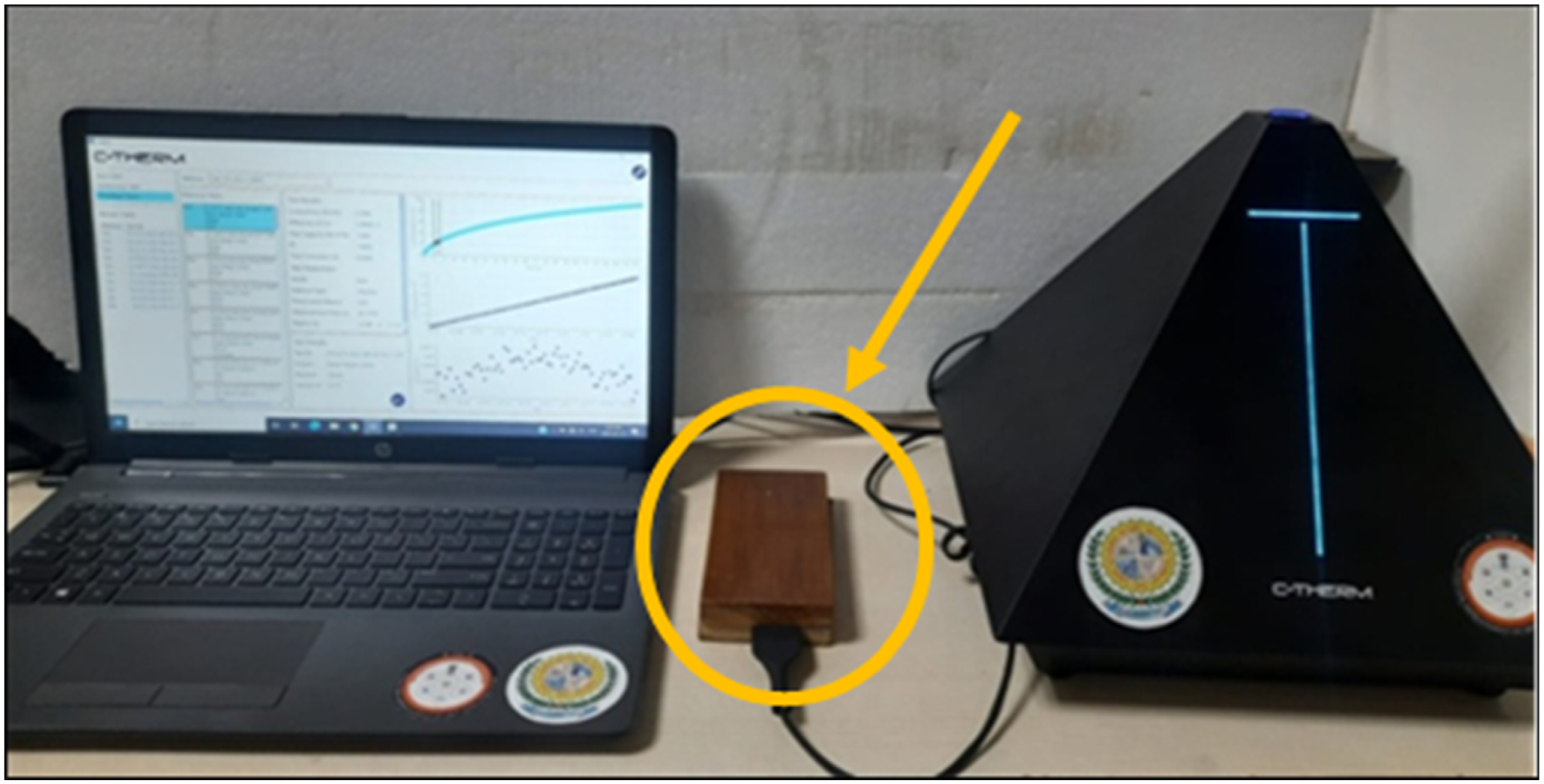

Chemical resistance test of PU coated wood panels
Acid, alkali, salt and solvent resistance test
Chemical resistance of PU coated wood panels.
Beverage (tea, coffee, and soft drinks) stain resistance test
Stain resistance is a term used to describe the ability of a surface for preventing becoming stained when comes into contact with liquids. Preventing liquid absorption is the fundamental objective of stain resistance. Beverage test of PU coated and uncoated wood panels carried out as Figure 10. The assessments of the liquid stain resistance for tea, coffee, and soft drinks were positive; moreover, PU-based wood coatings are easy to clean, and after wiping, there were no stains visible. While a spot was seen on the untreated surface of wood. Present result is similar with the reported study.
43
Beverage test of PU coated and uncoated wood panels.
Cigarette ignition resistance of PU coated wood panels
The performance of furniture under the conditions of exposure to a burning cigarette is assessed using the cigarette ignition resistance test Figures 10 and 11. The cigarette ignition resistance test was done as shown in Figure 11. The cigarette ignition resistance test was done by employing a burning cigarette on the surface of the polyurethane at different contact periods (1s, 10 s, and 30 s). If the contact period was for 1s or 10 s, the burning cigarette showed no effect on the PU coated wood panels. However, when the contact period 30 s, PU coated wood panels could be seen as a little black spot in both prepared coatings. This result is corresponds with reported literature.
49
Cigarette ignition resistance of PU coated wood panels.
Surface morphology of PU coated wood panels
Figure 12 shows the surface morphology of PU coated wood panels. Surface morphology demonstrated the consistency of the PUN coating on the wood panel without any surface voids being created, while some gaps were seen in the PUL coating that were formed by CO2 bubbles. The three distinct layers were seen in the cross-section of PU coated wood panels. The thickness of the PU coated wood panel outermost layer is about 528 μm for PUN and 20.3 μm for PUL. The thickness of the primer coat was determined around 520 μm for PUN and 8.34 μm for PUL. This suggests that the primer coat provides the basis for achieving a homogenous PU based wood coating. Surface morphology of (a) PUN coated wood panel surface (b) PUN coated wood panel cross section (c) PUL coated wood panel surface (d) PUL coated wood panel cross section.
Antibacterial properties of PU coated wood panels
Antibacterial properties of PU coated wood panels.
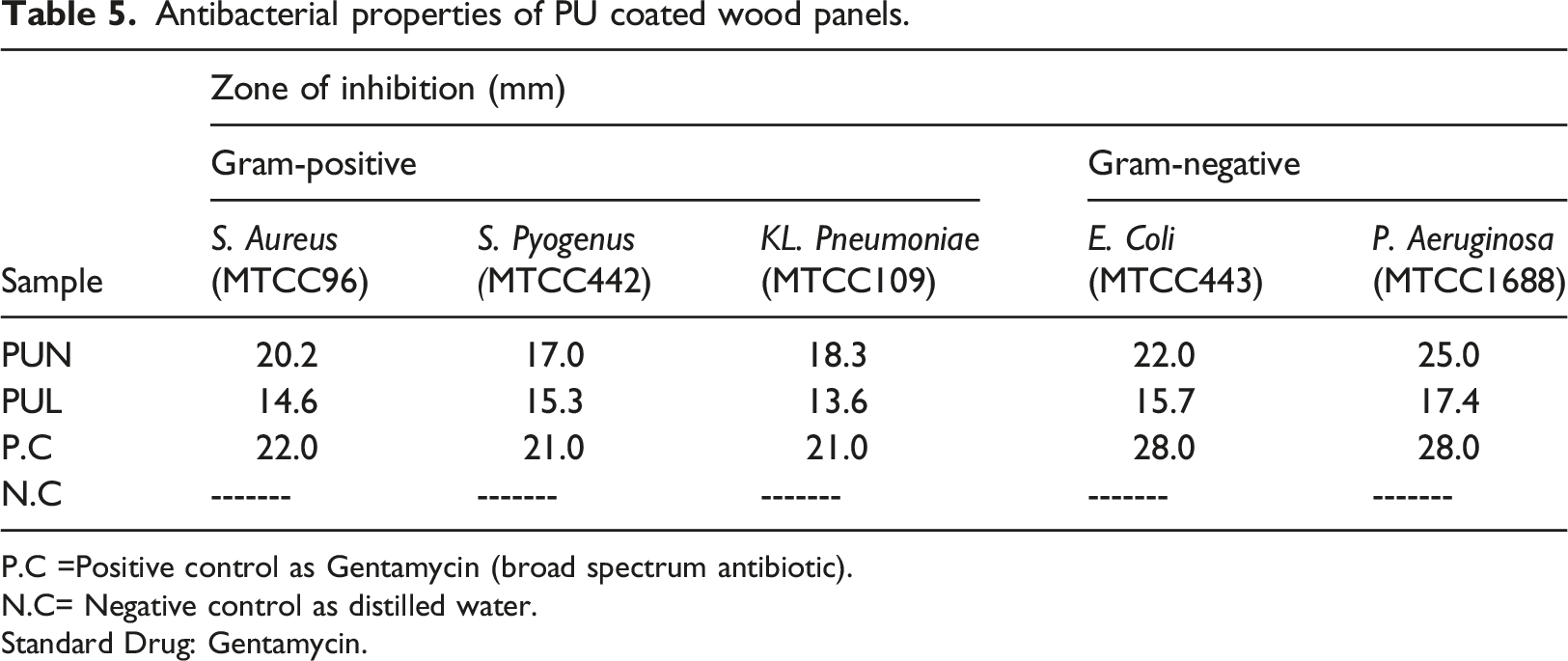
P.C =Positive control as Gentamycin (broad spectrum antibiotic).
N.C= Negative control as distilled water.
Standard Drug: Gentamycin.
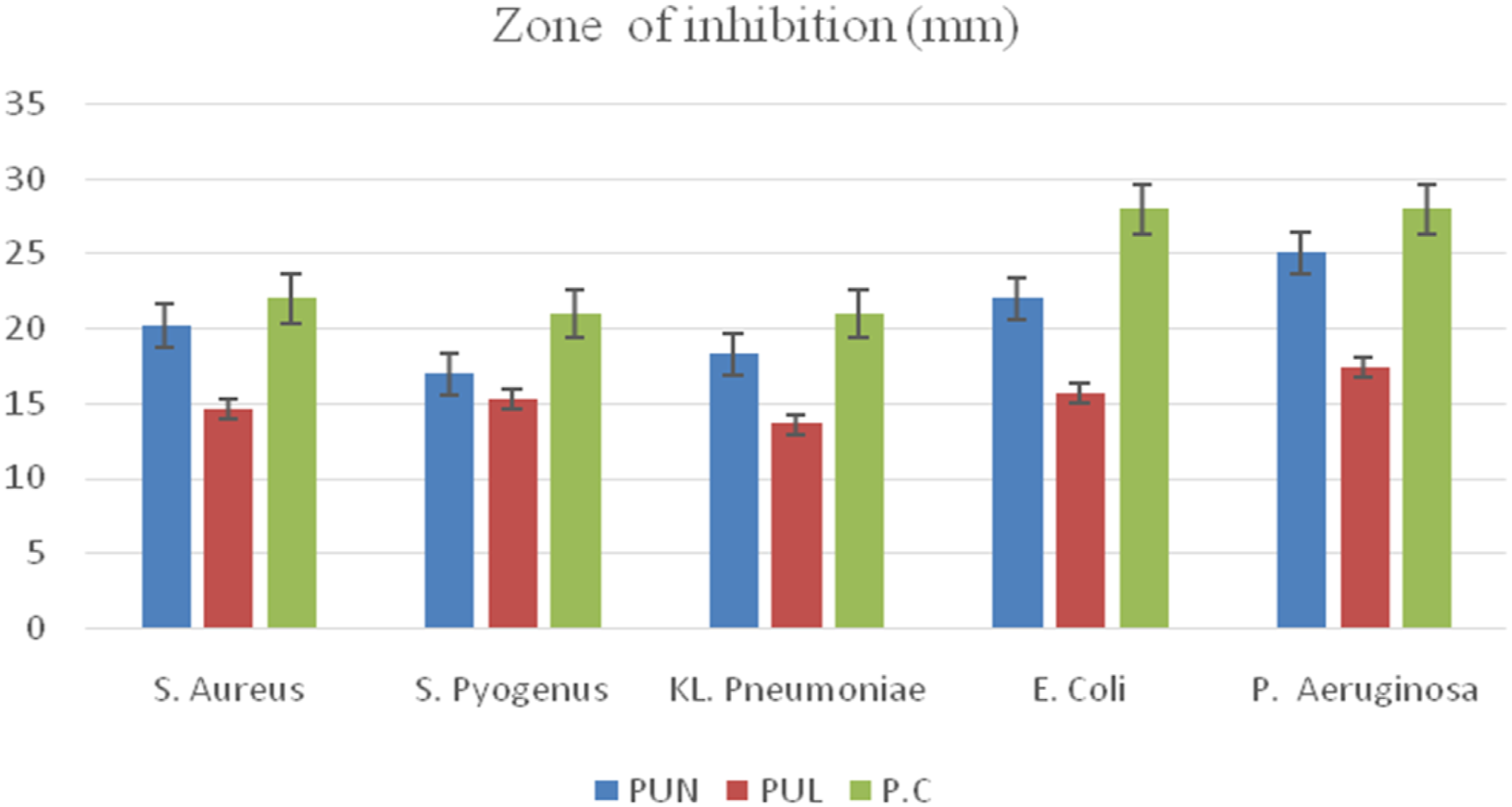
Antibacterial properties of PU coated wood panels.
Conclusion
The current trends in the coating industries is the substitution of petroleum-based materials by bio-based materials because of renewable, abundantly availability, cost-effective, biodegradable and non-toxic nature of bio-based materials. In the present work, cardanol as a renewable and economical resourcewas successfully employedto produce bio-polyolby per acid oxidation of triol and an attempt is made first time to separate the intermediates. The novel bio-polyol has been successfully prepared with high hydroxyl number and -OH functionally by reacting cardanol at phenolic site as well as at unsaturation sites of long alkyl fatty acid chain. ATR-FTIR, 1H-NMR and mass spectroscopy were used to confirm the bio-polyol. The obtained bio-polyol was evaluated and employed in the polyurethane based wood coating system. Different performance characteristics of the produced polyurethanes were investigated. The manufactured polyurethanes were found to have good mechanical, thermaland antibacterial properties. The polyurethanes that have been developed have a significant resistance to chemicals. The cardanol based bio-polyol has the potential to be a sustainable and environmentally friendly alternative to synthetic wood preservatives for building and construction. The present invention enhances the antibacterial activity of wood coating. The present bio-based polyurethanes wood coating can be used for various residential, commercial, and industrial construction applications.
Footnotes
Acknowledgements
The author thanks S. V. National Institute of Technology for providing the required research facilities. The author also thanks the Department of Surface Coating Technology, Institute of Science and Technology for Advanced Studies and Research (ISTAR) Vallabh Vidyanagar, Gujarat, India, for their assistance. MNIT Material Research Centre (MRC) Jaipur, India. Microcare lab., Surat, India for providing analytical facilities. The authors are grateful to Atul Ltd in Valsad, India, for providing the necessary materials for the research.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.