Abstract
With oil supplies, needed for plastic production, decreasing dramatically, there is a clear driver for alterative polymers from sustainable resources. Poultry feathers, containing ∼90% keratin, are one source of natural polymer with huge potential for biopolymer production. However, the presence of crosslinks, known as disulphide bonds, hinders processability. This paper reviews techniques to enable breakage of disulphide bonds through use of reduction agents (sodium sulphite and sodium sulphate) and hydrolysis. Samples were analysed using FTIR and DSC to quantify achievable bond breakage, effect on thermal properties and changes in protein concentration. A review on the effect of particle size on disulphide bond breakage was also conducted, along with quantifying the reformation of bonds post-processing. Finally, a bicinchoninic acid (BCA) protein assay was used to quantify changes to soluble protein content, key to predicting if biopolymer formation can occur. The results showed a final disulphide bond breakage of between 48% and 67% was achievable using these techniques. It was also shown that disulphide bond content exhibited up to 60% bond reformation post treatment. These reductions in disulphide bonds increased the thermoplastic nature and apparent protein content. Despite achieving the highest bond breakage percentage, hydrolysis caused degradation of useful proteins, rendering the material unsuitable for biopolymer production. Results suggested that treatment with sodium sulphite (4.3% wt. of feathers) and use of a small particle size (0–100 µm), sufficiently altered the properties of raw feathers to enable feather biopolymer production.
Introduction
Synthetic polymer production in the first decade of the 21st century has exceeded that from the previous 100 years combined. 1 In 2018, 6.9 billion barrels of oil were used globally to produce around 359 million tonnes of plastics. 2 This high oil use is due to the fact that 1.74 kg of crude oil is needed on average to produce 1 kg of raw plastic material. 3 This is caused by the high amounts of refining and processing needed to transform raw feedstock into a final material. It is this excessive need for petroleum oil feedstock that is creating the push for a reduction in plastic use. While there are various reported oil depletion scenarios (ranging from 15 to 150 years), there is agreement that it is a finite resource.4-6 This problem has led material scientists to explore the development of alternative polymer materials, many derived from waste products or sources to replace conventional oil-based polymers.7,8 These include biopolymers, defined a polymers derived from natural, globally available biomaterials (living organisms).7,9 Such materials include banana fibres, forestry waste, rice husk ash, poultry eggshells and litter ash10-13 which have been successfully transformed into new polymer materials. They are renewable, abundant, sustainable and often require no oil-based feedstock.14,15
Unlike the processing of crude oil into plastics, biopolymer production can require less energy and is considered more eco-friendly.16,17 A plastic PET bottle can take up to 450 years to degrade, 18 whereas biopolymers like polylactic acid (PLA) or those derived from waste products can degrade in around 6 months.8,19 The benefits of biopolymers now meant new companies, such as NatureWorks-PTT, Biomer and Total-Corbion,20-22 have been set up producing exclusively biopolymers like PLA and polyhydroxyalkanoates (PHA) from natural resources derived from crops (usually used as food sources).23,24 This has resulted in growth of crops purely for biopolymer production rather than for food, which has led to ethical issues with this type of polymer generation. 25 Researchers now aim to instead use materials that are currently considered as waste materials from a range of industries.
One such material that has been considered for biopolymer production is poultry feathers. The popularity for poultry meat products, along with advances in industrial technologies, means the poultry industry is the fastest growing agri-food sector worldwide. 26 Production globally has increased dramatically over the past decade with 42 billion more chickens being processed in 2018, compared to 2002. 27 This has resulted in an increase in the volume of poultry waste being generated, including poultry feathers. Around 5.5% (0.112 kg) of a chicken’s weight is feathers. 28 Annually 6.3 million tonnes of poultry feathers are produced as a waste product worldwide. Feathers are currently used for composting, fertiliser and to produce pet food or activated carbon,29,30 processes which do not utilise the full potential. Poultry feathers consist largely of keratin, which exists as a natural polymer31,32 and amounts for up to 90% of its dry weight. 33 The remainder consists of inorganics including compounds of sulphur, calcium and phosphorus.34,35 Feathers consists by weight of 50% quill (the hard stem of the feather) and 50% fibres (the light and soft branches), 36 both of which have different structures. Research has already demonstrated the feasible use of poultry feathers as a polymer filler with a range of plastics such as polypropylene, polyethylene and PLA37-39 and provided improvements in tensile and flexural properties in loadings of up to 55% wt. 37 Feathers as natural fibres are non-toxic and can reduce production costs. 40 The potential of feather keratin as a feedstock for biopolymer production, however, has not been fully realised.
Disulphide bonds in poultry feathers
However, the presence of disulphide bonds in feather keratin is a major hinderance to biopolymer formation.41,42 Disulphide bonds support all the higher level protein bonds and maintain protein structural integrity.
43
Without these bonds the secondary structures would lose their supporting links causing unfolding, leading to changes in properties and characteristics. Disulphide bonds (S–S) are generated by the reaction of two cysteine amino acids with oxygen, with water as a by-product.
44
The existence of disulphide bonds creates a crosslinking effect within feather protein restricting the ability for bonding (enabling polymer formation) by trapping useful material which would risk damaging equipment if processed in this manner. Verbeek and Van Den Berg
42
outlined that: To produce a thermoplastic material from proteins, cross-linking and non-covalent interactions have to be controlled.
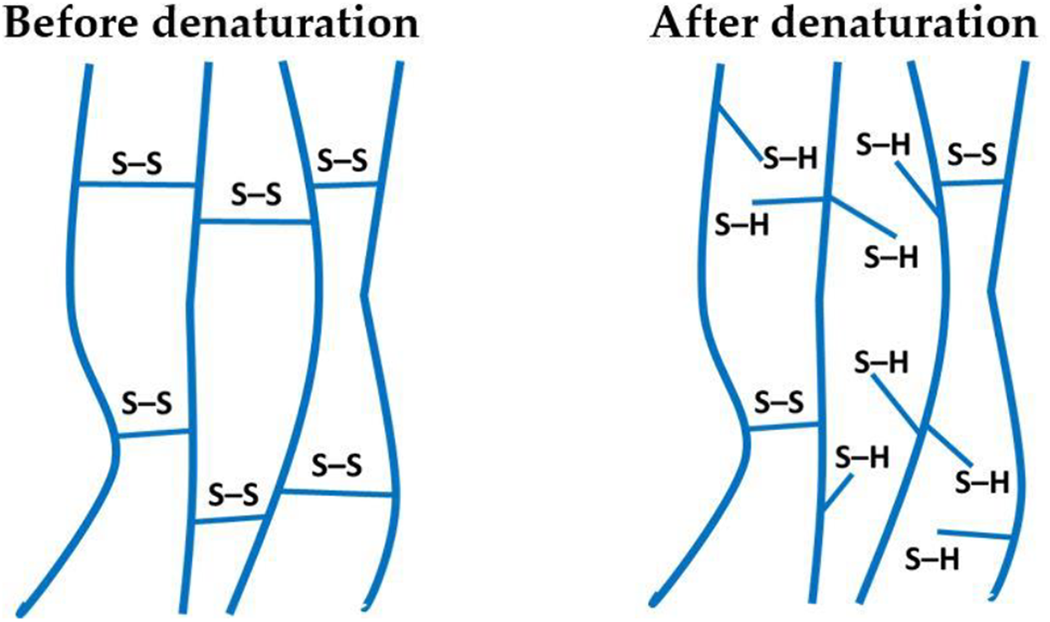
Structure of protein before and after denaturation.
To date, ionic liquids have been the most commonly investigated method for feather alteration for biopolymer production.45,46 This process dissolves and removes keratin for further processing. However the extraction processes used can result in a low protein yield, of 25–38% wt. of useful material, requires high chemical use and can only be conducted in small quantities (samples of ∼10 g). 44 Thermo-pressurisation is another process where heat and pressure is used to produce hydrolysed feathers (commonly used as pet food/fertiliser), allowing for permanent disulphide bond breakage. 47 The thermo-pressurisation process can be easily altered through changing pressure, temperature or moisture values to adjust levels of protein content and amino acid composition in feathers. 48 Breakdown of disulphide bonds can also be achieved using microbial enzymes to produce a similar material to thermo-pressurisation.49,50 However, this is a more complex and slower process, with upwards of 12 h needed to achieve degradation, compared to a maximum of 4 h for thermo-pressurisation.
The use of reduction agents is a process that allows greater control compared to ionic liquids or thermo-pressurisation. Idris et al. showed that sodium sulphite addition causes partial disulphide bond breakage, which increases as heat is introduced. 45 Orliac et al. and Ralston and Osswald also used sodium sulphite to break crosslinks in sunflower proteins and soy protein respectively.51,52 Both showed a decrease in material viscosity during processing, linked to greater protein unfolding and therefore increased chain movement (as no crosslinks restricting movement). Barone et al. and Ullah and Wu used sodium sulphite with poultry feathers to break disulphide bonds and enable easier processing.36,41,44 Both were able to produce feather polymer films through thermal processing. However, these investigations were carried out in small scale (samples of <10 g) and used only 50% wt. of feathers (using only quill or barbs). Moreover, the fundamental understanding of reduction agent addition on disulphide bond content and release of apparent (thermally processable) protein content has not been documented. Quantifying the latter can provide a key indicator of feasibility for thermal processing (extrusion etc.)
There is limited understanding in the literature of the disulphide bond breakage phenomenon, with little explanation of the effect of breakage on crosslinked proteins and their resulting behaviour. It is the major barrier to large scale thermal processing of feather-based biopolymers. This paper aims to 1) validate the feasibility of producing biopolymers from poultry feathers while 2) attaining a deeper understanding of disulphide bond breakdown, reformation and potential implications for thermal processing.
Materials and methods
Materials
Poultry feathers (Figure 2(a)) were obtained directly from the production line of Moy Park (poultry supplier, Armagh, UK) and rinsed with water until it ran clear (usually after 2–3 wash cycles). A Retsch SM 400XL cutting mill (Retsch, Germany) was used to produce a fine feather powder, consisting of fibres, with a determined particle size of D50 value 218.9 µm (Figure 2(b)).

Samples of (a) raw feathers, (b) milled feather powder and (c) hydrolysed feather meal.
Feather meal (hydrolysed feather – Figure 2(c)) was obtained from Farragh Proteins (Cavan, ROI), which had been processed at 150°C, under 4 bar for 4 h, with presence of stream, before being ground into a fine powder of D50 value 47.1 µm. No further processing was conducted.
Sodium sulphite (Sigma Aldrich, Reagent Ph. Eur., mol. wt. = 126.04 g/mol, density = 2.63 g/cm3) and sodium sulphate (Sigma Aldrich, ACS reagent, mol. wt. = 142.04 g/mol, density = 2.68 g/cm3) were used as reduction agents.
Raw material characterisation
In order to determine the structural properties and thermal profile of the milled feathers, characterisation using Fourier Transform Infrared spectroscopy (FTIR) and Differential Scanning Calorimetry (DSC) was conducted.
FTIR was undertaken using a Perkin-Elmer Fourier Transform Infrared Spectrometer (Perkin-Elmer, US) at a spectral resolution of 4 cm−1, at wavelengths of 550–4000 cm−1. DSC was conducted on a Perkin-Elmer Model DSC 6 (Perkin-Elmer, US), with samples weighing 6–9 mg, under a profile of 30–250°C, at a temperature change of 15°C per minute.
Inputs of experimental design
In order to determine a base-line procedure for the use of reduction agents, FTIR analysis of different set-up parameters was conducted to establish steps to maximise disulphide bond breakage. These parameters were:
Reduction agent content (varied sodium sulphite content of 1–9% wt. of feather): Heated at 100°C for 1 h.
Heating time (1, 6 and 24 h): Using 4% feather wt. sodium sulphite heated at 100°C.
Heating temperature (room, 60 and 100°C): Using 4% feather wt. sodium sulphite heated for 1 h.
Sodium sulphite was mixed with de-ionised water, in a 1:2 mixture, and the required amount added to a 2 g feather sample. FTIR readings were taken at a spectral resolution of 4 cm−1, in the disulphide bond region of 550–580 cm−1. The parameters experiencing the lowest absorbance (meaning least disulphide bond content) were selected and used for further investigation.
Disulphide breakage techniques
Sodium sulphite/sodium sulphate treatments were applied to milled feather using the determined baseline procedure (result of Inputs of experimental design section), while hydrolysed feather powder was used as received. FTIR and DSC analysis was carried out to previous values (Raw material characterisation section) to enable direct comparison to raw feathers.
Bond reformation post-processing
Literature previously theorised that partial disulphide bond reformation occurs overtime post-processing. This suggests that disulphide bond breakage may be higher within heated samples, with a bond reformation occurring as samples cool. If so, this means increased thermoplastic behaviour during processing. A study of disulphide bond reformation would determine if:
a final disulphide bond content is achieved straight after processing, or
a much lower disulphide bond content is achieved, but partial reformation (to final level) occurs with oxygen interaction.
To quantify bond reformation, FTIR readings at wavelengths of 550–580 cm−1 were then taken at intervals of 5, 15, 30, 60 and 120 min after removal from oven (heated at 100°C for 1 h), along with a final value after 24 h.
Repeatability of bond breakage
To determine the repeatability of the bond breakage process, DSC was repeated three times on a sodium sulphite treated sample, which was cooled at room temperature for 2 h between each run. A profile of 30–90°C, at a temperature change of 10°C per minute was used to avoid initiating degradation.
Effect of particle size
Feather powder was sieved using an Endecotts graduated sieve (Endecotts, UK) with five mesh screens of sizes 500, 400, 300, 200 and 100 µm. The contents of each sieve were collected into batches of each particle size range. Samples were then prepared by treating with sodium sulphite and analysed using FTIR and DSC as previously described.
Effect on protein concentration
Bicinchoninic acid (BCA) Protein Assay was used to quantify and outline changes to soluble protein content in feather keratin samples and determine the suitability of disulphide breakdown treatments. Four reduction agent treated feather samples (heated at 100°C for 1 h) of 5 g containing 4% wt. sodium sulphate and 2, 4 and 8% wt. sodium sulphite respectively were prepared. Raw and hydrolysed feather powder was also tested. Phosphate Buffered Saline (PBS) was used as a buffering agent.
Samples were then prepared for protein testing by mixing 0.1 g of feather product with 10 ml of PBS and heated to 37°C for 30 min to aid solubilisation. 1 µL of this solubilised feather sample was mixed with 4 µL of PBS to produce 5 µL in a 1:5 dilution. In order to determine the protein contents corresponding to the recorded absorbance values, a linear response curve (protein content vs. absorbance) was developed using standard samples of known Bovine Serum Albumin (BSA) protein concentration.
Samples were tested in triplicate and placed into a 96 well plate, with 250 µL of Bradford reagent added to each sample well. Absorbance values were recorded using a Tecan Infinite 200 plate reader (Tecan, Switzerland) at an absorbance of 595 nm (Figure 3). Protein absorbance values of known samples were averaged and used to produce a linear response curve. For feather samples, using the relationship between absorbance and protein content and excluding the effect of PBS addition, the output value was then multiplied by a correction coefficient to evaluate a final protein concentration.
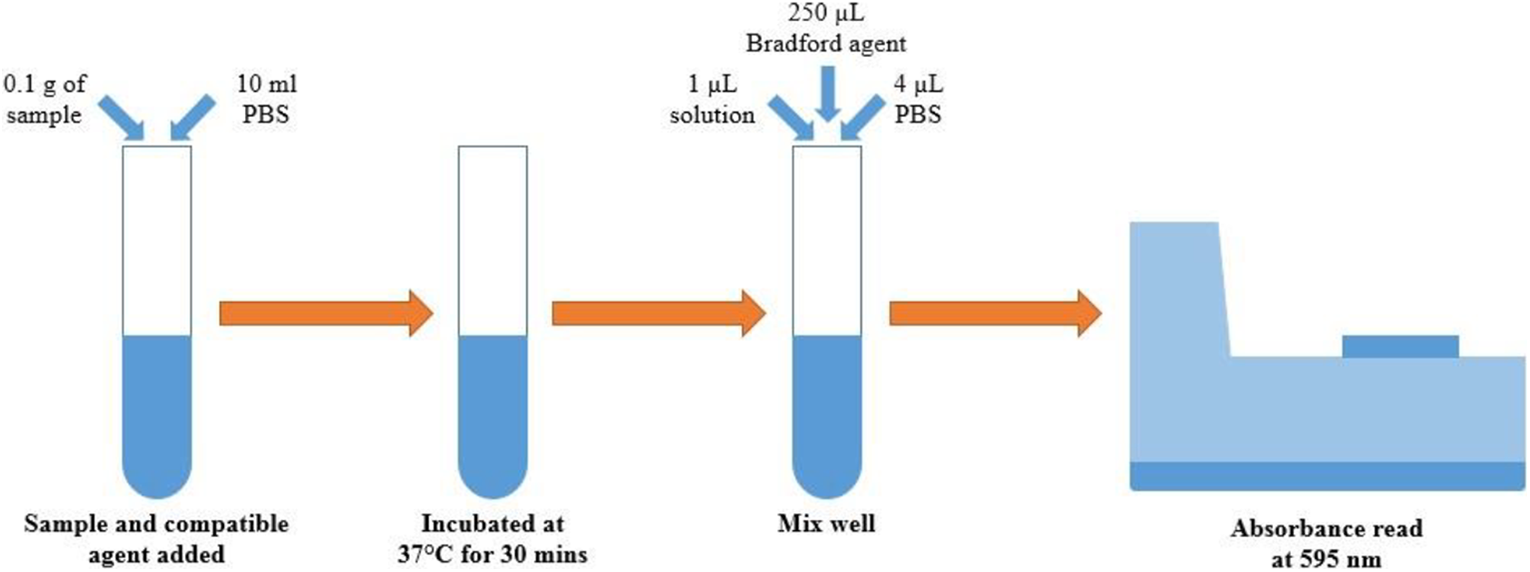
BCA protein assay procedure used.
Results and discussion
It is important at this point, to reiterate the aims of this investigation. 1) Validate using reduction agents and hydrolysis techniques the feasibility of producing poultry feathers biopolymers and 2) Attain a deeper understanding of the effects of bond breakage with respect to structural, processing and thermal behaviour.
Raw material characterisation
FTIR results showed several strong bonds were present within raw poultry feathers (Figure 4). Bonds corresponding to keratin proteins exist in the region of 1400–1800 cm−1, while disulphide bonds exist at around 550 cm−1.38,39
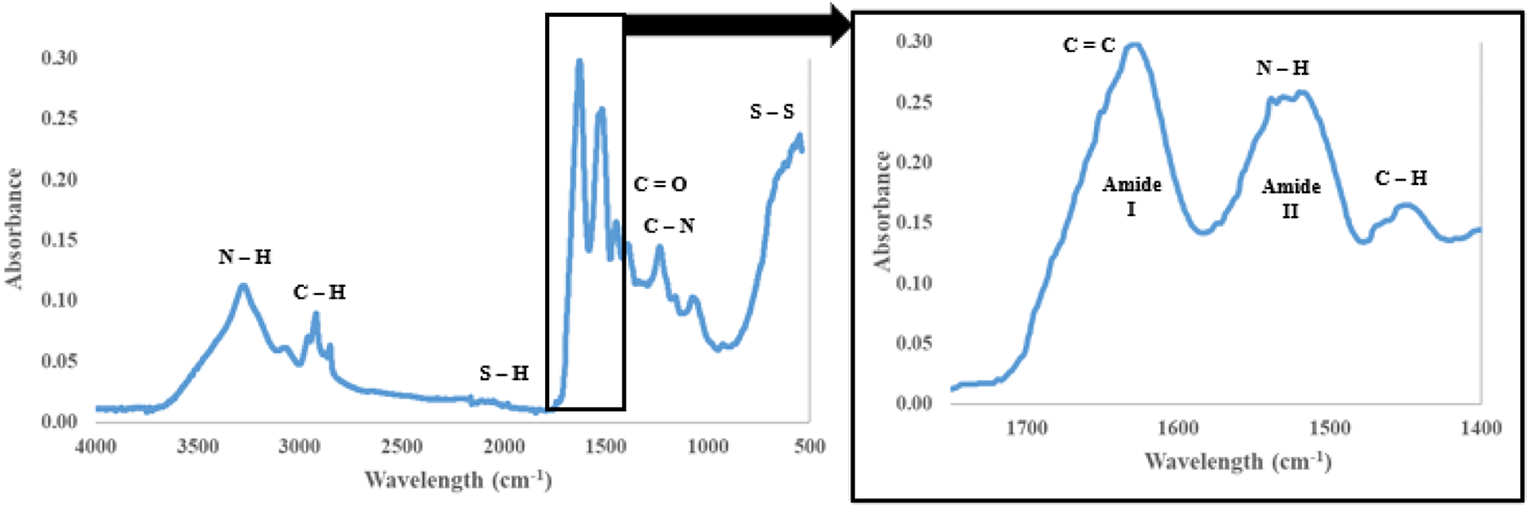
FTIR graphs of whole feathers.
This crosslinked polymer nature means raw feathers behave like a thermoset polymer. This is validated by the DSC results (Figure 5) which showed low heat flow and a flat curve below degradation temperature, corresponding to a thermoset nature. 53 A denaturation temperature of 71.2°C corresponds to the temperature where dissociation (denaturation) of disulphide bonds occurs.
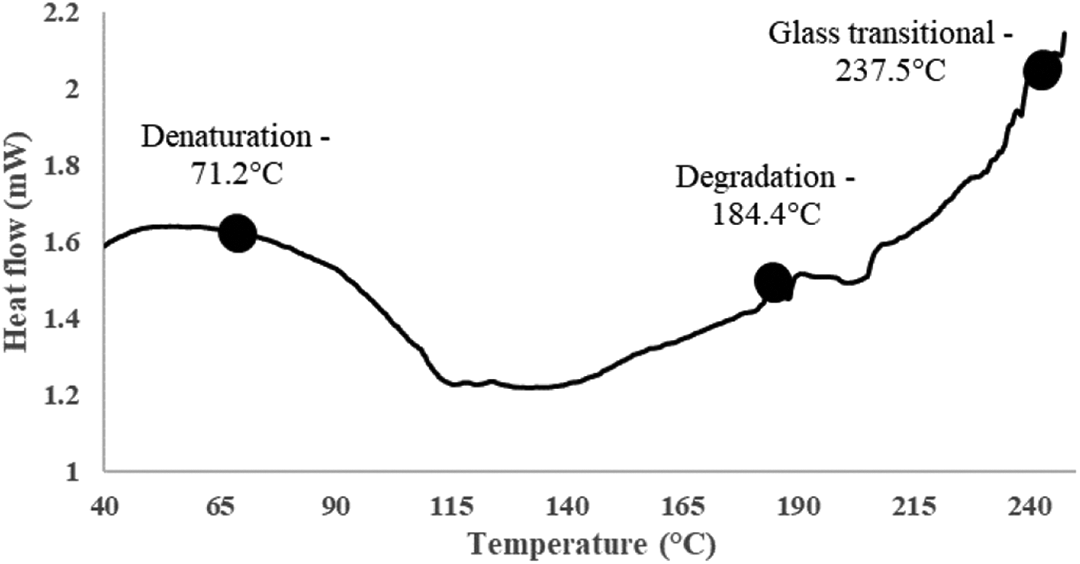
DSC scan of raw poultry feathers.
Inputs of experimental design
For sodium sulphite content, there was a linear relationship between reduction agent content and bond breakage up to 4% wt. feather (Figure 6(a)). Above this, disulphide bond content increased. Disulphide bond breakage was also higher for samples heated to 100°C (above denaturation temperature – Figure 5) compared to unheated (room) and 60°C samples (Figure 6(b)). Short term heating (1 h) provided lowest absorbance values, as longer time (6 and 24 h) tended to dry out reducing effectiveness (Figure 6(c)).
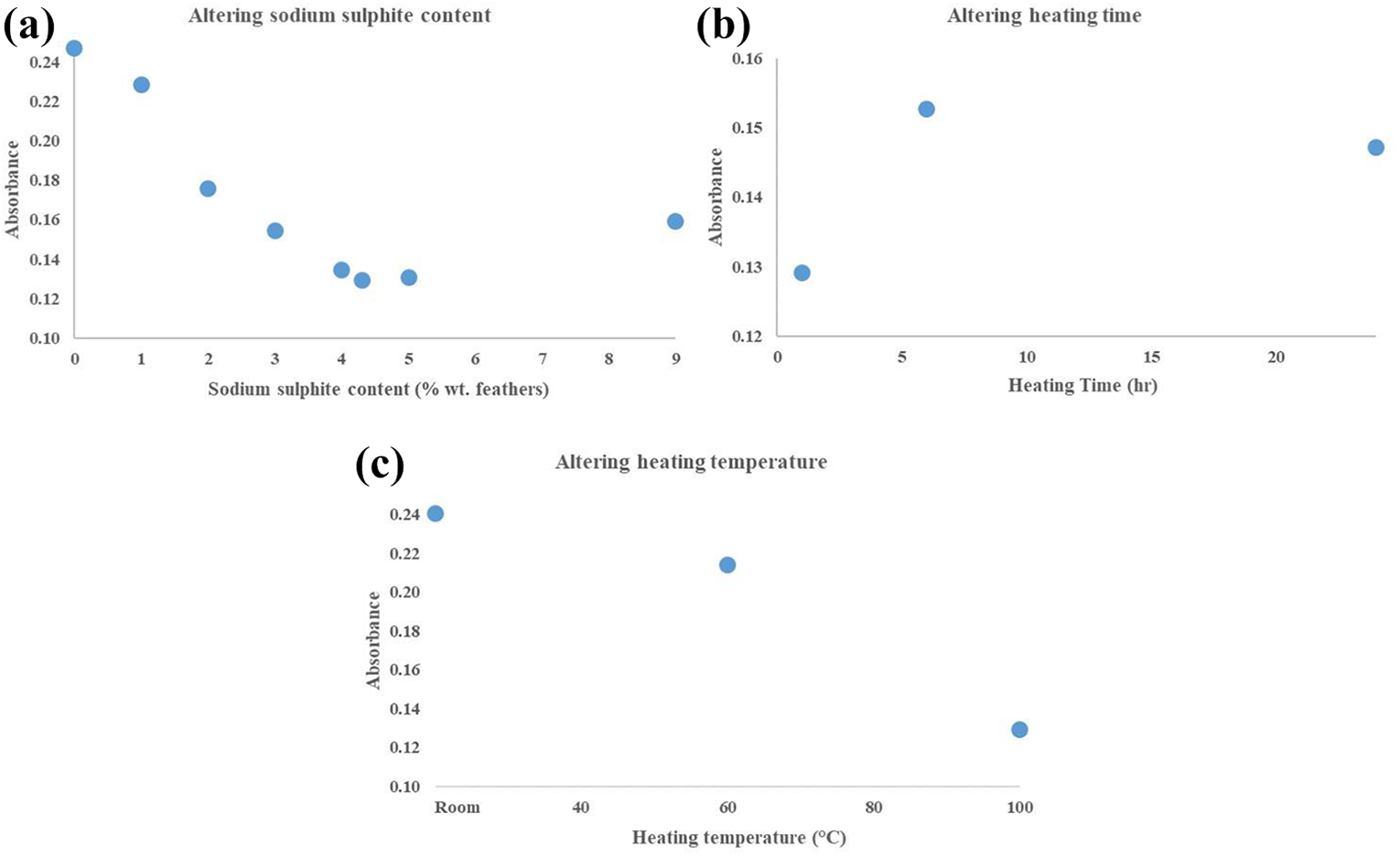
FTIR absorbance values with altering (a) sodium sulphite content (at temperature of 100°C for 1 h), (b) heating time (using 4% feather wt. sodium sulphite heated at 100°C) and (c) heating temperature (using 4% feather wt. sodium sulphite heated for 1 h) for selection of set-up procedure.
The results yielding the optimum outcome (lowest absorbance values) and selected as procedure for further investigations were:
A reduction agent content of 4% feather wt.
A heating time of 1 h.
A heating temperature of 100°C.
Disulphide breakage techniques
The FTIR and DSC graphs showed variation between the three treated feather samples (Appendix 1), with each technique enabling bond breakage of between 48% and 67% and improving thermoplastic nature compared to raw feathers (Table 1).
Absorbance recorded at disulphide bond region.
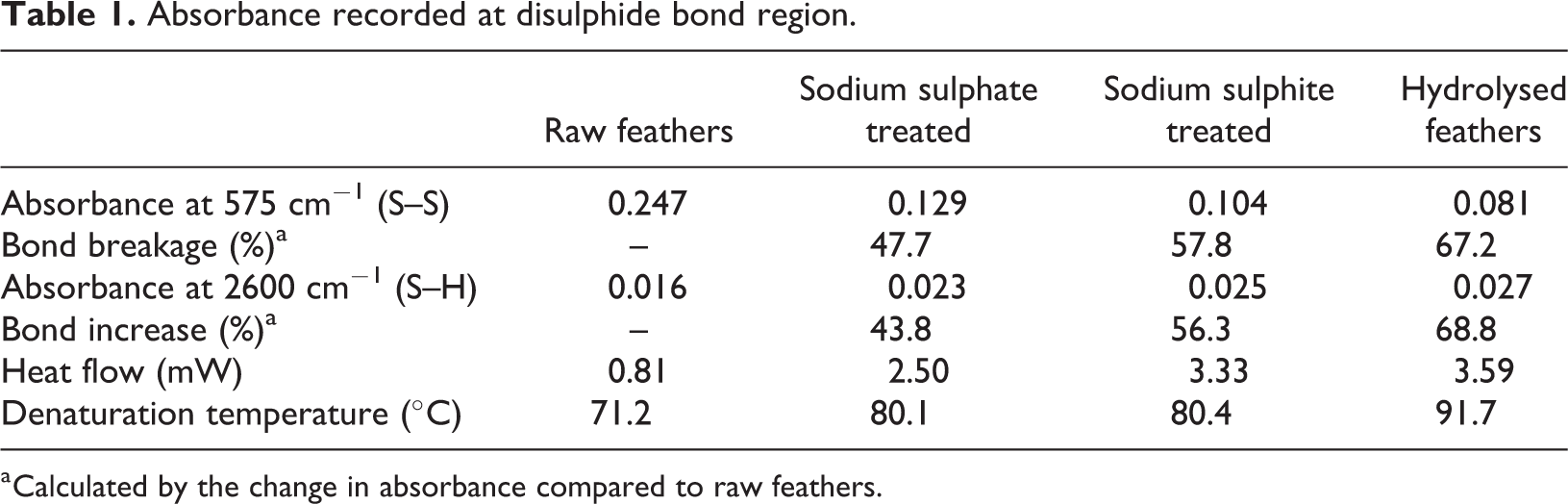
a Calculated by the change in absorbance compared to raw feathers.
The use of reduction agents in small quantities (4% feather wt.) achieved bond breakage of 57.8% and 47.7% using sodium sulphite and sulphate, respectively (Table 1). This process, with the presence of water, works by initiating disulphide breakage under heating. Hydrogen bonds, from water, join onto these broken bonds restricting reformation by forming S–H bonds (present at ∼2600 cm−1). The increase in S–H bond content is proportional to the decrease in disulphide bonds.
In determining set-up procedures, results showed bond breakage to be proportional to reduction agent content (up to 4% wt.), suggesting bond breakage can be controlled to desired values up to 57.8%.
Results showed disulphide bond content to be lower for hydrolysed feather compared to use of reduction agents (Table 1). This was due to the intensity of the heating process (with presence of moisture) causing degradation to the material, as opposed to denaturation, resulting in a disulphide bond loss of 67.2%.
The treatment of feather whether by reduction agents or hydrolysed resulted in increases to both heat flow and denaturation temperature, observed within DSC results. This is caused by the reduction in disulphide bonds and the significant effect on other protein and inorganic bonds in poultry feathers from these methods. FTIR results showed a direct relationship (R2 = 0.9975) between disulphide bond breakage and protein bond breakage (Figure 7(a)), implying that it is not possible to alter feather keratin into a thermoplastic material without unfolding and losing some of the higher structure (including alpha-helix and beta-sheet) protein content. The endothermic peak observed is the denaturation of feathers, 54 where protein strucutre breaks leading to a change in feather characteristics. The breakage of disulphide bonds means an increase in the level of unbonded chains within feathers, which facilitates protein molecules more freedom (molecular movement). 42 Treated samples have chains that are more active (free to move) due to a reduction in crosslinks (disulphide bonds – Figure 1) which have a tendancy to restrict molecular movement. Increased energy i.e. higher temperature, is needed to initiate denaturation due to the need to restrain these mobile polymer chains compared to a highly crosslinked materials. 55 Raw feathers require less energy, resulting in lower denaturation temperatures, 56 while increased disulphide bond breakage leads to increased denauration temperature. Raw feathers have a denaturation temperature of 71°C, whereas sodium sulphate and sodium sulphite samples denatured at ∼80°C, while hydrolysed was higher at 92°C (Table 1).
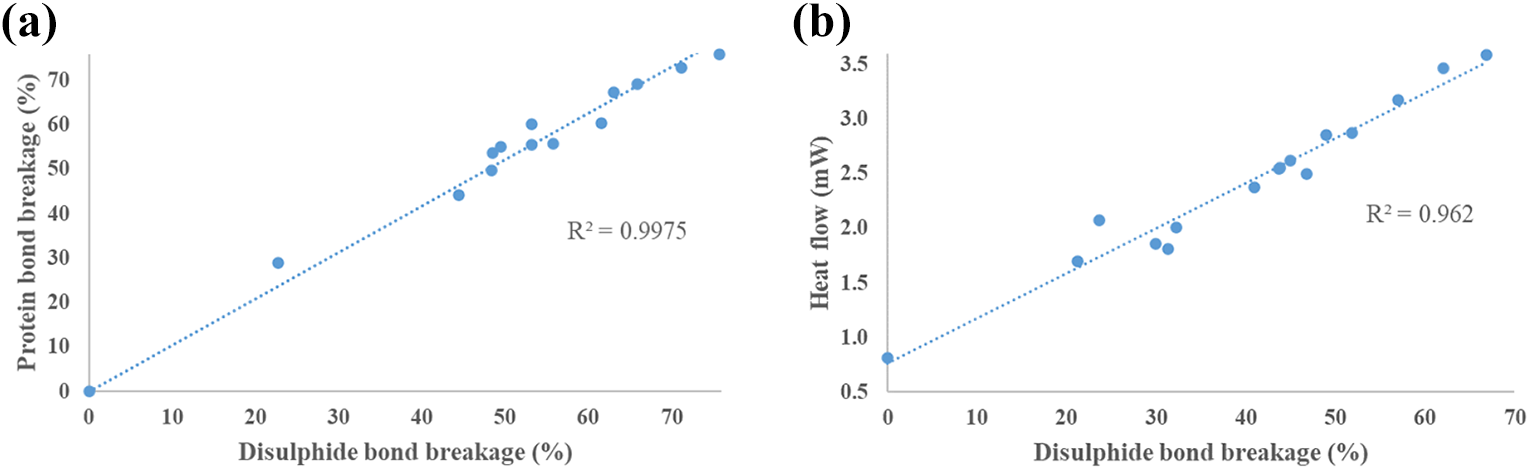
Effect of disulphide bond breakage on (a) protein bond breakage and (b) heat flow.
Although protein bonds are reduced, which can detrimentally affect the structure of raw feathers, an advantage of disulphide bond reduction is improved thermoplastic nature, which has benefits for processing. A directly proportional relationship (R2 = 0.962) exists between disulphide bond breakage and heat flow (Figure 7(b)). Higher heat flow values correspond to changes in polymers, whether melting, denaturation, etc. Thermoplastic materials have a high heat flow and sharp peaks, while thermoset materials have a broader curve and low heat flow, showing no changes at equivalent temperature. 53 The DSC curve for raw feathers (Appendix 1) showed a relatively flat curve corresponding to a thermoset. Whereas a sharp curve with clearly distinguishable peak was observed for treated samples, outlining transition to a thermoplastic nature. Heat flow values were highest for hydrolysed feather (highest disulphide bond breakage). Maximum heat flow for sodium sulphite samples was 33% higher than sodium sulphate samples (due to the reduced disulphide bond content), with all three treated samples significantly higher than raw feathers. These results show that altering disulphide bond content has a major effect on the final material structure and properties of poultry feathers.
Bond reformation post-processing
Disulphide bond content was lower for heated samples, with reformation of 10.8–59.9% occurring via reaction with oxygen during the cooling process (Figure 8, Table 2). Although previous research 41 theorised that partial reformation occurs, these are the first experimental results to prove and quantify the process. The process of renaturation (a exothermic reaction) 57 occurs where oxygen reacts with the joined hydrogen, to form water allowing for broken disulphide bonds to re-join. In all cases, disulphide bond content increased (reformation) linearly with time, showing that reformation starts with first contact with surrounding air and remains constant until the final bond content is reached.
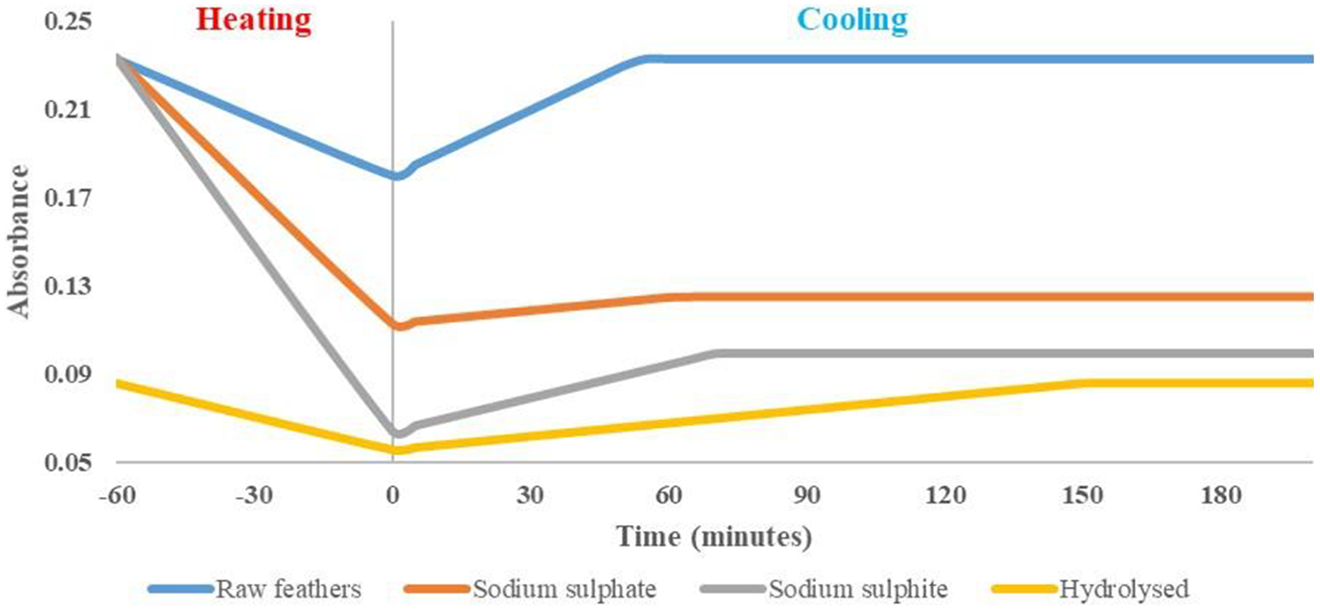
Bond reformation comparison of raw and treated feather materials.
Bond reformation data for feather materials.
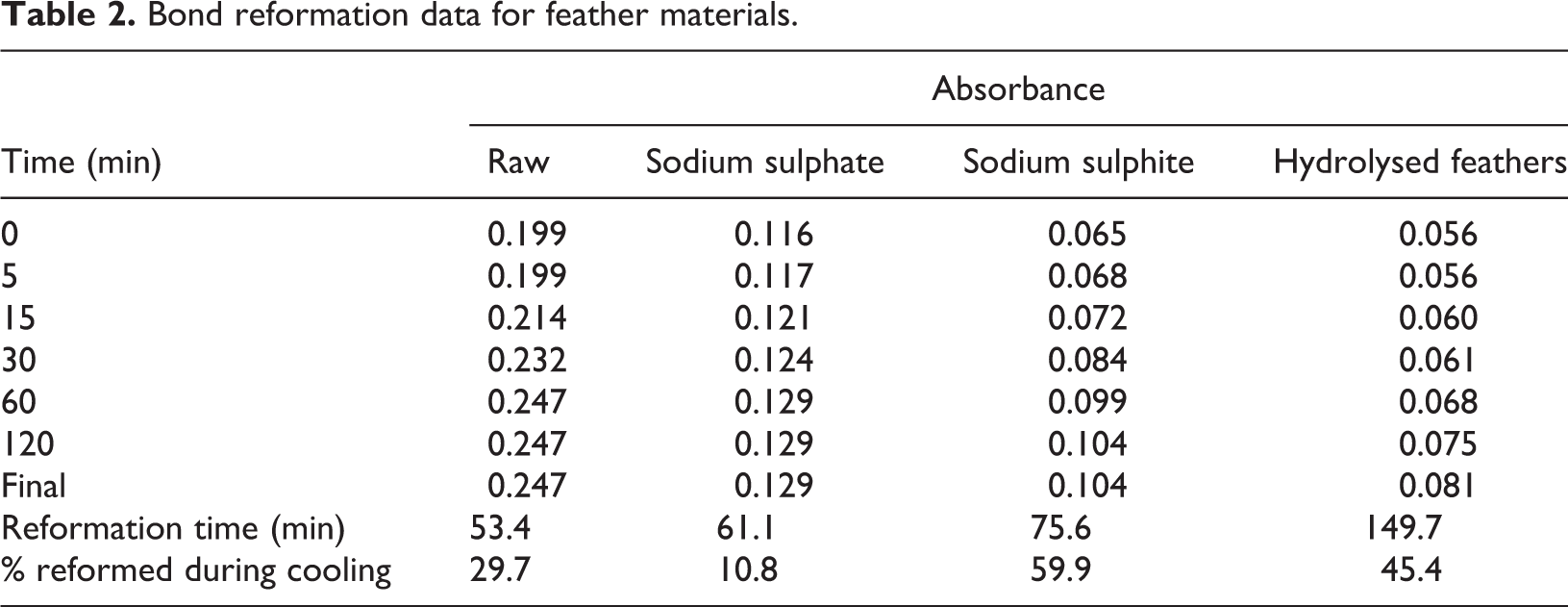
Under heating (to 100°C) alone, without the addition of water or reduction agents, a disulphide bond reduction of 29.7% was gained for raw feathers. This temperature is above the denaturation temperature, therefore causing cleavage of disulphide bonds. Such a reduction in disulphide bond content is insufficient for polymer formation. These results support the theory 58 that heat is required, particularly in the reduction agent processes, to break bonds and allow H-bonds to join. As no reduction agents were used in this sample, disulphide bond content returned to original value after 53.4 min.
The results showed an inverse relationship between disulphide bond content and reformation time. Raw feathers (highest disulphide content) had the shortest reformation time, 96 min quicker than hydrolysed feather samples (lowest disulphide content). This is caused by the higher protein bond content, increasing the rate of protein oxidation by enabling faster protein–protein interactions. Final disulphide bond content (and protein bond content) for raw feathers was 2.8 times higher than that of hydrolysed feathers, and reformation time was 2.8 times faster. As disulphide bond content increased, all remaining bonds, particularly protein bonds, increased proportionally, showing their use in maintaining and supporting the higher protein structures.
Disulphide bond breakage for heated samples was considerably higher for sodium sulphite samples in comparison to use of sodium sulphate (73.6% vs. 53.0% breakage compared to raw feathers). Under cooling sodium sulphite allows for greater bond reformation (59.9% vs. 10.8% for sodium sulphate). This shows sodium sulphite allows for considerably greater amounts of disulphide bond breakage (and associated benefits for polymer formation) during processing, a trend less noticeable using cooled samples (Table 1).
Bond reformation behaviour shows increased reduction of bonds during heating, meaning improved thermoplastic nature and easier processing, which are significant advantages for polymer processes such as extrusion and compression moulding. A further advantage is that, as the material cools, the reformation of disulphide bonds provides a crosslinked structure which can led to improved mechanical properties. 59
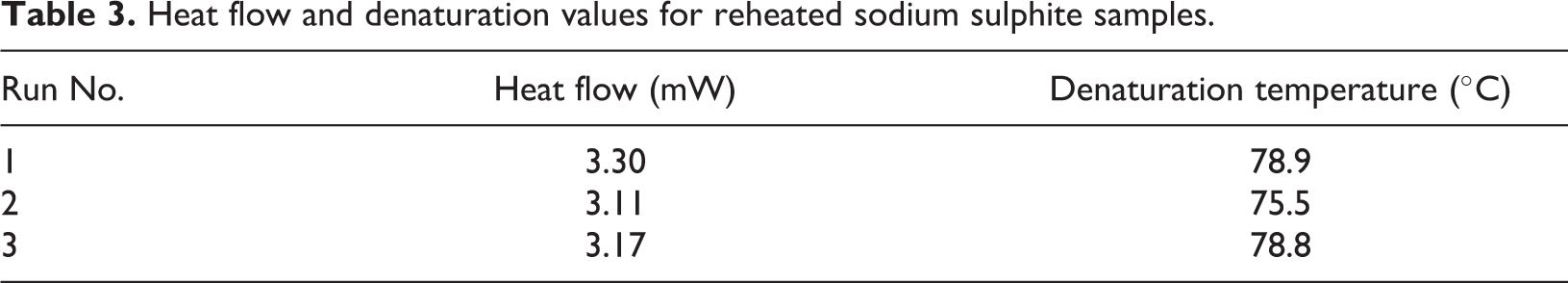
Repeatability of bond breakage
DSC results for the reheated sample showed consistency in both heat flow and denaturation temperature values for all three runs (Table 3). This showed the process of partial bond breakage and reformation could be repeated without any significant effect on final samples or processing properties. Although the sample in this form does not exist as a final polymer, this suggests potential for reprocessing/reuse for this type of polymer feedstock.
Heat flow and denaturation values for reheated sodium sulphite samples.
Effect of particle size
An increase in particle size resulted in uniform reduction in 1) percentage transmittance (inverse of absorbance) at ∼550 cm−1 (disulphide bonds), 2) denaturation temperature and 3) maximum heat flow value (Figure 9). Higher transmittance values (i.e. lower bond content) occurred for smaller particle sizes due to the greater surface area resulting in a greater reaction area (improving the disulphide breakage process). As particle size increases, this effect is reduced. The results showed that a smaller particle size provides increased heat flow, an important property for polymer processing. As disulphide bond content decreases, this enables more molecular movement (as disulphide bonds which hold everything together are broken) leading to an increase in the temperature needed to cause denaturation. 55 This unfolded nature also increases heat flow, meaning the material acts in a more thermoplastic manner.
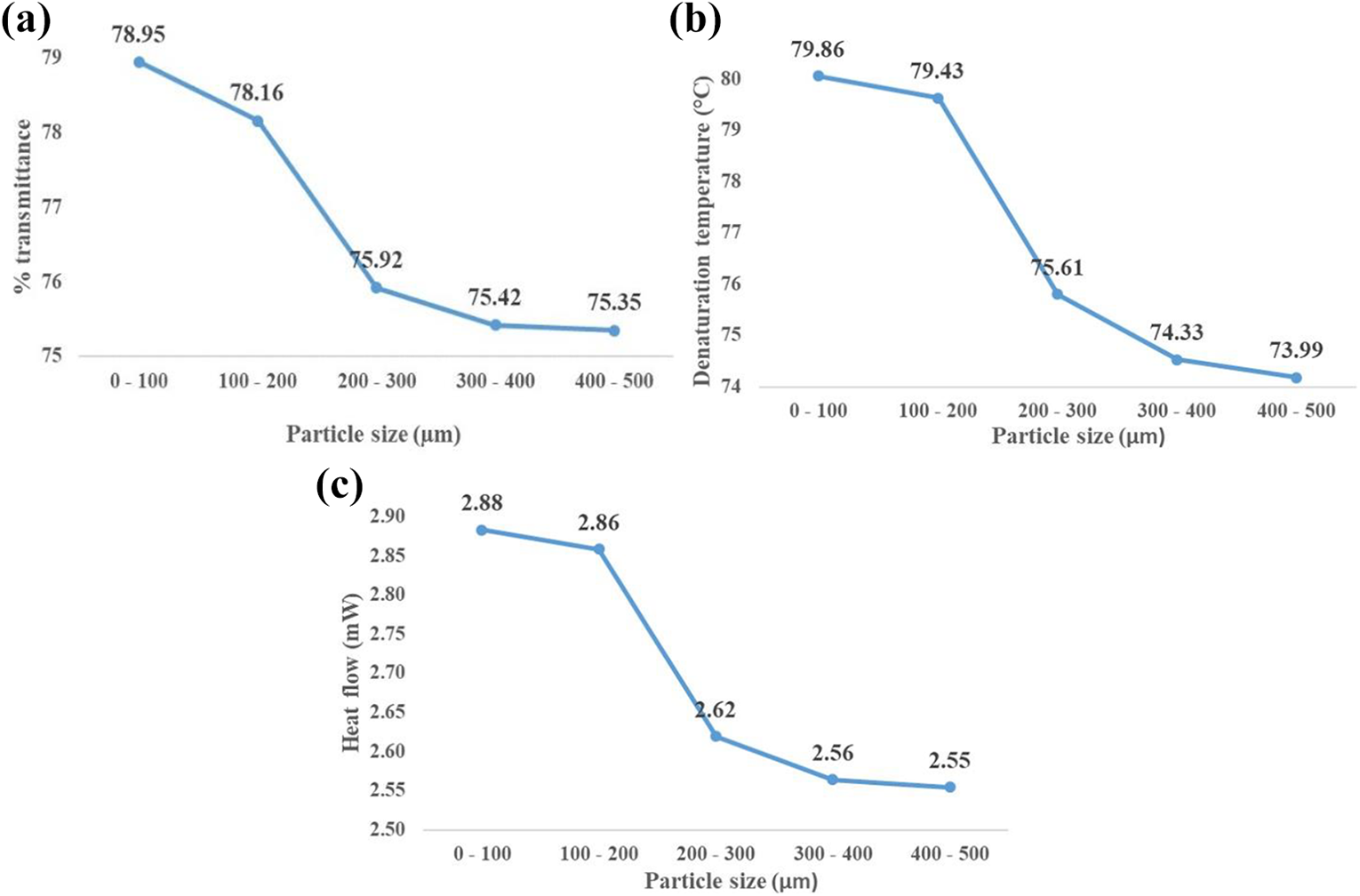
Effect of particle size on (a) disulphide bond transmittance, (b) denaturation temperature and (c) heat flow.
Effect on protein concentration
The protein content of varying protein samples was found to range from 11.7% to 36.5% (Table 4).
Protein absorbance data for feather samples.

The crosslinked nature of poultry feathers created difficulty in carrying out protein testing. A range of compatible agents commonly used in BCA protein assay were trialled but failed to work due to being insoluble with feather proteins. Literature commonly suggests raw feathers to have a protein content up to 90%,33,60 however protein assay showed a concentration of only 0.181 g per gram (18.1%). This lower apparent content is due to the structure and bond types (crosslinks) of the proteins. The protein assay used PBS to solubilise protein from the sample in order to quantify its content. However, the high disulphide bond content of raw feathers makes the amount of soluble protein very low. A large amount of protein is contained within these crosslinked structures and cannot be effectively solubilised, lowering the apparent concentration. This behaviour was also observed by Idris et al. in their work using ionic liquids, where only 25% extraction of potential protein from duck and turkey feathers was achieved.45,46
Only when these disulphide bonds are broken is more soluble protein released and the apparent concentration increased. Soluble protein was increased by 102% by using 4% feather wt. of sodium sulphite. Measurement of soluble protein content, and therefore protein assay concentration, is a method of assessing the suitability of the material for polymer generation. As sodium sulphite content increases, protein concentration and disulphide bond absorbance follow opposite trends (Figure 10). Both sets of results show the optimum level of sodium sulphite is 4% feather wt., which provides the lowest absorbance (i.e. highest disulphide bond content) and highest protein concentration. At this loading a maximum soluble protein concentration of 36.5% was obtained, providing sufficient material to enable protein biopolymer production.
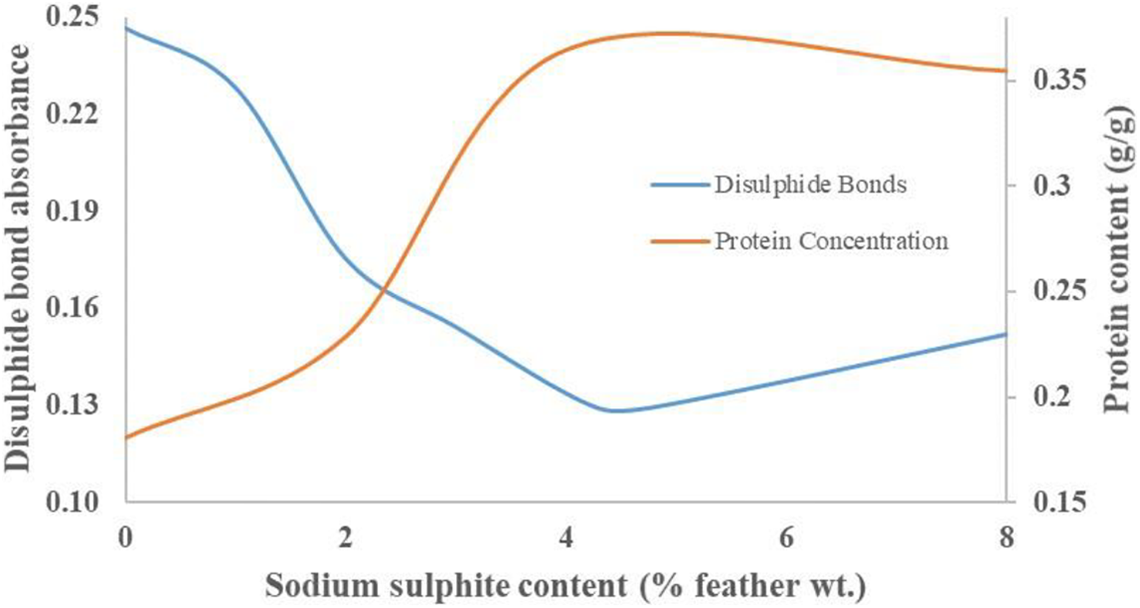
Changes to disulphide bond content and protein concentration with sodium sulphite content.
In comparison, hydrolysed feather had a low protein content (11.7%), lower than for raw feathers, due to the degradation caused by the high heating intensity of its production process. As it is the non-crosslinked free proteins that will be used to generate a bio-based material, the low content seen for hydrolysed feathers, would likely be insufficient to provide enough natural polymer for cohesive polymer generation.
Selection of optimal method
The use of sodium sulphite and sodium sulphate, as reduction agents, and hydrolysed feathers achieved high disulphide bond breakage, aiding the potential for biopolymer production. Sodium sulphate achieved lower breakage and protein concentration compared to sodium sulphite (Table 5). Hydrolysed feathers achieved similar bond breakage and heat flow values to sodium sulphite, however due to protein degradation occurring (due to the intensity of heating process) protein content was significantly lower.
Results for disulphide bond breakage techniques.
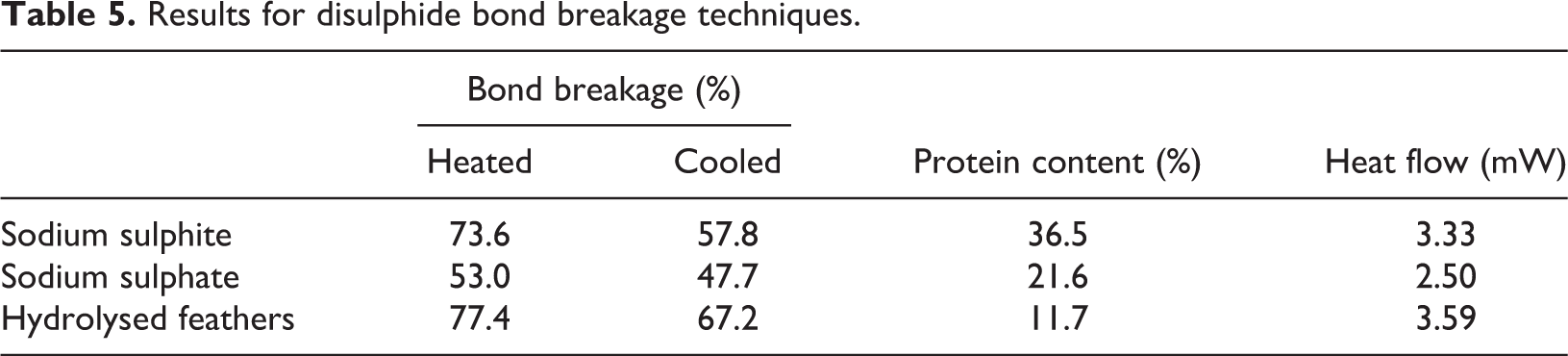
The low protein content for hydrolysed feathers provides insufficient material to enable polymer formation. Hydrolysed feathers also lost the raw appearance of poultry feathers and emitted a strong odour, compared to use of reduction agents, which can have a significant effect on final polymer properties, appearance and usage. The hydrolysed feathers for this study were gained from a meal producer using a standardised process. Theoretically the hydrolysis process could be adjusted to alter the final feather product, but with numerous variables (temperature, pressure, time and moisture content 48 ), it is extremely difficult to develop a specific material. This is in comparison to the controllable process using reduction agents. A linear relationship existed between reduction agent content and disulphide bond breakage up to 4% wt. content (Figure 6(a)).
Secondly, particle size was found to have a major impact of final results. Bond breakage improved by 17% using 0–100 µm particle size as opposed to 400–500 µm due to the greater surface area (and improved reaction). This suggests a small particle size should be used to maximise breakage, matching the outcome suggested by Barone et al. 41
Conclusions
The techniques of treating raw poultry feathers with reduction agents or hydrolysis enabled partial breakage of disulphide bonds, improved thermoplastic nature and increased apparent protein content. In comparison S–H bond content increased proportionally with reduction in disulphide bond content. Although highest breakage was achieved using hydrolysis, the degradation of protein meant a much lower protein content, which would significantly affect biopolymer formation. In comparison, use of reduction agents provided similar bond breakage values but maintained protein properties, with increased apparent content. Using of sodium sulphite provided more desirable properties compared to the use of sodium sulphate, resulting in it being chosen as the technique for further research.
Samples heated samples to 100°C exhibited much higher disulphide bond breakage (up to 60%), with reformation occurring during cooling with contact with oxygen (oxidation) proving the concept of bond reformation. This higher bond breakage improves processability but can enable post-processing crosslinking which helps improve final mechanical properties. Bond breakage was also aided by using a smaller particle size. Transmittance, heat flow and denaturation temperature all increased using a smaller particle size due to a greater surface area allowing for a greater reaction. Reprocessing of sodium sulphite samples showed minimal changes suggesting the denaturation/renaturation process can be repeated numerous times without any effect on final material properties. This also outlines reprocessing potential from the final polymer.
Use of these techniques enables significant alteration of raw poultry feathers to allow formation, with the addition of a plasticiser, into a biopolymer.
Footnotes
Acknowledgement
The authors would like to acknowledge Moy Park Ltd for their financial support and for the generous use of their resources through industry expertise and material supplies.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors would like to acknowledge Moy Park Ltd and the the Department for the Economy (DFE) for their financial support.
