Abstract
Type 2 diabetes mellitus (T2DM) is a chronic metabolic condition marked by insulin resistance, decreased insulin production, and persistent low-grade inflammation. The prevalence of T2DM has increased significantly in recent decades; as a result, it is now regarded as one of the fastest-growing public health concerns worldwide. Long-term micro- and macrovascular problems such as nephropathy, retinopathy, neuropathy, and cardiovascular disease can be caused by poor blood glucose control. Therefore, developing reliable diagnostic indicators for early diagnosis and investigating new treatment targets are critical for addressing the increasing prevalence of T2DM. Several novel diagnostic approaches have been created by targeting endogenous proteins, with fetuin-A being one of the most promising targets. Human fetuin-A, also referred to as alpha-2-Heremans Schmid glycoprotein, is a liver-produced glycoprotein that is abundantly secreted into the bloodstream and seems to be involved in insulin resistance, metabolic syndrome, and inflammation. Clinical studies have shown that circulating fetuin-A levels are closely associated with T2DM and its complications, underscoring its potential as both a biomarker and a therapeutic target. This narrative review provides a detailed overview of the evolving role of fetuin-A in the development of T2DM and its associated complications, as well as its future perspectives.
Plain language summary
Type 2 diabetes mellitus (T2DM) is a long-term condition in which the body does not use insulin properly (a problem called insulin resistance) and cannot make enough of it. This leads to high blood sugar levels and ongoing low-grade inflammation. Over time, poorly controlled diabetes can cause serious health problems, including kidney disease, eye disease, nerve damage, and heart disease. Because T2DM is becoming more common worldwide, finding better ways to detect it early and to prevent complications is very important. One protein of particular interest is fetuin-A, which is made in the liver and released into the blood. Fetuin-A has been linked to insulin resistance, inflammation, and metabolic syndrome. Studies show that circulating fetuin-A levels are closely linked with T2DM and its complications. Therefore, fetuin-A may serve as a useful biomarker (a measurable substance that signals disease risk or progression) and may also be a potential target for treatment. In this review, we look at the current evidence on the role of fetuin-A in T2DM and its complications, and discuss how it may be used in the future to improve patient care.
Keywords
Introduction
Diabetes mellitus (DM) is typically described as a metabolic condition characterized by elevated blood sugar levels, which may arise from issues with insulin secretion, insulin function, or a combination of both. Type 2 diabetes mellitus (T2DM) includes people who exhibit insulin resistance and typically have a relative deficiency in insulin production. 1 According to the 11th edition (2025) of the Diabetes Atlas, T2DM has become a significant global public health concern. An estimated 589 million adults aged 20–79 are currently living with the disease worldwide, a figure expected to increase to about 853 million by 2050. 2 There are a number of distinct mechanisms underlying the pathogenesis of T2DM. It is crucial to elucidate the intricate pathophysiology of this disease, which is important for the management and development of prevention strategies.
Fetuin-A, previously referred to as α2-Heremans-Schmid glycoprotein (AHSG) in humans and phosphorylated protein 63 (PP63) in rats, is a 64 kDa negatively charged plasma protein. It is encoded by a gene located on chromosome 3q27 and is primarily expressed by hepatocytes. 3 The concentration of fetuin-A in serum varies between 0.5 and 1.0 g/L, with a half-life of 1–2 days. 4 Fetuin-A has been demonstrated to play a crucial role in various physiological cellular functions, including the metabolism of proteins and fatty acids, the regulation of acute inflammatory responses, bone mineralization, and the metabolism of calcified matrices. It also influences neutrophil degranulation, lymphocyte recruitment, thyroid hormone activity, and calcium ion homeostasis, among other processes. 5
Glycoproteins such as fetuin-A have recently been proposed to play a role in the molecular connections between obesity, nonalcoholic fatty liver disease (NAFLD), insulin resistance, and metabolic syndrome, as they have been associated with the inhibition of insulin receptor activity, leading to a disruption of insulin signaling pathways.6,7 Beyond its metabolic functions, fetuin-A also plays a pivotal role in inflammatory processes.8,9 Since chronic low-grade inflammation underlies both T2DM and its complications,10–12 this shared inflammatory milieu highlights the importance of examining the role of fetuin-A in the pathogenesis of T2DM and its complications. Currently, fetuin-A has been identified as having a potential new role in the pathogenesis of T2DM. Given its pathological involvement in the development of T2DM, the clinical application of fetuin-A as a diagnostic marker and therapeutic target in T2DM is now underway. Therefore, this review article primarily focuses on the role of fetuin-A in the pathogenesis of T2DM and its potential clinical applications.
Methodology
This review included studies from research databases such as PubMed, PubMed Central, Web of Science, Google Scholar, and Cochrane Library. The keywords used included fetuin-A, AHSG, structural features of fetuin-A, the role of fetuin-A in T2DM, the relationship between fetuin-A and complications of T2DM, and the clinical application of fetuin-A in patients with T2DM. The studies included were published in or translated into English. There was no specific time frame for study inclusion, but the authors preferred recently published papers. However, studies on fetuin-B and those focused on type 1 DM, gestational DM, and other types of DM were not included in this review article.
Structural features of fetuin-A
Fetuin-A is primarily expressed in embryonic and adult liver cells, with lower levels in adipocytes and immune cells. 13 It is a heterodimeric plasma glycoprotein encoded by the AHSG gene on chromosome 3q27.3, which comprises multiple exons and introns. 14 The gene is transcribed into a single messenger ribonucleic acid (mRNA) strand coding for a 367-amino-acid preprotein (pre-fetuin-A). 15 This precursor contains two polypeptide chains: the A-chain (282 amino acids), the B-chain (27 amino acids), along with an 18-amino-acid signal sequence (SS) and a 40-amino-acid connecting peptide (CP),3,15 The A-chain of mature fetuin-A consists of three domains: two cystatin-like domains, D1 and D2, and a third domain, D3, which is further divided into the proline-rich subdomain D3a and the C-terminal subdomain D3b. D1 and D2 (116–118 residues) form α-helices, β-sheets, and reverse turns, comprising 29%, 24%, and 26% of the structure, respectively. Functionally, D1 has binding sites for calcium, TGF-β, and bone morphogenic protein; D2 hinders cysteine protease 16 ; and D3 binds to the insulin receptor. 17
Mature fetuin-A results from proteolytic processing and undergoes posttranslational modifications, including glycosylation, phosphorylation, and sulfation. 3 Processing yields a 321-residue heavy chain and a 27-residue light chain, linked by disulfide bonds. 18 Occasionally, cleavage omits 40 residues from the heavy chain’s C-terminus, with the B-chain identical to the light chain. 19 Fetuin-A has two phosphorylation sites (in the A-chain and CP), two N- and two O-glycosylation sites on the A-chain, and one O-glycosylation site on the B-chain.20,21 These modifications are likely to affect the expression, biological stability, and activity of fetuin-A. 22 In particular, phosphorylation plays a critical role in fetuin-A’s interaction with the insulin receptor (Figure 1).

Basic structure of fetuin-A.
The relationship of fetuin-A with T2DM
Numerous recent studies have highlighted a substantial association between fetuin-A and the progression of T2DM, suggesting its potential role in the pathogenesis of the disease, although its specific role remains uncertain. Evidence from prospective cohort studies indicates that elevated fetuin-A predicts the development of T2DM and contributes to disease progression by inhibiting insulin receptor signaling and promoting toll-like receptor 4 (TLR4)-mediated inflammation.17,23,24 Conversely, hepatic steatosis, inflammation, and metabolic dysregulation can elevate fetuin-A levels, suggesting that its increase may also result from the metabolic disturbances characteristic of T2DM and its complications.25–27 These findings underscore the need for large-scale experimental studies to clarify the relationship between fetuin-A and T2DM.
A significant amount of data indicates that individuals with T2DM exhibit significantly higher serum levels of fetuin-A compared to healthy controls. For instance, findings from the Health, Ageing and Body Composition Study (Health ABC) and the European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study demonstrated that individuals with elevated fetuin-A levels had an increased risk of developing diabetes. 28 Interestingly, a positive correlation between fetuin-A and T2DM has also been observed in individuals with elevated, yet nondiabetic, blood glucose levels. However, this association was not evident among individuals with strictly normal glucose levels.23,29 Supporting this, a systematic review and meta-analysis by Guo et al. 30 reported that each standard deviation increase in fetuin-A concentration was associated with a 23% higher risk of developing T2DM. Similarly, a multiethnic cohort study of middle-aged to older adults without clinically apparent cardiovascular disease (CVD) at baseline found that higher fetuin-A concentrations were significantly associated with increased diabetes risk in women. 31
In contrast to these findings, some studies have reported an inverse association, where fetuin-A levels were lower in individuals with T2DM compared to healthy counterparts.32–34 For example, Wojtysiak-Duma et al. 34 found significantly lower serum fetuin-A concentrations in patients with T2DM. This result was echoed by another case-control study, which also reported reduced fetuin-A levels in diabetic individuals. 32 Table 1 summarizes key studies exploring the relationship between fetuin-A levels and T2DM in the current literature.
A summary table on the relationship between fetuin-A and T2DM.

NGT, normal glucose tolerance; T2DM, type 2 diabetes mellitus.
The role of fetuin-A in the pathogenesis of T2DM
Several mechanisms have been proposed to explain the role of fetuin-A in the development of T2DM, although further research is warranted. One key mechanism is its involvement in insulin resistance, a central feature in T2DM pathogenesis. 47 Fetuin-A suppresses insulin receptor tyrosine kinase activity by blocking the autophosphorylation of the receptor and insulin receptor substrate-1 (IRS-1), thereby impairing insulin signaling and reducing insulin sensitivity in the liver and muscle tissues.22,48 In vitro studies using purified rat and bovine fetuin-A support this inhibitory effect. In humans, fetuin-A disrupts insulin-stimulated phosphorylation of the insulin receptor and IRS-1.49–51 Moreover, in rat adipocytes overexpressing human AHSG, fetuin-A significantly reduced basal and insulin-stimulated phosphorylation of Elk-1, a transcription factor downstream of mitogen-activated protein kinase (MAPK), without affecting glucose transport-4 (GLUT-4) translocation. 52 Conversely, Malin et al. 53 found that fetuin-A can directly impair glucose uptake by reducing GLUT-4 translocation in skeletal muscle.
Insulin receptor is a heterotetramer composed of two α and two β subunits. Insulin binds to the α-subunit, inducing a conformational change and autophosphorylation of tyrosine residues on the β-subunit, which activates downstream signaling pathways such as PI3K-Akt and Ras-MAPK. 54 Fetuin-A interacts with the tandem fibronectin type 3 (Fn3) domains located in the 194 amino acid residue extracellular region of the β-subunit of the insulin receptor, which is situated away from the high-affinity binding pocket produced by the two complementary α-subunits where insulin binds. Currently, only insulin and fetuin-A are known to bind directly to the receptor’s ectodomain, but with opposing effects: insulin activates, while fetuin-A inhibits, receptor function (Figure 2). 55

The role of fetuin-A in T2DM pathogenesis.
Fetuin-A has been strongly linked to obesity. In obese rats, fetuin-A gene expression is significantly elevated, 56 while fetuin-A knockout mice are resistant to diet-induced obesity, highlighting its critical role in weight regulation.57,58 In humans, several studies have shown a positive association between fetuin-A levels and both body mass index 39 and waist circumference. 58 Conversely, weight loss is associated with reduced fetuin-A levels, 28 suggesting its involvement in obesity-related metabolic disturbances.
In addition, fetuin-A is implicated in inflammation-associated insulin resistance. 59 Previous studies have shown that fetuin-A can stimulate the expression of proinflammatory cytokines at both mRNA and protein levels 9 and facilitates lipid-induced insulin resistance by enhancing the binding of free fatty acids (FFAs) to TLR4 via its terminal galactoside moiety. Fetuin-A can directly interact with the Leu100-Gly123 and Thr493-Thr516 residues in TLR4, promoting adipose tissue inflammation and, consequently, insulin resistance. 17 Consistent with this, a human study also confirmed the interaction between FFAs and fetuin-A in predicting insulin resistance. 60 Fetuin-A binds to FFAs and facilitates their interaction with TLR4 on adipocytes and macrophages, initiating proinflammatory signaling cascades such as the nuclear factor kappa B cells (NF-κB) and JNK pathways.17,61 This activation leads to the upregulation of cytokines, including tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and IL-6, all of which interfere with insulin receptor signaling and impair glucose uptake in peripheral tissues. For instance, fetuin-A stimulates the TNF-α production in monocytes and adipocytes. 9 Elevated TNF-α impairs insulin sensitivity and glucose metabolism, as shown in both animal and human studies,62,63 with regulation occurring through the tissue inhibitor of metalloproteases 3/TNF-α converting enzyme signaling axis.64,65 In addition to its role in TLR4 signaling, fetuin-A contributes directly to adipose tissue inflammation. It promotes the infiltration of monocytes into adipose depots and drives their polarization toward a proinflammatory M1 macrophage phenotype, thereby sustaining a local inflammatory environment. 66 This low-grade, chronic inflammation amplifies systemic insulin resistance and accelerates the progression of T2DM. 67
Finally, fetuin-A may suppress the expression of adiponectin, an adipokine known for its insulin-sensitizing and anti-inflammatory effects in adipose tissues. 9 By downregulating adiponectin, fetuin-A diminishes these protective effects, thereby contributing to insulin resistance and metabolic dysfunction.9,68 In summary, the collective evidence suggests that elevated fetuin-A levels contribute to insulin resistance, inflammation, obesity-related metabolic dysfunction, and ultimately, a higher risk of developing T2DM.
The relationship of fetuin-A with T2DM complications
The relationship between fetuin-A and complications of T2DM is complex. Both decreased and increased levels of fetuin-A have been associated with an increased risk of morbidity and mortality related to T2DM complications.69–72 Previous cross-sectional investigations revealed inverse relationships between fetuin-A levels and macrovascular conditions, such as peripheral arterial disease (PAD) and the formation of atherosclerotic plaques in individuals with T2DM.70,73,74 In addition, lower fetuin-A levels were linked to PAD in patients with advanced T2DM, independent of traditional cardiovascular risk factors, and tended to be even lower in those with more severe lower-extremity arterial calcification. 75 Conversely, longitudinal investigations such as the Rancho Bernardo Study and Cardiovascular Health Study reported that higher baseline fetuin-A levels in individuals with T2DM were associated with an increased risk of cardiovascular morbidity and mortality.71,76
With respect to diabetic nephropathy, Sherif et al. reported that patients with this condition had significantly lower mean fetuin-A levels compared to those without nephropathy. In addition, patients with macroalbuminuria exhibited notably lower fetuin-A levels than those with micro and normoalbuminuria. 77 These findings align with a previous prospective study that indicated lower fetuin-A levels are associated with a higher risk of developing diabetic nephropathy. 72 Furthermore, Mitkees et al. 78 discovered that diabetics without microalbuminuria had higher serum fetuin-A levels than those with microalbuminuria, revealing significant negative correlations between serum fetuin-A and albumin/creatinine ratios in both groups. Umapathy et al. 79 also reported that patients with persistent macroalbuminuria experienced a significant stepwise decline in circulating fetuin-A levels compared to those with micro- and normoalbuminuria. Conversely, a cross-sectional study indicated that diabetics with nephropathy had significantly higher fetuin-A levels than those without nephropathy. 43 Furthermore, T2DM patients with microalbuminuria had substantially elevated serum fetuin-A levels compared to those with normoalbuminuria, which was attributed to the influence of fetuin-A on insulin resistance, lipid profile abnormalities, and endothelial dysfunction. 80 Moreover, diabetic patients undergoing hemodialysis exhibited significantly higher fetuin-A levels than nondiabetic hemodialysis patients and healthy controls, suggesting that elevated circulating concentrations may contribute to the risk of kidney dysfunction. 81
In a prospective study, it was reported that lower fetuin-A levels are associated with an increased risk of diabetic retinopathy. 72 Similarly, another study found that serum fetuin-A levels were lower in cases of proliferative diabetic retinopathy compared to nonproliferative diabetic retinopathy and diabetics without retinopathy, although the differences among the groups were statistically insignificant. 77 However, in contrast to these findings, the fetuin-A levels in diabetic patients reportedly increased with the progression of diabetic retinopathy.82,83 In addition, Priya et al. 84 reported that T2DM patients with diabetic retinopathy exhibited higher circulating fetuin-A levels compared to those without this complication. On the other hand, a study by Mostafa et al. 85 suggested that fetuin-A plays a non-significant role in the etiopathogenesis of diabetic retinopathy.
Birukov et al. 72 found that lower levels of fetuin-A were associated with a higher incidence of neuropathy among diabetic patients. In a cross-sectional study, Sherif et al. 77 examined 160 patients with T2DM and reported that those with neuropathy had non-significantly lower mean fetuin-A levels compared to those without neuropathy. In contrast, Roos et al. 70 reported fetuin-A levels and their association with vascular disease in 149 T2DM patients with early diabetic nephropathy and found no association between fetuin-A levels and diabetic neuropathy. Finally, Jung et al. explored the associations between the levels of fetuin-A and diabetic microangiopathies (nephropathy, retinopathy, and peripheral neuropathy), including cardiac autonomic neuropathy in 172 T2DM subjects. Fetuin-A levels were not associated with diabetic neuropathy or cardiac autonomic neuropathy. 86
In light of these findings, the reasons for the differences in the effects of fetuin-A on macroangiopathies and microangiopathies remain unclear; several explanations have been suggested. One possibility is that the discrepancies may arise from differences in the study populations and the varying severity of complications among participants. 86 In addition, variations in fetuin-A concentrations suggest that fetuin-A secretion might serve as a feedback defense mechanism against vascular calcification in the early stages of diabetic and atherosclerotic diseases, while lipid disturbances and hyperinsulinemia could trigger hepatic release of fetuin-A. 34 Furthermore, it is essential to consider other factors that might account for the inconsistencies in the reports, such as the use of insulin-sensitizing medications like metformin, pioglitazone, and niacin; short-term dietary and exercise interventions; and differences in the ELISA kits used to measure serum fetuin-A levels. 87 These findings indicate that the relationship between fetuin-A and complications of T2DM is more complex; therefore, longitudinal studies with larger sample sizes are necessary to clarify the role of fetuin-A in the risk of developing T2DM-related complications. Table 2 summarizes the relationship between fetuin-A levels and T2DM complications.
Studies investigating the relationship of fetuin-A with T2DM complications.
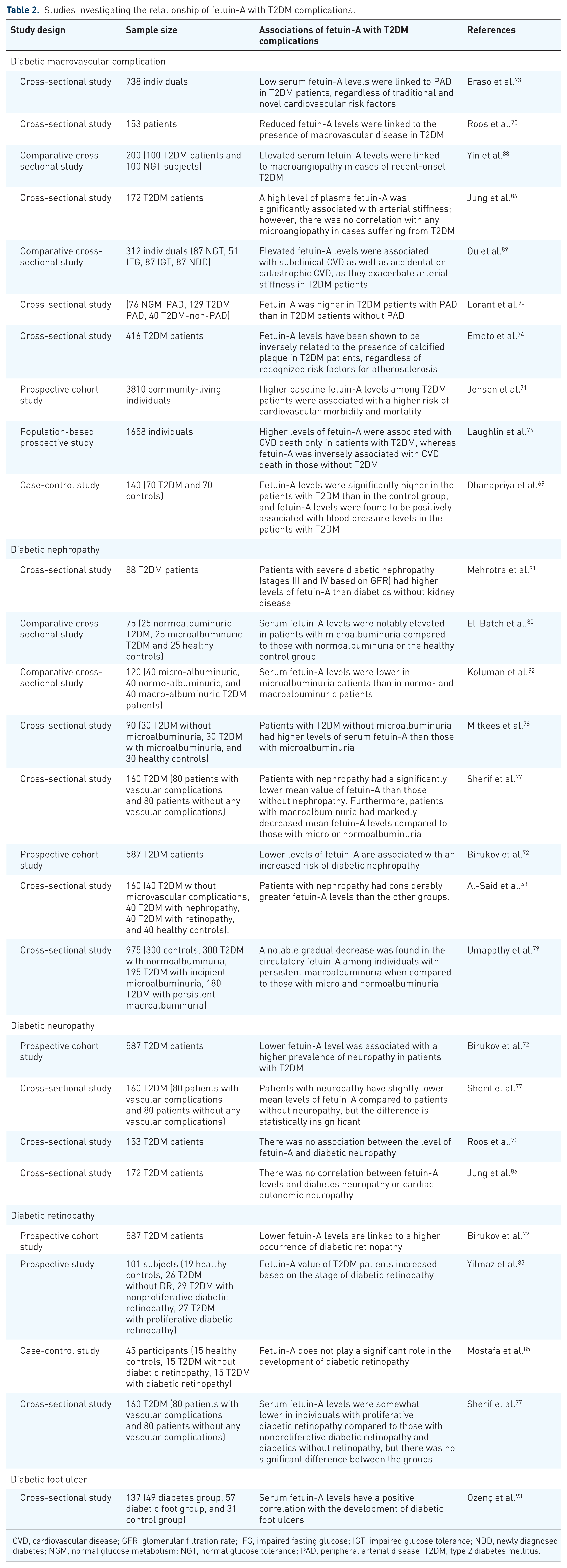
CVD, cardiovascular disease; GFR, glomerular filtration rate; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; NDD, newly diagnosed diabetes; NGM, normal glucose metabolism; NGT, normal glucose tolerance; PAD, peripheral arterial disease; T2DM, type 2 diabetes mellitus.
The potential diagnostic implications of fetuin-A in T2DM
A growing body of evidence suggests that fetuin-A may serve as a valuable diagnostic marker for various clinical conditions. Numerous studies have revealed a significant association between fetuin-A and T2DM, indicating its possible role in disease onset and progression.43,30,27 Additionally, other investigations have reported strong correlations between fetuin-A levels and T2DM-related complications.70,72,77 Collectively, these findings suggest that fetuin-A may function both as a diagnostic marker and as an indicator of T2DM severity in clinical practice.
Both preclinical and clinical studies have shown that elevated fetuin-A levels are a strong early predictor of various metabolic disorders, including obesity, T2DM, NAFLD, nonalcoholic steatohepatitis, and insulin resistance. 94 Demirbaş et al. 95 identified fetuin-A as an emerging risk factor for CVD and proposed a connection between high levels of fetuin-A and atherosclerosis, supporting its proatherogenic effects through the enhancement of insulin resistance. Conversely, a study has indicated that lower fetuin-A levels are associated with increased atherosclerosis and plaque formation. 74 Supporting this, a recent meta-analysis reported significantly lower circulating fetuin-A concentrations in patients with aortic valve calcification or stenosis. 96 These findings suggest that diminished fetuin-A may facilitate vascular calcification, underscoring its potential as a biomarker for both atherosclerosis and vascular calcification. 74
A study by Werida et al. 97 found a significant positive correlation between fetuin-A levels, fasting blood glucose, and the atherogenic index, suggesting that fetuin-A may serve as a novel biological marker for diagnosing dyslipidemia and other metabolic disorders. However, the fluctuating relationship between fetuin-A and both the early and advanced stages of atherosclerosis, along with the biphasic association with CVD development, may limit its utility as a diagnostic biomarker in clinical practice. 70 To establish fetuin-A as a reliable clinical tool, more comprehensive and rigorous research is required to clarify these inconsistencies and confirm its diagnostic value in CVD.
A recent clinical investigation found that elevated serum fetuin-A levels were associated with the development of NAFLD and T2DM, potentially through the activation of NF-κB, which inhibits IRS function. 98 Yamasandhi et al. 99 reported that fetuin-A levels were significantly higher in newly diagnosed T2DM patients with NAFLD compared to those without, suggesting that increased fetuin-A may predict NAFLD development. Similarly, Stephen et al. 47 found a positive association between fetuin-A and liver fat content after adjusting for age and sex. However, a negative association has been observed between fetuin-A and severe liver damage, possibly due to decreased hepatic production of fetuin-A in advanced disease stages. 87 These findings suggest that fetuin-A may serve as a potential biomarker in the progression of NAFLD, reflecting structural liver damage.
Regarding diabetic microvascular complications, fetuin-A has emerged as a novel urinary biomarker for monitoring the progression of diabetic nephropathy in T2DM patients. 100 In an analysis of the human urine proteome database, Magalhães et al. identified fetuin-A peptides as being associated with impaired kidney function in individuals with T2DM. Specifically, fetuin-A levels showed a negative correlation with the estimated glomerular filtration rate (eGFR) slope, indicating declining kidney function. Furthermore, fetuin-A may allow for earlier detection of kidney dysfunction compared to traditional markers, such as albuminuria, supporting its potential as a novel marker. 101 A large-scale proteomic analysis conducted as part of the Taiwan Renal Biomarker Study identified urinary concentrations of post-translationally modified, CP-containing fetuin-A fragments (uPTM-FetA) as promising markers of diabetic kidney disease in T2DM patients. 102 In prospective cohorts of patients with T2DM, uPTM-FetA demonstrates a strong association with the decline in renal function, independent of factors such as albuminuria, eGFR, age, sex, and other risk factors. 103 Consistent with this, emerging data indicate that uPTM-FetA may serve as a novel and sensitive biomarker for renal injury in chronic kidney disease (CKD) patients regardless of diabetic status. It could also help distinguish diabetic-related CKD from other causes, especially in complex cases or when a kidney biopsy is not feasible. 104 In terms of diabetic retinopathy, Li et al. investigated galectin-3 and fetuin-A levels in 100 T2DM patients, including 50 with and 50 without diabetic retinopathy, aged between 30 and 75 years. The study found elevated levels of both markers in patients with diabetic retinopathy, indicating their potential utility in predicting disease onset. When used together, galectin-3 and fetuin-A offered greater diagnostic value. 105 Supporting this, a cross-sectional study by Zhao et al. 82 demonstrated that serum and vitreous fetuin-A levels were significantly associated with the presence and progression of diabetic retinopathy. Furthermore, a recent meta-analysis by Das et al. 106 reported a significant increase in fetuin-A levels in patients with proliferative diabetic retinopathy. Similarly, another meta-analysis found that fetuin-A levels were significantly elevated in patients with diabetic retinopathy compared with both diabetic patients without diabetic retinopathy and nondiabetic controls. Fetuin-A concentrations were higher in individuals with proliferative diabetic retinopathy than in those with nonproliferative diabetic retinopathy. 107 Collectively, these findings provide strong evidence that circulating fetuin-A may serve as a novel biomarker for the diagnosis and monitoring of diabetic retinopathy. Regarding diabetic neuropathy, Karamfilova et al. assessed the relationship between fetuin-A and indicators of microangiopathy, including vibration perception threshold, neuropathy disability score, and autonomic neuropathy risk in 120 obese patients (40 patients without carbohydrate disturbances, 40 with prediabetes, and 40 with T2DM). The study revealed a positive correlation between fetuin-A and vibration perception threshold, and a negative correlation with the neuropathy disability score, highlighting a possible link between fetuin-A and diabetic neuropathy severity. 45
Beyond diabetes and its complications, recent studies have indicated that fetuin-A can be employed as a promising biological marker for cancer, 108 neurodegenerative diseases, 109 multiple sclerosis, 110 leukemia, 111 cognitive decline, 112 liver cirrhosis, 113 thoracic aortic aneurysms, 114 and COPD 115 in persons without T2DM.
Although fetuin-A is a promising potential diagnostic biomarker for T2DM and related conditions, several challenges hinder its practical use in clinical settings. These include the need for standardized assay methods, the high inter-individual and inter-population variability of fetuin-A levels, and the lack of clearly defined diagnostic cutoff values.116,117 Therefore, there is a pressing need to standardize fetuin-A measurement techniques and establish consistent reference ranges to enable its reliable use in clinical practice.
The potential therapeutic implications of fetuin-A in T2DM
Fetuin-A has been identified as a potential target for treating various diseases, and it may also serve as a biomarker. 118 Its growing importance in conditions such as T2DM underscores the necessity for therapies that can effectively manage fetuin-A levels. 27 Consequently, fetuin-A is predicted to emerge as a new therapeutic target for T2DM management. High levels of fetuin-A have been linked to insulin resistance and T2DM-related complications, such as CVD and NAFLD. 26 Studies have demonstrated that lowering fetuin-A levels or blocking its activity can enhance glucose tolerance and reduce systemic inflammation.87,119 This indicates that therapies aiming at modifying fetuin-A’s activity could be a promising new approach to treating insulin resistance and metabolic dysregulation in T2DM.
Considerable efforts are being made to develop effective therapies for regulating fetuin-A levels. Recent studies have shown that both pharmacological agents, such as metformin and pioglitazone and natural compounds, such as resveratrol and niacin, can modulate circulating fetuin-A levels.120,121 Specifically, treatment with pioglitazone and metformin has been associated with decreased fetuin-A levels. Esteghamati et al. conducted a 3-month follow-up study to compare the effects of metformin (1000 mg/day; 500 mg twice daily) and pioglitazone (30 mg/day; 15 mg twice daily) on circulating fetuin-A concentrations in patients with newly diagnosed T2DM. The findings revealed that pioglitazone significantly reduced fetuin-A levels independent of sex, whereas metformin exerted no measurable effect. Importantly, pioglitazone demonstrated superior efficacy in lowering fetuin-A levels, with the reduction being particularly pronounced in male patients. 120 Another study reported a non-significant decrease in fetuin-A levels in patients receiving metformin compared with newly diagnosed untreated T2DM patients, suggesting that the effect of metformin on fetuin-A may be limited. 122 Besides, it appears that the release of fetuin-A reduces adiponectin release, whereas dietary intake of resveratrol seems to enhance adiponectin levels. 123 Decreased adiponectin secretion has been associated with an increased risk of several disorders, including NAFLD, insulin resistance, and obesity.123,124 Thus, the interplay between fetuin-A and resveratrol may be mediated through their opposing effects on adiponectin release. Consistent with this, Lee et al. 125 reported that 4 weeks of resveratrol supplementation (8 mg/kg/day) increased serum adiponectin levels while simultaneously reducing fetuin-A levels in an in vivo study. Similarly, niacin treatment lowers serum fetuin-A levels, which coincides with favorable changes in blood lipids.121,126
In an in vivo study, Hsu et al. 127 demonstrated that a 12 weeks of apigenin (20 mg/kg/day) administration suppresses fetuin-A mRNA expression by inhibiting NF-κB activation, a key transcription factor involved in immune responses and insulin resistance. 128 Apigenin counteracted the palmitic acid-induced increase in fetuin-A expression and formation of the fetuin-A–insulin receptor complex, thereby restoring insulin signaling and enhancing glucose uptake in hepatocytes. 127 In a randomized study of 67 patients, participants were assigned to three 12-week treatment groups: glimepiride 4 mg with placebo (n = 22), glimepiride 4 mg with curcumin 1100 mg (n = 23), and glimepiride 4 mg with fenofibrate 160 mg (n = 22). Both curcumin and fenofibrate, as adjuncts to glimepiride, improved glycemic control, lipid profiles, and inflammatory markers (fetuin-A and sirtuin 1), with fenofibrate demonstrating a more pronounced reduction in circulating fetuin-A. 129 Werida et al. 97 evaluated 70 patients with T2DM, of whom 40 received rosuvastatin 10 mg/day and 30 received a placebo for a duration of 3 months. The rosuvastatin dose was administered in accordance with ACC/AHA recommendations. 130 Rosuvastatin therapy significantly improved glycemic control and reduced fetuin-A levels, suggesting its potential role in improving lipid profiles and atherogenic indices. Notably, rosuvastatin therapy demonstrated greater efficacy in reducing fetuin-A levels compared with the placebo group, highlighting its additional metabolic benefits beyond lipid-lowering effects. 97 Similarly, research by Nag et al. 131 found that vildagliptin dose-dependently suppressed palmitate-induced fetuin-A expression. Vildagliptin (20 mg/kg/day) inhibited the palmitate–fetuin-A-mediated activation of the TLR4–NF-κB pathway and decreased IL-1β release, thereby alleviating inflammation in pancreatic islet beta cells. Collectively, these findings support the hypothesis that targeting fetuin-A may offer substantial health benefits and represent a promising therapeutic strategy for more effective T2DM management.
Currently, numerous recent studies have shown that short-term dietary and exercise interventions can effectively reduce circulating fetuin-A levels in patients with T2DM.132,133 For instance, Farsani et al. reported that combined intervention of ursolic acid supplementation (250 mg/kg/day) and resistance training significantly reduced circulating fetuin-A levels, whereas each intervention alone failed to elicit such an effect. This observation underscores a potential synergistic interaction between nutritional and exercise-based strategies in modulating fetuin-A. 133 Nevertheless, several studies have also demonstrated that exercise interventions alone can lower fetuin-A.134–136 Keihanian et al. 135 reported that an 8 weeks program combining aerobic and resistance exercise effectively reduced serum fetuin-A levels in men with T2DM. Meta-analyses further support the role of exercise in improving glycemic outcomes by reducing fetuin-A levels in individuals with hyperglycemia and diabetes. 136 Moreover, in T2DM patients with nephropathy, moderate regular physical activity has proven more effective in lowering fetuin-A levels and improving associated renal impairment. 137 Several proposed processes explain why physical exercise reduces fetuin-A levels. The mechanisms underlying this effect may involve reductions in hepatic fat content and glucolipotoxicity, as well as mitigation of reactive oxygen species generation and proinflammatory signaling.138,139 In addition, exercise-induced activation of the Akt signaling pathway can enhance glucose tolerance and reduce insulin resistance. 140 Overall, these findings underscore fetuin-A’s potential as a therapeutic target for managing T2DM and related metabolic disorders. However, additional studies are needed to validate these findings and fully elucidate the molecular mechanisms involved.
Although this review integrates findings from multiple studies and offers insights into the relationship between fetuin-A levels and T2DM, thereby contributing to a better understanding of the role of fetuin-A in T2DM pathology and its potential clinical implications, it also has certain limitations. First, as a narrative review, it has a limited scope and does not provide a comprehensive examination of the subject matter. Additionally, the lack of direct comparisons between studies diminishes the ability to critically evaluate the consistency and reliability of the findings related to fetuin-A and T2DM, which could affect the robustness of the conclusions. Furthermore, as a narrative review, this work is inherently prone to potential biases in literature selection and interpretation, since it does not adhere to the predefined methodological rigor of a systematic review. Moreover, the conclusions drawn from this review may be influenced by the authors’ perspectives, as they are not based on a thorough analysis of all available evidence, potentially limiting the objectivity of the findings.
Conclusion and future perspectives
Numerous studies have found a link between fetuin-A and T2DM and its associated complications, but the findings are inconsistent and inconclusive. More extensive research is needed to reconcile conflicting results and gain a better understanding of fetuin-A’s potential role in T2DM development. Fetuin-A is currently the focus of research efforts due to its potential clinical significance for diagnosis and treatment. It is proposed that fetuin-A be used as a diagnostic tool for T2DM and its associated complications in medical practices in the coming years. However, issues such as the necessity for standardized tests, dealing with variability in fetuin-A levels among groups, and determining particular cutoff values for diagnostic purposes impede its practical use. In addition, fetuin-A has been identified as a possible therapeutic target in T2DM. Currently, some drugs and natural compounds such as metformin, pioglitazone, resveratrol, apigenin, fenofibrate, rosuvastatin, and vildagliptin have been recognized as potential agents for modulating fetuin-A levels in patients with T2DM. Several recent studies also showed that short-term dietary and exercise interventions lead to a decrease in serum fetuin-A levels. Future research should concentrate on understanding the molecular mechanisms underlying the observed relationships as well as investigating novel treatment targets for improving clinical outcomes in T2DM patients. Furthermore, large-scale, multicentered cohort studies are necessary to confirm the diagnostic accuracy and clinical utility of fetuin-A in patients with T2DM.
