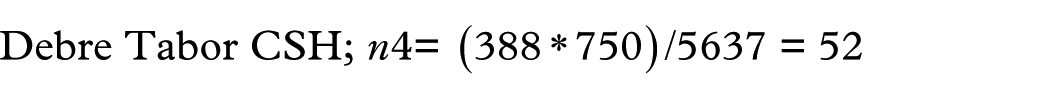Abstract
Background:
Optimal use of renin-angiotensin system (RAS) modulators plays a crucial role in improving the outcomes for chronic heart failure (CHF) patients with reduced ejection fraction (rEF). Despite their established benefits, there is limited evidence regarding real-world prescribing patterns, dose optimization, and factors influencing RAS modulator use in this population.
Objective:
This study aimed to evaluate the patterns of use, dose optimization, and associated factors affecting the administration of RAS modulators among CHF patients with rEF at Public Comprehensive Specialized Hospitals (PCSHs).
Design:
A hospital-based, multicenter cohort study was conducted from February 1, 2020, to May 31, 2024, at PCSHs among CHF patients with rEF.
Methods:
A total sample size of 385 patients was determined using a systematic random sampling technique at the Northwest Ethiopian PCSHs during the study period. Data were collected from medical records and interviews using standardized questionnaires. Data analysis was performed using SPSS version 27.0, and binary logistic regression analysis was employed to identify factors associated with the use and optimization of RAS modulators. The study strictly adhered to the most recent guideline recommendations from the American Heart Association (2022) and the European Society of Cardiology (2021).
Results:
Of 385 patients, 263 (68.3%) were prescribed RAS modulators; however, only 86 (32.7%) of these patients were receiving an optimal dose. Predictors significantly associated with the use of RAS modulators included a duration of CHF with rEF of ⩾3 years (AOR: 1.79, 95% CI: 1.02–3.15), the presence of ischemic heart disease (AOR: 8.23, 95% CI: 4.23–16), hypertension (AOR: 2, 95% CI: 1.09–3.69), diabetes mellitus (AOR: 7.34, 95% CI: 1.48–36.34), chronic kidney disease (AOR: 4.35, 95% CI: 1.32–14.34), and a furosemide dose of ⩾40 mg (AOR: 0.26, 95% CI: 0.013–0.49). Regarding suboptimal RAS modulator dosing, significant predictors identified were age ⩾65 years (AOR: 2.83, 95% CI: 1.46–5.50), a previous history of hospitalization (AOR: 2.05, 95% CI: 1.07–3.95), the use of diuretics (AOR: 5.34, 95% CI: 2.73–10.44), a furosemide dose of ⩾40 mg (AOR: 3.88, 95% CI: 1.89–7.97), and CHF with rEF for ⩾3 years (AOR: 0.31, 95% CI: 0.16–0.63).
Conclusions:
The majority of CHF patients with rEF received suboptimal doses of RAS modulators, with only one-third receiving optimal therapy. This highlights a critical gap in treatment that must be urgently addressed. Targeted interventions are needed to identify and mitigate modifiable predictors contributing to suboptimal dosing, thereby improving therapeutic outcomes and reducing the burden of CHF with rEF.
Keywords
Introduction
Heart failure (HF) is a complex and debilitating cardiovascular condition that can lead to significant morbidity and mortality worldwide.1–3 The progression of HF is associated with adaptive changes such as cardiac fibrosis, hypertrophy, apoptosis, and inflammation, which are driven by the activation of the “fetal” gene program along with the reduction of specific messenger ribonucleic acid (mRNA) expression patterns. This highlights the critical role of molecular mechanisms that regulate and decrease steady-state mRNA levels.4,5 Currently, it affects approximately 64 million people globally and accounts for nearly 26 million hospitalizations each year, underscoring its substantial burden on healthcare systems.1,6 The severity of HF is further highlighted by its high mortality rate, with about 59% of patients dying within 5 years of diagnosis. 7
In Ethiopia, the prevalence of HF is estimated to be around 1%–2% among the adult population, exerting a disproportionate strain on the country’s limited healthcare resources. This reflects broader global trends where HF prevalence and associated disability are increasing, particularly in low- and middle-income countries, due to factors such as aging populations and rising rates of coronary heart disease, previous heart attack, elevated uric acid, diabetes mellitus, and cardiomyopathy, the primary underlying causes of HF worldwide.8–14
In addition to these underlying causes, chronic inflammation is now seen as a key cause of cardiovascular diseases, including atherosclerosis, hypertension, and heart failure. It promotes problems with the blood vessel lining, activates immune cells, and makes plaques unstable. Ongoing inflammation signals cause changes in the heart and blood vessel structure, resulting in worse health outcomes. Important inflammatory substances, such as interleukins (IL-1β, IL-6), C-reactive protein, tumor necrosis factor-α, and nitrotyrosine, play crucial roles in these processes.4,15
Optimization of guideline-directed chronic HF medical therapy is highly recommended with every patient visit to improve outcomes in patients with HF with reduced ejection fraction. 16 These therapies typically include angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), angiotensin receptor-neprilysin inhibitors (ARNIs), mineralocorticoid receptor antagonists (MRAs), cardio-selective beta-blockers (BBs), and sodium-glucose transport protein 2 inhibitors (SGLT2i), among other interventions such as cardiac resynchronization therapy (CRT).11,17–19 These therapies mainly aim to slow the progression of HF and improve survival by preventing cardiac remodeling. By modulating the adaptive cardiac processes involved in fibrosis, hypertrophy, and apoptosis, anti-HF treatments can exert anti-remodeling effects, ultimately leading to better clinical outcomes for patients by improving the left ventricular ejection fraction and New York Heart Association (NYHA) classes.17,20,21
ACEIs, ARBs, and ARNIs collectively known as renin-angiotensin system (RAS) modulators, regarded as the cornerstone of standard care for chronic heart failure (CHF) patients with reduced ejection fraction (rEF)18,19 due to improve cardiac function and reduce cardiac remodeling by normalizing molecular pathways including calcium handling and oxidative stress, which may indirectly modulate growth hormone (GH) and insulin-like growth factor-I (IGF-I) signaling pathways involved in cardiac growth and repair. 22 RAS modulators help reduce hospitalizations and death in HF patients who do not respond to CRT with a defibrillator (CRTd) by modulating microRNAs (miRNAs) involved in cardiac remodeling.5,23 Research indicates that ARNI therapy can raise the chances of converting CRTd non-responders to responders due to its anti-remodeling effects related to these pathways. 23 Generally, these agents improve symptoms, enhance survival, decrease the frequency of hospital admissions, and reduce the incidence of hospitalization, morbidity, and mortality.24,25 Importantly, the clinical benefits of RAS modulators are dose-dependent, with greater efficacy observed at higher target doses. Therefore, up-titration to the maximum tolerated dose is critical to optimize patient outcomes and quality of life. According to the information provided in the prescribing information, titration should begin 2–4 weeks after hospital discharge.16,20,21,26
RAS modulators remain a cornerstone of CHF with rEF management, with guidelines recommending specific target doses, for example, 20–40 mg of Enalapril, 10 mg of Ramipril, or 97 mg sacubitril and 103 mg valsartan twice daily to achieve mortality and morbidity benefits.18–20 Compared to other RAS modulators, many observational studies and registries have shown that ARNI treatment in HF patients significantly reduces mortality and hospitalization rates. It also improves symptoms and promotes favorable cardiac reverse remodeling in various populations. For instance, real-world data from multiple cohorts show ARNI’s association with a 23%–66% relative risk reduction in primary clinical endpoints compared with standard therapy, with manageable safety profiles and improved health status.27–30
Despite this, global data reveal variable RAS modulator utilization rates: 65%–80% of patients in North America receive RAS modulators following hospitalization, 31 whereas rates in Ethiopia range from 62% to 75%, with Enalapril being the most commonly prescribed agent. 32 Although RAS modulators confer a 16%–20% reduction in mortality, underdosing remains a persistent issue. This is often due to adverse effects such as hypotension, renal impairment, and angioedema, the latter being more prevalent among women and Black individuals. 33 Clinical guidelines recommend cautious use of RAS modulators in patients with renal dysfunction or hyperkalemia and advocate for combination therapy with diuretics or beta-blockers, which are prescribed in approximately 49% and 34% of patients, respectively. 19
This study aimed to evaluate the patterns of RAS modulator utilization, dose optimization, and factors influencing these practices in patients with CHF with rEF. By addressing the gap between evidence-based recommendations and real-world clinical practice, the study seeks to improve the quality of care and patient outcomes in this population.
Methods and materials
Study design, period, and setting
A multicenter cohort study was conducted from February 1, 2020, to May 31, 2024, at the outpatient clinics of public comprehensive specialized hospitals (PCSH) in Northwest Ethiopia. The study involved five major hospitals: The University of Gondar, Tibebe Ghion, Felege Hiwot, Debre Tabor, and Debre Markos.
The University of Gondar Comprehensive Specialized Hospital (CSH) is located 750 km northwest of Addis Ababa and serves over 7 million people in the Amhara region. Felege Hiwot and Tibebe Ghion CSHs are both in Bahir Dar City, the regional capital, 565 km from Addis Ababa, and collectively provide healthcare to approximately 5 million people. Debre Tabor CSH, 667 km from Addis Ababa and 104 km from Bahir Dar, serves over 3 million individuals. Debre Markos CSH, located 300 km from Addis Ababa, serves an estimated population of 5 million.
Source and study population
The source population consisted of all adult CHF patients with rEF who had follow-up at the outpatient clinic of PCSHs, while the study population included CHF patients with rEF attending the outpatient clinic of PCSHs for follow-up during the study period.
Eligibility criteria
Inclusion criteria
Patients aged 18 years or older, with baseline information documented in their medical charts.
Outpatient CHF patients with at least 6 months of follow-up. 18
Patients with an ejection fraction (EF) of 40% or less, based on a recent echocardiogram performed within the last 2 years. 19
Patients who were willing to participate in the study.
Exclusion criteria
Patients with hearing or speaking problems who had no caregiver, as well as.
Severely ill patients were excluded from the study.
Sample size determination and sampling procedure
The sample size was calculated using a single population proportion formula, considering a 95% confidence interval (CI), a 5% margin of error, and the proportions of 74.7% and 35.7% for the utilization and dose optimization of RAS modulators among patients with CHF.
32
Therefore, the sample size can be 353 patients. By adding 10% non-respondents, the final sample size was 388 individuals. The total population of CHF patients with rEF during the study period across the five hospitals was 5637. Proportional allocation was used to ensure representativeness from each hospital based on the number of CHF patients: University of Gondar CSH (1750 patients), Tibebe Ghion CSH (1250), Felege Hiwot CSH (998), Debre Tabor CSH (750), and Debre Markos CSH (889). Having this minimum study sample size, the proportional allocation of samples to the total population of each PCSH ambulatory clinic was applied using the following formula (Figure 1):
A total calculated sample size of 388 was determined, comprising five groups (n1 + n2 + n3 +n4 + n5). In the final study, 385 CHF patients with rEF participated, representing a 99.2% response rate (385 out of 388). Three patients withdrew because of their unwillingness to participate. To select participants from each hospital, a systematic random sampling technique was used. Specifically, the sampling interval (k) was calculated by dividing the total number of CHF patients with rEF in the study area (5637) by the total sample size (388), yielding an interval of approximately 15 (k = 5637/388 = 15). This means that every 15th patient was systematically selected for inclusion after a randomly chosen start point, ensuring a representative and unbiased sample across the hospitals. The starting point was selected randomly from 1 to 15. Then, participants were interviewed, and concurrently, relevant data were recorded from the medical charts of every 15 patients until the requirement for the sample was fulfilled.

Sampling procedure at PCSHs in Northwest Ethiopia, 2024 (n = 385).
The baseline total number of heart failure cases for the preceding 4 months of the study period from the registration logbook was allocated to the respondents proportionally within the study areas (Figure 1).
Variables of the study
The dependent variables included the RAS modulator utilization pattern (underutilized and utilized) and dose optimization (suboptimal and optimal). The independent variables encompassed socio-demographic factors, comorbidities, medication side effects, clinical characteristics, duration of CHF with rEF, frequency of hospitalization, CHF with rEF medications, diuretic use, furosemide dosage, the number of current medications, and lab results.
Operational definitions
RAS modulators are considered “tolerated”: If BP is ⩾90/60 mmHg, serum creatinine is ⩽3 mg/dl, serum potassium is ⩽5.5 mg/dl, and there is no history or current indication of a cough or angioedema.18,19
Appropriate dose titration of RAS modulators: The time interval between dose titrations will be “appropriate” if the tolerated dose is titrated on time (every 2–4 weeks). However, if a dose is not titrated within every 2–4 weeks of the time interval, it is deemed “inappropriate” to do so.18,19
The optimal dose of RAS modulators is defined as the optimal dose of guideline-directed medical therapy(GDMT) used for HF patients. For angiotensin-converting enzyme inhibitors (ACEIs) medications, the guideline-recommended daily TD or maximum tolerable dose is defined as 20 mg/day for Enalapril, 150 mg/day for Captopril, and 20 mg/day for Lisinopril. If the patient is intolerant to ACEIs, ARBs are indicated, such as Valsartan 320 mg/day, Losartan 50–150 mg/day, and Candesartan 32 mg/day. ARNI is also indicated as a first-line treatment option in place of ACEI or ARBs for symptomatic CHF patients with rEF, such as 97 mg sacubitril and 103 mg valsartan twice daily.34–37
Suboptimal dose of RAS modulators: If a patient is receiving any dose of medication that is less than the guideline-recommended TD or maximum tolerable dose without any contraindications.34,38
Underutilized RAS modulators: If medications are not taken by the patients in the absence of contraindications.39,40
Overall treatment optimization of RAS modulators: When they are optimized, titrated appropriately (2–4 weeks), and the patients tolerate these medications.34,35
Data collection techniques and instruments
A structured data abstraction format, which was prepared by reviewing similar relevant studies,41–44 was used. The format contained the socio-demographic, clinical (medical), treatment (medication) characteristics, physical examination, and lab values of the patients. The socio-demographic characteristics of the patient, as well as some medication-related information, were retrieved by interviewing patients using the standardized questionnaire. Whereas the clinical, treatment-related characteristics, physical examination, and lab values of the patients were retrieved from the patients’ medical records retrospectively from February 1, 2020, to May 31, 2024 (study period).
Data quality assurance
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement.45–48 Therefore, the checklist from the relevant guidelines should be submitted as a Supplemental File. 49 The questionnaire was pretested by the Cronbach alpha test on 5% of the study participants (n = 20 patients) at UOG-CSH, Gondar, Ethiopia, before the actual data collection in the study area. These pretested participants were excluded from the part of the study to avoid bias. The reliability test from Cronbach’s α was measured and calculated (α = 0.86), and it was considered to have high internal consistency. The data were collected by five clinical pharmacists from February 1 to May 31, 2024 (data collection period). Three supervisors were recruited for the continuous supervision of the data collectors. The principal investigator trained the data collectors and supervisors for 3 days on the study’s objectives, ethical principles, adherence to the data abstraction format, and how and what data were collected from the patients both through interviews and using patient medical records.
Data analysis and management
The data were coded and entered into Epi-Data version 4.6 software (Centers for Disease Control and Prevention (CDC) in Denmark) and then exported to SPSS version 27.0 (IBM Corporation, Armonk, NY, USA) for analysis. Descriptive statistics was carried out and presented with frequency and graphical presentation. Outliers and multicollinearity of the variables were checked before running the bivariate and multivariable binary logistic regression analyses. Then, after all variables were shown, VIF <2.2 and VIF <1.25 were found in the utilization and dose optimization of RAS modulators, respectively; hence, there was no evidence of multicollinearity.
A binary logistic regression analysis (bivariate and multivariable binary logistic regression) was carried out to find the statistically significant factors. The variables that had statistically significant associations at p-values <0.20 with the dependent variable in the bi-variable logistic regression analysis were further considered candidates for a multivariable logistic regression to control for the possible effect of confounding variables. Lastly, statistically significant variables were established at p-value <0.05 in a multivariable binary logistic regression model, and an adjusted odds ratio (AOR) with a 95% confidence interval was reported to measure the strength of association. The model fitting information was checked by the Omnibus test (at p < 0.05), (χ2 = 98.2, df = 14, p < 0.001), and (χ2 = 101.87, df = 8, p < 0.001) in the utilization and dose optimization of RAS modulators, respectively, which were significant, so it indicates that the full model represents a significant improvement in fit over the null model. The Hosmer-Lemeshow test (at p-value >0.05) was used to determine the final model’s goodness-of-fit, and the results revealed that the model was well-fitted at p-value 0.518 and 0.20 in the utilization and dose optimization of RAS modulators, respectively. Nagelkerke’s (pseudo R 2 ) showed how much the independent variables explain the response variable (utilization and dose optimization of RAS modulators) by 31.7% and 44.8% in the model, respectively.
Ethical approval and consent to participate
Ethical clearance was obtained from the Institutional Review Board (IRB) of the University of Gondar, College of Medicine and Health Sciences, and Department of Clinical Pharmacy before conducting the study with a reference number (Ref: SOP 281/2024). A letter of permission was given by the clinical directorate of the outpatient clinic at the UOG-CSH. Both written and verbal informed consents were obtained from the patients. Participants who were unable to provide a signature themselves made a mark (such as an “X”) or provided a fingerprint on the signature line. This procedure was carried out in the presence of an impartial witness, who observed the entire oral presentation to ensure comprehension and voluntary agreement. The witness then signed the designated witness line on the long English form of consent to document the process. If the patients had hearing or speaking problems, informed consent was obtained from the patients’ caregivers. Confidentiality was ensured through no patient’s name or identifier was taken; rather, codes were used as identifiers. Information regarding patients was used for the study only and in adherence to the Declaration of Helsinki.
Results
Socio-demographic characteristics of the study participants
Of 388 selected samples, 385 CHF patients with rEF participated in this study, with a response rate of 99.2%. Out of the total participants, the majority, 208 (54%), were males. The majority of the study subjects lived in urban areas, 235 (61%). In addition to this, the mean (±SD) age of study subjects was 57.15 ± 19.90 years. The median age of the participants was 63 (18–92). Almost half, 199 (51.7%) of patients were unable to read and write (Table 1).
Socio-demographic characteristics of CHF patients with rEF at PCSHs, Northwest Ethiopia, 2024 (n = 385).

CHF, chronic heart failure; PCSH, public comprehensive specialized hospitals; rEF, reduced ejection fraction.
Clinical and health-related features of CHF patients with rEF
Nearly half, 188 (48.8%), of the patients had been hospitalized ⩾1 time in the preceding year. The mean (±SD) EF of the participants was 31.77 ± 8.10, and the majority of participants, 241 (62.6%), had EF between 31% and 40%. The systolic blood pressure (SBP) of the participants was 120.35 ± 18.79 (Table 2).
Clinical and health-related features of CHF patients with rEF at PCSHs, Northwest Ethiopia, 2024 (n = 385).

CHF, chronic heart failure; DBP, diastolic blood pressure; K+, potas; LVEF, left ventricular ejection fraction; PCSH, public comprehensive specialized hospitals; rEF, reduced ejection fraction; SBP, systolic blood pressure; SD, standard deviationsium.
Types of comorbidities associated with CHF patients with rEF
More than half, 220 (57.1%) of the patients had ⩾2 comorbidities. Hypertension, 168 (43.6%), and ischemic heart disease, 157 (40.8%), were the most common comorbidities, followed by valvular heart disease, 122 (31.7%) (Figure 2).
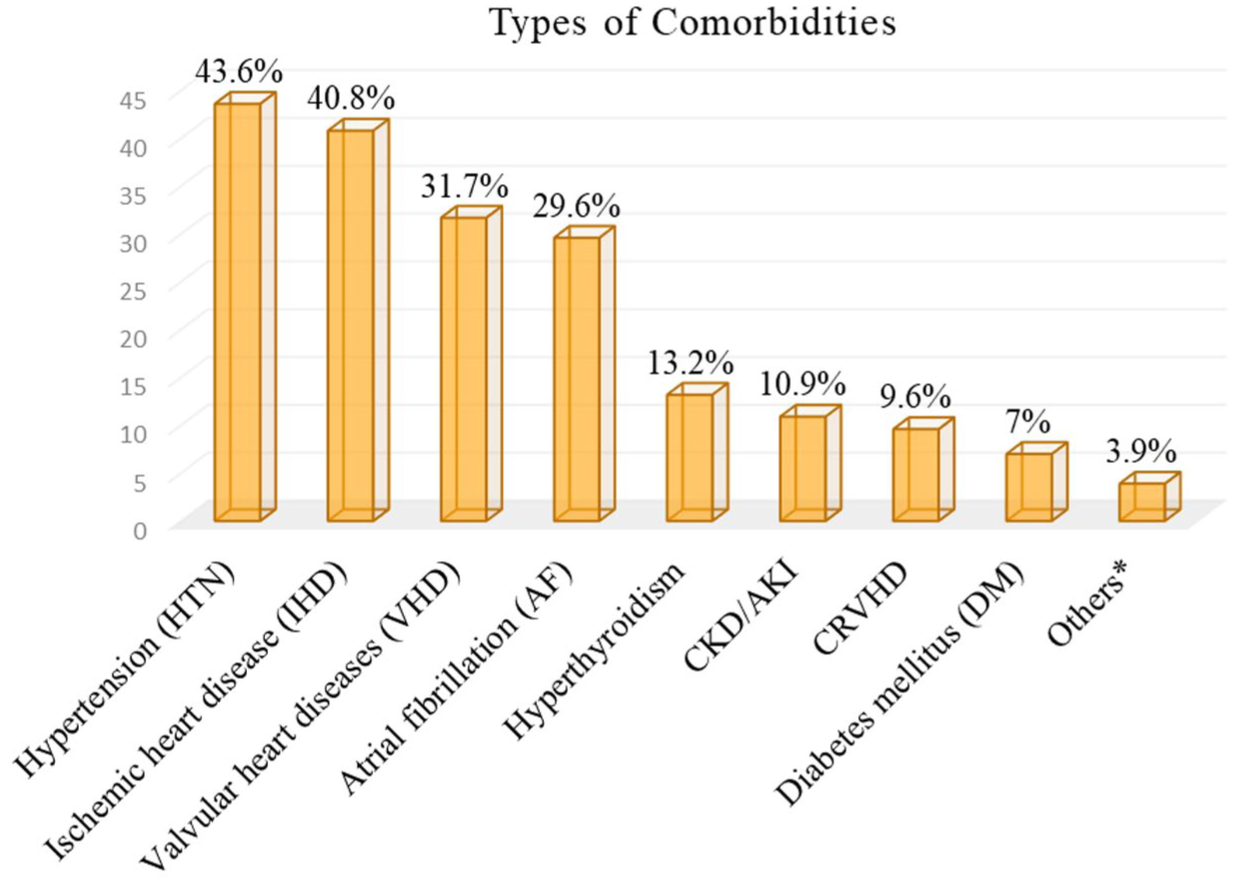
Types of comorbidities among CHF patients at PCSHs, Northwest Ethiopia, 2024 (n = 385).
CHF, chronic heart failure; CKD/AKI, chronic kidney disease/acute kidney injury; CRVHD, chronic rheumatic valvular heart disease; PCSH, public comprehensive specialized hospital.The NYHA class and ACCF/AHA staging system of CHF patients with rEF
The majority of participants were classified as NYHA functional class III, accounting for 209 patients (54.3%), followed by class IV with 125 patients (32.5%). Regarding ACCF/AHA stages, Stage C was the most prevalent, including 291 patients (75.6%), followed by Stage B with 48 patients (12.5%) (Figure 3).

Proportion of NYHA class and ACCF/AHA staging system among CHF patients with rEF taking RAS modulators at the ambulatory clinic of PCSH, Northwest, Ethiopia, 2024 (n = 385).
Management-related clinical aspects of CHF patients with rEF
The mean (±SD) number of HF medications prescribed per patient was 4.0 ± 1.685. The majority of patients, 50.6% were taking ⩾4 medications, while 49.6% were taking <4 medications at the same time. Loop diuretics (74.3%), BBs (68.6%), ACEIs/ARBs (68.3%), MRAs (48.1%), antiplatelet (47.5%), and statins (45.2%) were also the most frequently used HF medications (Figure 4).

Commonly prescribed medications among CHF patients at the outpatient clinic of PCSH, Northwest, Ethiopia, 2024 (n = 385).
Current utilization of RAS modulators among CHF patients with rEF
Among the total HF patients, 263 (68.3%) were prescribed RAS modulators. Of these, Enalapril was the most commonly prescribed medication, given to 250 patients (95%), followed by Captopril to 12 patients (4.6%), and losartan to 1 patient (0.4%). However, only 86 patients (32.7%) receiving RAS modulators were on an optimal dose, while the majority, 177 patients (67.3%), were on suboptimal doses. The mean daily doses for the medications were 13.7 mg for Enalapril, 55.9 mg for Captopril, and 25 mg for losartan (Table 3).
Treatment optimization of RAS modulators in CHF patients with rEF at PCSHS, Northwest Ethiopia, 2024 (n = 385).

CHF, chronic heart failure; IQR, interquartile range; PCSH, public comprehensive specialized hospitals; RAS, renin-angiotensin system; rEF, reduced ejection fraction; SD, standard deviation; TD, target dose (Figure 5).

Utilization of RAS modulators among CHF patients with rEF at the outpatient clinic of PCSH, Northwest, Ethiopia, 2024 (n = 385).
Treatment optimization and overall tolerability of the RAS modulators
Based on monitoring parameters such as potassium levels, atrial blood pressure, creatinine levels, cough, and angioedema, 87.5% of the study participants tolerated the RAS modulators (95% CI: 82.8–91.2). The mean (±SD) time to dose optimization of RAS modulators was 6.10 ± 1.87 weeks. Additionally, the median duration of RAS modulator use was 6 weeks, with an interquartile range (IQR) of 3–12 weeks (Table 4 and Figure 6).
Comparison of the overall toleration, timely titration, dose optimization, and overall RAS modulator treatment optimization during different periods of titration among CHF patients with rEF at the outpatient clinic of PCSHs, Northwest Ethiopia, 2024.

CHF, chronic heart failure; PCSH, public comprehensive specialized hospitals; RAS; renin-angiotensin system modulators; rEF, reduced ejection fraction.

Comparison of the overall toleration, timely titration, dose optimization, and overall RAS modulators treatment optimization during different periods of titration among CHF patients with rEF at the outpatient clinic of PCSHs, Northwest Ethiopia, 2024. (a) RAS modulators overall tolerability. (b) RAS modulators timely titration. (c) RAS modulators dose optimization. (d) Overall RAS modulators treatment optimization.
Dose optimization and reasons for not achieving target dosages and nonuse of RAS modulators
The main reasons for not achieving the target dose are ongoing dose adjustment, side effects (hypotension, cough, hyperkalemia), and sometimes no clear reason. The main reasons for nonuse are medical contraindications (low blood pressure, kidney issues, valvular heart disease, hyperkalemia) or alternative treatment requirements (Table 5 and Figure 7).
Reasons for not achieving target dosages and nonuse of RAS modulators among CHF patients with rEF at the outpatient clinic of PCSH, north-west Ethiopia, 2024 (n = 385).

CHF, chronic heart failure; PCSH, public comprehensive specialized hospitals; RAS; renin-angiotensin system modulators; rEF, reduced ejection fraction; TD, target dose.

Dose optimization of RAS modulators among CHF patients with rEF at the outpatient clinic of PCSH, Northwest, Ethiopia, 2024 (n = 263).
Predictors associated with the use of RAS modulators in CHF patients with rEF
In the bi-variable logistic regression analysis, duration of CHF ⩾3 years, IHD, HTN, DM, hyperthyroidism, CKD, previous history of hospitalization, frequency of hospitalization ⩾2, number of comorbidities ⩾2, age ⩾65 years, and dose of furosemide >40 mg were predictors associated with the utilization of RAS modulators (p < 0.2). However, after controlling for the confounding variables in the multivariable binary logistic regression analysis (p < 0.05), CHF with rEF ⩾3 years, IHD, HTN, DM, CKD, and dose of furosemide >40 mg were found to be statistically significant variables associated with the utilization of RAS modulators.
After adjusting for other covariates, the odds of using RAS modulators were 1.79 times higher among patients with CHF for ⩾3 years compared to those with a duration of less than 3 years (AOR: 1.79; 95% CI: 1.02–3.15, p = 0.043).
Similarly, CHF patients with rEF with comorbid ischemic heart disease (IHD) had 8.23 times higher odds of utilizing RAS modulators compared to those without IHD (AOR: 8.23; 95% CI: 4.23–16; p < 0.001). CHF patients with rEF with hypertension (HTN) were twice as likely to use RAS modulators as those without HTN (AOR: 2.00; 95% CI: 1.10–3.70; p = 0.025).
In addition, HF patients with diabetes mellitus (DM) had 7.34 times higher odds of RAS modulator use compared to patients without DM (AOR: 7.34; 95% CI: 1.48–36.43; p = 0.016). Those with chronic kidney disease (CKD) had 4.35 times higher odds of using RAS modulators than patients without CKD (AOR: 4.35; 95% CI: 1.32–14.34; p = 0.015).
Finally, among CHF patients with rEF, those receiving a furosemide dose of ⩾40 mg were 74% less likely to be utilized RAS modulators compared to patients receiving <40 mg of furosemide (AOR: 0.26; 95% CI: 0.13–0.49, p < 0.001) (Table 6).
Bivariate and multivariable binary logistic regression analysis of predictors associated with the utilization of RAS modulators among CHF patients with rEF at PCSHs, Northwest Ethiopia, 2024 (n = 385).

AOR, adjusted odds ratio; CHF, chronic heart failure; CI, confidence interval; CKD, chronic kidney disease; COR, crude odds ratio; DM, diabetes mellitus; HTN, hypertension; IHD, ischemic heart disease; PCSH, public comprehensive specialized hospital; RAS, renin-angiotensin system; rEF, reduced ejection fraction.
Predictors associated with the dose optimization of RAS modulators in CHF patients with rEF
In the bi-variable logistic regression analysis, age ⩾65 years, VHD, DM, number of comorbidities ⩾2, Number of hospitalizations, duration of CHF with rEF ⩾3 years, use of diuretics, and dose of furosemide >40 mg were predictors associated with dose optimization of RAS modulators (p < 0.20). However, after controlling for the confounding variables in the multivariable binary logistic regression analysis (p < 0.05), age ⩾65 years, previous history of hospitalization, duration of CHF with rEF ⩾3 years, use of diuretics, and dose of furosemide >40 mg were found to be statistically significant variables associated with suboptimal dose of RAS modulators.
After adjusting for other covariates, patients aged ⩾65 years had 2.83 times higher odds of receiving a suboptimal dose of RAS modulators compared with patients younger than 65 years (AOR: 2.83; 95% CI: 1.46–5.50, p = 0.002).
Similarly, the odds of a suboptimal dose of RAS modulators were 2.05 times higher among CHF patients with rEF who had a previous history of hospitalization, in contrast with those who had no history of hospitalization (AOR: 2.05; 95% CI: 1.07–3.95), p = 0.032).
CHF patients with rEF for ⩾3 years were 69% less likely to receive a suboptimal dose of RAS modulators compared with those with a duration of (<3 years) (AOR: 0.31; 95% CI: 0.16–0.63, p = 0.001).
In contrast, patients who were prescribed diuretics had 5.34 times higher odds of receiving a suboptimal dose of RAS modulators compared with those who did not receive diuretics (AOR: 5.34; 95% CI: 2.73–10.44; p < 0.001).
Similarly, patients receiving a furosemide dose of ⩾40 mg had 3.88 times higher odds of being on a suboptimal dose of RAS modulators than those receiving a <40 mg dose of furosemide (AOR: 3.88; 95% CI: 1.89–7.97; p < 0.001) (Table 7).
Bivariate and multivariable binary logistic regression analysis of predictors associated with the dose optimization of RAS modulators among CHF patients with rEF at PCSHs, Northwest Ethiopia, 2024 (n = 385).

AOR, adjusted odds ratio; CHF, chronic heart failure; CI, confidence interval; COR, crude odds ratio; DM, diabetes mellitus; PCSH, public comprehensive specialized hospitals; RAS, renin-angiotensin system modulators; rEF, reduced ejection fraction; VHD, valvular heart disease.
Discussion
Renin-angiotensin system modulators are the cornerstone of pharmacological therapy for patients with CHF with rEF, and these consist of ACEIs/ARB and ARNI. The clinical benefits of RAS modulators are the greatest when these medications are titrated in short follow-up intervals until target doses are reached to achieve optimal clinical benefits. 50
The result of this study showed that the use of RAS modulators was found to be 68.3% (95% CI: 63.4–72.9). This result was comparable to prior HF studies in Ethiopia: Tikur Anbessa Hospital (67%) and Bahir Dar (68.8%). This finding was also similar to studies conducted in Kenya (66.2%), Canada (65.3%), and Palestine (70.1%).51–56 On the other hand, this result was lower than studies done in Jimma, 74.9%. 32 This finding was also lower than studies done in Dubai (78%), Nigeria (83%), South Asia (93.3%), and the HF survey in Europe (88%).57–60 The possible reason for this difference could be the physician’s area of expertise. In addition, a major contributing factor to the lower utilization of RAS modulators at the study site may be the limited available resources. These constraints may affect several critical aspects, including limited access of essential medications, regular monitoring of renal function and electrolytes needed to safely initiate and titrate RAS inhibitors might be less available, even if the medication is available at private pharmacies, patients cannot afford it, lower involvement of clinical pharmacists in the hospital setting, and absence of prescribers’ team-work or less team-work spirit with clinical pharmacists.
In addition to resource constraints, there are additional critical factors contributing to the underutilization of RAS modulators in CHF with rEF that warrant further emphasis. These may include patient-related issues such as side effects and challenges with adherence, clinician-related factors, like therapeutic inertia (reluctance to change treatment) or concerns about adverse effects, and systemic issues like varying access to healthcare and how guidelines are shared further complicate the situation. Together, these resource limitations may significantly hinder optimal use of RAS-targeted therapies.
Although all CHF patients with rEF should be treated by RAS modulator therapy, according to evidence-based guidelines,19,35 31.7% (95% CI: (27.5–36.6) of the participants in this study were not taking these medications. This underutilization was supported by another similar study in Tikur Anbessa Hospital, Ethiopia (31.2%), 55 and Malaysia (29.3%). 61 The possible reasons for underutilization of these RAS modulators were medication unavailability, high medication cost, medication side effects, physicians’ lack of awareness about the guidelines, patients not being seen by an expert or trend physician at the ambulatory clinic, there being no cardiologist, and there being no separate cardiac outpatient clinic in the hospital.
Concerning the utilization of RAS modulators, the use of RAS modulators was positively associated with a duration of CHF with rEF ⩾3 years in this study, which was also supported by other studies in Tikur Anbessa Hospital, Addis Abeba, 53 and the Qualify Heart Failure Registry in Europe. 57 This may be because patients are encouraged to attend multiple clinic visits, and physicians can commence and increase the dosage of RAS modulators due to extended follow-up periods. Furthermore, patients with longer-term CHF with rEF got the opportunity to see other highly qualified physicians. The presence of ischemic heart disease was positively associated with the use of RAS modulators.
Apart from the Palatine study 51 and the Jimma study, 54 there was also a positive correlation found between the usage of RAS modulators and the existence of ischemic heart disease. This may be because RAS modulators are beneficial in ischemic heart disease. In this study, patients with hypertension were more likely to use RAS modulators. This was in line with the Palestine study. 51 Furthermore, there was a positive correlation found between the use of RAS modulators and the existence of diabetes mellitus. The extra indication and no protective impact of RAS modulators in hypertension and diabetes mellitus may provide a plausible explanation for the positive correlation between RAS modulators and hypertension and diabetes mellitus.
Regarding the dose optimization of RAS modulators, several studies reported that the clinical benefits of RAS modulators in systolic HF are dose-dependent, and a better benefit occurred at higher doses.35,62 Therefore, evidence-based guidelines recommend up-titration of RAS modulators to an optimal dose unless there is a tolerability problem.40,63 In contrast, in the present study, 67.3% (95% CI: 59.1–70.5) of the participants were receiving suboptimal doses of RAS modulators. Similarly, studies in Ethiopia: Jimma (64.7%) 32 and Bahir-Dar (69.4%) 55 had shown that most of the patients were below the TD, which was consistent with these findings.
The suboptimal use of RAS modulators may be attributed to several factors. These include physicians not scheduling timely follow-up appointments, limited access to and awareness of up-to-date clinical guidelines, and patients not being managed by specialists or experienced physicians in ambulatory clinics. Additionally, some physicians may not actively update their knowledge or may fear adverse effects associated with RAS modulators like hyperkalemia and hypotension. Patient-related issues, such as difficulty adhering to appointment dates, also contribute. Furthermore, a high patient load combined with a limited number of physicians can reduce the quality of care. Overall, inadequate physician knowledge and attitudes towards optimizing RAS modulator therapy play a significant role. Therefore, clinicians need to determine and prescribe the optimal dose to ensure effective treatment outcomes.
Although studies have shown that achieving the optimal dose of RAS modulators in older patients with HFrEF is associated with long-term survival, 20 older patients (age ⩾65) were more likely to take suboptimal doses of RAS modulators, in contrast to younger patients (age <65) in the present study. This is supported by similar studies in Niriayo et al., 32 Sweileh et al., 51 Gelaye et al., 55 and Tajeu et al. 64 The possible justifications may be fear of adverse drug reactions like hyperkalemia, hypotension, renal dysfunction, and cough, physicians’ misconceptions about dosing efficacy, physiological changes with aging, comorbidity burden, and polypharmacy. Suboptimal dose RAS modulators were significantly associated with patients who had a previous history of hospitalization. This is in line with similar studies.32,65,66 This is due to higher clinical risk and comorbidities, concerns about tolerability and adverse events, clinical inertia and conservative management, and low early initiation and under-prescription.67,68
Studies showed that diuretics play a crucial role in the management of CHF with rEF, primarily by relieving symptoms related to fluid overload and congestion.69–71 However, Studies showed that the use of diuretics and increased doses of diuretics was associated with a decrease in the utilization of guideline-directed medical therapies like ACEIs, ARBs, ARNIs, SGLT2i, and beta blockers. 72 More significantly, high dose of diuretics usually limits the treatment optimization of guideline-directed medical therapies that have mortality benefits in CHF patients, including RAS modulators. 18 In line with this, the use of a diuretic and the dose of furosemide (⩾40 mg) were significantly associated with the suboptimal dose of RAS modulators in this study. The possible reason may be due to volume depletion and renal hemodynamic changes caused by diuretics, combined with the pharmacological action of RAS modulators on renal vasculature, creating a clinical scenario where increasing RAS modulators doses becomes challenging, leading to suboptimal dosing in patients requiring higher diuretic therapy. Therefore, physicians should carefully balance diuretic dosing with gradual RAS modulator titration, use rational drug combinations, avoid nephrotoxic interactions, and closely monitor patients to optimize therapy and improve outcomes in HF patients.
Finally, the present study demonstrated that the duration of CHF with rEF (⩾3 years) was negatively associated with suboptimal doses of RAS modulators. This finding is supported by a study conducted in Komajda et al. 42 The possible justification stems from CHF patients with rEF with a longer history being more likely to have undergone gradual and careful optimization of their medical therapy over time, including up-titration of RAS modulators to target or near-target doses. This reflects accumulated clinical experience and tolerance development, allowing physicians to safely increase doses while monitoring for adverse effects. 67 In contrast, patients with a shorter duration of CHF with rEF may still be in earlier stages of treatment initiation or dose titration, leading to more frequent suboptimal dosing.
Moreover, longer disease duration often correlates with more stable clinical status and better management of comorbidities, which facilitates the use of higher doses of RAS modulators. Evidence supports that higher doses of these agents modestly reduce mortality and improve outcomes in heart failure with reduced ejection fraction, encouraging physicians to optimize dosing over time.18,19 Thus, the negative association suggests that as CHF with rEF duration increases, patients are more likely to receive guideline-recommended, higher doses of RAS modulators, reflecting ongoing treatment optimization and improved tolerability with chronic therapy.
The findings of this study suggest the following implications:
Practical Implications: The study highlights significant gaps in the optimal use and dosing of RAS modulators among patients with CHF with rEF, emphasizing the urgent need for targeted interventions in clinical practice. Efforts should be made to improve medication accessibility and affordability, especially in resource-limited settings, to ensure patients can receive and adhere to guideline-recommended therapies. Enhancing physician education and awareness on dose titration, supported by multidisciplinary teamwork including clinical pharmacists, is critical to optimizing therapy and improving patient outcomes. In addition, careful management of diuretic dosing alongside RAS modulators is essential to overcome barriers to optimal dosing and reduce adverse effects.
Theoretical Implications: These findings contribute to the evolving understanding of factors affecting guideline adherence and optimal pharmacotherapy in CHF with rEF. They underscore the complex interplay of patient characteristics (e.g., disease duration, comorbidities), healthcare system limitations, and clinician behavior in affecting treatment optimization. The study reinforces the model that chronic disease duration allows for gradual optimization of therapy, which may explain improved dosing over time. This adds empirical support to theories emphasizing personalized, longitudinal care in CHF with rEF management.
Policy Implications: The results call for health policy reforms aimed at improving infrastructure and resource allocation for HF management in low-resource settings. Policies should prioritize the procurement and subsidization of essential HF medications like RAS modulators to reduce economic barriers. Furthermore, integrated care models fostering collaboration among cardiologists, general physicians, and pharmacists should be promoted. Development and dissemination of locally adapted clinical guidelines, coupled with regular training and audit mechanisms, would enhance prescriber adherence and ultimately improve clinical outcomes. Monitoring systems to track dose titration and medication utilization can guide continuous quality improvement initiatives in HF care.
Together, these implications advance the field by translating evidence on medication utilization patterns into concrete strategies aimed at bridging gaps in HF treatment, thus contributing toward better morbidity and mortality outcomes globally.
Strengths and limitations of the study
One of the key strengths of this study is that it is a multi-center study. In this study, a comprehensive data collection approach was also used, which combined both retrospective reviews of patients’ medical records and direct patient interviews conducted during their appointment visits. This dual method allowed for more accurate detection and verification of the use and dosing of RAS modulators, ensuring that medication information reflected not only prescribed regimens but also actual patient adherence and current medication possession.
However, the study also has some limitations. Due to its observational study designs, much of the data relied on secondary sources such as medical records, which may be incomplete, inaccurate, or inconsistent due to documentation variability. Additionally, the retrospective nature of the study did not permit longitudinal follow-up of participants, limiting the ability to assess changes in medication dosing or clinical outcomes over time. While our analysis encompassed multiple determinants potentially affecting the utilization and dose optimization of RAS modulators, we did not evaluate the influence of healthcare professionals’ knowledge levels on their prescribing patterns and patient adherence to the drugs. Moreover, we were unable to examine patient-related factors, system-related factors, and physician-related factors that may also play significant roles. These factors may have introduced information bias and restricted the depth of analysis regarding treatment optimization and patient response. Even though the findings of this study are similar to those of many studies, it cannot be generalized to the general population because some important heart failure drugs like ARNI and SGLT2i are not available.
Conclusion
This study revealed that the duration of CHF with rEF ⩾3 years, comorbidity specifically (IHD, HTN, DM, and CKD), and dose of furosemide ⩾40 mg were significantly associated with the use of RAS modulators; while age ⩾65 years, previous history of hospitalization, duration of CHF with rEF ⩾3 years, use of diuretics, and dose of furosemide ⩾40 mg were found to be statistically significant predictors associated with suboptimal dose of RAS modulators. Hypotheses are suggested for the correlates of optimal dosing to be studied in larger samples of HF patients. Continuing education is needed for providers on the guideline-based management of HF.
Recommendation
For the health care system
Organize regular training programs and workshops for medical professionals focusing on the management of CHF with rEF, particularly emphasizing the initiation, titration, and monitoring of evidence-based RAS modulators.
Recruit and assign additional physicians to the HF and cardiology units to effectively manage the patient load and ensure that each patient receives adequate time and quality care.
Appoint at least one full-time cardiologist within the hospital to oversee the treatment of complex HF cases and provide expert consultation to general physicians.
Establish a dedicated cardiac clinic within the hospital, staffed with trained healthcare providers and equipped with the necessary diagnostic and therapeutic tools to deliver specialized care for cardiovascular patients.
Strengthen the pharmaceutical supply chain and procurement system to ensure the continuous availability of RAS modulators within hospital pharmacies.
Expand access to health insurance coverage for HF patients, potentially through partnerships with government programs or private insurers, to reduce out-of-pocket expenses and improve patients’ access to essential medications and follow-up services.
For health care providers
Implement mandatory training sessions and continuous medical education programs to ensure that all physicians consistently apply international guidelines (such as ESC-HF and AHA/ACC) in the management of HF, aiming to improve patient survival rates and minimize hospital readmissions.
Develop a standardized patient education protocol, where physicians must explain the importance of medication during each patient interaction and actively schedule the next follow-up appointment before the patient leaves the clinic.
For patients
Establish an appointment reminder system (via phone calls, SMS, or appointment cards) to ensure that patients are consistently reminded of their upcoming visits and encouraged not to miss their scheduled appointment dates.
Integrate a payment support system at the time of medication dispensing, where patients are guided to pay health insurance premiums during their appointment visits, helping to cover future medication costs and reducing financial barriers to treatment adherence.
For policymakers
Enhance Multidisciplinary Care Teams: Address the shortage of cardiologists and low pharmacist engagement by fostering multidisciplinary HF care teams that include cardiologists, clinical pharmacists, nurses, and other health professionals. This team-based approach can improve medication optimization and patient monitoring.
Implement Structured Medication Titration Protocols: Develop and enforce standardized protocols for the rapid and regular titration of RAS modulators to achieve optimal dosing, especially targeting older patients, those with a history of hospitalization, and those on diuretics or high-dose furosemide, who were identified as predictors of suboptimal dosing.
Educational Programs and Training: The Institute continues professional education for healthcare providers regarding the importance of achieving optimal dosing of RAS modulators in CHF with rEF management and addressing modifiable barriers informed by predictors identified in the study.
Patient Follow-up and Support Systems: Establish robust patient follow-up systems to monitor therapy adherence, side effects, and tolerability, enabling timely dose adjustments and reducing suboptimal treatment.
Resource Allocation and Access Improvement: Allocate resources strategically to overcome systemic barriers such as limited access to specialists, ensuring equitable availability of essential medications and clinical services.
Data-Driven Quality Improvement: Use data from ongoing clinical audits and research to identify gaps in RAS modulator use and dosing, and tailor interventions to local contexts.
For future researchers
Conduct a follow-up cohort study to investigate the relationship between RAS modulators dose optimization and key clinical outcomes such as mortality, hospitalization rates, and quality of life in patients with CHF with rEF.
Design targeted research studies to specifically identify whether patient-related factors (e.g., medication adherence, socioeconomic status) or system-related factors (e.g., medication availability, physician practice patterns) are the primary contributors to the suboptimal use of RAS modulators.
Implement large-scale prospective follow-up studies, enrolling diverse patient populations, to assess the impact of these identified factors on the successful optimization of RAS modulator therapy and to provide stronger evidence for future guideline improvements.
Conducting a qualitative study to explore the reasons for underutilization of RAS modulators. It is also advisable to perform statistical analyses of these qualitative findings, accompanied by sensitivity analyses that exclude patients with contraindications, including larger sample sizes, and more detailed data collection to achieve more robust results.
Supplemental Material
sj-docx-1-taj-10.1177_20406223251384514 – Supplemental material for Patterns of use and dose optimization of renin-angiotensin system modulators among chronic heart failure patients with reduced ejection fraction in resource-limited settings: a multicenter cohort study
Supplemental material, sj-docx-1-taj-10.1177_20406223251384514 for Patterns of use and dose optimization of renin-angiotensin system modulators among chronic heart failure patients with reduced ejection fraction in resource-limited settings: a multicenter cohort study by Sisay Sitotaw Anberbr, Samuel Berihun Dagnew, Getachew Yitayew Tarekegn, Fisseha Nigussie Dagnew, Zufan Alamrie Asmare, Teklie Mengie Ayele, Eskedar Dires Gebremeskel, Samuel Agegnew Wondm and Tilaye Arega Moges in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We, the authors, sincerely express our gratitude to the University of Gondar, College of Medicine and Health Sciences, School of Pharmacy, and Department of Clinical Pharmacy for providing the invaluable opportunity to conduct this research. We also extend our deepest appreciation to the data collectors for their dedication and to all the participants whose contributions were essential to the success of this study.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.