Abstract
Background:
Lymphomas represent various groups of cancers. Its treatment has a cytotoxic effect on all body tissues, especially female ovaries, yielding premature ovarian insufficiency.
Objectives:
This study aimed to compare the fertility and ovarian preserving role of gonadotropin-releasing hormone agonists (GnRH-a) when administered in conjunction with chemotherapy in women treated for lymphomas.
Design:
Only prospective clinical trials were included in this systematic review.
Data sources and methods:
We searched for pertinent studies in PubMed, Web of Science, Cochrane Library, Google Scholar, and Scopus till April 2025. Available prospective clinical trials comparing patients with Hodgkin lymphoma and non-Hodgkin lymphoma receiving concurrent chemotherapy and GnRH-a with patients receiving chemotherapy alone were included. The main outcomes were cyclic ovarian function (COF), pregnancy rate, follicle-stimulating hormone (FSH) level, luteinizing hormone level, estradiol level, and anti-Mullerian hormone (AMH) level. Cochrane’s risk of bias tool for clinical trials was used for the risk of bias assessment, and all the included studies were deemed of acceptable quality.
Results:
Twelve prospective clinical trials involving 733 women were analyzed, with 374 patients receiving GnRH-a and 359 as controls. The mean age of the GnRH-a group was 25.86 years, compared to 27.62 years in the control group. GnRH-a administration was associated with a notably higher COF, higher pregnancy rates, lower FSH levels, and higher AMH levels compared to controls. No significant difference between the groups was observed in estradiol levels.
Conclusion:
Co-administration of GnRH-a during chemotherapy in women with lymphoma appears to offer a protective effect against ovarian damage and supports fertility preservation in women with lymphoma. These findings advocate for the integration of GnRH-a into fertility preservation strategies, emphasizing its potential to improve long-term reproductive health in this vulnerable patient population. However, further high-quality, large-scale studies are warranted to standardize protocols and confirm these findings for broader clinical applications.
Keywords
Background
Lymphomas represent various groups of cancers that originate from the proliferation of lymphocytes at various maturation stages, such as T-cells, B-cells, and natural killer (NK) cells. It represents about 5% of all malignancies. 1 There are two main types of lymphomas, namely, Hodgkin lymphoma (HL; 10%) and non-Hodgkin lymphoma (NHL; 90%). These major types are further divided into classical and non-classical types in terms of HL, while NHL is further classified into NK, T-cell, and B-cell types. 2 Although there are no identified specific etiologies for lymphomas, various infectious, environmental, occupational, and genetic factors may play a major role in developing lymphomas. The main diagnostic tool for lymphoma is tissue biopsy. 3
HL has an overall 5-year survival of 90%, 4 while that of NHL is 75%. 5 Chemotherapy is a common cancer treatment with significant morbidity and mortality. 6 Evidence from previously published studies demonstrates that the treatment of HL with various effective chemotherapeutic agents such as adriamycin, bleomycin, vinblastine, and dacarbazine may enhance survival to about 90%. 7 Unfortunately, this chemotherapy causes cytotoxicity on body tissues, including the ovaries. Because female ovaries have a non-renewable and limited number of germ cells, premature ovarian insufficiency (POI), a serious complication that may lead to infertility, often occurs in young women after chemotherapy.8,9
Furthermore, estrogen production may be impaired, which results in premature menopausal symptoms, including amenorrhea, mood changes, headache, osteoporosis, night sweats, hot flushes, and vaginal dryness,10,11 which may negatively impact her self-esteem and quality of life. 11
Chemotherapeutic agents may increase oxidative stress, reduce ovarian blood flow, and cause apoptosis of the oocytes, leading to direct ovarian toxicity. 12 Alkylating drugs, such as cyclophosphamide, are known for their potency in inducing ovarian insufficiency. They modify base pairs, causing DNA cross-links, and leading to single-strand DNA breaks. 13 Cumulative dose and long treatment duration also have a significant role. 14 Thus, great efforts have been made to avoid these adverse effects of chemotherapy and preserve women’s fertility, such as cryopreserving oocytes and cryopreserving ovarian tissue.15,16
Given the profound psychological and reproductive implications, 17 effective fertility preservation strategies are urgently needed to mitigate these adverse outcomes. The effectiveness of gonadotropin-releasing hormone agonists (GnRH-a) to protect ovaries and preserve fertility throughout standard chemotherapy regimens has been studied carefully since it was first introduced in the 1980s.18–20 Several published studies showed that GnRH-a administration with chemotherapy might significantly decrease the cytotoxic effect on ovaries and the incidence of POI. 21 The proposed mechanisms for GnRH-a’s protection are the suppression of granulosa cell proliferation,22,23 the drug’s direct effects on the ovarian follicles and the pituitary gland, the decrease of gonadotropins secretion, and protection of germline stem cells.24–26 In addition, the cessation of ovary activity may lead to secondary hypoestrogenism and a decrease in ovarian blood flow. 27 However, there exists a degree of heterogeneity in the findings, partly due to variations in patient demographics, chemotherapy regimens, and study designs. Specifically, while some investigations have observed favorable hormonal profiles, such as lower follicle-stimulating hormone (FSH) levels and higher anti-Müllerian hormone (AMH) levels in the GnRH-a group, others have not recorded statistically significant differences in markers such as estradiol levels. This inconsistency underscores the necessity for a systematic review focused exclusively on the lymphoma population to reconcile these divergent findings and provide clearer guidance for clinical practice. Therefore, the current study aims to address this gap by critically evaluating prospective clinical trials that compare fertility outcomes, including cyclic ovarian function (COF), pregnancy rates, and hormonal profiles between women with lymphoma treated with chemotherapy alone and those receiving GnRH-a in combination with chemotherapy. By isolating data specific to lymphoma, the investigation seeks to clarify the efficacy of GnRH-a as a fertility preservation strategy and to optimize treatment protocols that may enhance reproductive outcomes.
Materials and methods
This systematic review was reported and conducted based on the recommendation of PRISMA 28 (Figure 1).
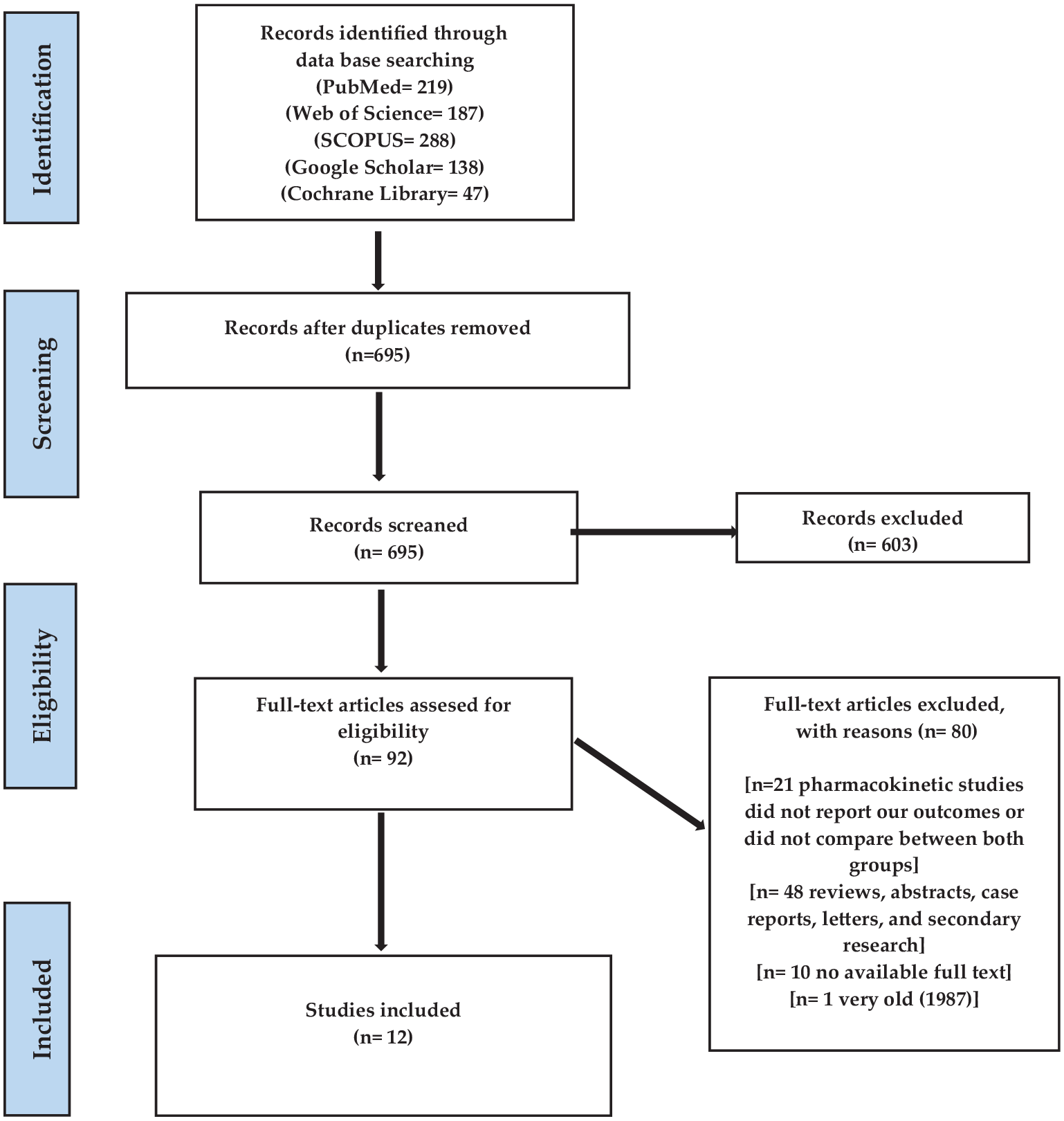
Study search strategy and article selection.
Data sources and searches
Regarding data sources, we searched for pertinent studies in PubMed, Web of Science, Cochrane Library, Google Scholar, and Scopus till April 2025 using the following keywords to develop a search strategy: “ovarian failure,” “ovarian dysfunction,” “GnRH-a,” “gonadotropin-releasing hormone agonist,” “lymphoma,” “Hodgkin lymphoma,” “non-Hodgkin lymphoma,” “chemotherapy,” “gonadotoxicity.”
Study selection and inclusion criteria
Two authors reviewed each study independently according to the following inclusion criteria:
Population
Premenopausal women with lymphoma who are receiving potentially ovarian toxic chemotherapeutic agents.
Intervention
Women who received GnRH-a in combination with chemotherapy.
Comparator
Women who did not receive GnRH-a.
Outcomes
COF, pregnancy rate, FSH level, luteinizing hormone (LH) level, estradiol (E2) level, and AMH levels.
Study design
Only prospective clinical trials, randomized or non-randomized, were included in this systematic review. We excluded observational studies, case reviews, case reports, and case series. Additionally, studies that did not report our outcomes, single-arm studies, and studies with no available full-text were excluded.
Data extraction and quality assessment
Three authors extracted three types of data from each included trial. The first category was baseline data, including study characteristics, demographic data of women, baseline FSH, baseline LH, baseline E2 level, and the type of lymphoma. The second category was extracting data on our outcomes, including COF, pregnancy rate, FSH level, E2 level, and AMH level. Finally, we extracted data required for the quality assessment of our studies. Quality assessment was conducted according to the Cochrane Risk of Bias tool, which assesses the risk of bias in clinical trials according to eight domains. 29
Results
General characteristics of the participants
A PRISMA flow diagram that demonstrates a literature search of ours is described in Figure 1. We identified 879 articles from the following databases: PubMed (n = 219), Web of Science (n = 187), SCOPUS (n = 288), Google Scholar (n = 138), and Cochrane Library (n = 47). After removing duplicates, 695 articles remained for the screening process. Two authors conducted the screening process independently and finally involved twelve clinical trials21,30–40 with the inclusion criteria of our systematic review. From these trials, we included 733 patients who had lymphoma treated with chemotherapy and suffering from ovarian insufficiency.
The included participants were divided into two cohorts: GnRH-a cohort that involved 374 participants and a control cohort with 359 participants. The mean age of the GnRH-a cohort was 25.86 ± 6.23 years, while that of the control cohort was 27.62 ± 6.73. A detailed summary of the involved studies and participants is demonstrated in Tables 1–3. Disease subtype characteristics are included in Table S1.
A detailed demographic, disease, and study design summary of the included studies and participants.

ABV, adriamycin, bleomycin, and vinblastine; ABVD, adriamycin, bleomycin, vinblastine, and dacarbazine; ACVBP, doxorubicin, cyclophosphamide, vindesine, bleomycin, prednisone; BEAC, bleomycin sulfate, etoposide phosphate, doxorubicin hydrochloride, and cyclophosphamide; BEACOPPesc, bleomycin sulfate, etoposide phosphate, doxorubicin hydrochloride, cyclophosphamide, vincristine sulfate, procarbazine hydrochloride, and prednisone-escalated; BEAM, carmustine, etoposide, cytarabine, melphalan; BMI, body mass index; BMT, bone marrow transplant; ChlVPP/ABVVP, chlorambucil, vinblastine, procarbazine, doxorubicin, bleomycin, vincristine and etoposide; CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone; C-MOPP, cyclophosphamide, vincristine, procarbazine, prednisone; COPP, cyclophosphamide, vincristine sulfate, and procarbazine hydrochloride, and cyclophosphamide; CVP, cyclophosphamide, vincristine sulfate, and prednisone; CVPP, cyclophosphamide and vinblastine, procarbazine, and prednisone; DHAP, dexamethasone, cytarabine, cisplatin; ESHAP, etoposide, methylprednisolone, cytarabine, cisplatin; GnRH-a, gonadotropin-releasing hormone agonists; HL, Hodgkin lymphoma; ICE, ifosfamide, carboplatin, and etoposide; MINE, mesna, ifosfamide, mitoxantrone, etoposide; MOPP, mechlorethamine hydrochloride, vincristine sulfate, procarbazine hydrochloride, and prednisone; NR, not reported; R-CHOEP, rituximab, cyclophosphamide, hydroxydaunorubicin, oncovin, etoposide, and prednisone; RCT, randomized controlled trial; R+/R− CHOP, rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone; SD, standard deviation; Stanford V, mechlorethamine, doxorubicin hydrochloride, vinblastine, vincristine, bleomycin, etoposide and prednisone.
A detailed baseline summary of the included studies and participants.

E2, estradiol; FSH, follicle-stimulating hormone; GnRH-a, gonadotropin-releasing hormone agonists; NR, not reported; SD, standard deviation.
A detailed biochemical and clinical outcome summary of the included studies and participants.

E2, estradiol; FSH, follicle-stimulating hormone; GnRH-a, gonadotropin-releasing hormone agonists; NR, not reported; POI, premature ovarian insufficiency; SD, standard deviation.
Risk of bias assessment
The quality of the included trials was assessed using Cochrane’s risk of bias (ROB) tool for clinical trials. 41 Five studies30–33,36–38 of the included trials reported randomization among the included participants, while the remaining seven trials21,31,32,35,39 were non-randomized. Regarding allocation concealment, five trials30–32,39,40 did not report enough data, and four33,36–38 were at low risk. According to the blinding of participants and personnel domain, 10 trials21,30–33,35,37–40 did not report enough data, 1 trial 36 was at low risk, and 1 trial 34 was at high risk. A detailed summary of the ROB evaluation is described in Figure 2.

Risk of bias assessment of included studies.
The analysis of the outcomes
Cyclic ovarian function
All the included studies21,30–40 reported data about the COF outcome. Only one study reported a higher percentage in the GnRHa group (20%) versus 19% of patients in the treatment and control groups, respectively, developed POI after 1 year. 36 On the other hand, the incidence of POI development was significantly higher in the control group than in the GnRHa group in six studies.31–35,37
After following up with the patients for a median of 70 months, a study reported that 12 out of 13 (92%) recovered their ovarian function, 35 whereas only one 40-year-old patient in the control group developed POI. Five patients out of 23 (21.7%) and 20 out of 22 (90.9%) in the GnRHa and control groups, respectively, developed POI in the previous study. 34 Only 2 out of 65 patients (3.1%) in the GnRHa group developed POI, versus 17 out of 46 (37%) in the control group in another study. 33 In a previous study, the incidence of POI was 53% in the control group, compared to 7% in the treatment group. 31 Similarly, 11 out of 18 control group patients experienced POI (61%) versus 1 out of 16 (6%) in the treatment group in another study. 32 Likewise, 31 patients (19.4%) in the treatment group versus 8 patients (25%) in the control group developed POI. 37 COF resumption was significantly related to GnRHa administration and younger age in another study. 21
Pregnancy rate
Only seven trials21,31,32,34,35,38,39 reported the rate of pregnancy among the involved women. The rate of pregnancy in the GnRH-a cohort was generally higher than in the control cohort. Spontaneous pregnancy occurred at a higher rate in the treatment group in five Studies.21,32,33,35,37 A previous study reported spontaneous pregnancies only in the study group (two patients conceived six times). 21 A higher rate of pregnancies was reported in another study in the GnRHa arm (17 of 32 patients (53.1%)), versus 15 of 35 patients (42.8%) in the control arm. 37 Eight patients had spontaneous pregnancy, and 12 babies in a previous study, 5 of them after GnRHa administration. 35 Similarly, 26 spontaneous pregnancies occurred in the study group versus 20 in the control group in another study. 33 Likewise, three patients in GnRH and two in the control group conceived. 32 On the other hand, no pregnancies were reported in either arm in one study. 30 In one study, three patients in the control group became pregnant versus two patients in the study group, 39 and in another study, pregnancy occurred only in the control group. 38
FSH level
FSH level was reported in seven trials.30,31,35–38,40 No difference in FSH levels was demonstrated in only one study after the administration of GnRHa. 37 On the other hand, treatment with GnRHa significantly alleviated chemotherapy-induced FSH elevation compared to the control group in six studies.21,32,34,36,38,40 The increased FSH was only temporary in three patients treated with GnRHa in a previous study. 33 . The median FSH level dropped to 7.2 IU/L after the GnRH-a/chemotherapy combination, compared to 31.5 IU/L in the control group in a previous trial. 32 The mean FSH values were lower in the GnRHa group during chemotherapy in a previous study. 36 However, after 6 months, this difference disappeared. The median FSH concentration in the study group was 6.34 U/L versus 92.29 U/L in the control group in a previous study. 21
Estradiol level
The E2 levels were reported in three trials.36–38 The E2 levels in both cohorts were similar.
AMH levels
Three trials30,36,37 mentioned the AMH level outcome. GnRH-a cohort was accompanied by higher levels of AMH than the control cohort. The mean AMH levels were significantly higher in the study group than in the control group in three previous studies.33,36,40 On the other hand, AMH level was higher in the control than in the treatment group in one study. 38 One study reported that the benefit of GnRH-a administration is age-limited. 33 However, one study reported that GnRH-a administration can’t significantly preserve ovarian function. 30
Discussion
In the last decades, although patients with HL and NHL showed marked improvement in cure rate, progression-free survival, and prognosis after aggressive chemotherapy, women often experience major side effects particularly relating to ovarian function. POI is a well-established common serious complication occurring in women receiving chemotherapy for the treatment of HL, NHL, and many other cancers.42–44 Ovarian damage may occur due to chemotherapy-induced follicle destruction, vascular lesions, cortical fibrosis, or accelerated atresia. 45 This necessitated investigating various options that aim to preserve a woman’s ovarian function and fertility. These options include the transposition of ovaries and cryopreservation of unfertilized oocytes, embryos, or ovarian tissue.46,47 Artificial ovary construction is one of the innovative techniques applied to preserve fertility in female cancer patients. 48 Furthermore, co-administration of GnRH-a with standard chemotherapy showed promising results in terms of reducing the gonadotoxic effect of chemotherapy and preserving fertility.33,49,50 They may also be utilized in parallel to non-pharmacological fertility preservation modalities. 51
Studying the efficacy of GnRHa administration in young females with lymphoma is important for preserving their fertility, despite the difficulty of its follow-up. 36 This systematic review included 12 studies involving 733 women with lymphomas who are receiving chemotherapy. The strength of our systematic review is that it is the first to assess the effectiveness of GnRH-a administration during the treatment period in avoiding chemotherapy-induced POI in patients with both HL and NHL, based on results obtained from randomized controlled trials (RCTs). Our findings demonstrated that patients who received GnRH-a had higher COF, pregnancy rate, and AMH plasma levels. Additionally, GnRH-a was associated with significantly lower plasma levels of FSH.
Gonadotoxic chemotherapy promotes the apoptosis of the nonresting follicles and decreases the plasma levels of sex steroids, causing an increase in FSH, increasing the maturation of preantral follicles, and their subsequent exposure to gonadotoxicity. This vicious cycle can be inhibited by the use of GnRH-a.31,52 FSH concentrations higher than 10 IU/L were associated with a reduction in ovarian reserve.53,54 However, FSH levels may show some variability among studies, even between young females with normal ovulatory cycles, due to interindividual variations, and it needs to be measured on a certain day of the menstrual cycle to be comparable. 30
AMH level can act as the most reliable indicator of ovarian aging and serve as a sensitive marker for evaluating the ovarian follicle pool.38,55 It is more strongly influenced by the ovarian follicular status than FSH and E2 and is not affected by the menstrual cycle stage.38,56 Its level starts to decrease after 30 years of age to reach 10 pmol/L after the age of 37 years. 57 At the age of 37, the follicular reserve may reach 25,000, which might not be sufficient, with a further accelerated rate of decrease. 58 Two patients with a relatively increased age (36 years old) in the GnRH-a group in a previous study developed POI, which suggests that maintaining follicles may be possible only at a younger age when the follicular reserve is higher. 33 GnRH-a administration may at least reduce the rate at which the chemotherapy-induced follicular loss occurs. 18 Moreover, our findings demonstrated that the administration of GnRH-a may not only preserve ovarian function but also preserve fertility and increase pregnancy rates. Although the ovarian reserve measured using AMH or FSH levels after a 5-year follow-up by Demeestere et al. did not differ significantly between the GnRHa group and the control group, pregnancy rates in the GnRH-a group were significantly higher (53% vs 43%). 37
Eventually, no difference was found between patients in both GnRH-a and control cohorts regarding estradiol levels. However, a previous systematic review and meta-analysis for breast cancer reported that the beneficial role of GnRH-a was limited to patients receiving chemotherapy regardless of the hormone receptor status in those women. They also concluded that GnRH-a should not replace the already proven fertility preservation techniques, such as embryo and gamete vitrification. 59 The exact protective mechanisms of continuous use of GnRH-a are still not well-known. However, it may protect ovaries and undifferentiated germ-line stem cells by interrupting LH and FSH secretions, thus temporarily decreasing ovarian function and follicular destruction, decreasing ovarian perfusion, and releasing anti-apoptotic molecules.33,49 However, hormonal protection may not be efficient when aggressive chemotherapy is used 30 as has been refuted by an in vitro experimental study on human ovarian tissues. 60
The long-term gonadoprotective effect of GnRHa in patients with HL was confirmed up to 15 years in a study by Blumenfeld et al. 33 Another previous study 37 assessed the long-term effect of GnRH-a (triptorelin plus norethisterone) on women with lymphoma. Their long-term follow-up found that the concurrent administration of GnRH-a and chemotherapy does not prevent POI or improve the pregnancy rate in survivors. However, they concluded that triptorelin might be utilized to inhibit the menorrhagia caused by thrombocytopenia associated with lymphoma. 36 This study was limited by the high rate of dropout, which reached about 50%. A previous study 61 performed another meta-analysis on 9 studies involving 765 women with different cancer types. They found that the use of GnRH-a was associated with a statistically significant reduction of the risk of POI, which is consistent with our results. Another study by Hickman et al., 62 reported results that are comparable to our findings. They demonstrated that GnRH-a had a preventive effect on the development of POI after receiving treatment with gonadotoxic chemotherapy. Besides, patients may show improvement in cardiovascular health, bone status, sexual function, and overall quality of life.
The study’s methodological framework is one of its major strengths, combining rigorous data collection. We provided the most recent large-scale study of survivors treated with or without GnRH-a to evaluate the efficacy of GnRH-a in preserving women’s fertility during chemotherapy. The majority of previously published studies assessed the effect on fertility and the gonadotoxic effect of chemotherapy in patients with HL, while data about patients with NHL and other lymphomas were limited. 63 Therefore, in our study, we included patients with HL, NHL, and other types of lymphomas, which is one of the major strong points of our systematic review. Additionally, the overall quality assessment of the included studies was good.
Our study has some limitations, such as the heterogeneity between trials, which can be attributed to multiple reasons, including differences in patients and treatment protocols, the different methods of defining POI according to the clinical trials, the relatively small sample size, and the variations of follow-up duration in each trial. Further higher-quality evidence is required to confirm the reliability of GnRH-a as a routine intervention in clinical practice and whether it should be given in addition to or instead of other fertility preservation modalities.
Conclusion
The systematic review reveals that the co-administration of GnRH-a during chemotherapy may provide a protective effect on ovarian function and enhance fertility outcomes in premenopausal women with lymphoma.
Most included studies support its association with improved COF, higher pregnancy rates, reduced FSH, and increased AMH levels, particularly among younger women. The exact protective mechanisms of continuous use of GnRH-a on the ovaries are not yet established. Before concluding that GnRH-a is effective in preserving fertility and preventing POI in women of reproductive age receiving gonadotoxic chemotherapy for the treatment of lymphoma, large, well-designed, long-term follow-up RCTs are required to minimize the ROB and compare the adjunctive ovarian preserving role of GnRH in female patients with lymphoma treated with chemotherapy. The review is strengthened by its inclusion of both HL and NHL patients and its focus on prospective clinical trials. However, heterogeneity across studies, including variability in definitions of POI, treatment protocols, and follow-up durations, limits the generalizability of results. In addition, due to this heterogeneity, we couldn’t conduct a meta-analysis. While GnRH-a presents a promising option for fertility preservation, especially when cryopreservation is not feasible, it should not replace established methods. Until the results of such clinical trials are obtainable, we suggest, in addition to the established gonadoprotective measures, such as oocyte/embryo cryopreservation, considering co-treatment with GnRH-a for achieving the best odds for fertility in reproductive-age women treated with chemotherapy for lymphomas.
Supplemental Material
sj-docx-1-taj-10.1177_20406223251356815 – Supplemental material for Preservation of ovarian endocrine function in patients with lymphoma: a systematic review
Supplemental material, sj-docx-1-taj-10.1177_20406223251356815 for Preservation of ovarian endocrine function in patients with lymphoma: a systematic review by Hebatallah Ahmed Mohamed Moustafa, Al Shaimaa Ibrahim Rabie, Ahmed Abdullah Elberry, Ahmed Hassan Shaaban, Amany Mohamed Alboghdadly, Hind Khalid Goresh, Muath Fahmi Najjar, Mabrouk AL-Rasheedi and Raghda R. S. Hussein in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223251356815 – Supplemental material for Preservation of ovarian endocrine function in patients with lymphoma: a systematic review
Supplemental material, sj-docx-2-taj-10.1177_20406223251356815 for Preservation of ovarian endocrine function in patients with lymphoma: a systematic review by Hebatallah Ahmed Mohamed Moustafa, Al Shaimaa Ibrahim Rabie, Ahmed Abdullah Elberry, Ahmed Hassan Shaaban, Amany Mohamed Alboghdadly, Hind Khalid Goresh, Muath Fahmi Najjar, Mabrouk AL-Rasheedi and Raghda R. S. Hussein in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
