Abstract
Background:
Antiretroviral therapy (ART) has significantly reduced morbidity and mortality among people living with HIV (PLWH). However, data on the burden of metabolic syndrome (MetS) in sub-Saharan Africa remains limited, particularly following the implementation of universal test-and-treat strategies and the widespread use of integrase inhibitor-based combinations.
Objective:
This study aimed to determine the prevalence and associated factors of MetS among adults receiving first-line ART in the Hawassa City Administration, southern Ethiopia.
Design:
A cross-sectional study.
Methods:
The study was conducted from January 2023 to May 2024, adapting the World Health Organization (WHO) stepwise approach to collect data. All study-relevant data were collected from participants using a pretested structured questionnaire. MetS was defined according to the 2009 harmonized criteria. A binary logistic regression analysis was conducted to identify predictors of MetS, with adjusted odds ratio (aOR) and 95% confidence intervals (CIs).
Results:
A total of 450 adults participated in the study, of whom 262 (58.2%) were females. The mean (standard deviation) age of the participants was 41.1(±9.7) years. The prevalence of MetS was 36.4% (95% CI: 32.2–41.6), with low high-density lipoprotein (HDL) cholesterol as the most frequent component observed in 368 (81.8%) participants. Age >50 years (aOR: 2.9; 95% CI: 1.4–6.2), alcohol use (aOR: 2.7; 95% CI: 1.2–6.4), body mass index ⩾25 kg/m² (aOR: 3.7; 95% CI: 1.9–7.1), triglyceride/HDL-cholesterol ratio (aOR: 1.5; 95% CI: 1.3–1.7), family history of hypertension (aOR: 2.1; 95% CI: 1.1–3.8), and high waist-height ratio (aOR: 5.4; 95% CI: 1.8–15.9) were significantly associated with MetS. However, dolutegravir-based first-line regimens were not significantly associated with MetS (p=0.482 for DTG initiation, and p=0.34 for switching to DTG).
Conclusion:
The noticeable prevalence of MetS among PLWH highlights its potential to increase cardiovascular risks. Therefore, routine screening of PLWH for components of MetS is essential to reduce the health risks associated with metabolic disorders. As most of the identified risk factors are modifiable, implementing lifestyle interventions is also imperative.
Plain language summary
In this study, we investigated the prevalence and determinants of metabolic syndrome (MetS) in adults with HIV on antiretroviral therapy in southern Ethiopia. We presented the prevalence of MetS and its key risk factors in this population. We observed a high prevalence of MetS, with low HDL cholesterol being the most frequently observed component. We also identified that older age, alcohol use, high body mass index, triglyceride/HDL cholesterol ratio, family history of hypertension and increased waist-height ratio were significantly associated with MetS. We found no significant association between dolutegravir-based regimens and MetS. Therefore, we recommend routine screening for MetS components and proactive management of related conditions. In addition, most of the identified risk factors are modifiable, interventions focused on diet, weight management and alcohol use could help reduce MetS in this population. Further studies are required to investigate additional underlying factors and the long-term metabolic risks associated with antiretroviral therapy.
Introduction
Metabolic syndrome (MetS) is characterized by a cluster of risk factors for cardiovascular diseases (CVDs) and metabolic disorders, which include abdominal obesity, high triglycerides (TGs), low high-density lipoprotein cholesterol (HDL-c), raised blood pressure or hypertension, hyperglycemia or diabetes mellitus, and insulin resistance (IR).1,2 Evidence indicates that individuals with MetS are at significantly increased risk of cardiovascular-related morbidity and mortality.3,4
Antiretroviral therapy (ART) has significantly improved life expectancy and reduced HIV/aquired immunodeficiency syndrome (AIDS)-related morbidity and mortality among people living with HIV (PLWH). However, along with this succsess, the prevalence of MetS is also rising in this population, posing individuals at significant risk for non-communicable diseases (NCDs). 5 Recent reports have indicated a significant prevalence of MetS among PLWH globally and in sub-Saharan Africa (SSA).6,7 A global meta-analysis estimated the pooled prevalence of MetS in PLWH to be 25.3% (95% CI: 22.9–27.6), with a higher rate of 25.6% (95% CI: 23.0–28.2) among those who received ART compared to 18.5% (95% CI: 15.5–21.4) in ART untreated individuals. 6 In SSA, studies reported a prevalence of MetS among PLWH of 23.3% (95% CI: 18.8–27.8) 5 and 21.5% (95% CI: 15.1–26.8), 8 both of which surpass the 12.0% (95% CI: 5.0–21.0) prevalence rate reported in individuals without HIV. 8
In Ethiopia, studies have reported the prevalence of MetS as 21.7% (95% CI: 19.36–24.0) using the National Cholesterol Education Program Adult Treatment Panel III (NCEP-ATP III) criteria, and 29.91% (95% CI: 21.54–38.28) using the International Diabetes Federation (IDF) criteria. 9 These rates were higher than those reported among the working adult population in Ethiopia, where the prevalence was 18.45% (95% CI: 13.89–23.01) 10 and 20.1% (95% CI: 17.8–22.4). 11 The development of MetS is complex and multifactorial, influenced by a range of contributing factors such as obesity,12–15 female sex, 14 the use of protease inhibitor (PI)-based ART regimens 12 and the duration of ART exposure. 12
In 2018, the integrase strand transfer inhibitors, particularly dolutegravir (DTG), were endorsed by the World Health Organization (WHO) as the preferred treatment option for PLWH. 16 Since then, DTG-based regimens have been widely distributed across SSA, including Ethiopia, substituting non-nucleoside reverse transcriptase inhibitors (NNRTIs) and PI-based ART regimens. 15 However, evidence regarding the effect of DTG-based regimens on MetS is inconsistent, with some studies raising concerns about metabolic issues associated with DTG, 17 such as dyslipidemia,18,19 elevated blood pressure,20,21 clinical weight gain22,23 and hyperglycemia.24,25 In contrast, other studies have reported a protective effect or reduced risk of metabolic dysregulation with the use of DTG-based regimens. 26
Early identification of cardiometabolic burden and its modifiable risk factors is essential for the prevention and management of cardiovascular risks among PLWH on ART, as evidenced by reliable epidemiological findings. However, there is a scarcity of data regarding individuals who initiated ART after the adoption of the universal test-and-treat strategy in Ethiopia, coupled with the widespread use of first-line DTG- based regimens that contain DTG. Therefore, this study aimed to determine the prevalence and associated factors of MetS among adults who initiated first-line ART and had been on treatment for at least 12 months in southern Ethiopia following the implementation of the test-and-treat strategy.
Materials and methods
Study setting, design, and study population
This cross-sectional study was conducted from January 5, 2023 to May 30, 2024 at health facilities in the Hawassa City Administration. Hawassa, the capital of the Sidama Region in southern Ethiopia, is 275 km away from Addis Ababa, the capital of Ethiopia. Eight health facilities in the city administration are providing ART services to over 6300 PLWH during the study period. The eligible participants were adult PLWH aged 18–65 years who had initiated ART and were actively receiving follow-up care at health facilities in the city administration.
Inclusion and exclusion criteria
This study included adults aged 18–65 years who initiated first-line ART after the implementation of the test-and-treat strategy in Ethiopia. In addition, participants had been on first-line ART for at least 12 months, achieved viral load suppression (HIV–ribonucleic acid (HIV–RNA)) below 1000 copies/milliliter, and demonstrated at least fair adherence to ART. Regarding first-line ART regimens, the study included individuals who initially started a regimen comprising two nucleos(t)ide reverse transcriptase inhibitors (N(t)RTIs) and one NNRTI and continued on this regimen (NNRTI-maintained group). It also included those who began with two N(t)RTIs plus one NNRTI and later switched to a regimen of two N(t)RTIs plus DTG (switched to the DTG group). Moreover, individuals who initiated and remained on a regimen of two N(t)RTIs plus DTG (DTG-maintained group) were included in the study. However, the study specifically excluded participants on first-line ART with a history of more than one treatment switch, as well as those who were pregnant or breastfeeding within the first 6 months of the study. In addition, participants who were critically ill or physically disabled, those with known liver or renal failure, and individuals with active opportunistic or severe infectious diseases were also excluded.
Sample size and sampling techniques
The sample size was calculated using Epi-Info version 7.2 developed by Centers for Disease Control and Prevention (CDC), based on a 23.9% prevalence of MetS among PLWH in SSA, 5 with a 5% margin of error and a 1.96 z-value for a 95% confidence interval (CI). In addition, risk factors significantly associated with MetS were considered to compute a maximally representative sample size,15,27 accounting for a 95% CI, 80% power, a 1:1 exposed-to-unexposed ratio, and odds ratio (OR). A sample size of 279 was calculated based on a prevalence of 23.9%. After adjusting for a 10% nonresponse rate and a design effect of 1.5, the final sample size was determined to be 465. This sample size was selected as it was considered larger and more representative of the study objectives than the sample size computed based on MetS risk factors. The sample size of 465 was proportionally allocated across each health facility based on the number of PLWH receiving first-line regimens at the respective study sites. Subsequently, a systematic random sampling technique was conducted to recruit study participants.
Data collection questionnaire
The data collection questionnaire was adapted from the WHO Stepwise Approach to Chronic Disease Risk Factor Surveillance tool 28 and recently published literature on MetS. The questionnaire was first developed in English and then translated into the Amharic language. It covers the following sections: sociodemographic and economic factors (such as sex, age, occupation, monthly income, marital status, residence, and education); anthropometric and related measurements (body weight, body mass index (BMI), waist circumference (WC), hip circumference (HC), neck circumference (NC), waist-to-hip ratio (WHR), waist-to-height ratio (WHtR), fat mass and visceral fat levels); clinical factors (comorbid conditions, self-reported recalling problem and family history of hypertension and diabetes); behavioral factors (alcohol use, smoking history, and level of physical activity and engagement); and HIV and its treatment-related variables (time since HIV infection, ART regimen type, treatment switches and the duration since ART initiation) and other relevant information. All data were collected by trained and experienced nurses and laboratory technologists working in the ART clinics of the respective health facilities.
Assessments and measurements
Sociodemographic, economic, anthropometric, body composition, clinical, behavioral, and other relevant data were collected through participant interviews and the review of their medical records using a pretested structured questionnaires. After a minimum of 10 min of rest in the data collection room, participants’ blood pressure was measured using a digital blood pressure monitor. This blood pressure apparatus has a diagnostic accuracy of ±3 mm of mercury (mmHg) for blood pressure and ±5% for pulse rate. Two readings were taken within a 5-min interval and the average was recorded. If the difference between the initial two readings exceeded 5 mmHg, a third reading was taken and the average of the last two readings was then recorded to assess the blood pressure status of each participant.
The WHO Stepwise approach was applied to collect data on body weight, height, and WC. 29 Body weight, visceral fat level (1–30 range), and body fat percentage (BFP) were measured using the Omron Full Body Sensor Body Composition Monitor with Scale (Omron HBF-514C; Omron Healthcare Ltd.). Measurements with this Omron bioelectrical impedance analyzer (BIA) were conducted in accordance with the manufacturer’s guidelines. 30 Standing height of the participants was measured with a Seca stadiometer (Hamburg, Germany) to the nearest 0.1 cm. Participants’ weight and height were measured while they stood barefoot and dressed in light clothing. BMI (in kg/m2) was calculated using the BIA during weight measurement by inputting each individual’s respective height into the device. The BMI was then categorized as underweight (<18.5 kg/m²), normal weight (18.5–24.9 kg/m²), overweight (25–29.9 kg/m²), and obesity (⩾30 kg/m²). 31 While fat mass was calculated from body weight and BFP as ((weight × BFP)/100).
WC and HC measurements were taken using a non-stretchable measuring tape and recorded to the nearest 0.1 cm. The WHR and WHtR were calculated by dividing WC by HC and WC by height, respectively. 32 Abdominal obesity was defined as a WC ⩾94 cm for males and ⩾80 cm for females, a WHR >0.90 in men and >0.85 in women, or a WHtR ⩾0.5. 32 NC was measured while participants stood upright with their arms relaxed at their sides. The head was positioned in the Frankfort horizontal plane, and the measurement was taken at the midpoint of the cervical spine and the mid-anterior neck, perpendicular to the neck’s long axis, just below the Adam’s apple for males. 33
Regarding substance use, current alcohol consumption and smoking refer to individuals who engaged in each behavior at least once during the month prior to the study. Ever alcohol drinking, smoking, and khat use denote respondents who have used each substance at any point in their lifetime. 34 In addition, the short form of the International Physical Activity Questionnaire (IPAQ) was used to assess participants’ physical activity levels. Total activity was measured in metabolic equivalent of task (MET)-minutes per week and categorized as low (<600 MET-min/week), moderate (600 to <3000 MET-min/week), or high (⩾3000 MET-min/week). 35
Blood sample collection, processing, and laboratory analysis
About 4–5 mL of venous blood was collected from each participant after an overnight fasting of 8–12 h. The samples were drawn into gel-based serum separator tubes, with each tube carefully labeled using the participant’s unique identification code. The blood samples were then left undisturbed at room temperature for a maximum of 20 min to allow for clot formation. Following this, they were promptly centrifuged at 3000 rotation per min (rpm) for 8–10 min. This process enabled serum separation from cells via a gel barrier, effectively creating a stable barrier between the two layers. Then, the serum samples were either analyzed promptly or transferred into Nunc tubes labeled with participants’ unique code and stored in the deep freezer at −20°C in the Hawassa University Comprehensive Specialized Hospital laboratory in case of a 1- or 2-week delay in laboratory analysis. The fasting blood glucose (FBG) and lipid profiles were analyzed using the Cobas 6000 series biochemistry analyzer (Roche diagnostics, Germany). TGs were measured using an enzymatic colorimetric method, FBG was measured with the hexokinase UV method, and HDL-c level was determined using a direct homogeneous enzymatic assay. Both blood samples collection and laboratory analysis were performed by trained laboratory technologists.
Outcome variable of the study
The primary outcome variable in this study was the prevalence of MetS among adults living with HIV who were on first-line ART. It is defined according to the 2009 harmonized criteria established by the Joint Interim Statement (JIS). A diagnosis of MetS in an individual requires the presence of at least three of the following components: (1) TGs ⩾150 mg/dL or treatment for high TGs; (2) HDL-c <40 mg/dL in males or <50 mg/dL in females, or treatment for low HDL-c; (3) BP ⩾130/85 mmHg or previously diagnosed hypertension or treatment with antihypertensive medication; (4) FBG ⩾100 mg/dL or previously diagnosed diabetes or treatment for high glucose; and (5) WC ⩾80 cm in females or ⩾94 cm in males. 4
Ensuring data quality
The questionnaire was initially developed in English and then translated into Amharic by a bilingual translator. Prior to data collection, a pretest was conducted on 46 the questionnaires, and necessary amendments were made based on the feedback obtained during the pretest. Data collectors received training before the study commenced. Test tube labels were cross-checked with participants’ unique identification numbers, and blood samples collection was carefully performed, including proper tourniquet application to prevent hemolysis. All laboratory procedures were executed in accordance with standard operating procedures. All data collection activities were overseen by the principal investigator.
Statistical analysis
Descriptive statistics were used to summarize the study participants across different variables. The chi-square test was used to assess the significance of categorical variables, while the independent t-test and Mann–Whitney U test were used to compare continuous quantitative data between study groups. In addition, binary logistic regression analysis was performed to evaluate the association between independent factors and the study outcome. Variables with a p-value <.20 in the bivariablete analysis were included in the multivariable analysis. In addition, variables such as sex and ART regimens, which have been previously identified as risk factors for MetS were included in the final model regardless of their significance in the bivariate analysis. Prior to multivariable analysis, multicollinearity was assessed using a variance inflation factor (VIF) to identify linear associations between predictors. variable with a VIF threshold of less than 10 were considered acceptable for this analysis. Whereas model fit was evaluated with the Hosmer–Lemeshow test, where a p-value >0.05 indicates a good fit. The strength of associations between the outcome and explanatory variables was reported using adjusted ORs (aORs) and 95% CI, with a p-value of <0.05 considered statistically significant. Data were entered into EpiData 3.1 and statistical analysis was performed using IBM Statistical Package for the Social Sciences (SPSS) Statistics version 27.0 (IBM Corp., Armonk, NY, USA).
Results
Sociodemographic and behavioral characteristics of the study participants
Of 465 eligible adult PLWH, 450 participated in the study (with a 96.8% response rate). Among the participants, 262 (58.2%) were female, and more than half were married. The majority of participants were resided in urban areas, and 262 (58.2%) reported alcohol consumption. Regarding nutritional status, nearly one-third were classified as overweight, while 61(13.5%) were categorized as obese (Table 1).
Characteristics of adult people living with HIV on antiretroviral treatment.

Physical activity (low: <600 MET-min/week; moderate: 600 to <3000 MET-min/week and high: ⩾3000 MET-min/week).
Anthropometric and clinical characteristics of the study participants
The mean age of the participants was 41.1 (standard deviation: 9.7) years and females were significantly younger than males. Females had significantly higher mean BMI, HC, HDL-c, and median fat mass compared to males. On the contrary, males had significantly increased mean systolic and diastolic blood pressure, WHR, NC, and median TG/HDL-c, visceral fat, and TGs than females. However, mean WC, FBG, and ART duration did not show significant variation between sexes (Table 2).
Anthropometric and clinical characteristics of adult people living with HIV on antiretroviral treatment.
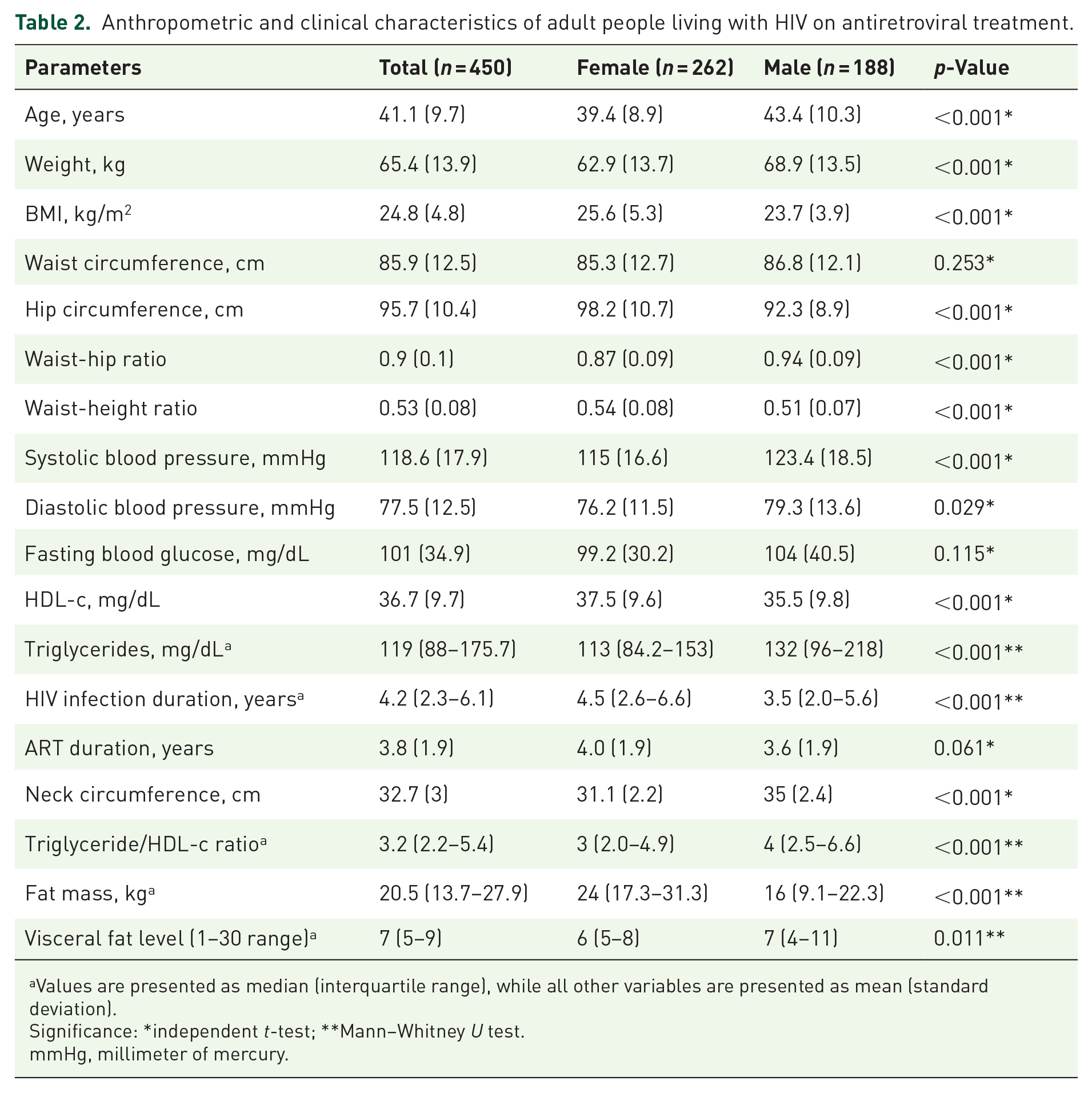
Values are presented as median (interquartile range), while all other variables are presented as mean (standard deviation).
Significance: *independent t-test; **Mann–Whitney U test.
mmHg, millimeter of mercury.
Two hundred twenty nine (50.6%) participants initiated DTG-based first-line ART regimens, while 221 (49.4%) participants initiated treatment with NNRTI-based first-line regimens, both with a similar backbone of two N(t)RTIs. Among those who initially started with NNRTI-based regimens, 204 of 221 (92.3%) were later switched to DTG-based first-line regimens, while 17 of 221 (7.7%) continued with NNRTI-based regimens.
Prevalence of MetS and its components
According to the 2009 harmonized (JIS) criteria, 422 (93.8%) participants had at least one component of MetS, with 81(18%) having three, 61 (13.6%) having four, and 22 (4.9%) having five components of MetS. The prevalence of two components of MetS was 29.4% (77/262) among female participants, accounting for 17.1% of all participants, and 22.9% (43/188) among male participants, representing 9.6% of all participants (Figure 1). Overall, 164 participants had MetS with a prevalence of 36.4% (95% CI: 32.2–41.6). Of the 164 participants with MetS, 73 (44.5%) were males and 91 (55.5%) were females. In terms of age groups, 42.7% (95% CI: 35.3–51.3) of participants aged 40–49 years and 54.7% (95% CI: 44.2–64.2) of those aged ⩾50 years had MetS (Figure 2). In addition, 62.4% (95% CI: 55–69.3) of participants with a BMI ⩾25 kg/m² had MetS. Among the 164 total cases of MetS: 79 (48.25%) were individuals who initiated and remained on a DTG-based regimen, while 77 (47%) were transitioned from an NNRTI-based first-line regimen to DTG, and 8 (4.9%) initiated and continued on NNRTI (EFV)-based regimen (Figure 3). Moreover, 15 (3.3%) participants on the efavirenz-based regimen, 165 (36.7%) who switched to a DTG-based regimen, and 188 (41.8%) who remained on the DTG-based regimen had low HDL-c (Figure S1).

Overview of the metabolic syndrome components in study participants stratified by sex.

Patterns of metabolic syndrome among people with HIV on first-line treatment in relation to age categories.
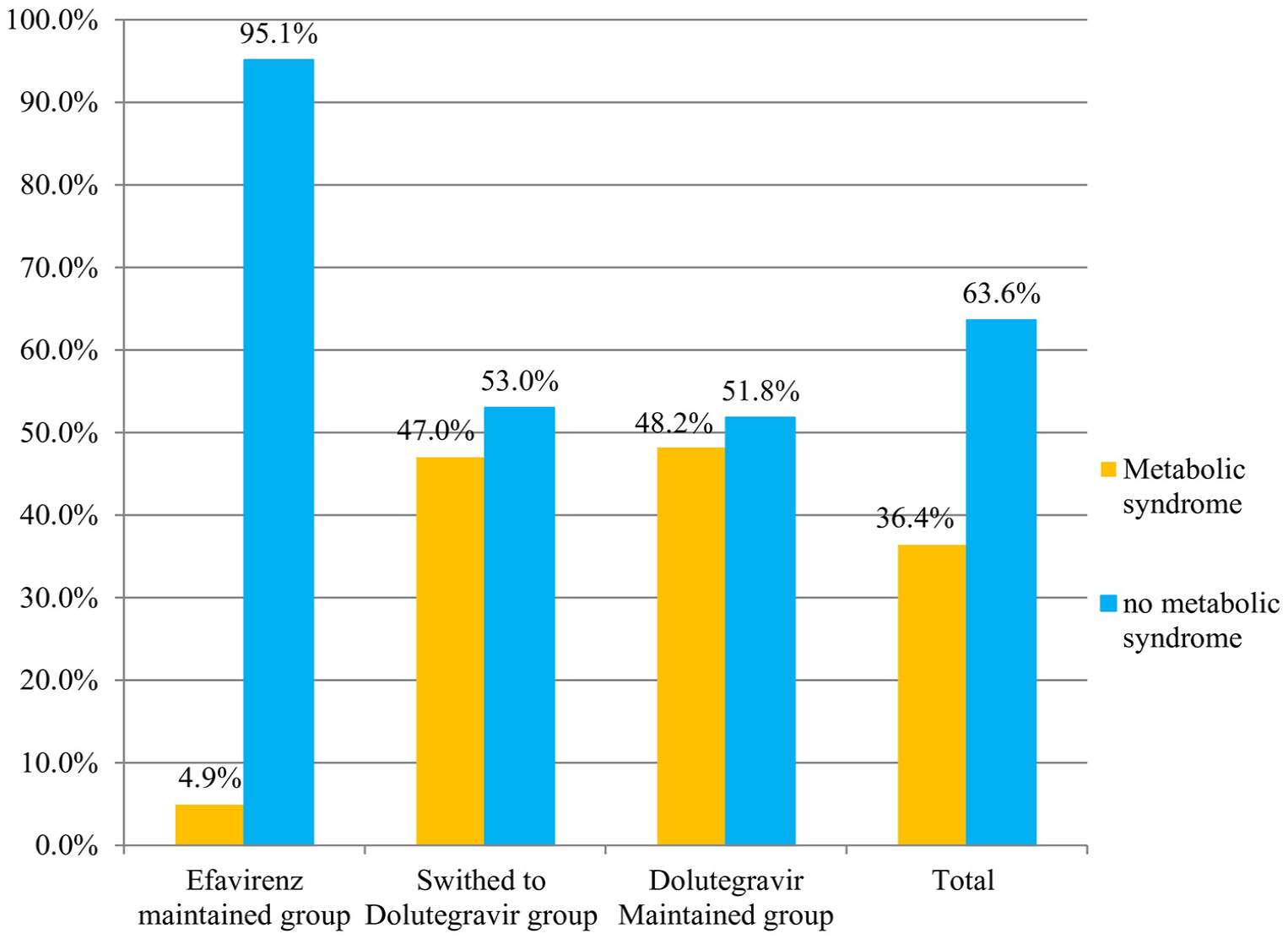
Patterns of metabolic syndrome among people with HIV on first-line treatment in relation to antiretroviral regimens.
The prevalence of individual components of MetS was as follows: 220 (44.4%) for high WC, 110 (24.4%) for raised FBG, 154 (34.2%) for high TGs, 143 (31.8%) for elevated blood pressure, and 368 (81.8%) for low HDL-c. In addition, females had significantly higher rates of abdominal obesity (51.5% vs 34.6%), low HDL-c (89.3% vs 71.3%), and high WHtR (70% vs 58%) compared to males, respectively. In contrast, males had significantly higher rates of elevated blood pressure (39.4% vs 26.3%), high TGs (43.6% vs 27.5%), and high WHR (69.1% vs 57.3%) compared to females, respectively (Table 3).
Prevalence of metabolic syndrome and its components among adult people living with HIV on first-line antiretroviral treatment.

HDL-c, high-density lipoprotein cholesterol.
Factors associated with MetS
We found that age >50 years, alcohol consumption, BMI ⩾25 kg/m², TG/HDL-c ratio, high WHtR, and a family history of hypertension were significantly associated with the prevalence of MetS. In contrast, DTG-based first-line regimens did not show a significant association with MetS when compared to NNRTI (efavirenz)-based regimens (Table 4).
Factors associated with metabolic syndrome among adults on first-line antiretroviral treatment.

Continuous covariate.
aOR, adjusted odds ratio; ART, antiretroviral therapy; cOR, crude odds ratio; ETB, Ethiopian birr; HDL-c, High-density lipoprotein cholesterol; TG, triglyceride.
Discussion
MetS was prevalent among adults on ART in southern Ethiopia, with a rate of 36.4% (95% CI: 32.2–41.6). Factors significantly associated with MetS included older age, alcohol consumption, BMI, high WHtR, a family history of hypertension, and a high TG/HDL-c. The use of DTG-based regimens did not show a significant association with MetS compared to those on efavirenz-based regimens.
The overall prevalence of MetS in the study was 36.4%, which is higher than the rates reported in systematic reviews and cross-sectional studies conducted in similar populations both globally and in Africa, where the prevalence ranged from 18.0% to 29.91% using IDF criteria,9,10,36,37 18.1% to 34.89% using ATP III criteria9,10,36,37 and from 13.9% to 33.1% using JIS.15,18,27,38,39 The finding was also higher than the prevalence reported among apparently healthy working adults in Ethiopia10,11,40 as well as in community-based and public servant studies,41,42 which ranged from 11.9% to 27.6% according to the IDF11,40–43 and from 11.2% to 18.5% according to ATP III.10,40–43 The discrepancies may be attributed to variations in diagnostic criteria, study timeframes, the effects of ART regimens on metabolic and cardiovascular health, and differences in the clinical and anthropometric characteristics of the populations. 44
Low HDL-c was the most frequent component of MetS in this study, with a prevalence of 81.8%. Similarly, several studies have identified low HDL-c as the most frequently observed component of MetS among PLWH.45,46 However, the finding was higher than the rates reported in previous studies, such as 64.1% in north-central Ethiopia, 47 75% in young adults and 58% in adults in Zambia, 48 and 72.1% in Uganda. 19 Differences in treatment duration 19 and the varying effects of ART medications may also explain the observed variations in HDL-c level across studies. 49
Sex was not associated with MetS in this study, which is consistent with reports from Nigeria 50 and Ethiopia. 51 In contrast, findings from apparently healthy general populations in Ethiopia40,42 and several similar studies conducted in PLWH14,36,52 have reported a significant association between female sex and MetS. This might be due to hormonal differences, abdominal fat distribution, level of physical activity engagement, and cultural factors, all of which differentially influence metabolic risk between sexes across studies.
In this study, more than 50% of participants over 50 years old had MetS, with older age being significantly associated with an increased risk of developing the condition. This finding was consistent with other studies conducted in Ethiopia.40–42 In support, aging may contribute to the loss of muscle mass and strength due to reduced physical activity, leading to increased fat accumulation and decreased lean mass, which likely contribute to the development of MetS. 53
A significant association between TG/HDL-c ratio and MetS was observed in this study, with each additional unit increase in TG/HDL-c corresponding to a 50% higher risk of developing MetS. The finding was consistent with previous studies that reported similar associations between TG/HDL-c and MetS, 54 further, highlighting its stronger association with the incidence of CVD. 55
In this study, 62.4% of individuals with a BMI ⩾25 kg/m² had MetS, and BMI was significantly associated with MetS. Similar findings have been reported in studies conducted both in Ethiopia and elsewhere.12–15,36,43 In addition, this study found a significant association between WHtR and MetS, which is consistent with previous studies that reported similar findings.54,56
Our study also identified a significant association of MetS with a family history of hypertension, which is in line with previous research showing that individuals with such a family history are more likely to have obesity, central obesity, and MetS. 57 This may be due to inherited genetic traits that predispose individuals to elevated blood pressure and other metabolic abnormalities, thus increasing the risk of MetS.
We observed a significant association between alcohol consumption and MetS, which is consistent with results from other studies.40,42,43 Alcohol may raise blood pressure and trigger metabolic changes linked to inflammation and obesity.58,59 In PLWH, at-risk alcohol use exacerbates cardiometabolic and neurocognitive issues, which, combined with chronic ART use, contribute to frailty and age-related comorbidities. 60 Overall, these findings strongly support the link between alcohol use and accelerated biological aging, as well as an increased risk for comorbidities. 60
Moreover, over 90% of participants with MetS were on DTG-based regimens in this study; however, no significant association was observed between DTG-based regimens and MetS. This finding is consistent with previous studies that reported similar results among individuals treated with DTG-based regimens.61,62 In contrast, several other studies have reported a significantly higher incidence of treatment-emergent MetS among individuals on DTG-based regimens compared to those on non-DTG regimens,17,52 with some also identifying a significant association between DTG and MetS. 63 The limited number of participants in the NNRTI group in our study, due to the national rollout of DTG treatment, may have contributed to the observed variation and influenced the interpretation of the association between DTG and MetS.
Strength and limitations of the study
A strength of our study is that it included all adults who initiated first-line ART across multiple health facilities following Ethiopia’s adoption of a universal test-and-treat strategy. This strategy prioritizes a positive HIV diagnosis for ART initiation, regardless of CD4+ count or WHO clinical stage, thus minimizing the inclusion of individuals with low CD4+ counts, advanced disease stages, or significant wasting syndrome, which could contribute to cardiometabolic complications. Additionally, the study included individuals who had undergone at most one switch within first-line regimens, reducing the potential for cardiometabolic complications associated with the use of multiple regimens or drugs. However, this study has several limitations. First, the cross-sectional design prevents establishing causal relationships between risk factors and MetS. Second, we did not conduct a nutritional assessment, despite its known association with MetS and cardiovascular risk. Third, the small number of participants on an NNRTI-based regimen may have limited our ability to fully interpret the association between ART regimens with MetS. Fourth, recall bias may have affected the accuracy of family history data, particularly regarding hypertension and diabetes, while reliance on self-reported data introduced potential bias and limitations in accuracy. Lastly, we used only the 2009 harmonized (JIS) criteria to assess MetS prevalence; different results may have been found if other criteria, such as WHO, NCEP-ATP III, or IDF, had been used.
Conclusion
We identified a high prevalence of MetS among adult PLWH in southern Ethiopia, with low HDL-c being the most frequently observed component. MetS was associated with both non-modifiable (age and family history of hypertension) and modifiable (BMI, WHtR, alcohol intake, and TG/HDL-c) risk factors, indicating a considerable cardiovascular risk. However, our study found no association between DTG-based first-line regimens and an increased risk of MetS.
Therefore, regular screening and monitoring for MetS components are essential to mitigate the health risks associated with metabolic abnormalities. As most of the identified risk factors are modifiable, effective lifestyle interventions are essential. In addition, implementing a comprehensive approach is essential for early diagnosis, preventing cardiovascular complications and enhancing incident management in this population. Furthermore, studies are required to investigate additional underlying factors and the long-term metabolic risks associated with ART.
Supplemental Material
sj-jpeg-1-taj-10.1177_20406223251346289 – Supplemental material for Prevalence and determinants of metabolic syndrome among adults living with HIV on first-line antiretroviral treatment in southern Ethiopia: a cross-sectional study
Supplemental material, sj-jpeg-1-taj-10.1177_20406223251346289 for Prevalence and determinants of metabolic syndrome among adults living with HIV on first-line antiretroviral treatment in southern Ethiopia: a cross-sectional study by Agete Tadewos Hirigo, Daniel Yilma, Ayalew Astatkie and Zelalem Debebe in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We sincerely appreciate the nurses, adherence workers, and laboratory staff at the ART clinics of the study health facilities, as well as all those who supported this study with materials and moral encouragement. Our heartfelt thanks also go to the study participants for their active involvement. In addition, we would like to acknowledge Addis Ababa University, Hawassa University, and the NORAD project for their financial support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
