Abstract
Background:
Sodium harmony is closely correlated with the progression of chronic kidney disease (CKD).
Objectives:
The current study aims to explore the significance of the circadian rhythm of urinary sodium excretion in CKD management and to help establish individualized salt restriction strategies.
Design:
This is a prospective study among inpatients with CKD.
Methods:
The study included 715 participants with CKD from the Third Affiliated Hospital of Sun Yat-Sen University. Multivariable Cox regression models and restricted cubic splines were employed to explore the independent and joint associations of 24-h urinary sodium and sodium rhythm with prognosis of CKD.
Results:
During a median follow-up of 6.92 years, 286 major renal events, 112 MACE, 160 deaths, and 321 composite events were documented. 24-h urinary sodium was not associated with any outcomes. However, there were dose–response associations of diurnal sodium/24-h urinary sodium with major renal events (poverall < 0.001, pnonlinearity = 0.016) and composite events (poverall < 0.001, pnonlinearity = 0.129). Compared with the combination of low 24-h sodium and normal sodium rhythm, the combination of high 24-h sodium and abnormal sodium rhythm had the highest risk of incident major renal events (hazard ratio, 1.62; 95% confidence interval, 1.04–2.51). A significant interaction was observed between sodium intake and sodium rhythm in associations with composite events (p for interaction = 0.015).
Conclusion:
The circadian rhythm of sodium excretion emerges as a novel and potentially more sensitive risk factor for the prognosis of CKD. Furthermore, the association between sodium intake and progression of CKD was notably pronounced among individuals with abnormal sodium rhythms. Efforts to implement individualized salt restriction strategies are warranted.
Plain language summary
Managing salt intake is essential for patients with CKD, but new research shows that when and how the body processes salt over 24 hours could be just as important as how much salt is consumed. The current study examined the effects of both 24-hour salt excretion and the rhythm of that excretion on kidney health, heart health, and overall survival. Interestingly, the total amount of salt excreted did not directly correlate with health outcomes. However, disruptions in the daily pattern of sodium excretion were associated with worse kidney outcomes and combined health events. Patients with both a high salt intake and irregular sodium rhythms had the highest risk of kidney failure. Additionally, salt intake and sodium rhythm appeared to interact, amplifying the risk of poor outcomes. The study highlights the importance of considering not only how much salt CKD patients consume but also how their bodies process it over the day. These findings suggest that tracking sodium rhythms may help personalize salt-reduction strategies, offering a new approach to improving CKD management and outcomes.
Introduction
Chronic kidney disease (CKD) is a progressive disease with no definitive cure, exhibiting high prevalence and mortality rates, affecting more than 10% of the global population and thereby increasing the global health burden.1–3 In 2021, CKD became the 11th leading cause of death worldwide. 4 There has been extensive research on biomarkers associated with CKD prognosis.3,5 Patients with CKD are often suggested to reduce their salt intake. 6 However, long-term low-salt diets may also lead to side effects, such as elevated blood lipids, insulin resistance, and activation of the Renin-angiotensin-aldosterone system (RAAS). 7 The impact of low-salt diets on the progression of CKD, cardiovascular diseases, and mortality is still conflicting.7–10
Various physiological functions of the kidney exhibit distinct circadian rhythms. 11 The renal blood flow, urine production, electrolyte excretion, and blood pressure exhibit daily oscillations. 12 Previous studies have indicated that renal function impairment disrupts the normal circadian rhythm of urinary sodium excretion. This disruption impairs diurnal natriuresis, leading to increased pressure natriuresis and compensatory nocturnal urinary sodium excretion.13,14 Consequently, the disruption is associated with elevated nocturnal blood pressure and results in a typical nondipping pattern,13,15 which is associated with poor prognosis among individuals with CKD.16,17 Additionally, our previous study also found that abnormal circadian rhythm of urinary sodium excretion correlates closely with hypertension target organ damage in Chinese patients with CKD. 18 However, the prospective evidence on the association between sodium rhythm and prognosis of CKD is lacking. Furthermore, the sodium intake and sodium rhythm might be interactional with renal function. The impaired renal sodium excretory capacity and disrupted circadian rhythm might be remarkable when the salt intake is higher than appropriate. 19 The joint effects and interaction of sodium intake and circadian rhythm of urinary sodium excretion on the prognosis of CKD remain unknown.
Therefore, to address these important gaps, the current study prospectively aims to investigate the independent dose–response associations and joint effects of sodium intake and the circadian rhythm of urinary sodium excretion with the risk of major renal events, major adverse cardiovascular events (MACE), and all-cause mortality among individuals with CKD.
Methods
Study population
We recruited participants with CKD aged 14–75 years from the Third Affiliated Hospital of Sun Yat-Sen University between July 2010 and December 2019. 20 The exclusion criteria were: other causes resulting in secondary hypertension such as renovascular hypertension, Cushing’s syndrome or aldosteronism; sustained BP higher than 200/120 mmHg; had taken an anti-hypertensive medication (before BP data and urinary sodium were collected, short-acting amlodipine was used according to the patient’s condition) or a diuretic drug in the previous month; had taken a salt restriction intervention or self-reported poor appetite; treatment with corticosteroids or hormones; acute changes in the estimated glomerular filtration rate (eGFR) > 30% in the previous 3 months; maintenance dialysis; kidney transplantation; history of drug or alcohol abuse; employment involving night work or shiftwork; acquired immunodeficiency syndrome; inability to communicate or comply with all the study requirements; pregnancy; and refusal to provide informed consent. All participants provided written informed consent before enrollment in the study. eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration equation 21 :
For women,
For men,
Exposures
Exposures in the current study were sodium intake and the circadian rhythm of urinary sodium excretion. 24-h urinary sodium excretion is considered the gold standard measurement for assessing salt intake. 22 We collected urine samples from 07:00 to 22:00 (diurnal) and between 22:00 and 07:00 (nocturnal), respectively. The patients were asked to void their bladders at 07:00 and 22:00 to ensure valid results. The concentration of sodium in the urine was measured with flame spectrophotometry. The urine sodium excretion had been normalized to urine volume. 24-h urinary sodium excretion was calculated as the sum of diurnal and nocturnal values. We divided it into two groups according to the guideline-recommended level (2 g/d): high (24-h urinary sodium ⩾ 85.47 mmol) and low (24-h urinary sodium <85.47 mmol). We assessed the circadian rhythm of urinary sodium excretion using the ratio of diurnal to 24-h urinary sodium. We used the median value (0.62) as the cutoff to define the rhythm categories, resulting in two groups: normal (ratio ⩾ 0.62) and abnormal (ratio < 0.62).
Outcomes
The primary outcomes were major renal events (including death as a result of kidney disease, requirement for dialysis or transplantation, or doubling of serum creatinine), MACE (death or hospitalization due to cardiovascular diseases), all-cause mortality, and composite events. On March 1, 2023, we conducted censoring and computed the follow-up duration from the baseline date to the occurrence of the earliest event among death, first event, or cessation.
Covariates
We considered the following characteristics as the potential covariates: age (continuous, years), sex (male/female), BMI (continuous, kg/m2), smoking (yes/no), drinking (yes/no), diabetes (yes/no), systolic blood pressure (SBP, continuous, mmHg), diastolic blood pressure (DBP, continuous, mmHg), creatinine (continuous, mmol/L), and low-density lipoprotein cholesterol (LDL-C, continuous, mmol/L). SBP and DBP were measured using 24-h ambulatory blood pressure monitoring, and the average value of ambulatory blood pressure throughout the day was calculated. The laboratory data were measured with a 7180 Biochemical Automatic Analyzer (Hitachi, Tokyo, Japan).
Statistical analysis
Baseline characteristics were presented as mean (standard deviation, SD) or mean (interquartile range, IQR) for continuous variables. Frequencies and percentages were used for categorical variables. Missing data were multiply imputed using the “mice” R package to maximize the statistical power, assuming that data were conditionally missing at random. 23
We used Kaplan–Meier curves to plot the cumulative incidence of major renal events, MACE, all-cause mortality, and composite events by 24-h sodium and sodium rhythm. The continuous dose–response analyses assessed the shape of the associations of 24-h sodium and sodium rhythm with the renal event, MACE, all-cause mortality, and composite event. We used restricted cubic splines with three knots to assess the possible linear and nonlinear associations. Departures from linearity were examined using a Wald test. Additionally, we performed two multivariable-adjusted Cox proportional hazards regression models to estimate the hazard ratio (HR) for the associations of 24-h sodium and sodium rhythm categories with incident renal event, MACE, all-cause mortality, and composite event. Model 1 was adjusted for age, sex, BMI, smoking and drinking; Model 2 was additionally adjusted for diabetes, SBP, DBP, creatinine, and LDL-C.
To test the joint association between 24-h urinary sodium and circadian rhythm of urinary sodium excretion, we first examined their interaction in relation to the risk of major renal events, MACE, all-cause mortality, and composite events. Furthermore, we subdivided the overall sample into four groups according to the 24-h urinary sodium and circadian rhythm of urinary sodium excretion. We used multivariable Cox models to estimate the joint association between 24-h urinary sodium and circadian rhythm with incident major events, MACE, all-cause mortality, and composite events, with the group having low 24-h urinary sodium and normal sodium rhythm used as a reference. Additionally, we repeated Cox regression and restricted cubic spline analyses to investigate the association between 24-h urinary sodium and the incidence of major renal events, MACE, all-cause mortality, and composite events among participants with normal or abnormal sodium rhythm.
Statistical analyses were performed using R software Version 4.3.1 (R Development Core Team, Vienna, Austria). A value of p < 0.05 (two-sided test) was considered statistically significant.
Results
Baseline characteristics
As shown in Table 1, the present study included 715 participants (56.9% male, mean age: 45.7 years), 167 (23.3%) were classified as having low 24-h sodium, and 357 (50.0%) were categorized as having an abnormal sodium rhythm. Individuals with higher 24-h sodium levels were more likely to be male, had a higher BMI, and worse blood pressure control. Those with an abnormal sodium rhythm were older, more likely to be female, and had unhealthier lifestyles and worse health conditions.
Baseline characteristics among patients with CKD stratified by 24-h urinary sodium and sodium rhythm.
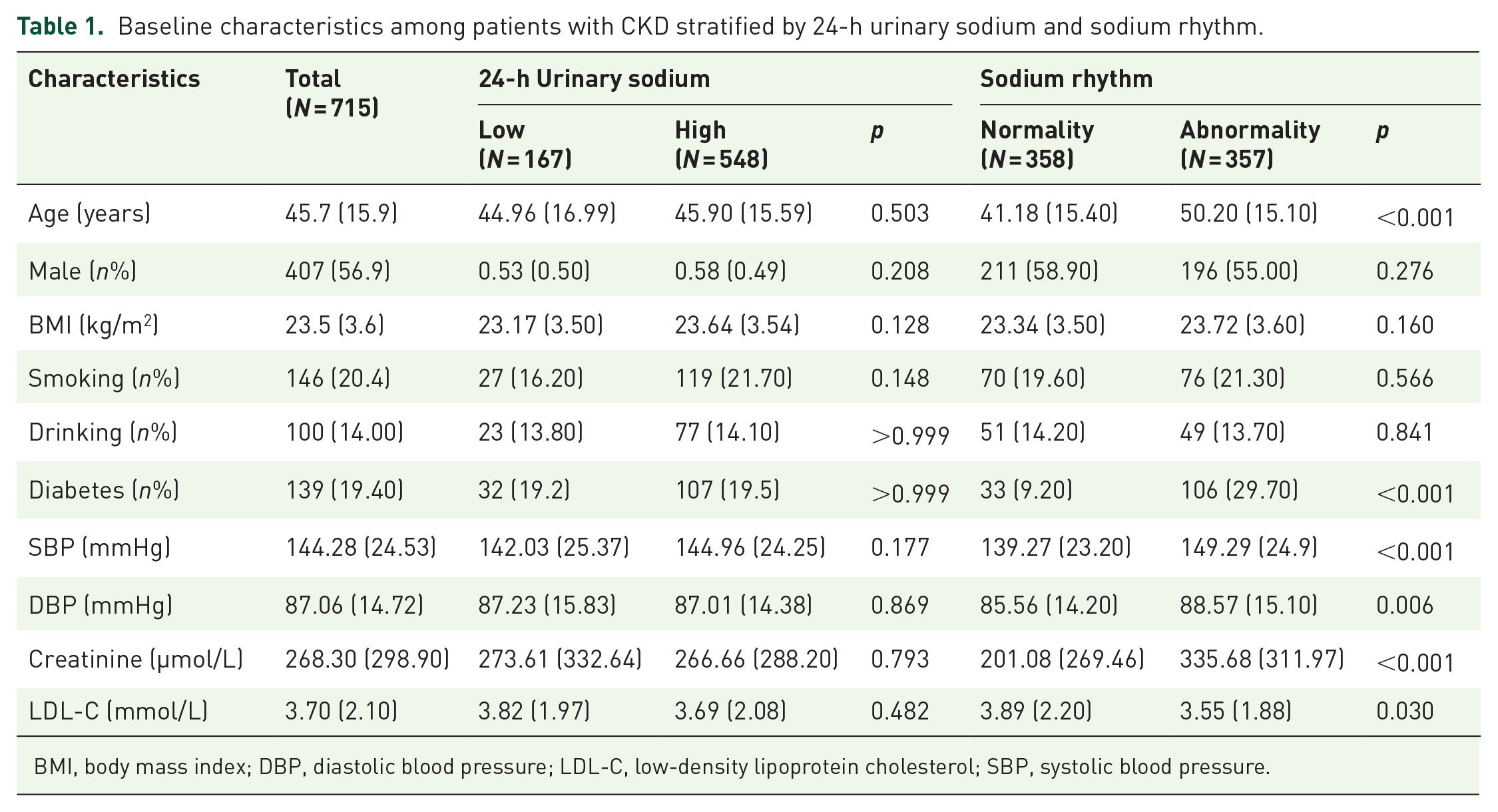
BMI, body mass index; DBP, diastolic blood pressure; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure.
Independent associations of 24-h sodium and sodium rhythm with major renal events, MACE, and all-cause mortality
The cumulative incidence of major renal events, MACE, all-cause mortality, and composite events according to 24-h sodium and sodium rhythm is presented in Figure 1. During a median follow-up of 6.92 years, 286 major renal events, 112 MACE, 160 deaths, and 321 composite events were documented. As shown in Figure 2, for dose–response associations, 24-h urinary sodium was not associated with any outcomes (all poverall > 0.05, pnonlinearity > 0.05). However, there was a nonlinear dose–response association between diurnal sodium/24-h urinary sodium and major renal events (poverall < 0.001, pnonlinearity = 0.016). In terms of composite events, we found a linear association (poverall < 0.001, pnonlinearity = 0.129; Figure 3). For categorical analysis (Tables 2 and 3), compared with individuals with a normal sodium rhythm, those with an abnormal sodium rhythm had increased risks of major renal events (HR, 2.37; 95% confidence interval (CI), 1.85–3.02), MACE (HR, 2.23; 95% CI, 1.51–3.30), all-cause mortality (HR, 2.57; 95% CI, 1.83–3.60), and composite events (HR, 2.44; 95% CI, 1.94-3.07) in the univariate models. In the fully adjusted models, the associations for major renal events (HR, 1.65; 95% CI, 1.28–2.14) and composite events (HR, 1.53; 95% CI, 1.20–1.94) were still significant.

Cumulative rate of incident renal event, MACE, all-cause mortality, and composite event according to 24-h sodium and sodium rhythm.

Dose–response associations of 24-h urinary sodium with major renal events, MACE, and all-cause mortality.

Dose–response associations of sodium rhythm (diurnal/24-h urinary sodium) with major renal events, MACE, and all-cause mortality.
Association of 24-h urinary sodium with major renal events, MACE, and all-cause mortality.

Model 1 was adjusted for age and sex.
Model 2 was additionally adjusted for BMI, smoking, drinking, history of diabetes, SBP, DBP, creatinine, and LDL-C.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; MACE, major adverse cardiovascular events; SBP, systolic blood pressure.
Association of sodium rhythm (diurnal/24-h urinary sodium) with major renal events, MACE, and all-cause mortality.

Model 1 was adjusted for age and sex.
Model 2 was additionally adjusted for BMI, smoking, drinking, history of diabetes, SBP, DBP, creatinine, and LDL-C.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; LDL-C, low-density lipoprotein cholesterol; MACE, major adverse cardiovascular events; SBP, systolic blood pressure.
Joint associations of 24-h sodium and sodium rhythm on major renal events, MACE, and all-cause mortality
Figure 4 illustrates a potential nonlinear dose–response association of 24-h urinary sodium with major renal events (poverall = 0.033, pnonlinearity = 0.009) and composite events (pnonlinearity = 0.020) among individuals with abnormal sodium rhythm. As shown in Table 4, compared with the combination of low 24-h sodium and normal sodium rhythm, a combination of high 24-h sodium and abnormal sodium rhythm had the highest risk of incident major renal events (HR, 1.62; 95% CI, 1.04–2.51). There was a significant interaction (p for interaction = 0.015) between 24-h sodium and sodium rhythm in associations with composite events (Table 5). Compared with individuals with recommended levels of 24-h sodium, higher 24-h sodium was associated with increased risk of incident major renal events (HR, 1.52; 95% CI, 1.05–2.22) and composite events (HR, 1.47; 95% CI, 1.02–2.11) among individuals with abnormal sodium rhythm. For MACE and all-cause mortality, there were no interactions or joint effects of 24-h sodium and sodium rhythm.

Dose–response associations of 24-h urinary sodium with major renal events, MACE, and all-cause mortality stratified by sodium rhythm.
Joint associations of 24-h sodium and sodium rhythm (diurnal/24-h urinary sodium) on major renal events, MACE, and all-cause mortality.

Model 1 was adjusted for age and sex.
Model 2 was additionally adjusted for BMI, smoking, drinking, history of diabetes, SBP, DBP, creatinine, and LDL-C.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; MACE, major adverse cardiovascular events; SBP, systolic blood pressure.
Associations of 24-h urinary sodium on major renal events, MACE, and all-cause mortality stratified by sodium rhythm.

Model 1 was adjusted for age and sex.
Model 2 was additionally adjusted for BMI, smoking, drinking, history of diabetes, SBP, DBP, creatinine, and LDL-C.
BMI, body mass index; CI, confidence interval; DBP, diastolic blood pressure; HR, hazard ratio; LDL-C, low-density lipoprotein cholesterol; MACE, major adverse cardiovascular events; SBP, systolic blood pressure.
Discussion
In this population-based prospective cohort study, we found that circadian rhythm of urinary sodium excretion, but not sodium intake, was associated with major renal events, and composite events among individuals with CKD. The circadian rhythm of urinary sodium excretion was a more sensitive risk factor for the prognosis of CKD than sodium intake. In addition, the association between sodium intake and incident composite events following CKD was more remarkable among individuals with abnormal sodium rhythm.
Sodium intake is related to extracellular fluid volume, and an increase in fluid volume leads to increased blood pressure. 22 Increased sodium excretion in the kidney requires elevated blood pressure in the glomeruli, resulting in hypertension, which is described as the pressure-natriuresis relationship. 24 Previous large-population-based cohort studies using data from the UK Biobank found that salt intake increased the risk of incident CKD. 25 Several studies also reported that kidney function decline is related to urinary sodium excretion in patients with CKD.26,27 However, a meta-analysis including 16 observational studies and 20 intervention studies concluded that there was no robust evidence suggesting that long-term reduction of salt intake prevents CKD or delays its progression. 28
Previous studies have reported the associations of sodium intake with health-related risk, but the results were inconsistent. Sodium intake was previously reported to be associated with cardiovascular risk in linear, “U-shape,” or “J-shape” patterns,29,30 which implied that both higher and lower sodium intake were associated with cardiovascular events. Another explanation was the different measurement methods of salt intake. 31 Our study measured it using 24-h urine, which is considered the gold standard. 32 In terms of individuals with CKD, Yasuyuki Nagasawa thought that salt intake had dual effects on prognosis, both negative and positive effects. 33 A prospective cohort study of patients with CKD from seven locations in the United States enrolled in the Chronic Renal Insufficiency Cohort Study and a dose–response meta-analysis both reported that higher urinary sodium excretion was associated with increased risk of cardiovascular events.8,34 Furthermore, previous cohort studies found that sodium intake increased the risk of kidney outcomes and all-cause mortality.35,36 However, sodium intake was also reported to have no association with CKD development and long-term survival among CKD patients.37,38 The inconsistency might be attributed to different response to sodium intake; thus, individualized salt restriction is urgently needed.
To our knowledge, the current study is the first prospective study to report the association between circadian rhythm of sodium and prognosis of CKD. Renal control of sodium homeostasis is essential for long-term blood pressure control. Electrolyte reabsorption and excretion, including sodium and potassium, have rhythms that rely on circadian as well as reactive adaptations.39–41 Previous cross-sectional studies and ours both found that abnormal circadian rhythm of sodium excretion was associated with higher nocturnal blood pressure and target organ damage,15,18 which is correlated closely with the prognosis of CKD. 16 Furthermore, our findings extended the literature by establishing dose–response associations between urinary sodium and prognosis of CKD among patients with abnormal sodium rhythm but not among those with normal sodium rhythm.
Although the mechanism underlying the joint associations of abnormal rhythm of sodium excretion and sodium intake on prognosis of CKD still needs to be elucidated, several possible mechanisms may partly explain the associations. Blood pressure drops by 10%–20% during the night in healthy subjects. 42 Conversely, in patients with the salt-sensitive type of hypertension, nighttime blood pressure does not fall, resulting in a “nondipper” pattern. Patients with CKD, due to either reduced ultrafiltration capacity or enhanced tubular sodium reabsorption, are more likely to develop the salt-sensitive type of hypertension. 13 When salt intake is excessive the defect in sodium excretory capability becomes evident, resulting in abnormal circadian rhythm of renal function and blood pressure. 19 It has been suggested that a blunted decline in nocturnal BP is associated with sympathetic overactivity, 43 chronic inflammation, 44 increased arterial stiffness, 45 and hyperaldosteronism. 46
From a public health perspective, our findings highlight the circadian rhythm of sodium excretion as a novel and more sensitive risk factor for the prognosis of CKD. For individuals with CKD, the KDIGO guidelines recommend a sodium intake of <2 g/day mainly due to the cost-effectiveness of salt restriction. However, due to the lack of large-scale case-control studies and prospective cohort studies, the recommendation strength in the KDIGO guidelines is only 1C. 47 For interventions targeting individuals and communities, we should focus on not only sodium intake but also the rhythm of sodium excretion for CKD management. Furthermore, the response to salt intake was different among patients with varied characteristics. 13 The recommended total salt intake should be individualized, with the circadian rhythm of urinary sodium excretion taken into consideration. Our findings suggest a potential personalized salt restriction strategy, where restricting salt intake in patients with urinary sodium rhythm disruption may delay the progression of kidney diseases.
The strengths of the current study include long-term follow-up, prospective study design, and careful control of covariates. In addition, we measured sodium intake with 24-h urines, which is considered the gold standard measurement. 32 However, there are also several limitations. First, we selected the study population from the inpatient population, which may lead to admission rate bias. Second, residual confounding and follow-up bias cannot be completely excluded due to the limitations of the observational study design. Third, the current study consisted only of the Chinese population, which limited the generalizability of our findings to other populations. Fourth, we measured 24-h urines only once and failed to investigate the associations between changes in urinary sodium and the prognosis of CKD. Fifth, while we adjusted for the mean value of ambulatory blood pressure, the potential influence of blood pressure patterns was not considered in our analysis. Finally, the sample size was determined based on hospitalized patients who met the eligibility criteria, with those lost to follow-up being excluded. Therefore, the sample size was not predetermined.
Conclusion
In conclusion, the circadian rhythm of sodium excretion appears to be a novel and potentially more sensitive risk factor for the prognosis of CKD. Furthermore, there is significant public health potential in directing salt intake interventions toward individuals with irregular sodium rhythms to mitigate adverse outcomes linked to CKD progression. Therefore, there is a clear need to implement personalized salt restriction strategies.
