Abstract
Background:
Rosacea is a common inflammatory disease. Four clinical rosacea subtypes have been described, with the papulo-pustular being the most common. At present, the only FDA-approved treatment for papulo-pustular rosacea is low-dose doxycycline. Retinoids, in particular isotretinoin, have shown for decades positive outcomes in the treatment of rosacea, but due to the lack of robust evidence, they are still considered an off-label treatment for this condition.
Objectives:
Summarize the current evidence in the literature regarding the role of topical and systemic retinoids in the treatment of papulo-pustular rosacea.
Design:
Systematic review and meta-analysis.
Data source and methods:
A systematic MEDLINE, EMBASE, and Cochrane search of relevant publications on this topic was carried out. Randomized and non-randomized studies regarding topical and systemic retinoid treatment for rosacea were included. We excluded not relevant studies, case reports, reviews, and non-English language studies. We have used RoB2 and ROBINS-1 tool for assessing risk of bias in randomized and non-randomized trials, respectively.
Results:
Due to the heterogeneity in study design and outcomes reporting, the standardization of our results is limited, but the findings from this systematic review with meta-analysis indicate that retinoids, particularly isotretinoin, are a promising treatment option for papulopustular rosacea, with a favorable tolerability and safety profile. On the other hand, available data for topical retinoid therapy are less definitive and more contradictory, with only two randomized studies reporting opposite outcomes.
Conclusion:
Retinoids may be a valid treatment option for rosacea, but larger, randomized controlled trials are needed to establish standardized dosing regimens and long-term safety profiles.
Introduction
Rosacea
Rosacea is a common, multifactorial, inflammatory skin disease with a relevant burden on psychological well-being. 1 It is clinically characterized by recurrent episodes of flushing, persistent erythema, telangiectasia, or papulo-pustules, configuring a multiphasic phenotype spectrum of disease with associated burning, pain, or migraine-like symptoms.2–4 Rosacea is thought to affect equally women and men, with a global prevalence recently estimated to be 5.5% of the general population, and can occur at any age.5–7 Therapeutic options for rosacea include patient education, establishment of a personalized skincare routine, topical and oral medications, as well as lasers/light-based therapies. 8
Four rosacea clinical subtypes have historically been described: erythemato-telangiectatic, papulo-pustular, phymatous, and ocular rosacea. 9
Most studies investigating the effectiveness of therapies for rosacea are conducted in patients with the papulo-pustular rosacea, which is the most prevalent disease subtype. 2 Papules and pustules in rosacea have the characteristic centrofacial distribution and tend to wax and wane without scarring, emphasizing the value of maintenance therapy. 4 Current treatment for this subset of rosacea largely depends upon the clinical severity and varies from topical treatment such as ivermectin (1%), metronidazole (0.75%–1%), azelaic acid (15%), sodium sulfacetamide 10%, and sulfur 5%. 10 At present, the only FDA-approved systemic therapy for papulo-pustular rosacea is low-dose doxycycline. 11 Considering the high prevalence of the disease, its relapsing-remitting course, and the limited therapeutic armamentarium, it is highly desirable to find new therapies either through the development of new molecules or the identification of clinically effective off-label drugs.
Retinoids are vitamin A derivatives widely used for acne vulgaris and psoriasis and have been demonstrated to be a valid treatment option for various skin conditions like: photoaging, actinic keratosis, and ichthyoses, since a long time ago.12–14 The effectiveness of these compounds in treating rosacea is still up for debate, as several studies have reported mixed results. Even though some authors have shown positive outcomes, the use of retinoids in rosacea is restricted due to several concerns, mainly regarding local side effects and a lack of strong evidence from large-scale trials.12,15,16
The use of retinoids is largely considered a successful but still unapproved (off-label) treatment for rosacea.1,17,18 The evidence on topical retinoid therapy for rosacea is intriguing but limited, as there are fewer studies, which generally lack standardization regarding the vehicle, formulation, and therapeutic regimen. In this review, we analytically searched for any evidence in the literature regarding the efficacy of retinoids, either topical or systemic, for rosacea therapy, trying to shed some light on a relevant topic that probably deserves more scientific contribution so as to reach a stronger level of evidence.
Methods
Search strategy
A comprehensive literature search was carried out according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. 19 The MEDLINE (PubMed), EMBASE, and Cochrane Library electronic databases were systematically searched using the following combination of terms: “rosacea” and “retinoids” and “isotretinoin.” There was no limit to the search in terms of publication date, and the most recent search was run in January 2025. A manual search of reference lists was also performed.
Study selection
We included randomized controlled trials (RCTs), relevant open-label studies, retrospective, double-blind and placebo-controlled, randomized comparison trials conducted on patients with papulo-pustular rosacea treated with both topical and systemic retinoids. Exclusion criteria included editorial letters, case reports, reviews, meta-analysis, and non-English language studies.
Data extraction
Two authors (A.S., F.F.) independently reviewed studies for inclusion and exclusion criteria. The following data were extracted from each of the included studies: type of study, date of publication, topical/systemic/association therapy with retinoids, number of patients treated, and results of the studies. This study was designed to summarize the current evidence in the literature regarding the role of topical and systemic retinoids in the treatment of papulo-pustular rosacea.
Statistical analysis
We performed a proportional meta-analysis using MedCalc 14.8.1 software (MedCalc Software bvba, Ostend, Belgium; http://www.medcalc.org; 2014), applying the Freeman-Tukey transformation (square root arc-sine transformation) to calculate the weighted overall proportion. 20
The meta-analysis of mean differences was performed to compare the reduction in the number of lesions between the treatment and control groups. Standardized mean differences (SMD) were calculated for each study with corresponding 95% confidence intervals (CI). Both fixed-effects and random-effects models were used for pooled estimates. Heterogeneity across studies was assessed using Cochran’s Q test and the I2 statistic.
The proportions (expressed in percentage) with 95% CIs of the symptoms identified by each study were included in the meta-analysis.
The overall proportion with 95% CI was calculated using both the random-effects model and the fixed-effects model.
The fixed-effects model assumes that all included studies have a similar effect, so the summary effect is an estimate of the weight of similar effects in the studies.
The random-effects model assumes that effects vary among studies, and the summary analysis is a weighted average reported in different studies.
The forest plot is a graphical representation in which, for each study included in the meta-analysis, values related to the effect size and CI are presented.
The forest plot also includes the weighted effect size of prevalence of treatment patients for type of studies, with a 95% CI.
The size of the marker (square) represents the weight of each study; studies with a smaller patient sample will have less weight.
The overall effect is represented in the plot by a diamond: its width represents precision, and its position represents the estimate of effects.
Heterogeneity among studies based on various types of studies was estimated using Cochran’s Q statistic test and the I2 index. Heterogeneity was considered significant when p < 0.01 for the Q statistic (to assess whether observed variance exceeds expected variance).
For interpreting the I2 index of given heterogeneity (I2 = 100% x(Q−df)/Q), we have: I2 = 0%–25%, homogeneous; I2 = 25%–50%, moderate heterogeneity; I2 = 50%–75%, large heterogeneity; I2 = 75%–100%, extreme heterogeneity.
Results
Study findings: systemic treatment
Table 1 displays a summary of study findings. The use of retinoids as a potential treatment option for papulo-pustular rosacea has been demonstrated in two multicentric randomized double-blinded clinical trials.21,22
Analyzed studies.
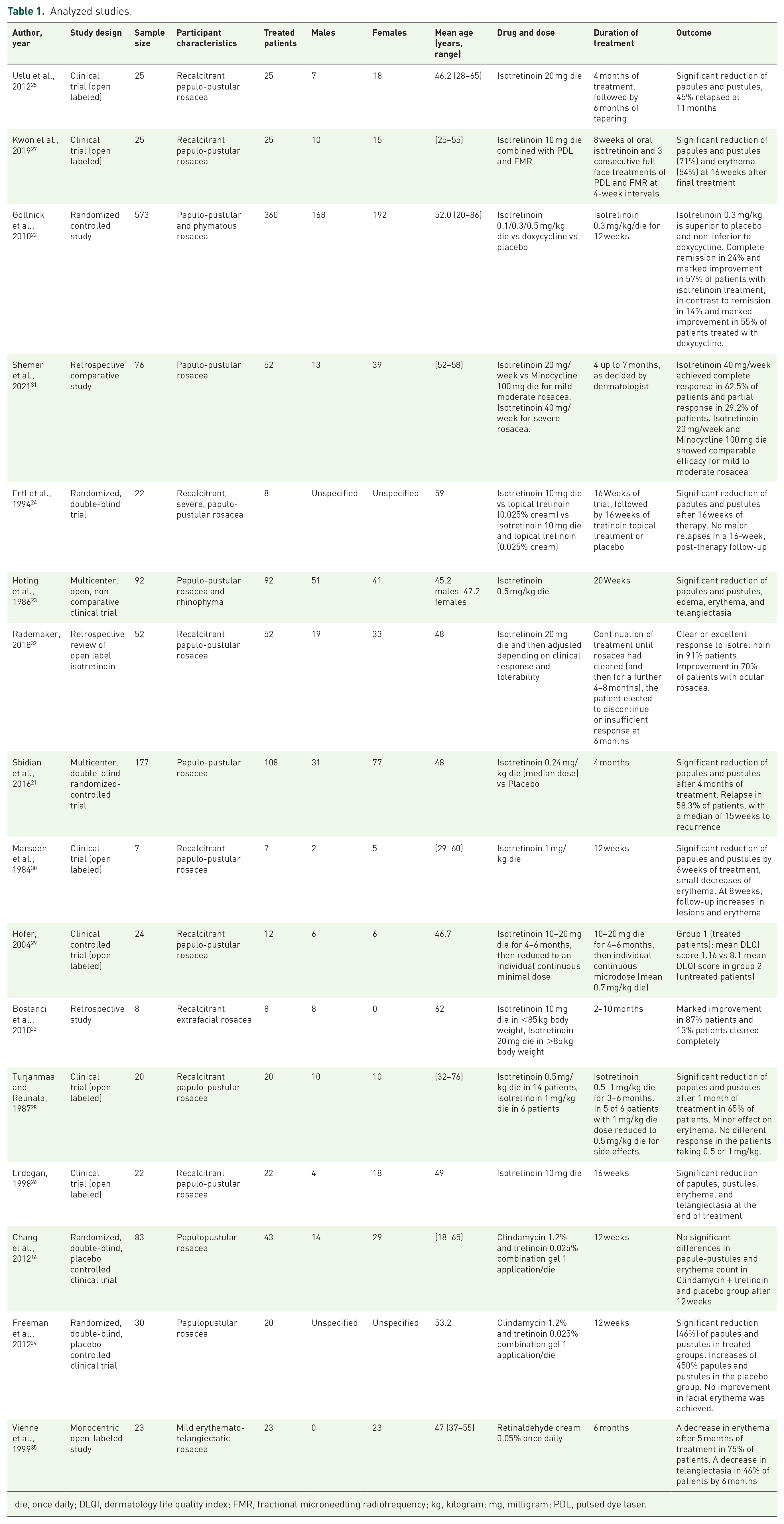
die, once daily; DLQI, dermatology life quality index; FMR, fractional microneedling radiofrequency; kg, kilogram; mg, milligram; PDL, pulsed dye laser.
In the first methodically valid, dose-finding, and efficacy study trial, Gollnick et al. showed that isotretinoin dosage of 0.3 mg/kg/day for 12 weeks is superior to placebo in reduction of number of papules and pustules in rosacea. Moreover, this isotretinoin dosage was not inferior to doxycycline treatment. 22
The second multicenter, placebo-controlled, randomized trial by Sbidian et al., including 177 patients with papulo-pustular rosacea, has shown significant reduction of papules and pustules after 4 months of treatment with oral isotretinoin 0.25 mg/kg/day. Rosacea relapsed in 58% of patients with a median of 15 weeks to recurrence. 21 This high relapse rate likely implies that longer therapeutic periods are necessary to more effectively and stably reverse the pathogenetic mechanisms of rosacea. A multicenter open-label noncomparative clinical trial including 92 patients with papulo-pustular rosacea and rhinophyma, has shown that isotretinoin 0.5 mg/kg/day for 20 weeks reduces papules, pustules, erythema, edema, and telangiectasia. 23
Ertl et al. randomized 22 subjects with recalcitrant severe papulo-pustular rosacea to oral isotretinoin 10 mg/day or topical tretinoin 0.025% once daily or isotretinoin 10 mg/day and topical tretinoin. After 16 weeks of therapy, all three groups showed a significant decrease over baseline in the number of papules and pustules. At week 4, maximal or near-maximal responses were noted in the isotretinoin group and in the isotretinoin-tretinoin combination group. Such improvement took instead 8 weeks in the tretinoin group, highlighting that oral isotretinoin may act faster than topical tretinoin. 24
Two open labeled clinical studies showed the efficacy of oral isotretinoin in the treatment of recalcitrant papulo-pustular rosacea with a daily dose of 10 mg/day for 16 weeks and 20 mg/day for 4 months followed by 6 months of tapering, respectively.25,26
In another study by Kwon et al., 27 a low-dose isotretinoin treatment (10 mg/day for 8 weeks) was combined with Pulsed Dye Laser (PDL) and Fractional Microneedling Radiofrequency (FMR) with a significant reduction of papules and pustules.
Furthermore, Turjanmaa and Reunala conducted an open labeled clinical trial including 20 patients with recalcitrant rosacea, of whom 14 underwent isotretinoin treatment 0.5 mg/kg/day and 6 on 1 mg/kg/day. Both groups showed significant reduction in papules and pustules, minor improvement in erythema. Interestingly, no difference in response to 1 mg/kg/die or 0.5 mg/kg/day isotretinoin treatment was noted. 28
Patient-reported outcome is a fundamental parameter when dealing with rosacea. Hofer conducted a clinical open-labeled study comparing dermatology life quality index (DLQI) in two groups of patients with recalcitrant papulo-pustular rosacea. Twelve patients were treated with oral isotretinoin 10–20 mg/day for 4–6 months, then reduced to an individual continuous microdose (mean 0.7 mg/kg/day) and were compared to a placebo group made up of 12 patients. The treated group presented a DLQI 1.6 compared to 8.3 of the placebo group. 29
Marsden et al. 30 reported a significant reduction of papules and pustules in seven patients treated with oral isotretinoin 1 mg/kg/day for 12 weeks, with a clinical improvement that was already evident in the first 6 weeks of treatment.
Shemer et al. demonstrated in a retrospective comparative study on 76 patients that the use of a weekly low-dose isotretinoin (20 mg weekly) is an effective treatment for papulopustular rosacea, comparable to Minocycline 100 mg/day in patients with mild to moderate rosacea. Moreover, a weekly dose of 40 mg for severe rosacea achieved a complete or partial response in almost all treated patients. 31
In another retrospective study, Rademaker reported a positive effect of oral isotretinoin in a dose of 20 mg administered daily until clinical improvement. In this study, the daily dose of isotretinoin was adjusted according to clinical response and tolerability. 32
Bostanci et al. performed a retrospective study on eight patients who suffered from recalcitrant extrafacial rosacea using isotretinoin 10 mg/day in patients with <80 kg body weight and 20 mg/day in patients with >80 kg body weight, with a duration of treatment ranging from 2 to 10 months. Both treatments resulted in marked improvement of rosacea in 87% of patients and in complete improvement in 13% of patients. 33
Study findings: topical treatment
Three studies focused on the use of topical retinoids in patients with rosacea, as shown in Table 1. Chang et al. conducted a randomized, double-blind, placebo-controlled trial on 83 patients with papulo-pustular rosacea treated daily with topical clindamycin 1.2% in combination with topical tretinoin 0.025% compared to a placebo group. This study showed that after 12 weeks of treatment, there was no difference in papules and pustules reduction between the treatment group and placebo group. 16
On the other hand, a randomized, double-blind trial conducted on 30 patients described a significant improvement in patients with papulo-pustular rosacea treated with topical clindamycin 1.2% in combination with topical tretinoin 0.025% compared to a placebo group. 34
Another multicentric open-labeled study included 23 patients with mild erythemato-telangiectatic rosacea. Patients were treated with topical retinaldehyde cream 0.05% once daily for 6 months and achieved a decrease of erythema and telangiectasia after 5–6 months. 35
Study findings: Meta-analysis
Table 1 shows the detailed characteristics of the eligible studies. Sixteen studies, involving 1259 patients and 877 with treatment, were assessed for the proportional meta-analysis.
The majority of patients were female 506 (59.6%) and the mean age was 50.6, 2 studies did not specify the gender and 4 the mean age.
The results of the proportional meta-analysis, including the combined proportion (95% CI) are summarized in Supplemental Table 3, with estimates of the overall proportion shown in the Forest Plot (Figure 1). The overall response rate to treatment was 69.73% (95% CI: 65.72%–73.52%) with a fixed-effects model and 70.19% (95% CI: 58.16%–80.94%) with a random-effects model. Heterogeneity among studies was significant (I2 = 87.05%). For all types of treatment, the meta-analysis showed an SMD of −3.179 (95% CI: −3.595 to −2.762, p < 0.001) with a fixed-effects model and −8.231 (95% CI: −15.032 to −1.430, p = 0.018) with a random-effects model. Heterogeneity among studies was high (I2 = 99.36%). For systemic treatments only, the SMD was −7.798 (95% CI: −8.593 to −7.002, p < 0.001) with a fixed-effects model and −10.532 (95% CI: −20.589 to −0.476, p = 0.040) with a random-effects model (I2 = 99.32%; Figure 2). The high heterogeneity among studies could influence the reliability and generalizability of the results. Overall, the results indicate that systemic retinoids, particularly isotretinoin, represent a promising therapeutic option for papulopustular rosacea, with a favorable safety and tolerability profile. However, data on the effects of topical retinoids are less conclusive and show conflicting results.

Proportional meta-analysis of included studies. Aggregate percentage of patients with a response rate to treatment. Markers represent grouped effects. The position of the diamond represents the estimated effect size, and the width of the diamond reflects the precision of the estimate.
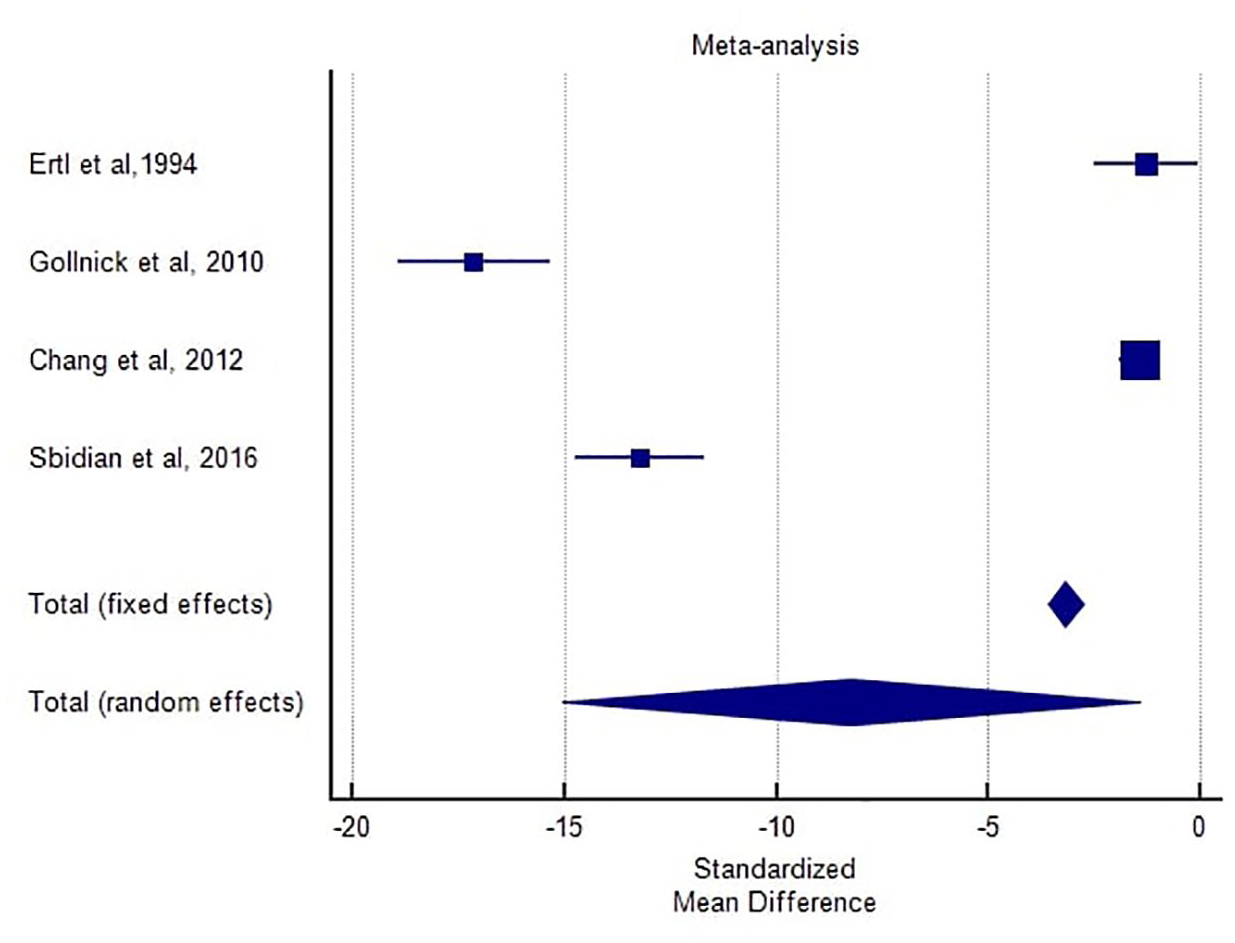
Forest plot comparing the reduction in lesion count in patients receiving isotretinoin monotherapy compared to topical monotherapy.
Search results
Figure 3 shows the PRISMA flow diagram for the systematic review detailing the studies that met inclusion and exclusion criteria. A total number of 226 articles was initially yielded. Following title/abstract screening and exclusion of case reports, case series, reviews, guidelines, and non-English studies, 91 articles were selected for eligibility. In the second round of screening, the reviewers assessed for eligibility those studies based on the full text and predefined inclusion criteria. Finally, 16 articles were selected for the final review. Thirteen of them regarded systemic treatment, and 4 considered topical treatment for rosacea. One article focused on both topical and systemic treatment for rosacea.

Workflow diagram describing the systematic selection of studies for inclusion in the present review (PRISMA flow chart).
Risk of bias of included studies
We assessed bias in our included studies using RoB2 and ROBINS-1, widely accepted tools for evaluating methodological quality and bias risk in systematic reviews. These tools cover crucial domains like participant selection, group comparability, and exposure/outcome ascertainment. Our team independently conducted bias assessments for each study, resolving discrepancies through consensus (Supplemental Tables 1 and 2). We generally observed a low risk of bias in randomized trials, with only one out of five studies showing potential deviations from intended interventions. However, a higher risk of bias was documented for non-randomized trials.
Discussion
The findings from this systematic review indicate that retinoids, particularly isotretinoin, show promise as a treatment option for papulopustular rosacea, especially in cases refractory to conventional therapies.
This review examined 16 studies involving 877 participants, of whom 506 (57.70%) were female, as shown in Table 1. Of all the patients, 791 were treated with oral isotretinoin at different dosages. The average daily dosage was 20 mg, and the average medication duration was 12 weeks, ranging from 8 weeks to 10 months.
Due to the heterogeneity in study design and outcomes reporting, the standardization of our results is limited, but we can conclude that most patients with rosacea showed a clinical response to oral isotretinoin at variable dosages, resulting in a drastic reduction in papules and pustules count.
In the analyzed studies tolerability of isotretinoin was good, and severe side effects were uncommon. In one of the largest studies we considered, Gollnick et al. showed that low-dose (0.3 mg/kg/day) isotretinoin in the treatment of rosacea has a similar safety profile as when it is used to treat acne. Therefore, routine laboratory monitoring, which comprises lipids and liver function tests, and the adoption of an adequate pregnancy prevention program for fertile female patients, are recommended.22,36 The majority of patients experienced xerosis of the skin (31%), cheilitis (57%), and dry eyes (14%) and transient dyslipidemia (11%) that are well-known side effects of retinoids therapy. Eye drops and calming lotions were sufficient to effectively control the side effects specific to retinoid therapy. Ocular side effects were usually nonsevere, and they did not cause therapy discontinuation. Interestingly, none of the analyzed studies reported a significant worsening or an exacerbation of concurrent ocular rosacea manifestation during isotretinoin therapy. In fact, regarding ocular rosacea symptoms, Rademaker 32 reported that, when rosacea associated with xerophthalmia was present, it improved significantly during low-dose isotretinoin therapy.
The available data for topical retinoid therapy for rosacea are less definitive and more contradictory, with only two randomized studies reporting opposite outcomes. Topical tretinoin 0.025% combined with clindamycin 1.2% turned out to be effective in the study by Freeman conducted on 30 patients, but according to the study by Chang et al., conducted on 83 patients, the same drug was shown to be non-superior to placebo. Apart from sample size, there wasn’t a significant difference between the study designs.
Multiple studies have demonstrated that both low-dose and conventional-dose isotretinoin can significantly reduce the number of papules and pustules, with some studies suggesting that isotretinoin can be as effective as doxycycline, the currently FDA-approved systemic therapy for rosacea.
The mechanism of action of isotretinoin in treating rosacea appears to be multifaceted. Its ability to modulate gene expression, reduce sebaceous gland size, and exert anti-inflammatory effects is crucial in addressing the pathophysiological aspects of rosacea. The reduction in TLR2 signaling and increased expression of cathelicidin suggest a positive impact on the immune dysregulation associated with rosacea.
Despite these promising results, the use of isotretinoin in rosacea remains off-label, and its application is often limited by concerns over side effects. The studies reviewed highlight the importance of tailoring the dose to minimize adverse effects while maintaining therapeutic efficacy. Common side effects of isotretinoin, such as dry skin, cheilitis, and potential teratogenicity, necessitate careful patient selection and monitoring during treatment.
Topical retinoids also demonstrate efficacy in reducing the symptoms of rosacea. Studies indicate that topical tretinoin can be effective, particularly when combined with oral isotretinoin. However, the onset of action for topical retinoids may be slower compared to oral isotretinoin, requiring a longer duration of treatment to achieve maximal effects.
The recurrence of rosacea symptoms after discontinuation of isotretinoin, as reported in several studies, suggests the need for maintenance therapy. The potential for combining retinoids with other treatment modalities, such as PDL and FMR, offers a promising avenue for enhancing treatment outcomes and reducing relapse rates.
Conclusion
In conclusion, retinoids, especially isotretinoin, represent a viable treatment option for papulopustular rosacea, with the potential to provide significant improvement in symptoms. Further large-scale, RCTs are needed to establish standardized dosing regimens and long-term safety profiles. The integration of retinoids into the therapeutic arsenal for rosacea should be considered, particularly for patients who do not respond adequately to existing treatments.
Supplemental Material
sj-docx-1-taj-10.1177_20406223251339964 – Supplemental material for Rosacea and treatment with retinoids: a systematic review and meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223251339964 for Rosacea and treatment with retinoids: a systematic review and meta-analysis by Alberto Sticchi, Flavio Fiorito, Shaniko Kaleci, Alessia Paganelli, Marco Manfredini and Caterina Longo in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223251339964 – Supplemental material for Rosacea and treatment with retinoids: a systematic review and meta-analysis
Supplemental material, sj-docx-2-taj-10.1177_20406223251339964 for Rosacea and treatment with retinoids: a systematic review and meta-analysis by Alberto Sticchi, Flavio Fiorito, Shaniko Kaleci, Alessia Paganelli, Marco Manfredini and Caterina Longo in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-3-taj-10.1177_20406223251339964 – Supplemental material for Rosacea and treatment with retinoids: a systematic review and meta-analysis
Supplemental material, sj-docx-3-taj-10.1177_20406223251339964 for Rosacea and treatment with retinoids: a systematic review and meta-analysis by Alberto Sticchi, Flavio Fiorito, Shaniko Kaleci, Alessia Paganelli, Marco Manfredini and Caterina Longo in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-4-taj-10.1177_20406223251339964 – Supplemental material for Rosacea and treatment with retinoids: a systematic review and meta-analysis
Supplemental material, sj-docx-4-taj-10.1177_20406223251339964 for Rosacea and treatment with retinoids: a systematic review and meta-analysis by Alberto Sticchi, Flavio Fiorito, Shaniko Kaleci, Alessia Paganelli, Marco Manfredini and Caterina Longo in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
