Abstract
Background:
Klotho is a kidney-derived protein that is involved in various kidney diseases. The role of serum soluble Klotho (sKlotho) in the anemia of patients undergoing hemodialysis has not been well characterized.
Objective:
We aimed to characterize the relationship between sKlotho and hemoglobin (Hb) levels in this group of patients.
Design:
A single-center cross-sectional study of 208 patients undergoing maintenance hemodialysis (MHD) and 50 healthy controls was performed between June 1 and 31, 2023.
Methods:
Demographic information and biomedical parameters, such as age, body mass index, medication use, and their Hb, albumin, interleukin-6, and sKlotho concentrations, were obtained. Patients undergoing MHD were allocated to a group that achieved the Hb target (⩾110 g/L) and a group that did not (<110 g/L). Correlation analysis and multivariate logistic and linear regression analyses were performed to evaluate the relationship of sKlotho with Hb concentration.
Results:
Participants undergoing MHD had lower Hb and sKlotho concentrations than controls. Those who had not achieved the target Hb level were given fewer erythropoiesis-stimulating agents and had lower sKlotho and albumin concentrations, but higher interleukin-6 concentrations, than those who had achieved the Hb target. The sKlotho concentration positively correlated with the Hb concentration and was inversely associated with the incidence of a lack of achievement of the target Hb level. Multivariate logistic regression models revealed that there was a close association between sKlotho and a lack of achievement of the target Hb level after adjustment for potential confounders (odds ratio: 0.335, 95% confidence interval: 0.142–0.791, p = 0.013). This relationship was closer on multivariate linear regression analysis when sKlotho was included as a continuous variable.
Conclusion:
The circulating sKlotho concentration is very low, but deficiency of this protein is independently associated with a high risk of anemia in patients undergoing MHD. Therefore, the routine monitoring of sKlotho concentration might be useful in the management of renal anemia in such patients.
Plain language summary
Anemia is a common and serious problem associated with long-term kidney disease. The identification of specific methods of monitoring and treating anemia associated with kidney disease is important. Klotho is a protein from the kidney that may be involved in the development of anemia. However, there have been few studies regarding the relationship of sKlotho with anemia in patients with such kidney disease, and especially those undergoing blood filtration (hemodialysis). We aimed to characterize the relationship between sKlotho and anemia in this group of patients. We studied 208 patients undergoing hemodialysis and 50 healthy people. The average hemoglobin (Hb) concentration was 22.9% lower and the average sKlotho concentration was 33.4% lower in the patients undergoing hemodialysis than in the healthy people. Furthermore, the Hb concentration was 23.3% lower and the sKlotho concentration was 15.7% lower in the participants with a Hb concentration <110 g/L than in those with a higher concentration. In addition, the Hb and sKlotho concentrations were closely related, and there was a close relationship between sKlotho and anemia. This suggests that the routine measurement of blood sKlotho concentration might help doctors treat anemia in patients with long-term kidney disease.
Introduction
Anemia is a common and critical complication of chronic kidney disease (CKD) and its incidence increases as CKD progresses, 1 such that it affects almost all patients with advanced CKD and those who are undergoing dialysis.1,2 The presence and severity of anemia have significant effects on clinical outcomes, including the progression of CKD, 3 cardiovascular events, 4 and all-cause mortality. 5 Therefore, a deeper understanding of the mechanism underlying renal anemia is crucial to facilitate the management of anemia and the screening for potential therapeutic targets. It has been demonstrated that multiple factors are implicated in renal anemia, including absolute or relative deficiency of erythropoietin (EPO), iron status, hematopoietic raw materials, inflammation, and oxidative stress.6,7 Emerging evidence suggests that abnormal Klotho expression is also associated with the development of renal anemia, and this molecule has been proposed to be a novel player in patients with anemia.8,9
Klotho was first identified as a longevity protein in 1997 10 and encodes three products: α-Klotho, β-Klotho, and γ-Klotho. 11 β-Klotho is a transmembrane protein that is primarily expressed in the kidney, 12 is an obligate subunit of fibroblast growth factor 23 (FGF-23), and promotes phosphate excretion together with FGF-23. 13 Soluble Klotho (sKlotho) is obtained by cleavage of the membrane form of α-Klotho and has endocrine effects on multiple organs. 14 It has been well documented that the sKlotho concentration gradually decreases with the progression of CKD15–17 and is associated with poor kidney outcomes.16,18,19 As a result of this, sKlotho has been proposed to be useful for the diagnosis of CKD and the prognostic assessment of patients. 20
Klotho deficiency and anemia are characteristic features of CKD, and Klotho knockdown in mice results in abnormalities in hematopoiesis, suggesting that Klotho plays a role in the development of hematopoiesis and anemia. 21 Several recent studies have investigated the relationship between sKlotho and anemia in patients with CKD, but have yielded inconsistent results.22–24 Thus, the role of sKlotho in the anemia associated with CKD is uncertain. Therefore, in the present study, we aimed to characterize the relationship of sKlotho concentration with the hemoglobin (Hb) concentrations of patients undergoing maintenance hemodialysis (MHD).
Methodology
Study sample
The study was approved by the Medical Ethics Committee of the Affiliated Kunshan Hospital of Jiangsu University (approval number: 2023-03-018-K01) and was conducted in accordance with the principles of the Declaration of Helsinki. Informed written consent was obtained from the participants. The study is reported according to the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material). 25 A single-center study was performed at the Blood Purification Center of Kunshan First People’s Hospital. A total of 208 patients who were undergoing MHD were enrolled after 249 individuals were screened between June 1 and 31, 2023. A total of 50 healthy volunteers were also recruited as controls. Participants aged >18 years who had been undergoing dialysis for >3 months were initially regarded as eligible. Participants who had hematologic disorders, active malignancy, hemorrhage, or acute inflammation; those who had undergone major surgical procedures or blood transfusions; and those who died within 3 months were excluded.
Study design
We performed an observational, case–control study. During enrollment, demographic and clinical information, including age, sex, body mass index (BMI), medication history, duration of dialysis, dry mass, and comorbidities, was collected. The use of medication for the treatment of renal anemia, including EPO, erythropoiesis-stimulating agents (ESAs), and iron supplements, was also recorded. Owing to China’s reimbursement policies and for economic reasons, renal anemia was treated using ESAs in the vast majority of the patient samples. The concentrations of circulating substances, including Hb, glucose, albumin (Alb), urea nitrogen, creatinine (Cr), calcium (Ca), phosphorus (P), intact parathormone (iPTH), ferritin, and interleukin-6 (IL-6), were measured using standard laboratory techniques, and the urea clearance index (Kt/V) was calculated. sKlotho concentrations were measured using a commercial human sKlotho ELISA kit (Immuno-Biological Laboratories, Gunma, Japan), according to the manufacturer’s instructions. Venous blood samples were collected after 12 h of fasting and prior to dialysis. All the participants were administered ESAs, iron supplements, or other agents regularly on the basis of their Hb concentrations, and ferritin. Demographic information was initially collected, and biochemical datasets, including for the sKlotho concentration, were compared between the participants undergoing MHD and the control participants. Those undergoing MHD were then allocated to two groups on the basis of their Hb concentrations: those who achieved an Hb concentration of ⩾110 g/L and those who did not. The sKlotho concentrations and other parameters were subsequently measured and compared between these two groups.
Statistical analysis
The baseline characteristics of the participants are presented as descriptive statistics. Categorical data are expressed as frequencies and percentages, and comparisons were made using the Chi-square test. Continuous datasets are summarized according to whether or not they were normally distributed: normally distributed data are presented as mean ± standard deviation (SD) and were compared using Student’s t-test, and non-normally distributed data are presented as median (interquartile range) and were compared using the Mann–Whitney U-test. Non-normally distributed data were ln-transformed for subsequent correlation analysis. Spearman or Pearson correlation testing was employed to evaluate the relationships between sKlotho concentration, other parameters, and Hb concentration. In addition, multivariate logistic regression and linear regression models were used to evaluate the relationship between the sKlotho and Hb concentrations. Statistical analyses were performed using SPSS v.23.0 (IBM, Inc., Armonk, NY, USA), with p < 0.05 being considered to indicate statistical significance.
Results
Characteristics of the participants
A total of 208 participants undergoing MHD and 50 healthy controls, who were matched for age and sex, were considered to be eligible and enrolled in the study. The two groups contained participants with comparable age, sex distribution, and BMI. Compared with the controls, the participants undergoing MHD had lower Hb, Alb, and sKlotho concentrations, and higher urea, Cr, and IL-6 concentrations. Specifically, the mean Hb concentration was 22.9% lower and the mean sKlotho concentration was 33.4% lower in the MHD group. A flowchart of the study is shown in Figure 1 and the characteristics of the participants are presented in Table 1.

Flow chart of participant recruitment and exclusion.
Comparison of the biochemical data between the control and MHD groups at baseline.

Data are presented as mean ± SD for normally distributed data and median (25th, 75th percentile) for non-normally-distributed data.
Alb, albumin; BMI, body mass index; Bun, blood urea nitrogen; Ca, calcium; Cr, creatine; Hb, hemoglobin; IL-6, interleukin-6; MHD, maintenance hemodialysis; P, phosphorus.
Comparison of the participants who did or did not achieve the target Hb concentration
Of the 208 participants undergoing MHD, 60.1% had not achieved the target Hb concentration, according to the definition stated above. Those with a low Hb concentration had significantly lower Alb, Ca, P, and sKlotho concentrations and calcium–phosphorus product (Ca × P), were less likely to be male, were less likely to be administering an ESA, and had a higher IL-6 concentration. Specifically, the Hb concentration was 23.3% lower and the sKlotho concentration was 15.7% lower in the participants who had not achieved the Hb target than in those who had. There were no significant differences in the age, BMI, duration of dialysis, iPTH or ferritin concentration, Kt/V, or the prevalence of comorbidities, such as hypertension and diabetes, between the groups (Table 2).
Comparison of biochemical and demographic data for patients with and without anemia.

Data are presented as mean ± SD for normally distributed data and median (25th, 75th percentile) for non-normally distributed data.
Alb, albumin; BMI, body mass index; Bun, blood urea nitrogen; Ca, calcium; Cr, creatine; ESA, erythropoiesis-stimulating agents; Hb, hemoglobin; HIF-PHI, hypoxia-inducible factor prolyl hydroxylase inhibitor; IL-6, interleukin-6; iPTH, intact parathyroid hormone; Kt/V, urea clearance index; MHD, maintenance hemodialysis; P, phosphorus.
Correlations of clinical indices with the Hb concentration
Correlation analysis revealed positive associations of the Hb concentration with the male sex, ESA use, Alb concentration, Ca × P, and the Ln sKlotho concentration (Figure 2 and Table 3); and a significant negative association with the Ln IL-6 concentration (Figure 3 and Table 3). There were no associations of age, BMI, diabetes, hypertension, duration of dialysis, dry mass, Kt/V, Ln iPTH, Ln ferritin, or P with the Hb concentration (Table 3).

Correlation of the Hb and sKlotho concentrations.
Correlations of Hb with clinical and biochemical parameters.

Alb, albumin; BMI, body mass index; Ca, calcium; ESA, erythropoiesis-stimulating agents; Hb, hemoglobin; IL-6, interleukin-6; iPTH, intact parathyroid hormone; Kt/V, urea clearance index; P, phosphorus.

Correlation of the Hb and IL-6 concentrations.
The relationship between the sKlotho and Hb concentrations, characterized using regression analysis
The participants undergoing MHD were allocated to groups on the basis of whether their Hb concentrations were ⩾110 or <110 g/L (those who had achieved the target concentration and those who had not, respectively). Univariate logistic regression analysis revealed that the sex distribution, the use of ESA, Alb concentration, Ln IL-6 concentration, Ca × P, and Ln sKlotho concentration were associated with a lack of achievement of the target Hb concentration when Hb was used as a categorical variable, as shown in Table 4. On multivariate analysis, Ln sKlotho concentration was found to be independently associated with a lack of achievement of the target Hb concentration (odds ratio (OR): 0.343, 95% confidence interval (CI): 0.146–0.804, p = 0.014) in Model 1, after adjustment for sex distribution, ESA use, Alb concentration, and Ca × P. This close association was also demonstrated in Model 2 (OR: 0.335, 95% CI: 0.142–0.791, p = 0.013) after adjustment for other potential confounding factors (the variables adjusted for in Model 1, plus age and Kt/V), as shown in Table 5. Moreover, these relationships were also identified using multivariate linear regression (standardized b coefficient: 0.155, 95% CI: 1.450–13.348, p = 0.015), with Hb as a continuous variable (Table 6).
Results of the univariate logistic regression analysis to identify factors associated with the incidence of a lack of achievement of the target Hb concentration.
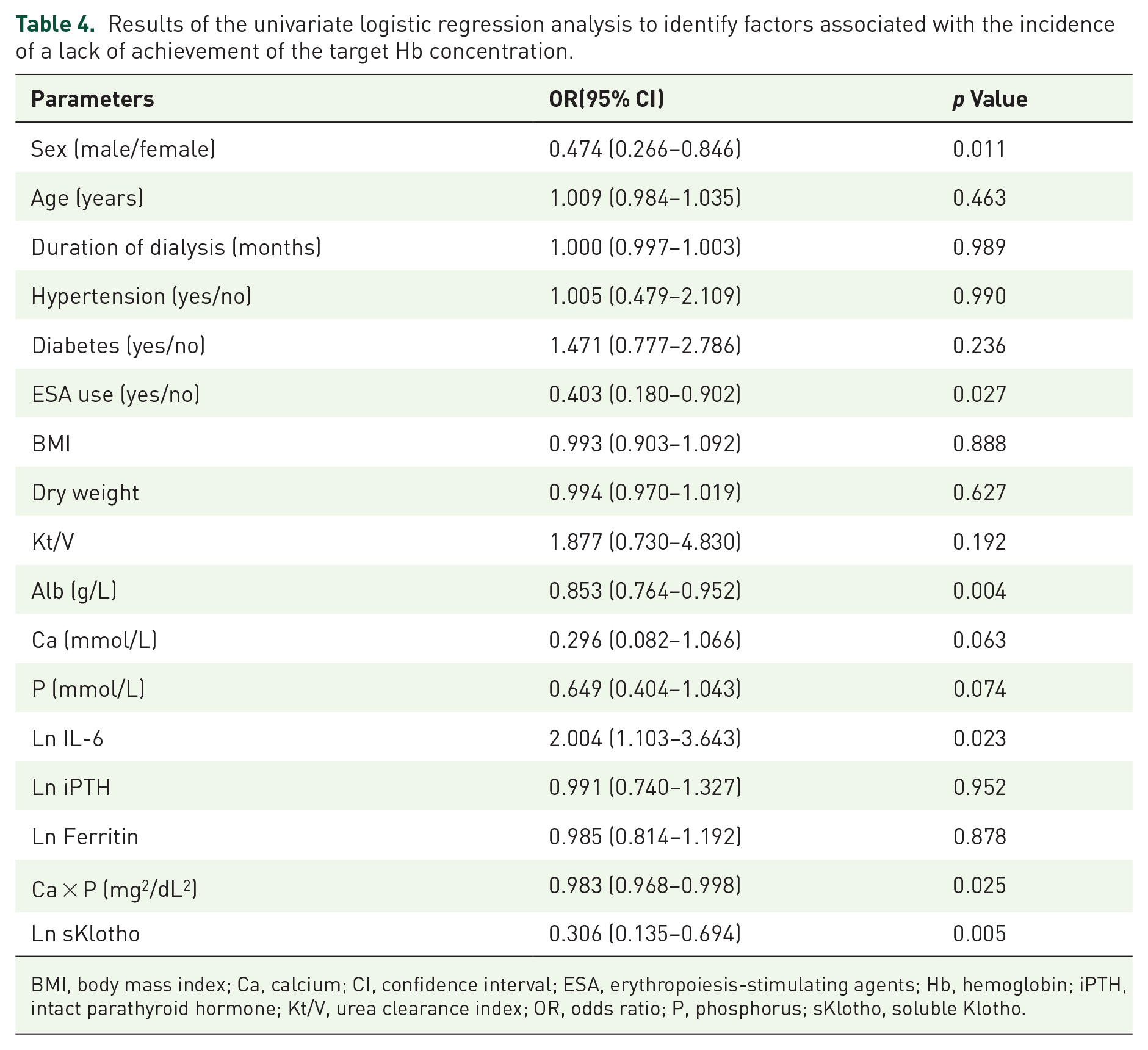
BMI, body mass index; Ca, calcium; CI, confidence interval; ESA, erythropoiesis-stimulating agents; Hb, hemoglobin; iPTH, intact parathyroid hormone; Kt/V, urea clearance index; OR, odds ratio; P, phosphorus; sKlotho, soluble Klotho.
Results of the multivariable logistic regression analysis to identify factors associated with the incidence of a lack of achievement of the target Hb concentration.

Model 1 was adjusted for sex, ESA use, Alb, Ca × P, Ln IL-6, and Ln sKlotho; Model 2 was adjusted for the parameters in Model 1 + age and Kt/V.
Alb, albumin; Ca, calcium; CI, confidence interval; ESA, erythropoiesis-stimulating agents; Hb, hemoglobin; IL-6, interleukin-6; Kt/V, urea clearance index; OR, odds ratio; P, phosphorus; sKlotho, soluble Klotho.
Results of the multivariable linear regression analysis to identify factors associated with the Hb concentration.

The model was adjusted for sex, age, Kt/V, Alb, ESA use, Ca × P, Ln IL-6, and Ln sKlotho.
Alb, albumin; Ca, calcium; CI, confidence interval; ESA, erythropoiesis-stimulating agents; Hb, hemoglobin; IL-6, interleukin-6; Kt/V, urea clearance index; P, phosphorus; sKlotho, soluble Klotho.
Discussion
In the present study, we have evaluated the relationship between the serum sKlotho concentration and Hb in patients undergoing MHD. We have shown that the sKlotho concentration is very low in such patients, and moreover, that there is an inverse association between the two, irrespective of whether Hb is regarded as a categorical or continuous variable, and after adjustment for a number of potential confounders. Thus, a low sKlotho concentration is independently associated with a higher risk of not achieving the target Hb concentration in patients undergoing MHD, even when low ESA usage is accounted for.
Anemia is a frequent complication of CKD, and the prevalence of anemia is extremely high in patients with advanced CKD, whether or not they are undergoing dialysis.26–28 Numerous clinical studies have demonstrated that anemia is associated with adverse renal and nonrenal outcomes, such as a rapid decline in kidney function, a high risk of hospitalization, poor quality of life, and cardiovascular events.4,29 Therefore, maintaining or increasing the Hb concentration to the target (110–130 g/L) is an important component of the management of renal anemia. Conventionally, oral or intravenous iron, EPO, and ESAs are used to increase the Hb concentration as part of the standard treatment for renal anemia. 30 In recent years, hypoxia-inducible factor prolyl hydroxylase inhibitors have been approved for the treatment of renal anemia, owing to their ability to promote EPO production, inhibit inflammation, reduce oxidative stress, and regulate iron metabolism, leading to some amelioration of renal anemia.31–33 Despite the optimization of these strategies, the prevalence of anemia remains high and many patients with CKD fail to achieve their Hb goal for various reasons.27,34 This presents a considerable challenge in the treatment of renal anemia and reflects the complexity of the etiology and pathogenesis of renal anemia. 6 Therefore, it is important to identify novel targets for the management or treatment of renal anemia. In addition to the well-characterized etiologic factors, including iron deficiency, low EPO production, malnutrition, inflammation, and high hepcidin concentration, 6 recent studies have shown that other factors may also contribute to renal anemia.34–36 In particular, Klotho is increasingly considered to be involved in the etiology of renal anemia.9,23
Growing evidence is demonstrating that sKlotho is not only a biomarker but also involved in the progression of CKD and the development of related complications. Although sKlotho was first identified more than two decades ago, the effect of this protein on erythropoiesis is not fully understood. A recent study of 13,357 middle-aged and older adults with normal kidney function that evaluated the relationship between sKlotho and anemia showed that the incidence of anemia decreased by 10.9% for each 100-pg/mL increase in its concentration and that this relationship was not affected by kidney function. 23 In this study, the authors provided solid evidence regarding the association of sKlotho with anemia in the general population. sKlotho deficiency and anemia are common features of CKD, and the correction of anemia is followed by the restoration of sKlotho concentrations, 24 indicating that there might be a relationship between sKlotho and anemia in patients with CKD.
Milovanov et al. 37 previously investigated the risk factors for anemia in 79 patients with CKD stages I or II, and found that patients with anemia had lower sKlotho concentrations and that changes in sKlotho paralleled the changes in Hb concentration. Thus, a low sKlotho concentration was hypothesized to be a risk factor for, or a marker of, renal anemia in early CKD. 37 The same authors subsequently performed a 1-year longitudinal study to evaluate the relationship between the 2 in 64 patients with CKD stages 3B–4 and found that when anemia is ameliorated by EPO treatment, there is a marked increase in sKlotho concentration. 24 Another recent study of 100 healthy individuals and 239 patients with CKD that was performed to characterize the relationship between changes in sKlotho concentration and anemia 8 showed that a low sKlotho concentration is associated with the severity of renal anemia using multivariate models and after adjustment for potential confounders. Importantly, the amelioration of anemia through the treatment of bone metabolic disorders has been shown to be associated with an increase in sKlotho concentration. 8 These findings show that the sKlotho concentration is associated with the incidence and severity of anemia in patients with CKD. Therefore, sKlotho is a risk factor for, and represents a candidate biomarker for use in the diagnosis and monitoring of, renal anemia.
Despite these findings, studies of the association of sKlotho with anemia in patients with CKD who are undergoing MHD have yielded conflicting results. Yu et al. 38 previously conducted a cross-sectional study of 330 patients undergoing MHD to interrogate a possible link between sKlotho and atherosclerosis. They found positive correlations of sKlotho concentration with Hb, Ca × P, and the quantity of carotid atherosclerotic plaque. However, another recent 1-year longitudinal study of 91 patients undergoing MHD that was performed by Topal and Guney failed to confirm this association. They found that the sKlotho concentration did not correlate with the mean Hb concentration or the rate of change of Hb on multivariate linear regression analysis. 22 The lack of consistency in these findings has left uncertainty regarding the relationship between sKlotho and anemia in patients undergoing MHD, and therefore further studies are necessary. In the present study, we evaluated the relationship between sKlotho and Hb concentration in such patients. We found that the sKlotho concentration was 33.5% lower in patients undergoing MHD than in controls, and it was also a further 15.7% lower in those who had not achieved the Hb target than in those who had. This significant relationship of sKlotho with Hb was identified using both univariate and multivariate regression analysis. This implies that sKlotho may be a useful biomarker to assist with the management of renal anemia in patients undergoing MHD. For instance, measurement of the sKlotho concentration could help to identify patients undergoing MHD who are at risk of anemia and those who may benefit from specific treatments. Moreover, tracking the trend in sKlotho concentration over time could help assess the effectiveness of ongoing treatment strategies for renal anemia.
The mechanism underlying these associations may be multifaceted. First, Klotho has a direct effect on EPO production. Klotho ablation in mice results in increases in Hb concentration and red blood cell count, and these effects partially depend on an increase in EPO synthesis, secondary to the activation of hypoxia-inducible factors. 21 Intriguingly, EPO has also been reported to prevent a decrease in Klotho concentration by inhibiting nuclear factor-kB production in the presence of uremia. 39 This may imply that a high Klotho concentration is a compensatory response that aims to ameliorate the effects of renal anemia, suggesting that a negative feedback mechanism may be responsible for the interaction between Klotho and EPO. 9 Second, iron deficiency is a common cause of renal anemia in patients with CKD, and Klotho appears to play a role in iron metabolism. Iron-dextran administration reduces renal Klotho expression, and iron chelation attenuates the reduction in Klotho that is induced by angiotensin II in the kidney. 40 Consistent with this, a clinical study of 70 patients at various stages of CKD revealed close correlations of sKlotho with ferritin and the transferrin saturation percentage. 41 These findings suggest that Klotho may have an effect on iron metabolism. However, we did not identify this relationship in the present study, at least in part because of the low ferritin concentrations and low doses of iron administered.
CKD is recognized to be an inflammatory condition,42,43 and the development of anemia is associated with an enhancement of the inflammatory response.44,45 In addition, Klotho has previously been shown to have an inhibitory effect on inflammation. 46 Consistent with this, we found that the circulating concentration of the proinflammatory cytokine IL-6 was high and inversely correlated with the Hb and sKlotho concentrations in patients with anemia. The sKlotho-induced amelioration of inflammation may also be associated with an amelioration of renal anemia. 9 Finally, Klotho also modulates oxidative stress, energy metabolism, and sex hormone concentrations,47–49 and therefore it may also have an indirect effect on anemia through these mechanisms. However, the evaluation of these possible mechanisms was beyond the scope of the present study. Taking these findings together, the role of Klotho in anemia is complex and has not been well characterized in the few studies conducted to date. Therefore, the exact mechanisms involved require further study.
The present study had several limitations that should be addressed. First, we acknowledge the difficulty in clarifying the causal relationships of EPO and Hb with sKlotho, or vice versa, owing to the limited number of studies that have been performed. For example, one previous study showed that Klotho ablation contributes to EPO production, 21 but consistent findings were not obtained in a more recent study. 50 Although an amelioration of anemia is accompanied by a restoration of sKlotho concentration, a low sKlotho concentration in patients with CKD may be ascribed to the influences of factors other than Hb. 51 Furthermore, the effects of sKlotho on EPO or Hb could be affected by the presence of other regulators. 51 This may be a possible explanation for the lack of consistency in previous findings.21,50 In fact, there may be a complex interaction between Klotho and EPO that requires further exploration in future research. Second, the study was cross-sectional in nature and longitudinal changes in the sKlotho and Hb concentrations were not assessed. Therefore, causal relationships between the variables of interest cannot be inferred. Third, it was a single-center study for which a sample size was not calculated a priori, which weakens its external validity. Finally, other metabolic parameters, such as FGF-23, active vitamin D, and indicators of redox status, were not measured, and therefore effects of these potential confounders on the relationship between sKlotho and anemia could not be excluded.
Conclusion
Taken together, the present findings indicate that a low sKlotho concentration is associated with a higher risk of not achieving an appropriate Hb concentration in patients undergoing MHD, even after adjustment for low ESA usage. This may imply that sKlotho may represent a useful biomarker for renal anemia, and the routine monitoring of its concentration may provide useful information regarding renal anemia in patients with MHD. Owing to the limitations of the present study, the complex relationship between sKlotho and Hb should be further characterized in future studies.
Supplemental Material
sj-docx-1-taj-10.1177_20406223251318481 – Supplemental material for Relationship of sKlotho with hemoglobin level in patients undergoing maintenance hemodialysis: a case–control study
Supplemental material, sj-docx-1-taj-10.1177_20406223251318481 for Relationship of sKlotho with hemoglobin level in patients undergoing maintenance hemodialysis: a case–control study by Qi-Feng Liu, Zhuo-Yi Sun, Xiao-Fang Tang, Li-Xia Yu and Sha-Sha Li in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
