Abstract
Background:
Severe upper extremity paresis due to stroke is a significant clinical sequela. Neuromuscular electrical stimulation (NMES)-based rehabilitation has demonstrated promising results along with cortical plasticity. Transcranial alternating current stimulation (tACS) has gained attention due to its unique ability to entrain endogenous oscillatory brain rhythms with injected AC frequency, offering the potential for modifying brain conditions to enhance rehabilitative interventions. Because repetitive motor execution in rehabilitation training requires a smooth transition of the brain state despite often being impaired secondary to stroke, combining NMES and tACS may offer better treatment efficacy.
Aim:
This study proposes a phase I/II trial of an outpatient comprehensive rehabilitative treatment combining the integrated volitional-control electrical stimulation (IVES), a closed-loop NMES, and the timing-specified focal tACS in individualized beta frequency (dynamic-precision tACS) targeting severe hand paresis in patients with chronic stroke, aiming to demonstrate the feasibility of combination treatment.
Design:
Double-blind randomized cross-over trial.
Methods:
The repetitive facilitative finger extension training utilizing closed-loop NMES is combined with dynamic-precision tACS on the primary motor cortex to assist post-movement beta-rebound. Together with regular occupational therapy, we propose a comprehensive outpatient neurorehabilitative regimen. Here, a total of 10 sessions will be conducted using a cross-over design using real and sham tACS.
Analysis:
The perception and fatigue from stimulation will be investigated as the primary outcomes. The efficacy of improving sensorimotor function and their background physiological mechanisms will be evaluated as the secondary outcomes.
Discussion:
This phase I/II trial will be the first to combine tACS and neurorehabilitation using functional electrical stimulation. A weekly outpatient protocol with cheap devices may offer a new treatment paradigm toward functional recovery for chronic stroke patients with severe upper extremity paresis.
Ethics and trial registration:
This study was approved by the Ethics Committee of Kyorin University Faculty of Medicine (814-01). The trial was registered in a public database: UMIN000048274.
Keywords
Background
Rehabilitation is essential in restoring function for individuals affected by neuromuscular diseases such as stroke and spinal cord injury. However, its effectiveness can be limited, prompting the exploration of various combination therapies known as neurorehabilitation, aimed at augmenting the effects of rehabilitation through advancements in brain science. Noninvasive transcranial brain stimulation (NTBS) is a promising approach that involves stimulating the brain cortex using methods like electric current, magnetism, and ultrasound without invasive procedures. NTBS has attracted wide attention as a noninvasive combinatorial measure for rehabilitation to modulate cortical function suited to motor execution and learning. 1 Two types of transcranial electric current stimulation (tCS), which have the advantages of simplicity and high practicality, are available: transcranial direct current stimulation (tDCS) with direct current and transcranial alternating current stimulation (tACS) with alternating current (AC). While tDCS has been widely studied for decades, its clinical application has been hindered by variability in efficacy. On the other hand, tACS investigations were started approximately 10 years ago. 2
The effects of tACS rely on a phenomenon called entrainment of brain oscillations. Brain oscillation, also known as neural oscillation, is a fundamental physiological activity of neuronal cells in the brain acquired through electroencephalography (EEG) or magnetoencephalography. 3 In the entrainment phenomenon, the brain’s endogenous rhythm synchronizes with the frequency of exogenous AC applied via the electrodes attached to the scalp. 4 Endogenous brain oscillatory activities are associated with ongoing various functions. 5 For instance, brain oscillations in the alpha band (8–13 Hz) have been linked to visuomotor integration and learning, 6 while beta-band rhythms (14–30 Hz) are associated with cortical excitability. 7 Additionally, premovement oscillation8,9 and event-related desynchronization (ERD) 5 play pivotal roles in defining sequential motor execution. Based on these associations, researchers are exploring the use of tACS to artificially enhance alpha- or beta-band rhythms, aiming to improve motor function or improve memory consolidation and augment the effects of rehabilitation training. 10 A meta-analysis showed that beta-tACS stronger than 1 mA yields a robust M1 excitability. 11 Despite these promising prospects, most studies on tACS for this purpose remain in the preclinical stage, and only a few researchers have reported its clinical application for stroke rehabilitation, including gait, 12 aphasia, 13 and hemispatial neglect. 14 Since the application of tACS for stroke patients is limited, and a recent review even avoided a detailed comparison between tDCS and tACS, the underlying mechanisms and possibility for further utilization of tACS remain to be elucidated. 15
Naros et al. first applied tACS to stroke patients in a motor imagery task, which serves as a model similar to rehabilitation, while no study has applied tACS to an actual stroke rehabilitation. In their study, they stimulated the primary motor cortex of the impaired hemisphere continuously or intermittently using low-intensity beta-band tACS at 20 Hz and 1.1 mA in chronic stroke patients with upper limb paralysis. Continuous tACS was applied throughout the entire imagery session. Conversely, intermittent tACS was administered to restore the prepared state before each motor imagery session, while motor imagery was performed during the off-phase of tACS. They found that intermittent tACS led to an enhancement in ERD. 16 While Naros et al. did not mention in their research article, the restoration of the prepared state is also known as post-movement beta-rebound (PMBR) of motor cortical oscillation. Researchers revealed that PMBR is impaired in stroke patients and might related to motor function and motor learning ability.17,18 Therefore, intermittent tACS may improve motor learning via enforcing PMBR. This hypothesis is recently tested in the context of modulating voluntary movement using 15 min continuous tACS for the primary motor cortex using a classical montage in healthy participants. 19
Neuromuscular electrical stimulation (NMES) is a well-established stroke neurorehabilitative method. Particularly, a closed-loop NMES, integrated volitional control electrical stimulation (IVES), is one of the representatives to induce sensory and motor functional recovery of the upper extremity accompanying the plasticity of cortical and local spinal circuits.20,21 Fujiwara et al. 20 demonstrated that a closed-loop NMES-based 3-week inpatient rehabilitation program, which incorporates functional and task-specific occupational therapy (OT), reduced short intracortical inhibition in the affected hemisphere, and led to motor functional recovery in chronic stroke patients. Additionally, Tashiro et al. showed a reduction in sensory processing time, represented by the duration between central somatosensory evoked potential (SEP) peaks, and restoration of peak deficiency in line with sensory functional recovery after the same treatment regimen. 21 A meta-analysis showed that such electromyography (EMG)-related NMES has a robust short-term effect on restoring wrist and hand impairments. 22 These findings indicated that the combination of closed-loop NMES and OT rehabilitation has significant potential for facilitating functional recovery in chronic stroke patients in combination with tACS. Based on these outcomes, we adopted a comprehensive rehabilitation program that includes a tACS-NMES combinatory session, followed by a manual OT session in an outpatient setting.
In this study, our objective is to investigate the application of tACS combined with NMES training. The goal is to establish a more effective neurorehabilitation approach that enhances the effects of NMES training with tACS by targeting the paretic finger extensors in chronic stroke patients. We named this trial as Synchronized Application of IVES and tACS study (SAITAC). Saitac is taken from the old Japanese-Chinese saying Saitaku-Douki, which means symphonic cooperation as the chick clicks egg shell from the inside, the parent bird cracks it from the outside. As this study represents the first to combine tACS with another electrical stimulation method for stroke patients, we have designated it as a phase I/II trial. Thus, the primary focus of this experiment is to address safety issues and check the feasibility associated with this approach. We will evaluate the influence of real tACS in comparison to sham tACS a novel neuromodulatory factor. Additionally, we aim to explore the physiological aftereffects of tACS, particularly entrainment, while also assessing any differences in functional recovery outcomes among participants. The experiment is planned as follows.
Methods
Inclusion and exclusion criteria and requested items
Individuals who meet the following selection and exclusion criteria demonstrate an understanding of the research, and provided informed consent will be included in this trial. We will adopt enrolment strategies via the Internet as well as referral consultation from other hospitals and clinics. The enrolment process will be implemented by the principal investigator, which will cease 6 months before the end of the study period.
The inclusion criteria are as follows: (1) age between 20 and 80 years, (2) patients with hemiplegia due to first-ever stroke caused by a unilateral hemispheric lesion, (3) stroke in the chronic phase at least 180 days after the onset, (4) patients who have completed rehabilitation and are currently living at home, (5) upper extremity function falls in the range that ⩾2 in the Stroke Impairment Assessment Set (SIAS) knee–mouth subtest for proximal function AND ⩾1 b in the SIAS finger-function subtest for the distal function23,24; briefly, the patient can elevate paretic hand to the height of their nipples, and can extend his or her paretic fingers to a visible extent, (6) no severe contracture of the upper limb, (7) ability to attend electrical stimulation and outpatient rehabilitation sessions (totaling approximately 2.0 h) at the required frequency.
The exclusion criteria are as follows: (1) Participants with contraindications to exercise therapy and those ineligible for the study due to the following conditions: (i) serious cardiac disease, including unstable angina, recent myocardial infarction, uncompensated congestive heart failure, acute pulmonary heart, poorly controlled arrhythmia, severe aortic stenosis, active myocarditis, endocarditis, etc.; (ii) uncontrolled hypertension; (iii) serious pulmonary disease, including recent pulmonary embolism, acute pulmonary heart, and severe pulmonary hypertension; (iv) complications of severe hepatic or renal dysfunction, metabolic disorders (e.g., acute thyroiditis); (v) acute systemic illness or fever; (vi) complications of serious orthopedic disease that would interfere with exercise; (vii) cognitive function or higher brain dysfunction to the extent that comprehension of the research’s purpose and methods is difficult. (2) Participants requiring caution with electrical stimulation due to the following conditions: (i) lesions involving the cortex; (ii) history of head or spinal cord surgery or head trauma with impaired consciousness; (iii) presence of metal (except titanium) or electronic implants in the brain, intracranial, or internal body (e.g., foreign body in the eye, aneurysm clips, cochlear implants, deep brain stimulator, or cardiac pacemaker); (iv) history of epilepsy, convulsions, or sudden abnormalities in EEG study before trial inclusion, or having a close relative with epilepsy; (v) use of drugs that affect the nervous system (e.g., anti-convulsant drugs and major and minor tranquilizers); (vi) presence of skin problems such as dermatitis, psoriasis, and eczema, on or around the electrode-applied area; (vii) pregnancy or possibility of being pregnant; (viii) previous experience of physical discomfort due to tCS or transcranial magnetic stimulation (TMS).
The following items are requested from each participant: (1) not to participate in any advanced rehabilitation program or neurorehabilitation program for 3 months prior to joining the study. Examples of such programs include transcranial repetitive magnetic stimulation therapy, functional electrical stimulation therapy, constraint-induced movement therapy, and repetitive facilitation therapy; (2) not to receive any upper extremity rehabilitation program during the intervention period of the current study.
Study design and schedule
In this phase I/II clinical study, we adopted an AB/BA crossover design to address safety concerns and assess test treatment endurance for each participant, in terms of noninferiority, considering the variability in stroke lesions and individual backgrounds. This single-center study is conducted in an outpatient clinic of an academic hospital. Participants, occupational therapists implementing OT training, and assessors will be blinded to the condition; however, the investigator who implements the tACS part cannot be blinded. The entire intervention consists of 10 intervention days (days 1–10), during which real and sham tACS will be administered for 5 consecutive days either on days 1–5 or days 6–10. Participants will be randomly assigned to two groups: real-to-sham (R-S), where tACS will be applied during the first 5 days and sham tACS during the next 5 days, or sham-to-real (S-R), where the order is reversed (Figure 1(a)), according to computer-generated random number in a sequential manner. Each single-day intervention session will comprise a 20-min session of repetitive finger extension training with closed-loop NMES combined with intermittent beta-tACS conditioning, followed by 40 min of OT. The tACS-NMES session will consist of 50 repetitive finger extension training cycles (Figure 1(b)). Each training cycle will last 24 s, including 5 s of tACS with 5 s of ramp-up and ramp-down phases, where the tACS intensity will be gradually increased and decreased, and 5 s of finger extension attempts during the off-phase of tACS. The preparation of electrodes and devices in sham condition will be totally identical to the real, and the injected current lacks the main beta-tACS at 1.1 mA but includes ramp-up and ramp-down phases (Figure 1(c)).

The overall protocol. (a) The experimental timeline depicts 10 weekly interventions, consisting of 5 consecutive single-day interventions with either real (blue boxes) or sham (red boxes) tACS. Each participant will be randomly allocated to one of two orders: R-S or S-R. Assessment days are scheduled before day 1, in the middle of day 5 and 6, and after day 10. (b) A detailed view of the single-day intervention is presented. Following the preparation, a 20-min session of repetitive finger extension training using dynamic precision tACS and closed-loop NMES (tACS-NMES) and 40 min of OT are implemented. (c) The tACS-NMES session encompasses 50 repetitive finger extension cycles, each lasting 24 s. During this phase, while the participants maintain a relaxed body state, beta tACS is applied for 5 s with 5-s ramp-up and ramp-down phases over the motor cortex. In contrast, the sham stimulation consists only of ramp-up and ramp-down phases and lacks the main stimulation with 1.1 mA. Then, finger extension efforts supported by closed-loop NMES are implemented for 5 s during the off-phase of tACS.
While some researchers have reported that a 48-h interval is sufficient for washout between sessions, 25 we plan to have a sufficient washout period for previous brain stimulation. Thus, each intervention will be held weekly, as a 1-week washout period is widely used in tCS studies, 26 and we will apply a 5-day interval as the shortest period considering the convenience of the participants. 27 We consider that weekly intervention is not only adequate to reduce the aftereffects of previous tACS but also practical for an outpatient rehabilitation program designed for stroke patients. In addition, as a crossover study, we will set 2 weeks of interval between two different interventions (real and sham conditions) to ensure any lasting or cumulative effects remain. 28
NMES training combined with tACS
A closed-loop NMES, known as IVES (IVES®; OG Wellness, Okayama, Japan), will be used in this study. Briefly, this device detects EMG activity and provides electrical stimulation to the same muscle, even when the muscle’s strength is too weak to induce substantial muscle contraction. 29 This enables the device to stimulate the target muscle based on the amount of detected endogenous EMG activity of the same muscle, resulting in instantaneous and substantial muscle contraction. In addition, the device has a separate stimulation-only electrode to induce synergistic contraction of different muscles. In this study, we applied a protocol to stimulate the extensor digitum communis muscle (EDC) and extensor indicis pollicis using voluntary EMG activity detected from the EDC to facilitate finger extension movement (Figure 2). 30 Thus, the weak voluntary muscular activity of the finger extensors is amplified into substantial volitional movement through simultaneous electrical stimulation. At the beginning of each single-day intervention, we will optimize the sensitivity of EMG detection and the intensity of electrical stimulation as needed. In this experiment, the voluntary activity triggering electrical stimulation is induced via the patient’s effort at an off-phase of tACS based on the cue provided by the experimenter during a cycle of 24 s (Figure 1(c)). During the 20 min tACS-NMES session, the detected EMG activities over 50 finger extension efforts will be recorded on a scale of 0–5 as a displayed light up at 5°, to determine the transition in muscle output. Previous inpatient rehabilitation programs using this method have demonstrated significant motor and sensory recovery in patients with chronic stroke, accompanied by plastic changes in the motor and sensory cortex.20,21 However, to our knowledge, no studies have reported its effectiveness in outpatient rehabilitation.
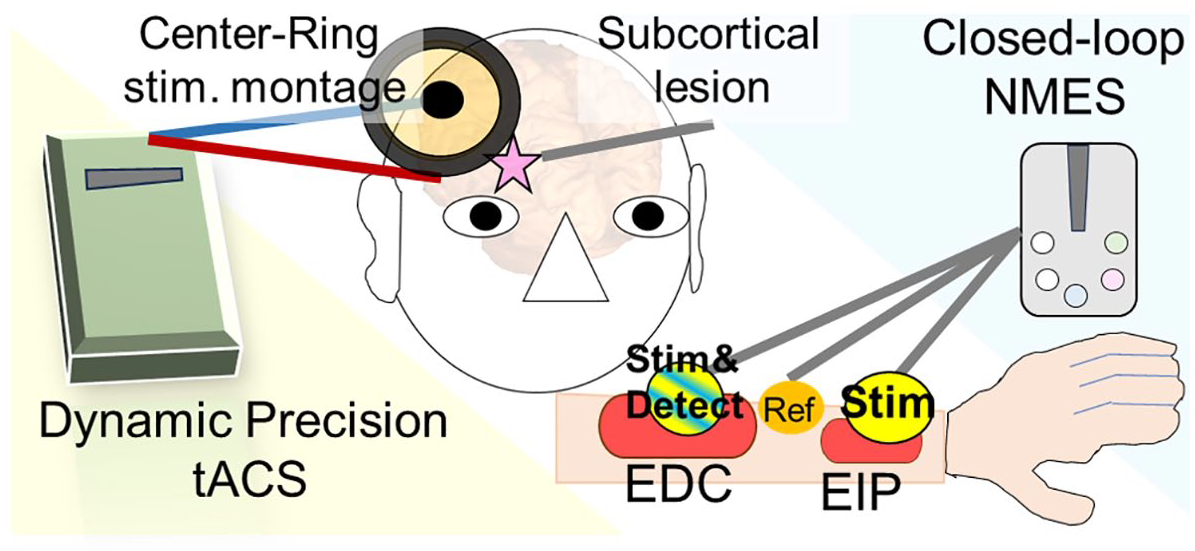
An experimental setting is shown in the case with a right subcortical lesion presenting with left hemiparesis. The center and ring electrodes placed at C3h or C4h over the affected hemisphere are used for radially oriented tACS. The impedance will be kept under 5 kΩ. The closed-loop NMES device is attached to the paretic arm to amplify finger extension function with an EMG detection/stimulation electrode over the belly of EDC muscle, reference electrode over the tendon of EDC, and stimulation electrode over the belly of EIP muscle. The parameters of NMES, sensitivity of EMG detection and intensity of electrical stimulation, are optimized at the beginning of every single-day intervention as needed.
A radially oriented stimulation montage will be used, employing center and ring electrodes, which are composed of a ring-shaped surrounding rubber electrode with a diameter of 100 mm and a width of 12.5 mm and a circular rubber electrode with a diameter of 34 mm in the center. 31 These electrodes will be placed at C3h or C4h, ipsilateral to the subcortical lesion, following the international 10-5 system (Figure 2), which corresponds to hand motor hot-spot. 32 To attach the electrodes, we will use Ten20 paste (Weaver, Denver, CO, USA). We opt to use C3h or C4h instead of motor hotspots detected by TMS or magnetic resonance imaging guidance because we focus on the broad clinical application of this method. Before the session, the skin surface will be cleaned using Nuprep (Weaver), and we will maintain the impedance below 5 kΩ throughout the intervention. Beta-tACS will be applied using Nurostym (Brainbox, Cardiff, UK). We will adopt a tACS protocol similar to that used by Naros and Gharabaghi, 16 incorporating an intermittent finger extension training task using NMES, rather than a motor imagery task in the previous study. The individualized beta frequency of each patient, acquired through EEG at eyes opening prior to the study, will be used for the tACS stimulation. The frequencies will be manually determined by board-certificated EEG specialists in 0.5 increments. Each stimulation block will last for 24 s and consists of 5 s of 1.1 mA beta-band tACS accompanied by 5 s of ramp-up and ramp-down, 5 s of finger extension, and 4 s of rest; no current will be applied for 9 s. This short block will be repeated 50 times, resulting in 20 min of tACS-NMES training (Figure 1(c)). In the cases of harm, patient request, or worsening disease, we will cease the intervention immediately. Unblinding will be allowed upon the patients’ request in such cases.
OT training
OT training will be conducted using conventional methods: joint range of motion training, muscle strengthening exercises, and dexterity training, including task-specific training. We believe that arbitrary modifications to the OT training after the tACS-NMES session are not preferable since the main purpose of this study is to establish a new neurorehabilitative approach that enhances the effects of conventional training. Therefore, occupational therapists, who will be blinded to the type of stimulation (real or sham), will have the freedom to design the rehabilitation program relatively independently. The only restriction placed on them will be to avoid physical therapies such as electrical, ultrasonic, and magnetic stimulation during OT sessions.
Assessments
Assessments of adverse events: primary outcomes
We will record the recruitment rate, which is composed of the number of recruitment attempts, I/C conducted, and I/C obtained, the adherence rate derived from the stoppage of the tACS session, and the drop-out rate, together with any other findings or event during sessions. To assess adverse events and participants’ perceptions, we will use a modified questionnaire form on an open platform. 33 The questionnaire will include analogous scales to measure dizziness, nausea, metallic taste, phosphene, and tickling-tingling sensation of the scalp while omitting detailed questions about stimulation sensation, as they may be challenging and time-consuming for older persons with stroke sequela. Instead, we will include other analogous scales to evaluate tiredness, sleepiness, headache, tinnitus, and taste abnormalities, as well as abnormal sensations in the arm, and to detect any reported side effects. 34 The questionnaire will be administered on every intervention day. In addition, the transition of the muscle output corresponding to 50 finger extension attempts will be recorded as an index of muscle fatigue.
To evaluate fatigue, we will use the Japanese version of the Profile of Mood Status, 2nd edition (POMS2), as a subjective assessment. Participants will complete the POMS2 questionnaire on days 1, 5, 6, and 10 upon arrival at the outpatient clinic and immediately before returning home. In addition, we will use 3-min heart rate variability (HRV) recordings, obtained using a smartwatch (Polar Vantage v2; Polar, Kempele, Finland) worn on the nonparalyzed side and an accompanying heart rate monitor (H-10; Polar). HRV assessments will be conducted before and immediately after the tACS-NMES session, as well as after OT training on every intervention day, to provide an objective assessment of fatigue. The POMS2 is a widely used fatigue rating scale with established reliability and validity, and it is suitable for application to stroke patients. 35
Behavioral functional assessments: secondary outcomes
Blinded evaluations of the following batteries will be conducted before the intervention (day 0), after the first half of the program (day 5/6), and at the end of the whole intervention (day 11) by occupational therapists or rehabilitation physicians: (i) SIAS, which possesses scale reliability and predictive validity for stroke rehabilitation,23,24 (ii) Fugl-Meyer assessment for the upper extremity, 36 and (iii) Motor Activity Log-14 to record the quantity and quality of paretic hand participation in activities of daily living 37 since participation is a determinant of longitudinal functional recovery after the intervention, 38 (iv) Modified Ashworth Scale to assess spasticity, 39 (v) sensory assessments with the Semmes–Weinstein monofilament test, 40 thumb-localizing test, 21 two-point discrimination test, and vibration test using 30- or 256-Hz tuning forks. 41
Electrophysiological assessments: secondary outcomes
All electrophysiological assessments will be conducted by the Japanese Society of Clinical Neurophysiology board-certified EEG and EMG specialists. The following assessments will be performed: (i) EEG: To monitor entrainment by tACS, EEG recordings will be taken from C3h and C4h, where the tACS electrodes will be centered, with the other side as the control. A hybrid electrode that enables both EEG recording and tACS stimulation (DONUT, DOuble-electrodes for Nouha U TCS) will be used for quick recording after tACS. 42 Reference electrodes will be placed on both ear lobes, and the average will be used as the reference. The ground electrode will be placed at the C7 spinous process. The impedances of all the electrodes will be maintained under 5 kΩ during recording. EEG recordings will be performed using Neuropack (Nihon-Kohden, Tokyo, Japan) before and just after the tACS-NMES session and after OT training on every intervention day. The power of EEG at the range of applied tACS frequency ±0.5 Hz will be extracted using a custom MATLAB script (2022b; Mathworks, Natick, MA, USA). (ii) Surface EMG recording: To assess voluntary muscle activity, surface EMG recording from the EDC and flexor carpi radialis (FCR) muscle will be evaluated. 43 (iii) H-reflex and reciprocal inhibition of wrist flexor: To evaluate the spasticity of the wrist flexors, reciprocal inhibition of the FCR muscle will be assessed as previously described. 20 Surface EMG and reciprocal inhibition will be evaluated using Neuropack (Nihon-Kohden) on days 1, 5, 6, and 10 upon arrival at the outpatient clinic and just before returning home. (iv) SEP: To evaluate sensory cortical plasticity, SEP corresponding to median nerve stimulation will be assessed before the intervention (day 0), after the first half of the program (day 5/6), and at the end of the whole intervention (day 11). Active electrodes will be placed at Cp3, Cp4, F3, and F4. Reference values will be determined by averaging the signals from bilateral earlobe electrodes. N13 in the median nerve will be assessed at the C5 spinal level. The ground electrode will be placed at Fz. The impedance of each electrode will be maintained under 5 kΩ. In the SEP recording, median nerves will be alternately stimulated at the wrist at a frequency of 2.05 Hz (i.e., 1.025 Hz for each side) with a square wave with a 0.2-ms duration at a stimulation intensity that induces visible yet minimal muscular contraction of the abductor pollicis brevis muscle (approximately 6–10 mA). A 500-sweep addition-averaging method will be used. 21 Although the finger extensors, the target muscles of our training paradigm, are dominated by the Radial nerve, the electrical stimulation of NMES may induce functional modification like sensory adaptation to this nerve due to its anatomical position. 44 In contrast, the Medial nerve, which dominates antagonistic muscles to extensors and is free of direct electrical stimulation, will sense repetitive passive movements during the interventions. Therefore, we consider the Median nerve to be the most sensitive nerve monitoring the training effect.
Sample size calculation
This study is a phase I/II trial designed to investigate adverse events and feasibility. The 60-min rehabilitative intervention, including the OT session at the end, may be influenced by fatigue when intermittent tACS is combined with finger extension tasks using closed-loop NMES at the beginning. To determine the sample size, we will consider fatigue induction, as there are no prior studies reporting tACS adverse events in similar cases, and the stimulation protocol is well within the safety range for healthy participants. 34 POMS2, a widely used assessment tool for fatigue in psychiatry and rehabilitation medicine, was chosen for this purpose. Ishida et al. 45 compared rTMS applied to the right dorsolateral prefrontal cortex with sham stimulation in 16 healthy participants, and a significant reduction in the fatigue-inertia (FI) score of POMS2 was observed. Nakashima et al. 46 compared the motor imagery task of lifting a 500-mL plastic bottle and a 1.5-L plastic bottle in 12 healthy participants and reported that the latter condition significantly increased POMS2-FI scores. Although the standard deviation is not specified in these papers, the relatively homogeneous nature of healthy participants suggests that a sample size of N = 20 will be necessary for this study.
Statistical analysis
The comparisons between two data sets, that is, the demographic information, the change of outcomes over the pre-post intervention of R-S and S-R groups, the subjective feeling of tACS via the questionnaire, the POMS2 subscores, and the values of functional and electrophysiological assessments between pre- and post-intervention, will be done with paired parametric or nonparametric T-test. The transition of physiological values monitored during the intervention, namely, the detected EMG output with IVES, HRV, and EEG power, will be analyzed with two-way repeated measures ANOVAs, along with point-by-point comparisons with T-tests. We will remove outliers more than ±2 S.D. in the values of electrophysiology, suggesting some error in the acquisition. Missing data will be analyzed using pair-wise deletion. We will set the significance level at 0.05. Subgroup analysis will not be intended. We will omit cases with protocol nonadherence. All statistical analyses will be implemented using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a modified version of R commander, a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). 47
Discussion
Combination effect of tACS and rehabilitation
We believe that tACS can enhance rehabilitation effects in two main ways. First, there is a direct interaction between the alternately and repetitively applied tACS and finger extension training using NMES. That is, tACS can aid in the reconditioning, thus beta rebound, of the primary motor cortex, facilitating the execution of the finger extension task. Second, the intermittent tACS application may have lasting effects that assist in the implementation of OT sessions thereafter.
To the best of our knowledge, there has been a limited number of evidence which are applicable to an estimation of the direct interaction between tACS and NMES in neurorehabilitation. Naros and Gharabaghi 16 previously demonstrated the effect of intermittent tACS preconditioning coupled with motor imagery tasks. In our study, we will be using a focal stimulation montage to apply beta tACS, which has demonstrated a higher effect on upregulating corticospinal excitability compared to the classical C3-to-Supra-Orbital montage. 31 While Kolmos et al. 48 recently published their protocol of focal tDCS for stroke neurorehabilitation using a central anode and four equidistant electrodes, the current study will be the first to use focal tACS. The feature of tACS in the current trial is applied in a precision stimulation manner in terms of individualized AC frequency and focal stimulation montage, as well as the specified stimulation timing in a sequence of movements. Therefore, we call this method dynamic precision tACS. We have chosen to use a 50-times finger extension task instead of motor imagery, as repetitive facilitative exercise has been shown to promote functional recovery in stroke patients. 49 Ohnishi et al. 50 recently showed the combinatorial effect of repetitive facilitation exercise and peripheral electrical stimulation therapy for severe upper limb paresis in an inpatient setting with high-dose intervention. Moreover, researchers have reported the combinatorial effect of tDCS and neurorehabilitation with NMES.51,52 Overall, we would expect a treatment effect of our protocol to integrate tACS and NMES to undergo repetitive facilitative training even on an outpatient basis.
Moreover, it is plausible that the aftereffect of intermittent beta-tACS enhances the effect of OT. A meta-analysis reported that beta-tACS can upregulate corticospinal excitability, 11 and its lasting effect has been estimated to be up to 60 min.31,53 However, the aftereffect of intermittent stimulation may differ from that of continuous stimulation. To explore this, we plan to monitor the lasting effect of tACS using EEG immediately after the tACS-NMES session and again after the 40-min OT session held at the end of the intervention. On the other hand, the participants may notice the abrupt stoppage and restart of ramp-up and down current in the sham condition. However, we consider that it is necessary to extract a pure effect of the main part of tACS to assess the adverse effect and that the sensation of sham stimulation could be considered a sort of foreign body sensation as of electrodes in a sense that is inevitable and does not convey solid effect theoretically.
Safety concern
The 2017 International Society for Clinical Neurophysiology guidelines have established the safety of low-intensity tCS, including tACS. 34 According to these guidelines, no serious side effects have been reported when the stimulation intensity is ⩽10 mA, and even minor side effects such as headache are not typically observed if the stimulation intensity does not exceed 2 mA for 60 min per day. Here, we will use intermittent stimulation with a total duration of 250 s (5 s/cycle, 50 cycles). Additionally, we will employ a focal stimulation montage, which targets only the intact primary motor cortex and avoids unnecessary areas, to enhance safety during tACS application. 31 Although our participants have a brain lesion in the ipsilateral hemisphere, we have carefully excluded individuals with cortical lesions or any risk of seizures, ensuring safety against convulsions during tACS. However, our protocol uses a stimulation intensity of 2.2 mA peak-to-peak, which is slightly above the range recommended in the guidelines, indicating that minor issues should be monitored.
Another peripheral electrical stimulation will be applied to the forearm during the trial. However, existing studies on tDCS that combine peripheral stimulation have shown that such dual-site electrical stimulation is generally safe. For example, studies combining tDCS with upper extremity NMES, similar to our protocol, have reported no remarkable adverse effects. 52 Additionally, other combinations of tDCS with radial nerve stimulation in acute patients 51 and tDCS and NMES targeting cervical muscles to treat dysphagia, which have a closer stimulation site, have also demonstrated safety with minimal side effects. 54 Moreover, we will apply tACS and NMES alternately to avoid crosstalk; NMES will be applied during the off-phase of tACS, and the NMES device will be turned off during the tACS phase. Considering the chronicity of stroke, 51 remoteness of stimulation sites, 54 and heterochronicity of the two stimuli, as well as the focal nature of the central stimulation, we believe that the safety of our trial will be ensured.
Does fatigue matter for the implementation of rehabilitation?
The effects of tACS on fatigue have a bilateral character. First, the consequences of tACS may limit the endurance of stroke patients during OT sessions. Second, intermittent tACS might improve the efficiency of finger extension training using closed-loop NMES and help implement repetitive facilitation training.
Subclinical fatigue, which is not distinct from tiredness, has been observed in healthy participants who underwent continuous tACS in pilot experiments. Although tACS may not induce apparent “fatigue” as a side effect, it could contribute to tiredness in stroke patients, who are already vulnerable to fatigue. 55 Therefore, one concern is that fatigue induced by tACS may hinder the implementation of OT rehabilitation.
However, intermittent tACS may also ameliorate fatigue when combined with repetitive finger extension training using NMES. Fatigue during NMES training has been reported by some researchers. Meadmore et al. 56 reported that functional electrical stimulation, of which NMES comprises one part, targeting upper extremity paresis secondary to stroke, induced fatigue weakening of the muscle response over the course of the intervention session. While such fatigue seems to be related to event-related EEG changes, 57 Naros and Gharabaghi 16 demonstrated that intermittent beta-TACS improved event-related EEG responses in stroke patients during motor imagery tasks. Thus, precision dynamic, intermittent, tACS can assist in repetitive motor execution and suppress fatigue.
Feasibility and perspective
Progress in outpatient rehabilitation programs is crucial for achieving functional recovery in individuals with hemiparetic stroke especially in the chronic phase. While public insurance systems may cover low-frequency rehabilitation programs, such as for 1 h 1–2 times per week, 45 these services often focus on maintaining residual function and fitness, leaving a gap in specialized neurorehabilitation for severe upper limb paresis. Therefore, it is essential to propose effective neurorehabilitative approaches aiming at functional recovery that can be implemented in various settings, including general welfare institutes, small and local clinics, and nonspecialized hospitals. Here, “feasibility” encompasses three aspects: economic, social, and legal. The devices used in this study must be sufficiently inexpensive to be available at these institutes. The protocol must be sufficiently short and infrequent to be implemented through public or popular insurance systems. Rehabilitation therapists must allow interventions. We consider that the devices used in this study are available at a relatively reasonable price: 15,000 USD for the tACS device and 4000 USD for the NMES device; therefore, they are economically feasible. Second, the current study protocol, consisting of 20 min of tACS-NMES and 40 min of OT, generally falls within the range of the general insurance system. This suggests that the protocol is socially feasible and can be implemented through public or popular insurance systems. Lastly, from a legal perspective, tACS can be considered a part of electrical stimulation, a form of physical therapy that physiotherapists or occupational therapists are permitted to implement as a part of rehabilitation. Considering these aspects, the current study protocol holds promise and is worth investigating further. If the results are favorable, this method has the potential to be widely adopted as a treatment option for severe upper limb paresis in patients with chronic stroke, making a significant impact on stroke rehabilitation in various medical and welfare settings.
Footnotes
Acknowledgements
We appreciate the sincere support of Mr Osamu Takahashi for his technical advisory for electrophysiological assessments and Ms Yuna Dan and Ms Mitsuyo Ikeda for their specialist’s advisory for occupational therapy for severe upper extremity paresis in stroke patients.
