Abstract
Background:
Psoriatic arthritis (PsA) is a chronic systemic inflammatory disease that affects up to 30% of patients with psoriasis. Diagnosis and treatment could be improved by implementing an interdisciplinary dermatological-rheumatological consultation (IDRC).
Objectives:
This study aimed to assess the effect of a face-to-face IDRC involving both a dermatologist and a rheumatologist evaluating patients in a single visit, on disease activity and burden in patients with PsA.
Design:
Prospective, single-center, cohort study.
Methods:
202 patients with psoriasis were enrolled, among whom 115 individuals with psoriasis and musculoskeletal symptoms underwent an IDRC. Disease manifestations, comorbidities, and both objective and subjective disease activity scores were evaluated.
Results:
Out of the participants, 56 were diagnosed with definite PsA, while the remaining 146 had psoriasis. Nail involvement was associated with axial PsA (odds ratio 4.11; 95% CI 1.22–13.82; p = 0.02). Patients with PsA often experienced a prolonged time to diagnosis (mean 187 weeks) and had a significant psychosocial burden (mean Hospital Anxiety and Depression Index Scale [HADS]-Anxiety score of 7.66 and mean HADS-Depression score of 5.63). Post-IDRC, both objective and subjective disease parameters showed improvement, and patients required less time for consultations with healthcare professionals compared to before the IDRC.
Conclusion:
These findings suggest that an IDRC approach could effectively expedite and optimize the diagnosis and treatment of patients with psoriasis and musculoskeletal symptoms.
Plain language summary
Psoriatic arthritis is a chronic inflammatory disease that occurs in one in three patients with psoriasis. Finding the right diagnosis and treatment is challenging and often time-consuming, so it is crucial to find ways to establish care models which accelerate and improve this care. In our presented study, we established a face-to-face consultation with a dermatologist and a rheumatologist consulting each patient in a single visit and assessed the effects of this consultation on diagnosis, disease activity and patient burden. We studied 202 patients with psoriasis. Out of these, 115 showed both skin and musculoskeletal symptoms and therefore underwent our interdisciplinary consultation. We looked at their symptoms, other health issues, and how they felt about their disease. We found that 56 of them had definite psoriatic arthritis, while the rest had psoriasis alone. Nail problems were linked to inflammation in the axial skeleton. People with psoriatic arthritis often got diagnosed late (after about 3-4 years) and felt a lot of stress. Our interdisciplinary consultation enabled a fast diagnosis and improved treatment - patients had fewer symptoms and higher quality of life. They also needed less time with doctors. These findings suggest that interdisciplinary care can improve diagnosis and treatment in patients with psoriasis and musculoskeletal symptoms.
Keywords
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease affecting about 0.3% of the general population. 1 It develops in 20%–30% of psoriasis, 2 often emerging about 10 years after the initial psoriasis diagnosis. 3 PsA involves the skin, enthesis, and synovium, and can also impact various systems resulting in cardiovascular diseases, uveitis, or inflammatory bowel disease. It imposes a significant psychosocial burden due to pain, stigma, sleep disorders, and reduced work productivity. 4
Despite advancements in its classification,5,6 timely diagnosis of PsA remains challenging due to the heterogeneous manifestations and the lack of validated diagnostic markers. The conventional approach of separate consultations with dermatologists and rheumatologists often leads to delays in diagnosis and treatment.7,8 In a population-based study, 58% of PsA patients were not receiving systemic therapy, despite 88% reporting a history of inflammatory joint pain, strongly suggesting the presence of PsA disease activity. 9 This is particularly alarming, as delayed treatment is associated with increased joint damage10,11 with even a 6-month delay in diagnosis resulting in poorer radiographic and functional outcomes. 12
To improve this condition, several models of interdisciplinary care for PsA have emerged, such as parallel care (dermatology and rheumatology consultations that follow each other), preferential circuit (two specialists work separately and refer to each other only when necessary), and face-to-face care (both specialists assess the patient together). 13 Both the European Alliance of Associations for Rheumatology (EULAR) and expert groups recommend the implementation of interdisciplinary care for PsA.14,15 Studies also suggest that patients prefer combined visits with both specialists 16 . However, there is limited research on the impact of multidisciplinary care on disease activity and patient well-being.16,17
In a previous study, multidisciplinary care for patients with psoriasis and musculoskeletal pain was associated with an improved definitive diagnosis of PsA, high adherence to follow-up consultations, treatment modifications, and a trend toward enhanced psoriasis-related quality of life. 18 In the present analysis, we provide specific insights into our cohort of PsA patients, detailing the distribution of PsA manifestations, subgroup analyses examining associations between different manifestations, and the impact of interdisciplinary consultations on their management and disease activity.
Materials and methods
Study population and design
In this prospective, single-center, cohort study, patients with psoriasis, not previously diagnosed with PsA, were screened at dermatology/ rheumatology outpatient clinics (University Medical Center Mannheim) and were enrolled between August 2020 and May 2021. Inclusion criteria were a diagnosis of psoriasis, age over 18 years and written informed consent. Exclusion criteria were an inability to provide consent or a lack of German proficiency. Patients were assessed at baseline and 6 months later. This report focuses on outcomes in patients newly diagnosed with PsA following multidisciplinary consultation.
Multidisciplinary consultation
Patients underwent assessments in specially arranged outpatient clinic sessions employing an Interdisciplinary Dermatological-Rheumatological Consultation (IDRC) utilizing the face-to-face approach. PsA was classified according to CASPAR classification criteria. 6 Data collected included demographic information, disease onset, psoriasis subtype, skin/nail lesions, presence of musculoskeletal symptoms (in joints, tendons, and fingers), past/current therapies, comorbidities, adherence and compliance, and money spent and time lost due to treatment (e.g., for transportation cost and prescription charges). Patient-reported outcomes (PRO) were measured using validated tools: Dermatology Life Quality Index score (DLQI), a PRO used to assess quality of life and disease-related reduction of quality of life 19 and Hospital Anxiety and Depression Scale (HADS), a validated PRO for screening patients for elevated levels of anxiety and depression. 20 Disease severity was evaluated using Psoriasis Area and Surface Index (PASI), 66/68 joint count, Clinical Disease Activity Index (CDAI), Simplified Disease Activity Index (SDAI), Axial Spondyloarthritis Disease Activity Score (ASDAS), Disease Activity Index for Psoriatic Arthritis (DAPSA), Bath Ankylosing Spondylitis Functional Index (BASFI) and Leeds Dactylitis Index (LDI). Blood tests included rheumatoid factor, ESR, and CRP. Musculoskeletal manifestations were classified based on the clinical judgment of rheumatology experts, often supported by imaging, with MRI being mandatory to assess axial inflammation. Topical and systemic anti-inflammatory therapy was assessed and adjusted according to consensus between dermatology/psoriasis and rheumatology/PsA experts. The time to diagnosis was calculated based on the patient-reported initial onset of musculoskeletal symptoms, which were later confirmed as inflammatory musculoskeletal manifestations for PsA, and the onset of psoriatic skin changes for psoriasis. Disease manifestations were diagnosed at the discretion of psoriasis and rheumatology experts based on clinical judgment. Musculoskeletal manifestations were often supported by imaging, with MRI being mandatory to assess axial inflammation.
Statistical analysis
Loss to follow-up was managed by initially contacting participants, and subsequently excluding cases with missing data from analyses of changes in disease activity. Microsoft Excel (version 16.0, Microsoft Corporation, Redmond, WA, USA), GraphPad PRISM (version 10.0, GraphPad Software, Inc., Boston, MA, USA) and SAS (version 9.4M8, SAS Institute, Cary, NC, USA) were used for statistical analysis. Baseline characteristics were based on descriptive data (mean, standard deviation, frequencies). Statistical significance was determined using the U-test, Kruskall–Wallis, and Chi-Square test. Multivariable logistic regressions were used to calculate ORs and 95% CI for comorbidities and disease patterns.
Correlation was assessed using Spearman’s R test.
Results
Patient demographic and disease characteristics
A total of 257 psoriasis patients were screened, and 202 were included in the study (Figure 1). Among these, 115 had musculoskeletal symptoms. PsA was diagnosed in 56 patients (28% with psoriasis patients and 49% of patients with musculoskeletal symptoms), while 146 patients had no evidence of the inflammatory musculoskeletal manifestation of PsA (psoriasis only). Most patients (149 (73.8%), 73.2% (Pso), 74.0% (PsA)) attended all visits.
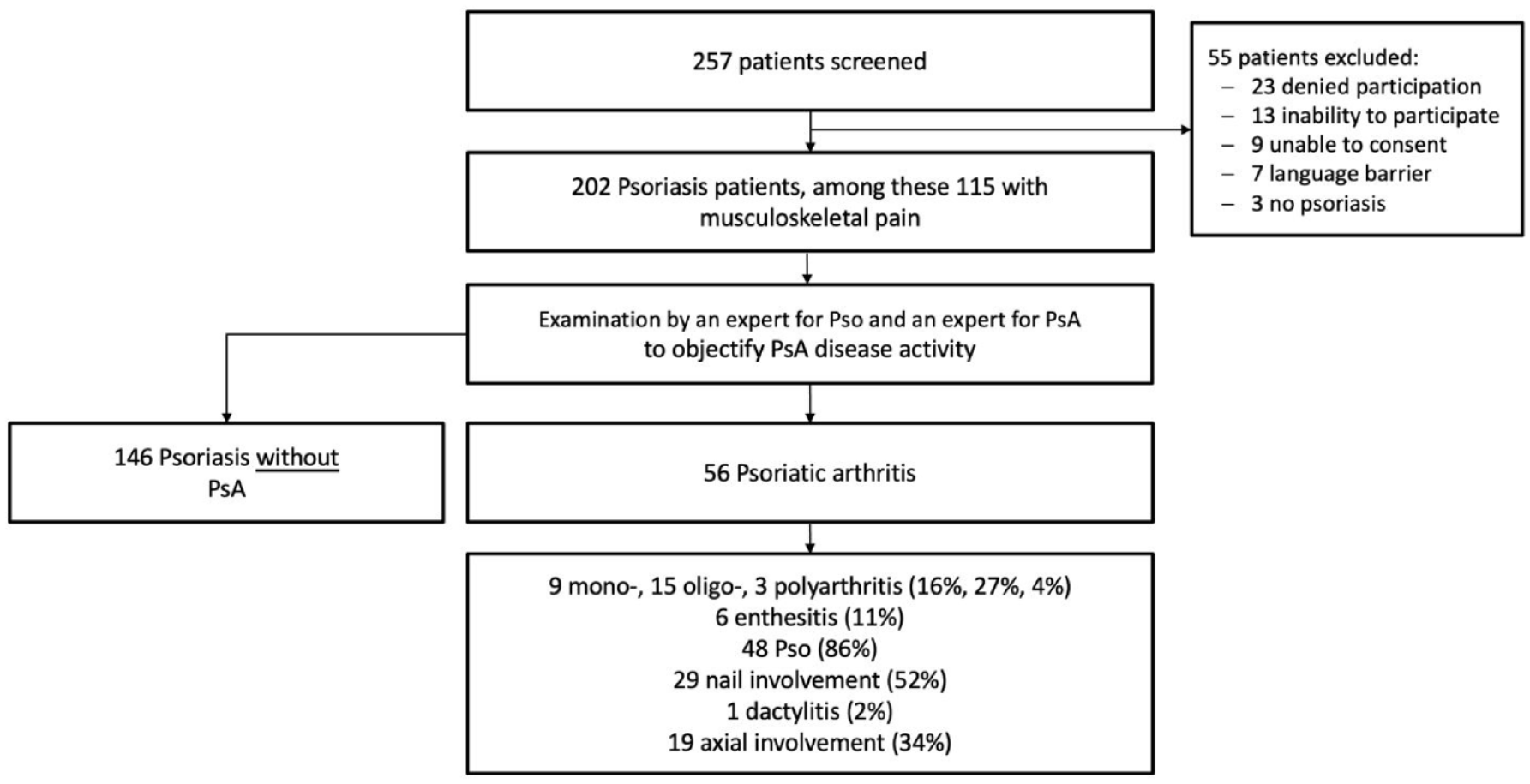
Flowchart of included patients. Patients can have several active disease manifestations at once.
Baseline demographic characteristics such as age, sex, or BMI showed no significant differences between psoriasis and PsA patients (Table 1). However, PsA patients more frequently received bDMARDs (63% vs 42%, p = 0.03) and MTX (15% vs 3%, p = 0.002). Notably, PASI scores were significantly higher in psoriasis patients than in PsA patients (6.08 vs 3.15, p> = 0.002).
Baseline patient demographics and characteristics.

Data are presented as mean and standard deviation.
Inpatient admission for PsA-/ psoriasis (Pso) -related symptoms.
Time from Pso and PsA symptoms onset to diagnosis.
bDMARD, biological disease-modifying anti-rheumatic drug; BMI, body mass index; csDMARD, conventional synthetic disease-modifying anti-rheumatic drug; PASI, Psoriasis Area and Surface Index; PsA, Psoriatic arthritis; RF, rheumatoid factor; VAS, visual analog scale.
The most common comorbidities in both psoriasis and PsA patients were hypertension (34% and 38%, p = 0.72), obesity (27% and 25%, p = 0.83), and depression/anxiety (14% and 11%, p = 0.60) (Table 2). Notably, PsA disease activity was associated with higher rates of diabetes (OR, 4.00; 95% CI, 1.02–15.6; p = 0.03) and dyslipidemia (OR, 1.96; 95% CI, 1.63–2.36 p = 0.01).
Comorbidities.

Comorbidities in psoriasis (Pso) and PsA patients are shown as absolute numbers (frequencies).
MACE, major adverse cardiovascular events; PsA, Psoriatic arthritis.
Active psoriasis was observed similarly in both psoriasis and PsA patients (86% vs 87%, respectively; p = 0.93), while nail involvement was more frequent in patients with PsA (18% vs 52%, respectively; p = 0.002). PsA patients exhibited axial involvement (34%), enthesitis (11%), and dactylitis (2%). Among PsA patients with arthritis, 9 had monoarthritis, 15 had oligoarthritis, and 3 had polyarthritis (Figure 1).
Concurrent disease activity with more than one subgroup of disease activity was prevalent among most patients, 27% of patients exhibited 3 or more disease activity subgroups (Figure 2). In subgroup analyses, nail psoriasis was not significantly associated with enthesitis or arthritis types in PsA patients. However, nail involvement was associated with a 4-fold increased the risk of axial involvement (14/19 with axial involvement (74%); OR 4.11; 95% CI, 1.22–13.82; p = 0.02), and also linked to enthesitis (67%; OR 2.00; 95% CI 0.33–11.92; p = 0.67), polyarthritis (100%), monoarthritis (50%), and oligoarthritis (46%; overall OR 1.56; 95% CI, 0.54–4.49; p = 0.43).

Distribution of extraarticular disease manifestations represented by pictograms and frequencies including combinations thereof among arthritis subtypes (different colors) in patients with psoriatic arthritis (n = 56).
Compared to psoriasis, PsA patients had significantly higher scores for anxiety (HADS-A; 5.84 vs 7.66; p < 0.01) and depression (HADS-D; 4.37 vs 5.63; p = 0.05).
Impact of IDRC on diagnosis and treatment
PsA patients (n = 56) experienced an average time to diagnosis of 183 weeks. In our IDRC, we were able to diagnose or exclude PsA with a mean waiting time of 6.25 ± 7.85 weeks. Treatment was changed in 22/42 (52%) of patients, the most frequent treatment change was implementing bDMARDs (6/42; 14%). The ‘time spent due to consulting doctors’ was reduced from 11.29 h per month before IDRC in patients with PsA to 6.51 h per month after (p = 0.001). ‘Money spent on treatment in the last 6 months’ was reduced from an average of €127 to €79 (p = 0.35). Overall patient satisfaction with the IDRC was high, rated at 7.8 on a numeric rating scale (1–10).
Effect of IDRC on psoriasis and PsA outcomes
The PASI decreased from 6.08 ± 8.26 at baseline to 1.91 ± 3.75 at 6 months (p < 0.001 for change from baseline) in psoriasis patients and from 3.15 ± 5.54 to 2.22 ± 4.32 (p = 0.35) in PsA patients (Figure 3). Similarly, the DLQI improved from 6.62 ± 7.61 to 3.87 ± 4.76 (p = 0.001) in psoriasis patients and from 6.86 ± 7.73 to 6.00 ± 7.58 (p = 0.58) in PsA patients. The between-group difference in DLQI at 6 months indicated a significantly worse quality of life for PsA patients compared to psoriasis patients (6.00 vs 3.87, p = 0.04).
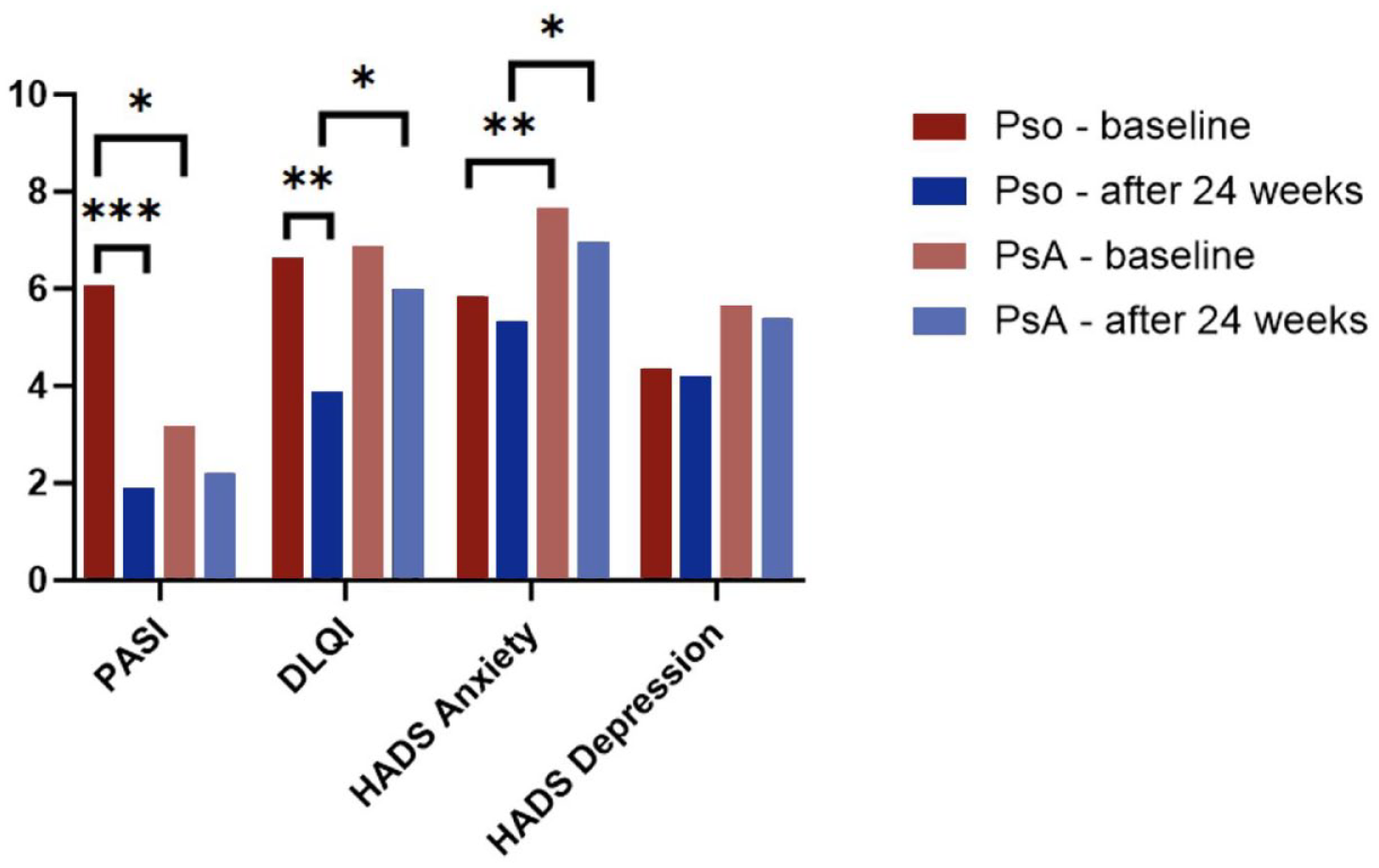
Changes in disease activity and PROs for psoriasis (Pso) and PsA patients at baseline and 6 months later.
In PsA patients, the CDAI and SDAI scores showed a significant reduction in disease activity over 6 months (12.16 vs 5.27 (p < 0.001) and 19.50 vs 10.61 [p = 0.002], respectively) (Figure 4). The BASFI decreased from 4.37 to 2.95 (p = 0.32). CDAI and ASDAS mean scores at 6 months correlated significantly with HADS-D depression scores (R = 0.28; p < 0.01 and R = 0.25; p = 0.01, respectively).

Disease activity in PsA patients at baseline and after 6 months (n = 56).
Dactylitis resolved in all patients after 6 months. The average frequency of sport activities increased (2.4 h/week to 3.5 h/week (p> = 0.30).
Discussion
This prospective study aimed to evaluate how multidisciplinary care affects timely PsA diagnosis/exclusion, patient satisfaction, and disease activity. In addition, we assessed all PsA manifestations in detail to identify potential correlations between different manifestations.
In our cohort, which includes patients seen at a university dermatology center and therefore more likely to have prominent skin symptoms, 27% of psoriasis patients and nearly half (49%) of those with musculoskeletal symptoms were diagnosed with PsA upon further investigation. These findings are consistent with the average PsA prevalence of 23.8% in psoriasis reported in a meta-analysis. 21 The higher prevalence observed in our study compared with some other studies22,23 may be due to the comprehensive, thorough prospective assessments, including musculoskeletal imaging, performed by expert rheumatologists to establish the diagnosis of PsA.
Notably, our cohort presented a high prevalence of cardiovascular comorbidities, obesity, and anxiety/depression, consistent with previous meta-analysis data. 24 More than half of PsA patients (52%) showed nail involvement, which was associated with a 4-fold increased risk of axial involvement. This association, together with data from the Corrona Psoriatic Arthritis/Spondyloarthritis Registry demonstrating that axial involvement was associated with worse nail psoriasis, 25 reinforces the importance of assessing nails in (suspected) PsA and spondylarthritis intensively.
Patients often experienced long waiting times until receiving a correct diagnosis of PsA, on average 143 weeks. Swift diagnosis and treatment are crucial to mitigate joint damage and improve patient quality of life. 10 Our face-to-face multidisciplinary care expedited diagnosis or exclusion of PsA within an average of 6.25 weeks after inquiry, optimizing care for all PsA manifestations as the dermatologist and rheumatologist collaborated on treatment options, for example, topical agents in combination with DMARDs. This approach also notably reduced waiting times and disease burden as patients received comprehensive care from both specialists at the same visit. Conventional referral systems from dermatologists to rheumatologists were reported to have an average waiting time of 85 days, 26 prolonging the diagnostic process and potential treatment changes.
Patients’ time spent visiting outpatient clinics for their dermatology and rheumatology consultations nearly halved (11.29 vs 6.51 hours) in our cohort. Reduced frequency of doctor’s visits has been associated with improved quality of life. 27 Moreover, patients reported decreased treatment expenses during the IDRC observation period compared to pre-IDRC spending. Overall patient satisfaction with our IDRC was high, averaging 7.8 out of 10, which is particularly meaningful given the chronic nature of PsA and its substantial impact on the patient’s disease burden.
Although the time between symptom onset and diagnosis was quite long, averaging 187 (±393) weeks, only 5% of patients showed joint erosions, which might be explained by subclinical or mild synovitis prior to diagnosis.
The VAS pain scores in the psoriasis cohort were remarkably high. A possible explanation for this could be that patients were included in the consultation or study specifically to investigate possible PsA due to musculoskeletal complaints, or to clarify the presence of psoriasis in those with pre-existing musculoskeletal inflammation.
Of the 59 patients with psoriasis and musculoskeletal pain, no definitive signs of PsA or other rheumatic diseases were identified. In one case, osteoarthrosis was identified as the primary cause of pain. While some patients may have degenerative diseases or somatoform pain disorders, most did not exhibit symptoms of classic fibromyalgia.
Interestingly, our PsA cohort had higher rates of biologic use than the Pso cohort, despite observational evidence suggesting that biologics may prevent the onset of PsA. 28 We attribute this to indication bias, as patients with PsA (particularly those with non-severe psoriasis) are more likely to receive targeted therapies. Long-term prospective studies are needed to further investigate this association in Pso patients who have not yet developed PsA.
Throughout our study period, disease activity in PsA patients notably decreased. This improvement, together with faster PsA diagnosis compared to conventional consultations, indicates that an interdisciplinary consultation effectively reduces inflammation, preventing long-term joint damage, and improves patients’ quality of life. The IDRC was associated with substantial improvements in rheumatic disease activity scores for PsA patients, a critical aspect as reducing objective disease measures not only enhances life quality but also mitigates long-term joint damage. 11
Limitations
Our study was limited by several factors. Small subgroups resulted in reduced statistical power, potentially resulting in missed effects of interest. Additionally, as an exploratory study, we lacked a “usual care” group for comparison, although we attempted to partially address this by comparing “before” and “after” data, as the status of the patient before the IDRC intervention represented “usual care.”
The study took place during the COVID pandemic, impacting patients’ overall quality of life due to pandemic-related restrictions and effects on mental health. These restrictions also contributed to increased dropouts in our study.
Conclusion
Our study offers valuable insights into the epidemiological characteristics of PsA patients and the impact of interdisciplinary consultations. The IDRC facilitated rapid PsA diagnosis, associated with reduced patient burden and decreased disease activity. This consultation model appears to be a valuable tool to improve the diagnosis and management of PsA patients.
