Abstract
Background:
One-third of intractable epilepsy patients have no visually identifiable focus for neurosurgery based on imaging tests [magnetic resonance imaging (MRI)-negative cases]. Stereo-electroencephalography-guided radio-frequency thermocoagulation (SEEG-guided RF-TC) is utilized in the clinical treatment of epilepsy to lower the incidence of complications post-open surgery.
Objective:
This study aimed to identify prognostic factors and long-term seizure outcomes in SEEG-guided RF-TC for patients with MRI-negative epilepsy.
Design:
This was a single-center retrospective cohort study.
Methods:
We included 30 patients who had undergone SEEG-guided RF-TC at Sanbo Brain Hospital, Capital Medical University, from April 2015 to December 2019. The probability of remaining seizure-free and the plotted survival curves were analyzed. Prognostic factors were analyzed using log-rank tests in univariate analysis and the Cox regression model in multivariate analysis.
Results:
With a mean time of 31.07 ± 2.64 months (median 30.00, interquartile range: 18.00–40.00 months), 11 out of 30 patients (36.7%) were classified as International League Against Epilepsy class 1 in the last follow-up. The mean time of remaining seizure-free was 21.33 ± 4.55 months [95% confidence interval (CI) 12.41–30.25], and the median time was 3.00 ± 0.54 months (95% CI 1.94–4.06). Despite falling in the initial year, the probability of remaining seizure-free gradually stabilizes in the subsequent years. The patients were more likely to obtain seizure freedom when the epileptogenic zone was located in the insular lobe or with one focus on the limbic system (p = 0.034, hazard ratio 5.019, 95% CI 1.125–22.387).
Conclusion:
Our findings may be applied to guide individualized surgical interventions and help clinicians make better decisions.
Keywords
Introduction
Epilepsy affects approximately 70 million individuals worldwide, 30% of whom are intractable.1,2 Resective surgery is an effective treatment for intractable epilepsy3–6; however, postoperative complications are uncommon but should not be ignored. 7 One of the most critical procedures for its preoperative evaluation is the delineation of the epileptogenic zone (EZ), where magnetic resonance imaging (MRI) plays a key role in identifying potential EZ. Nevertheless, one-third of patients have no visually identifiable focus for neurosurgery on MRI, for which they are labeled MRI-negative cases. 8
Despite the increasing accuracy in the identification of EZ in noninvasive tests,9,10 stereo-electroencephalography (SEEG) evaluation was also seen to be advantageous in delineating EZ since it was first applied by Bancaud and Talairach in the 1960s, 11 especially in MRI-negative patients. 12 SEEG-guided radiofrequency-thermocoagulation (SEEG-guided RF-TC) is a minimally invasive surgical procedure that can be performed directly after SEEG without causing additional risks. Compared to conventional resection surgery, SEEG-guided RF-TC was seen to have lower complication rates, better preservation of cerebral function, and shorter hospitalization.13,14 In contrast with other minimally invasive treatments, SEEG-guided RF-TC remains distinctive because of the convenience of performing it post-SEEG and the unique advantages of SEEG in MRI-negative patients. 15
Some studies13,14,16–22 have demonstrated the efficacy and safety of SEEG-guided RF-TC, especially in hypothalamic hamartomas (HH) 21 and nodular heterotopy, such as periventricular nodular heterotopias (PNHs). 18 Despite some studies reporting that the seizure outcome in the MRI-negative group was worse than that in the MRI-positive group,13,23 the seizure outcomes and prognostic predictors of SEEG-guided RF-TC in MRI-negative patients remain unclear. In response to this, we reviewed data from MRI-negative patients who underwent SEEG-guided RF-TC at the epilepsy center of the institution to further determine the seizure outcomes and prognostic predictors associated with this procedure.
Methods
Patient selection
This retrospective cohort study was conducted using detailed data from all MRI-negative patients who had undergone SEEG-guided RF-TC between April 2015 and December 2019 at the epilepsy center in Sanbo Brain Hospital, Capital Medical University. The inclusion criteria were as follows: (1) patients whose preoperative MRI was negative, (2) patients who underwent SEEG-guided RF-TC in our center, and (3) patients who were followed up for more than 1 year. Patients who lacked data on the electrodes of SEEG or SEEG-guided RF-TC, those who had undergone surgery before SEEG-guided RF-TC, and those who were lost to follow-up were all excluded from this review. This study included all eligible patients who underwent SEEG-guided RF-TC procedures in our center since its launch. As a result, a power analysis was not conducted to justify the sample size chosen for this study.
This study was compliant with the principles outlined in the Declaration of Helsinki and was approved by the Ethics Committee of Sanbo Brain Hospital, Capital Medical University (SBNK-2017-15-01).
MRI-negative definition
All patients underwent scans using either a 1.5-T or 3.0-T MRI (Philips) before proceeding with SEEG. The MRI scan included axial T1-, T2-, diffusion-weighted imaging, coronal, sagittal, and axial T2 fluid-attenuated inversion recovery images. In this study, we defined ‘MRI-negative epilepsy’ as a disorder where recurrent unprovoked seizures occur with or without secondary generalization in the absence of an epileptogenic lesion on visual inspection of the MRI. 24 We first collected the patients whose MRI imaging had no abnormal findings referencing the reports from the neuroradiologists and then confirmed again by one neuroradiologist, one neurologist, and three neurosurgeons. These MRI results were reported independently and blindly by two neuroradiologists. However, these two neuroradiologists had viewed the patients’ clinical information before reporting the MRI results.
Preoperative evaluation
To locate the EZ, previous medical records of patients who underwent SEEG-guided RF-TC were obtained and reviewed, and a comprehensive evaluation of their detailed demographic and clinical variables was conducted. Epilepsy types were assessed according to the patient’s complaints and based on the International League Against Epilepsy (ILAE) classification of epilepsies25,26 by two neurologists.
Noninvasive tests aside from MRI were initially performed, and these included computed tomography (CT), long-term scalp video-electroencephalography (VEEG), magnetic source imaging (MSI), and 18 F-fluorodeoxyglucose positron emission tomography (PET). A 64- or 128-channel scalp VEEG used the standard 10–20 system of electrodes, where a neurophysiologist reported the results, and another expert reviewed them. Every scalp VEEG recording included more than three habitual seizures, and VEEG pattern definitions were based on the study by Jeha et al. 27 The magnetoencephalography (MEG) used a 306-channel whole-head system (Elekta Neuromag® 306-channel MEG systems, Finland), which runs for 60 min per patient and requires more than five spikes for a localization result. The MEG image was then co-registered with the preoperative MRI to evaluate the anatomic location (MSI). Patients who underwent PET scanning (PET/CT PoleStar m660, Sinounion, China), on the other hand, were seizure-free for at least 2 days before the procedure.
The invasive evaluation was done based on the following indications stated in the French Guidelines on SEEG 28 : (1) EZ could not be identified using noninvasive tests; (2) EZ overlapped with eloquent cortical areas; and (3) the necessity to evaluate the possibility of resection or SEEG-guided RF-TC. Every trajectory blueprint of SEEG was designed by one neurologist and two neurosurgeons based on the results of the noninvasive tests. The basic rule was to not only cover and delineate the boundaries of every region of interest as well as its pathway of discharge transmission but also to avoid critical vessels to ensure the safety of the SEEG blueprint.
Intracranial electrodes implantation
Electrodes (5–18 contacts, length: 2 mm, diameter: 0.8 mm, 1.5 mm apart, Depth Coagulation Electrodes, ALCIS, France) were implanted with the aid of a robotic stereotactic assistant (ROSA [Medtech, Montpellier, France]) based on the trajectory planning results and was performed under general anesthesia. CT was performed 6 h postoperatively to confirm the placement of every electrode in the targeted space and check for the presence of intracranial bleeding.
Subsequently, all patients were monitored using a 128-channel VEEG (Nicolet, USA) post-implantation. After capturing at least three habitual seizures, a session of functional cortical and subcortical mappings was performed to reproduce the clinical ictal manifestations entirely or in part. 23
Once the monitoring was completed, the recorded material was viewed by two neurophysiologists, one of whom recorded the results while the other reviewed it. The ‘focus of the limbic system’ was defined through ictal SEEG results showing that, besides the insular lobe, only one electrode recorded the epileptiform discharges located at the specific neuroanatomical site of the limbic system: the cortex contained in the great limbic lobe of Broca (the olfactory cortex, prepyriform area, hippocampal gyrus and hippocampus, and the parasplenial, cingulate, and subcallosal gyri) as well as ‘nuclear structures’ (including the amygdala, septal nuclei, hypothalamus, epithalamus, anterior thalamic nucleus, and parts of the basal ganglia). 29 In addition, irritative zone, seizure onset zone (SOZ), and EZ were roughly the same as those defined in the study by Rosenow and Luders. 30
Radiofrequency-thermocoagulation
According to the recordings made by the SEEG, SEEG-guided RF-TC was offered to patients in whom 31 (1) the EZ overlapped with eloquent cortical areas, (2) the EZ was very focal (optional treatment), or (3) the EZ site was located deeply or widely (palliative treatment). The basic function of the anatomic targets of SEEG-guided RF-TC is to facilitate the selective destruction of the EZ or critical nodes in the epileptogenic network. The site was considered eligible for SEEG-guided RF-TC through SEEG results showing bipolar recordings through two adjacent contacts of the same electrode as well as evidence of spike-wave discharges or low-amplitude fast patterns at the SOZ or crucial nodes of the network transmitting, 16 and ‘critical nodes’ were defined as those the contacts monitored with low-amplitude fast pattern in the onset and early time.
In brief, the SEEG-guided RF-TC procedure used a radio-frequency generator system (RF Lesion Generator R-2000B M1, Beiqi, Beijing, China) that connected the selective electrodes using the following parameters: progressively raised current power from 1.5 W up to 8.32 W within 60 s and the current intensity and voltage changes were determined by the delivered power. Then, we were expecting that the impedance rapidly increased until they spontaneously collapsed. These parameters could increase the tissue temperature to 78–82°C and produce an ovoid lesion with a long axis of approximately 7 mm between the two selective contacts.17,23,32 SEEG was performed to observe changes in the epileptiform discharges (ED) pre- and post-SEEG-guided RF-TC. CT was also performed 24 h after the procedure, and if the patient was assessed to be comfortable, they were subsequently discharged within 1–3 days. All patients were prescribed mannitol or steroids. Incidentally, SEEG-guided RF-TC was performed in the operating room without anesthesia.
Follow-up and seizure outcomes
After SEEG-guided RF-TC, patients who still had seizure episodes were advised to undergo open surgery 3 months later. The region of resection was based on the EZ boundaries identified by the SEEG, which also contains the SEEG-guided RF-TC lesion. At the beginning and end of the surgery, the surgeons used electrocorticography to confirm the extent of surgery to be done and if the resection was complete. In these patients, we assumed that the seizure outcomes of SEEG-guided RF-TC were concordant with the results of the last follow-up before open surgery in the univariate and multivariate analyses.
Post-SEEG-guided RF-TC patients who remained seizure-free were scheduled to visit after the first 3 months, 6 months, and 1 year postoperatively, followed by yearly visits as required. The last follow-up was completed by telephone, or by re-examination at the outpatient clinic, in December 2020. The follow-up assessment included seizure outcomes, seizure type and frequency, the period from surgery to the first seizure occurrence post-SEEG-guided RF-TC, use of anti-epileptic drugs, and occurrence of other complications. Seizure outcomes were assessed according to the ILAE seizure outcome classification 33 and seizure freedom was defined as ILAE class 1. Responders were defined as those with at least a 50% reduction in seizure frequency as compared with the pre-procedure period. 13 In addition, to reduce the location error due to postoperative cerebral edema [Figure 1(f)], we used MRI imaging after 3 months [Figure 1(g)] to determine the cerebral regions.
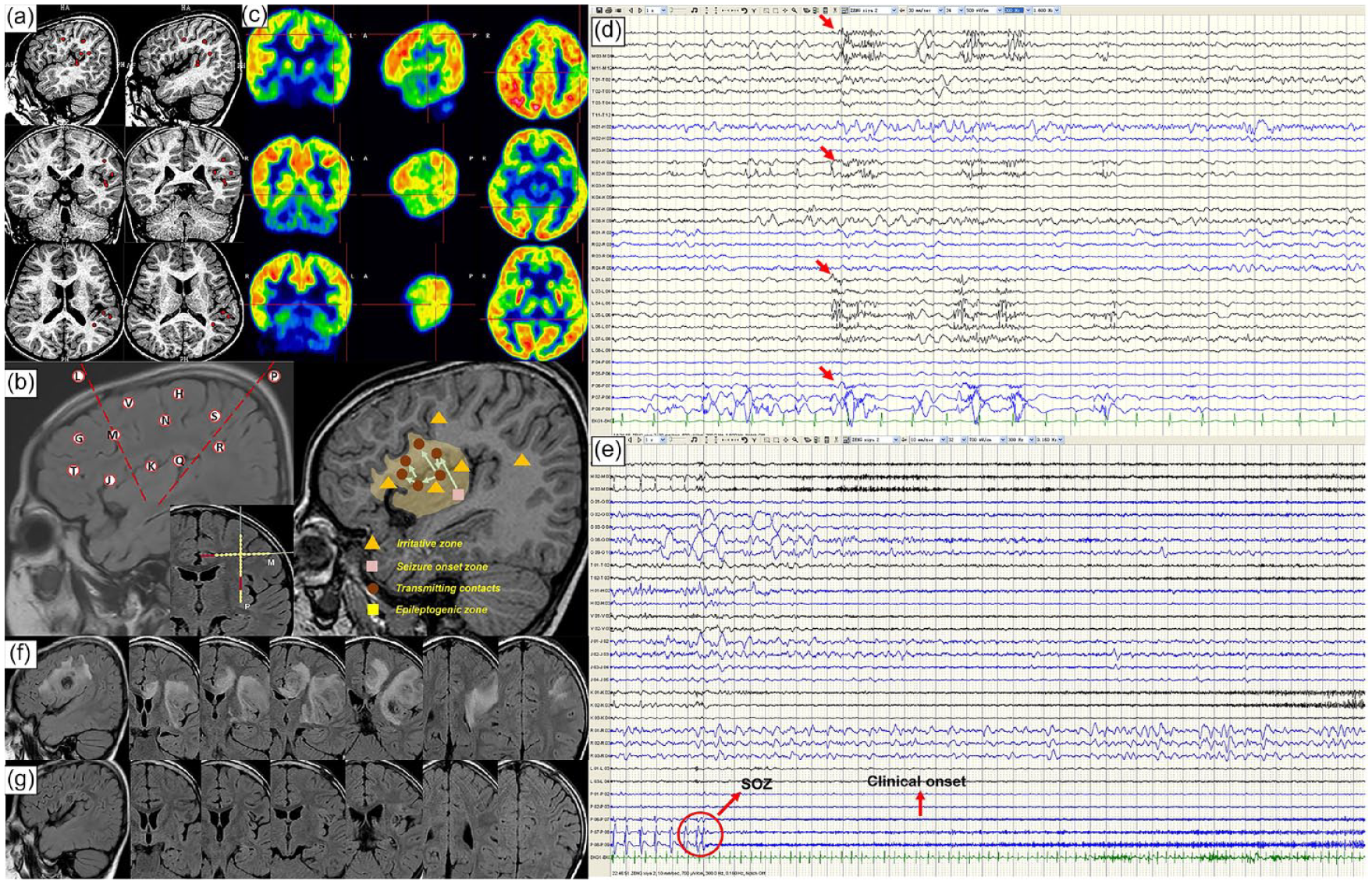
Illustration of some pre- and postoperative data for patient 5. An approximate 6-year-old right-handed female with a 72-month history of intractable epilepsy visited our center for preoperative evaluation. (a) MSI showing no significant epileptiform discharges [dispersedly involving the left posterior lateral fissure around (red plots)] and PET showing significant hypometabolism in the left frontal and temporal lobe. (b) The general sites of every electrode in MRI scanning, and K, L, M, and P performed SEEG-guided RF-TC. The red contacts mean the selective contacts of SEEG-guided RF-TC in M and P electrodes. (c) Electroclinical data elicited by SEEG allowing us to define the irritative zone (yellow triangles), the seizure onset zone (pink quadrangle), the transmitting contacts during the ictal period (brown plots), and the epileptogenic zone (blonde area). (d) Interictal SEEG showing some spike-wave discharges (red arrow) involving abundant areas. (e). Ictal SEEG showing the seizure onset zone characterized by the low-amplitude fast pattern (red circle). (f) and (g) Showing the MRI imaging post-SEEG-guided RF-TC at 3 days and in 3 months, respectively.
Statistical analysis
For the baseline characteristics, including detailed demographics and clinical data, numerical qualitative variables were described using median [interquartile ranges (IQRs)] and compared using the Mann–Whitney U test due to small sample sizes. Categorical variables were described using frequencies (percentages, %) and compared using Pearson’s or Fisher’s exact chi-square test, if necessary. The parameter of survival analysis is ‘time to event’. We defined ‘time’ as the period from SEEG-guided RF-TC to the first postoperative seizure in the non-seizure-free group and to the time of the last follow-up in the seizure-free group, and ‘event’ as the patient’s relapse post-SEEG-guided RF-TC. Kaplan–Meier analysis was used to analyze the estimated cumulative probability of remaining seizure-free and plotted survival curves compared by log-rank (Mantel-Cox) tests.
To analyze prognostic predictors, a Cox regression model was applied. All variables associated with seizure freedom by a p value ⩽ 0.2 in univariable Cox regression analysis were included in the multivariable Cox regression model. A two-sided p value < 0.05 was considered statistically significant in all comparisons. Statistical analyses were performed using IBM SPSS version 24 (IBM Corp., Armonk, NY, USA).
Results
We analyzed a total of 425 patients who underwent SEEG at the comprehensive epilepsy center of Sanbo Brain Hospital, Capital Medical University, from April 2015 to December 2019. After the selection process, as shown in Figure 2, a total of 30 patients were included in our cohort. The partial demographics and SEEG data are shown in Table 1. The preoperative MRI, intracranial electrode implantation design, and the results of SEEG monitoring, as well as the post-SEEG-guided RF-TC MRI of patient 5, are shown in Figure 1 for reference.

The flow chart describes the procedure of exclusion and inclusion in our study.
The partial demography and SEEG data of the 30 patients.

a, anterior; B, bilateral; Cg, cingulate gyrus; Cp, central-parietal; EZ, epileptogenic zone; F, frontal; FM, female; FU, follow-up; GTCS, generalized tonic-clonic seizure; H, hippocampus; i, inferior; I, insular; ILAE, International League Against Epilepsy seizure outcome classification; l, lateral; L, left; M, male; m, mesial; No., number of; O, occipital; Op, opercular; P, parietal; p, posterior; Pl, paracentral lobule; Pt., patient; R, right.; s, superior.; SEEG, stereo-electroencephalography.; SEEG-guided RF-TC, SEEG-guided radio-frequency thermocoagulation; SMA, supplementary motor area; T, temporal.; VNS, vagal nerve stimulation.
Baseline of patients’ characteristics
In our cohort, 60% (18/30) of the patients were male, and 40% (12/30) were female. All patients were followed up for more than 1 year, with a mean time of 31.07 ± 2.64 months (median 30.00, IQR: 18.00–40.00 months). The mean age at seizure onset was 150.70 ± 20.72 months (median 123.50, IQR: 48.75–264.50), and the mean age at SEEG-guided RF-TC was 212.50 ± 22.18 months (median 192.00, IQR: 90.25 ± 303.00). The mean number of SEEG-guided RF-TC electrodes was 3.43 ± 0.27 (median 3.00, IQR: 2.00–4.25), and the mean number of SEEG-guided RF-TC pairs was 9.50 ± 1.29 (median 7.50, IQR: 4.00–13.00).
The detailed baseline demographic and clinical characteristics are summarized in Table 2 and classified under seizure freedom or non-seizure freedom. The risk factors for epilepsy were made up of three patients with viral encephalitis, two with febrile convulsion, and one with head trauma in this cohort. Three patients did not undergo MSI. Upon seizure outcome analysis, only the characteristics of the anatomic targets of the SEEG-guided RF-TC in the EZ (p = 0.004) were statistically significant. By contrast, no statistical significance was observed between the other characteristics and seizure outcome.
Baseline clinical characteristics of the population study according to the seizure outcome post-SEEG-guided RF-TC.

Numerical variables were described by the median (interquartile range, IQR), and categorical variables by frequencies (percentages, %). p Values of numerical variables were shown for the Mann–Whitney U test, and Pearson or Fisher’s exact chi-square tests were used in categorical variables.
Bold characteristics are statistically significant values.
Mann–Whitney U test. $Fisher’s exact chi-square test. ‡Pearson chi-square test.
AEDs, anti-epilepsy drugs; ET, 18 F-fluorodeoxyglucose positron emission tomography; MSI, magnetic source imaging; NED, no epileptogenic discharge; No., number of; NSF, non-seizure-free; SEEG, stereo-electroencephalography; SEEG-guided RF-TC, SEEG-guided radio-frequency thermocoagulation; SF, seizure-free; SOZ, seizure onset zone; VEEG, video-electroencephalography.
Seizure outcomes and complications
Figure 3(a) shows seizure outcomes in the cohort at 3 months, 6 month, 1 year, and the last follow-up. By the last follow-up, 11 of 30 patients (36.7%) were ILAE class 1, nine patients (30.0%) were ILAE classes 4 and 5, and one patient (3.3%) was ILAE class 6. No patient was in ILAE classes 2 and 3. Notably, among the 19 patients who still had seizure episodes post-SEEG-guided RF-TC, 12 underwent secondary surgery (1 vagal nerve stimulation and 11 secondary open surgery), where the seizure outcomes of SEEG-guided RF-TC were defined as the results of the last follow-up before secondary surgery.
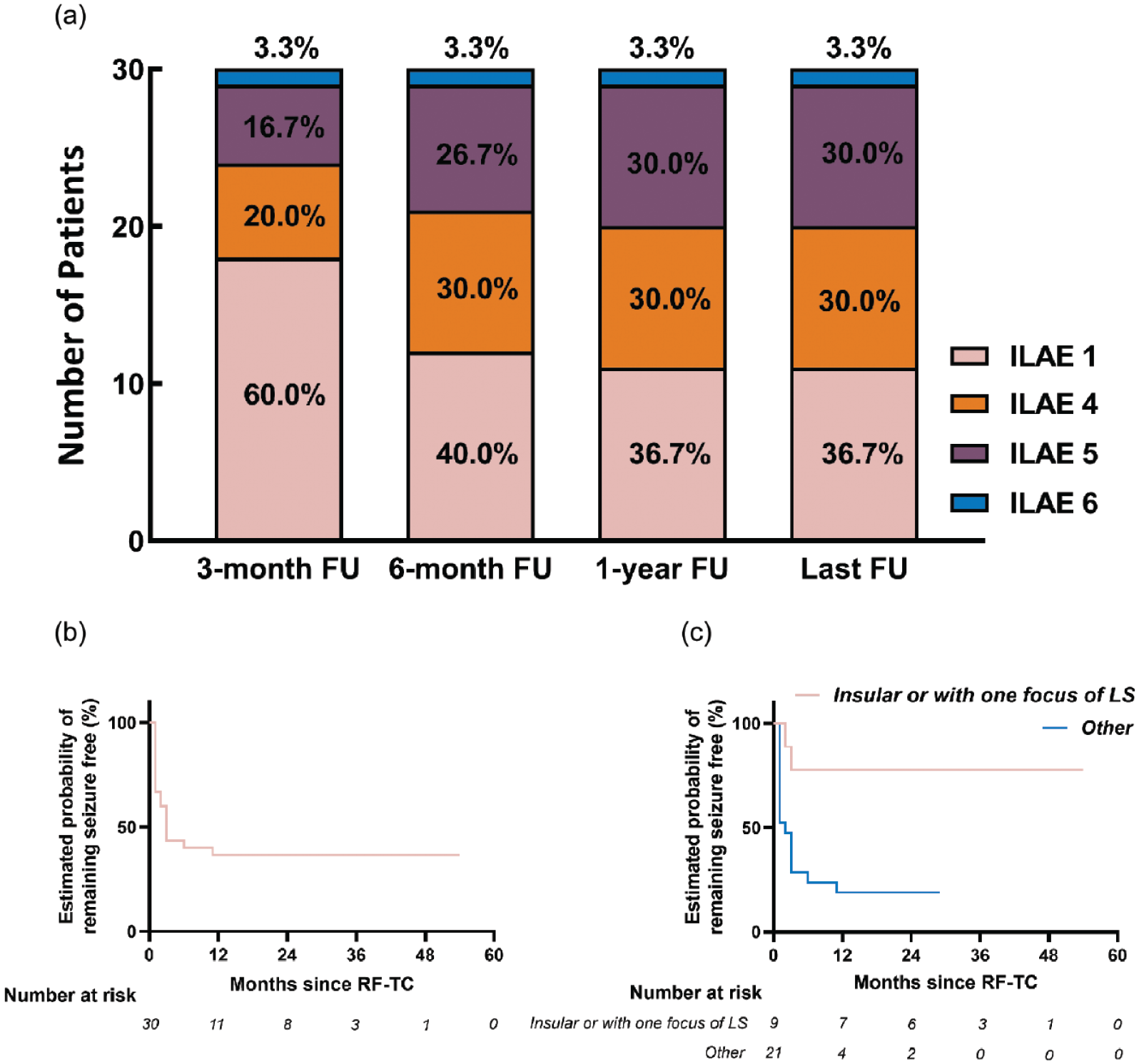
Seizure outcomes SEEG-guided RF-TC according to ILAE classification. (a) Illustrating the outcomes at 3 months, 6 months, 1 year, and the last FU, respectively. (b) Illustrating the survival curve for seizure freedom for the cohort of all 30 patients in this study. (c) Illustrating the survival curves for seizure freedom for anatomic targets of the SEEG-guided RF-TC in the epileptogenic zone.
The Kaplan–Meier method was used to estimate the cumulative probability of remaining seizure-free and plotted survival curves, as summarized in Supplemental Table A.1. The probabilities at 3-month, 6-month and 1-year follow-up were 43.3% [95% confidence interval (CI) 25.66–60.94], 40.0% (95% CI 22.56–57.44), and 36.7% (95% CI 19.45–53.95), respectively. The mean time of remaining seizure freedom post-SEEG-guided RF-TC was 21.33 ± 4.55 months (95% CI 12.41–30.25), and the median time was 3.00 ± 0.54 months (95% CI 1.94–4.06). In addition, despite falling in the initial year, the probability of remaining seizure-free gradually stabilizes in the subsequent years. Figure 3(b) shows the survival curve for seizure freedom among the 30 patients included in this cohort.
The detailed seizure outcomes after open resection are shown in Supplemental Table A.2. The positive predictive value (PPV) of being responders 3 months after SEEG-guided RF-TC and the seizure-free rate post-open surgery was 100%; however, there was no statistically significant difference between the responders’ post-SEEG-guided RF-TC and the outcomes after open surgery (p = 0.194).
Furthermore, none of these 30 patients had permanent neurologic or cognitive complications from the perioperative period to the last follow-up.
Prognostic analysis
In the univariate analysis, Supplemental Table A.3 shows the results between independent characteristics and seizure outcomes by log-rank tests. The following characteristics were seen to statistically influence the seizure outcomes: age at onset (p = 0.027), age at surgery (p = 0.029), number of SEEG electrodes (p = 0.029), and anatomic targets of the SEEG-guided RF-TC in the EZ (p = 0.004). No statistically significant association between the other characteristics and seizure outcome was observed. Figure 3(c) shows the survival curves for seizure freedom according to the anatomic targets of the SEEG-guided RF-TC in the EZ.
In the multivariate analysis, we performed univariate Cox regression analysis first, and then all statistically significant and adjusted variables with p values ⩽0.2 were included in the multivariate Cox regression model. The final model included anatomic targets of the SEEG-guided RF-TC in the EZ [p = 0.024, hazard ratio (HR) 5.483, 95% CI 1.252–24.020] and was adjusted for PET (p = 0.191, HR 1.842, 95% CI 0.737–4.601) (Supplemental Table A.4). The multivariate Cox regression analysis in Table 3 shows that only the anatomic targets of the SEEG-guided RF-TC in the EZ were seen to be statistically significant (p = 0.034, HR 5.019, 95% CI 1.125–22.387).
p Values, HRs, and 95% CIs of variables correlating with postoperative seizure-freedom in univariate and multivariate Cox regression analyses.
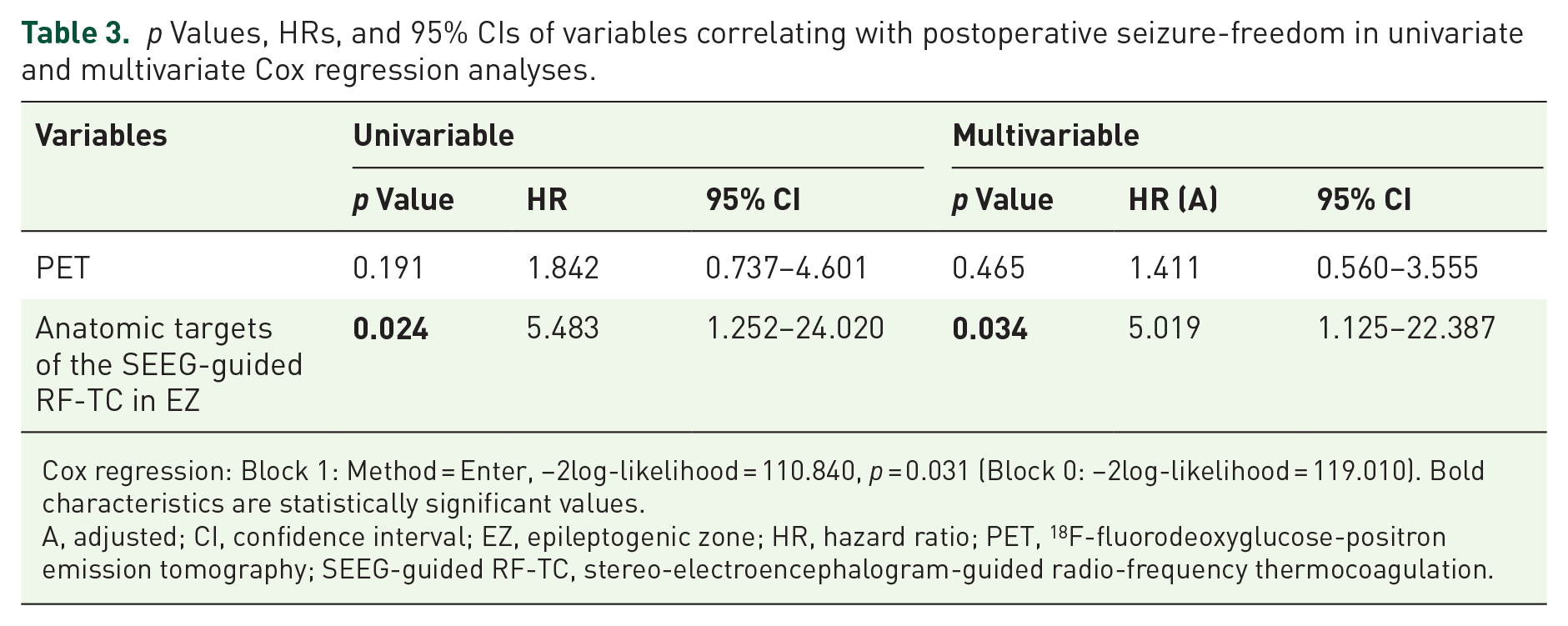
Cox regression: Block 1: Method = Enter, −2log-likelihood = 110.840, p = 0.031 (Block 0: −2log-likelihood = 119.010). Bold characteristics are statistically significant values.
A, adjusted; CI, confidence interval; EZ, epileptogenic zone; HR, hazard ratio; PET, 18 F-fluorodeoxyglucose-positron emission tomography; SEEG-guided RF-TC, stereo-electroencephalogram-guided radio-frequency thermocoagulation.
Discussion
Efficacy and safety
In this study, we demonstrated the efficacy of SEEG-guided RF-TC in patients with MRI-negative intractable epilepsy. Approximately two-fifths of patients could obtain long-term seizure freedom; meanwhile, it has gradually stabilized in the subsequent years, despite some falling in the initial year.
So et al. 34 believed that, during the whole preoperative evaluation, the understanding of MRI results was the basis of developing plans to identify the EZ and the range of resection and predict the seizure outcomes. Namely, we took it for granted that 1.5T MRI-negative patients should be reserved since these patients were treated as MRI negative during the whole evaluation. Thus, 1.5T MRI-negative patients in this cohort were finally reserved. Compared with lower field strengths, 7T MRI has an increased signal-to-noise ratio and susceptibility effects, thereby providing better image contrast, higher spatial resolution, and stronger susceptibility contrast. 35 Complete resection of the 7T-identified lesion was associated with seizure freedom, and 7T MRI post-processing simultaneously yielded 25% more lesions than 3T MRI post-processing and showed noticeable improvement in 46%. 36 Unfortunately, data on 7T MRI could not be reported in our study because we lacked the relevant equipment in our center. The authors are devoted to expanding the application of 7T MRI to future patients as soon as possible.
Previous studies13,15,17,18,21,23,37 have reported that SEEG-guided RF-TC could be an alternative treatment for epilepsy, especially in those with minimal foci, including HH 21 and PHNs. 18 However, the outcomes from the MRI-negative group were unsatisfactory.13,23 Cossu et al. 23 found that in the non-lesional group, five of 46 patients (10.9%) obtained seizure freedom, and the other five patients (10.9%) were considered as having sustained worthwhile improvement. Our results were better compared to the findings of Cossu et al.; 23 one potential reason for better outcomes was that the lesion was too highly circumscribed to visually identify by 1.5-T or 3.0-T MRI but could be completely destructed. In addition, better results could be contributed to the progressive understanding and development of SEEG-guided RF-TC techniques and also reflect the importance of candidates, thus including a more reasonable patient selection process when EZ was very focal. However, our SEEG-guided RF-TC results had less seizure freedom outcomes compared to the approximately 59% of seizure freedom in open operation for MRI-negative epilepsy, 38 also less than the overall range of 40–60% among patients with MRI-negative TLE,39,40 46% (12/26) among patients with 3T MRI-negative focal epilepsy by Kogiasa et al. and 58.3% (28/48) in 1, 2, and 5 years postoperatively, respectively, by Mariani et al., 41 potentially because without visually identifiable focal clues, trajectory blueprint of SEEG depended on the patients’ semiology, and other noninvasive tests (VEEG, MSI, and PET), which may not optimally define the EZ but only the sample portion. In other words, the real EZ of the worsening patients might not be destroyed completely. Meanwhile, that is the reason why some patients responded initially, but later the seizures worsened, thereby requiring secondary open surgery.
Compared to SEEG-guided RF-TC, RF-TC could be performed as a secondary or stand-alone procedure with dedicated RF probes after SEEG, which might achieve a more complete or larger volume of ablation. Dimova et al. 31 suggested that RF-TC was offered to patients eligible for resective surgery and in whom the EZ seemed very focal (optional treatment). Overall, the proportion of seizure freedom was 39% after SEEG-guided RF-TC in a recent meta-analysis. 42 However, the proportion could be increased to 53% 20 or 76.2% 14 if selected patients received SEEG-guided RF-TC as an optional treatment after the EZs had been identified. Notably, in consideration of the possibility of performing SEEG-guided RF-TC, the authors designed every trajectory blueprint optimally to achieve the greatest volume of thermos-lesions. SEEG-guided RF-TC was not performed as an optional treatment in most patients due to a lack of lesions on MRI. In addition, the long-term probability of seizure freedom was 36.7% in our study, which was significantly lower than the efficacy when RF-TC was treated as a curative method and slightly lower than in the meta-analysis.
In addition, the results of the earlier meta-analysis 13 showed that the mean ± SD number of RF-TCs performed in each patient ranged from 5 ± 4.3 to 12.5 ± 9.9 at the study level and from 1 to 71 at the patient level. The latest meta-analysis 42 had no data on the number of RF-TCs. The number of RF-TCs in our study was 9.50 ± 1.29 (median 7.50, IQR: 4.00–13.00), which was slightly different from the previous study. We designed every trajectory blueprint optimally to achieve the greatest volume of thermos-lesions in consideration of the possibility of performing SEEG-guided RF-TC, which might have resulted in quantitative differences in the results. Meanwhile, Gao et al. 43 reported that the mean ± SD number of RF-TC contacts was 18.6 ± 3.2 (median 13.0, range 3–52). In 19 MRI-negative patients, the probability of seizure freedom and response at 1 year after SEEG-guided RF-TC was 15.8% (95% CI 5.6–44.6%) and 52.6% (95% CI 34.4–80.6%), respectively. Meanwhile, the group of 10 patients with MEG clusters was 30% (95% CI 11.6–77.3%), which was slightly lower than the 36.7% in our study; it is possible this difference is due to the small sample size.
In terms of responders, our study had almost the same results as that of the study by Bourdillon et al. 16 The PPV in our study suggested that the responders from SEEG-guided RF-TC have an increased chance of achieving seizure freedom after secondary open surgery; however, no statistically significant association between responders and seizure outcome was seen due to the small sample sizes (only 11 patients underwent secondary resection). Incidentally, ‘the ramping down period’, where patients had some seizures after the procedure and became seizure-free later, was not observed during the duration between SEEG-guided RF-TC and open surgery. We speculated that the reason might be that SEEG-guided RF-TC efficacy should be immediate, while the latency to seizure freedom is only a consideration for radiosurgery (gamma knife) treatment because it takes time for the radiation exposure to result in the lesion.
Generally, the patients were prescribed mannitol or steroids according to the complaints of symptomatic raised intracranial pressure, the volume of thermos-lesions, and the range of post-thermocoagulation edema. In consideration of the possibility of performing SEEG-guided RF-TC, the authors designed every trajectory blueprint optimally to achieve the greatest volume of thermos-lesions. Hence, we suspected that peri-lesion edema may be caused by the large number of RF-TC pairs and three-dimensional cross-bonding RF-TC.
The treatment of EZ located at the eloquent cortex has always been challenging. In our study, no permanent neurologic or cognitive complications were observed in any patient since SEEG-guided RF-TC was guided by SEEG and performed in minimal cortical sites. To ensure the safety of SEEG-guided RF-TC and secondary open surgery when EZ is located at the eloquent cortex, the patients would perform a sodium amytal test, or WADA test, to evaluate whether function had been transferred to other areas. Moreover, before the standardized SEEG-guided RF-TC, the patients would undergo preliminary SEEG-guided RF-TC (2.0 W) with low power to confirm those without any complaints. In addition, some complications (e.g. when patients have visual field defects, some cognitive disorder, or neurological dysfunction that they were unaware of) would be detected by only standard tests but overlooked through a telephone interview. Lastly, the focal stimulation of SEEG-guided RF-TC did not elicit any clinical response due to its small cortical involvement. 15 Hence, SEEG-guided RF-TC can be considered a potential option, with a favorable risk-benefit ratio, in the treatment of MRI-negative epilepsy with an EZ located close to a functional area or in an area that is poorly accessible to conventional operation (e.g. insular lobe).
Prognostic analysis
We tested all the potential characteristics by log-rank tests in the univariate analysis and the Cox regression model in the multivariate analysis. The results showed that the characteristics of the SEEG-guided RF-TC anatomic targets in the EZ located in the insular lobe or with one focus of the limbic system were statistically significant in both univariate and multivariate analyses.
With the involvement of the insular lobe, it is crucial to locate the EZ accurately by SEEG for MRI-negative epilepsy patients. Due to the intricate connection between the insular lobe and other brain areas as well as its anatomical location in the depth of the Sylvian fissure, 44 patients with insular lobe epilepsy could present with abundances of semiological features and its EZ can be poorly accessible to conventional operation. Therefore, Alomar et al. 45 suggested that insular lobe epilepsy was the most common indication for intractable epilepsy by SEEG. Meanwhile, Mullatti et al. 20 illustrated the efficacy of RF-TC in the insular lobe and suggested that the optimal volume of RF-TC, which is around 2 cm3, offered the best compromise between efficacy and safety. Hence, combined with our results and the convenience of SEEG-guided RF-TC, SEEG-guided RF-TC can be considered as an alternative treatment for MRI-negative epilepsy with insular lobe-located anatomic targets.
Goddard et al. 46 found that not all brain areas had the capability to be kindled by stimulation, but the limbic system was seen to be the most responsive. These results indicate that the limbic system contains connections with other areas, including the insular lobe. Therefore, we speculated that EDs are transmitted to the symptomatogenic zone and generate relative seizures through the abundance of underlying networks between the insular lobe and the limbic system. When the surgeons destroyed the critical nodes completely in the insular lobe or with the focus on the limbic system, the transmission within the networks was cut off. However, due to the limitation of lesion volume covered by SEEG-guided RF-TC (diameter 7 mm per contact), destruction of the critical nodes is hardly achieved when the EZ includes more than two foci of the limbic system.
Mullatti et al. 20 suggested that the optimal volume of RF-TC, which is around 2 cm3, offered the best compromise between efficacy and safety. Perry et al. 47 reported the seizure outcomes of 20 children with insular epilepsy who performed laser interstitial thermotherapy (LiTT), of which 70% were MRI negative. At a mean follow-up of 20.4 months, 50% of the children achieved seizure freedom. Unfortunately, the authors did not elucidate the predictive factors. Moreover, a meta-analysis 48 showed that LiTT had a higher rate of seizure freedom than SEEG-guided RF-TC [65% (95% CI 56%–74%) versus 23% (95% CI 10%–39%)], whose reason might be that the volume of lesion ablation by LiTT is larger than SEEG-guided RF-TC. Our results showed no statistically significant association between the number of SEEG-guided RF-TC electrodes or pairs and seizure outcomes. The potential reason is that to obtain a larger volume of SEEG-guided RF-TC lesions, we added two contacts, which were adjacent to the contacts of fast activity as the target of SEEG-guided RF-TC. This method may reduce the statistical weight of the patients who could have obtained seizure freedom by destroying a smaller volume. Hence, we have difficulty in confirming the relationship between the volume of insular ablation and seizure outcomes. However, the reason for an MRI-negative result may indicate that the lesion was too highly circumscribed to visually identify by 1.5-T or 3.0-T MRI. Furthermore, the seizure outcomes in our study were better than the results of the meta-analysis by Bourdillon et al. 13 Hence, to our experience, we agree with the view by Mullatti et al. 20 that, under the premise of accurately identifying the EZ, patients were more likely to obtain satisfactory outcomes when the EZ was highly circumscribed epileptogenic lesions. In other words, the possible cause for 11 patients’ seizure freedom in our study may be that the EZs were minimal such that SEEG-guided RF-TC covered them either completely or ‘a hard core, lowest threshold epileptogenic area’. Conversely, incomplete thermos-lesions were insufficient for seizure freedom, and the remaining patients relapsed after SEEG-guided RF-TC.
Comparison with other treatment options
The development of stereotactic minimally invasive surgery is progressing49–57 due to the higher incidence of postoperative complications in open surgery. However, open surgery still remains one of the most effective treatments for intractable epilepsy patients.3–6
To our knowledge, there are no studies concerning MR-guided LiTT (MRgLiTT) for MRI-negative refractory epilepsy. Gireesh et al. 58 reported the seizure outcomes of nine MRI-negative patients with insular epilepsy and/or cingulate gyrus epilepsy who had performed LiTT. In this study, six of the nine patients (66.7%) achieved seizure freedom after LiTT, including four of five patients (80%) with insular epilepsy. SEEG-related complications in nine patients, including one case of asymptomatic subdural hematoma, one case of Intraparenchymal bleed, and one case of cerebrospinal fluid leak. No permanent deficits were noted after LiTT. In our cohort, seven of nine insular patients (77.8%) (insular lobe or with one focus of the limbic system, Table 2) were seizure-free. A meta-analysis study 59 attempted to compare minimally invasive with traditional open surgery for refractory mesial temporal lobe epilepsy. The results showed that the patients who reached Engle class 1 had no statistical significance between MRgLiTT and SEEG-guided RF-TC, and the proportion of the patients was 57% and 44%, respectively. In addition, the rate of complications was 3.8% and 3.7%, respectively. However, there is a tendency toward more favorable outcomes with MRgLiTT than SEEG-guided RF-TC. Furthermore, the long diameter of the lesion by MRgLiTT ranges from 20 to 25 mm, and surgeons could monitor it in real time in case the temperature is higher than the warning critical point. Therefore, due to the convenience, we recommend SEEG-guided RF-TC when EZ could be destructed completely after evaluation; otherwise, MRgLiTT may be a better option.
In brief, compared to other minimally invasive surgeries, SEEG-guided RF-TC advantages and conveniences are as follows: (1) Abundant information could be obtained from SEEG to better delineate the boundaries of the EZ, especially in MRI-negative epilepsy patients whose EZ is hardly located by noninvasive evaluation so that almost every patient was advised to perform SEEG; (2) SEEG-guided RF-TC can be performed directly post-SEEG, which avoids changes in the primary location due to the secondary placement of the therapeutic apparatus; and (3) SEEG-guided RF-TC can be performed without anesthesia, which decreases the overall risk of the procedure.
However, some limitations of SEEG-guided RF-TC remain to be unsolved. One of its major limitations is the coagulated volume per contact-making SEEG-guided RF-TC, a palliative treatment option in most cases. Fan et al. 14 designed an optimized blueprint containing three-dimensional cross-bonding of adjacent contacts in electrodes for mesial temporal lobe epilepsy with hippocampal sclerosis, which may provide more ideas for optimization. Another limitation is the difficulty of pathological diagnosis in recurring patients that require secondary surgery due to the SEEG-guided RF-TC lesion. Hence, combined with only 58% of responders, 13 we conservatively suggested that SEEG-guided RF-TC should be performed only after meeting the inclusion criteria and in SEEG-guided RF-TC candidates when the EZs were very focal, or some other MRI-positive patients (HH, PNHs, tuberous sclerosis complex with focal and explicit epileptogenic tuber, and focal cortical dysplasia with small volume) in case that the confirmation of pathological diagnosis was not available, despite the results showed a PPV of >90% in both our study and that of Bourdillon et al. 16
Limitations
Several limitations were encountered in the conduct of this study. First, due to the lack of visually identifiable focal clues, the trajectory blueprint of SEEG depended on the patients’ semiology and other noninvasive tests (VEEG, MSI, and PET), which may not optimally define the EZ but only the sample portion. More importantly, our study is a single-center retrospective study wherein the method of acquiring seizure outcomes was through a telephone interview conducted with patients and their families; hence, the bias of follow-up is unavoidable. In addition, in the clinical characteristics of Anatomic targets of the SEEG-guided RF-TC in EZ (Table 2), the subgroup, Other, was a rough portion and the sample size was too small, which might open to bias. Besides, the relationship between the volume of ablation and seizure outcomes remained unclear. Lastly, insular lobe epilepsy fills most of this cohort, and our sample size is small; therefore, our results need to be further confirmed by an investigation with a larger number of patients.
Conclusion
In conclusion, we demonstrated that SEEG-guided RF-TC can be a potential option for the treatment of MRI-negative epilepsy. Despite falling in the initial year, the probability of remaining seizure-free gradually stabilizes in the subsequent years. Moreover, patients with an EZ located in the insular lobe or with one focus on the limbic system have higher probabilities of obtaining seizure freedom. Our findings may be applied to guide individualized surgical interventions and help clinicians make better decisions.
Supplemental Material
sj-docx-1-taj-10.1177_20406223241236258 – Supplemental material for Predictors of seizure outcomes in stereo-electroencephalography-guided radio-frequency thermocoagulation for MRI-negative epilepsy
Supplemental material, sj-docx-1-taj-10.1177_20406223241236258 for Predictors of seizure outcomes in stereo-electroencephalography-guided radio-frequency thermocoagulation for MRI-negative epilepsy by Qi Huang, Pandeng Xie, Jian Zhou, Haoran Ding, Zhao Liu, Tianfu Li, Yuguang Guan, Mengyang Wang, Jing Wang, Pengfei Teng, Mingwang Zhu, Kaiqiang Ma, Han Wu, Guoming Luan and Feng Zhai in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-xlsx-2-taj-10.1177_20406223241236258 – Supplemental material for Predictors of seizure outcomes in stereo-electroencephalography-guided radio-frequency thermocoagulation for MRI-negative epilepsy
Supplemental material, sj-xlsx-2-taj-10.1177_20406223241236258 for Predictors of seizure outcomes in stereo-electroencephalography-guided radio-frequency thermocoagulation for MRI-negative epilepsy by Qi Huang, Pandeng Xie, Jian Zhou, Haoran Ding, Zhao Liu, Tianfu Li, Yuguang Guan, Mengyang Wang, Jing Wang, Pengfei Teng, Mingwang Zhu, Kaiqiang Ma, Han Wu, Guoming Luan and Feng Zhai in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
