Abstract
Background:
Diffuse low-grade gliomas (DLGGs) are prone to invade the frontal lobes, with seizures being the most common symptom. However, limited attention has been paid to surgical outcomes and their predictors in patients with frontal DLGG-related epilepsy.
Objective:
This study aimed to analyze predictors of postoperative seizure outcomes in patients with frontal DLGG-related epilepsy.
Design:
This is a single-center retrospective study.
Methods:
This study retrospectively collected data of 115 patients with frontal DLGG-related epilepsy who underwent resective surgery between January 2014 and January 2021. Patients were categorized into favorable and unfavorable seizure outcome groups based on the International League Against Epilepsy (ILAE) classification. Univariate and multivariate analyses were used to identify potential predictors of seizure outcomes.
Results:
The mean follow-up was 4.11 ± 2.06 years, and 77.4% (89 of 115) of patients were seizure-free. Permanent neurological deficits were observed in 7.0% (8 of 115) of patients. Univariate and multivariate analyses revealed that total tumor removal [odds ratio (OR), 0.31; 95% confidence interval (CI), 0.12–0.82; p = 0.018] and older age at seizure onset (OR, 0.96; 95% CI, 0.93–0.99; p = 0.042) were independent predictors of favorable seizure outcomes.
Conclusion:
Surgical resection is an effective treatment for frontal DLGG-related epilepsy. Favorable seizure outcomes are more likely to be achieved in patients with complete tumor removal and those with older age at seizure onset.
Introduction
Diffuse low-grade gliomas (DLGGs) arise from the supporting glial cells of the central nervous system and are classified as World Health Organization (WHO) grade II tumors based on their histopathologic features. 1 Its slow growth results in a relatively long survival in patients with DLGG after resective surgery.2,3 Thus, factors that may affect the quality of life are important in surgical planning. DLGGs have a predilection for invading the frontal lobes. 4 Previous studies have mainly focused on the involvement of the motor area, supplementary motor area, and Broca’s area in patients with frontal DLGG, which may directly influence postoperative neurological deficits and the quality of life of the patient after resective surgery.5–7
Seizures are the most common symptom in patients with DLGG and the first symptom in most patients with DLGG.8,9 Persistent seizures may not only decrease the quality of life but also deteriorate the oncological outcomes and cognitive functions of the patients.10–13 Seizure outcomes according to tumor location have been reported for temporal lobe DLGG because of their high epileptogenicity.14,15 Frontal lobe epilepsy is the second most common type of surgically treated focal epilepsy after temporal lobe epilepsy and could potentially cause executive dysfunction and behavioral problems. However, limited attention has been paid to seizure outcomes and their predictors in patients with frontal DLGG-related epilepsy. Despite some previous studies in patients with frontal DLGG-related epilepsy, the results of cases with tumor location other than frontal lobe had limited applicability in patients with frontal DLGG-related epilepsy.
This study aimed to report the surgical outcomes and analyze the prognostic factors of frontal DLGG-related epilepsy in a case series of 115 patients who underwent resective surgery as treatment.
Methods
Study design and patient cohort
This study retrospectively collected data of patients with frontal DLGG-related epilepsy who underwent resective surgery at the Department of Surgery, Xiangya Hospital, Central South University, between January 2014 and January 2021. Patients who met the following criteria were included in this study: had a diagnosis of DLGG (WHO grade II astrocytoma or oligodendroglioma) based on postoperative pathological examination; had a DLGG located in the frontal lobe; had preoperative epilepsy associated with DLGG; and underwent follow-up of more than 12 months after surgery. The exclusion criteria were as follows: had a postoperative pathological diagnosis that was not DLGG; did not have preoperative epilepsy, or epilepsy was found to be not associated with the DLGG after preoperative evaluation; had a DLGG that involved brain tissue beyond the frontal lobes; and had a follow-up of less than 12 months. The flow chart of patients is shown in Figure 1.
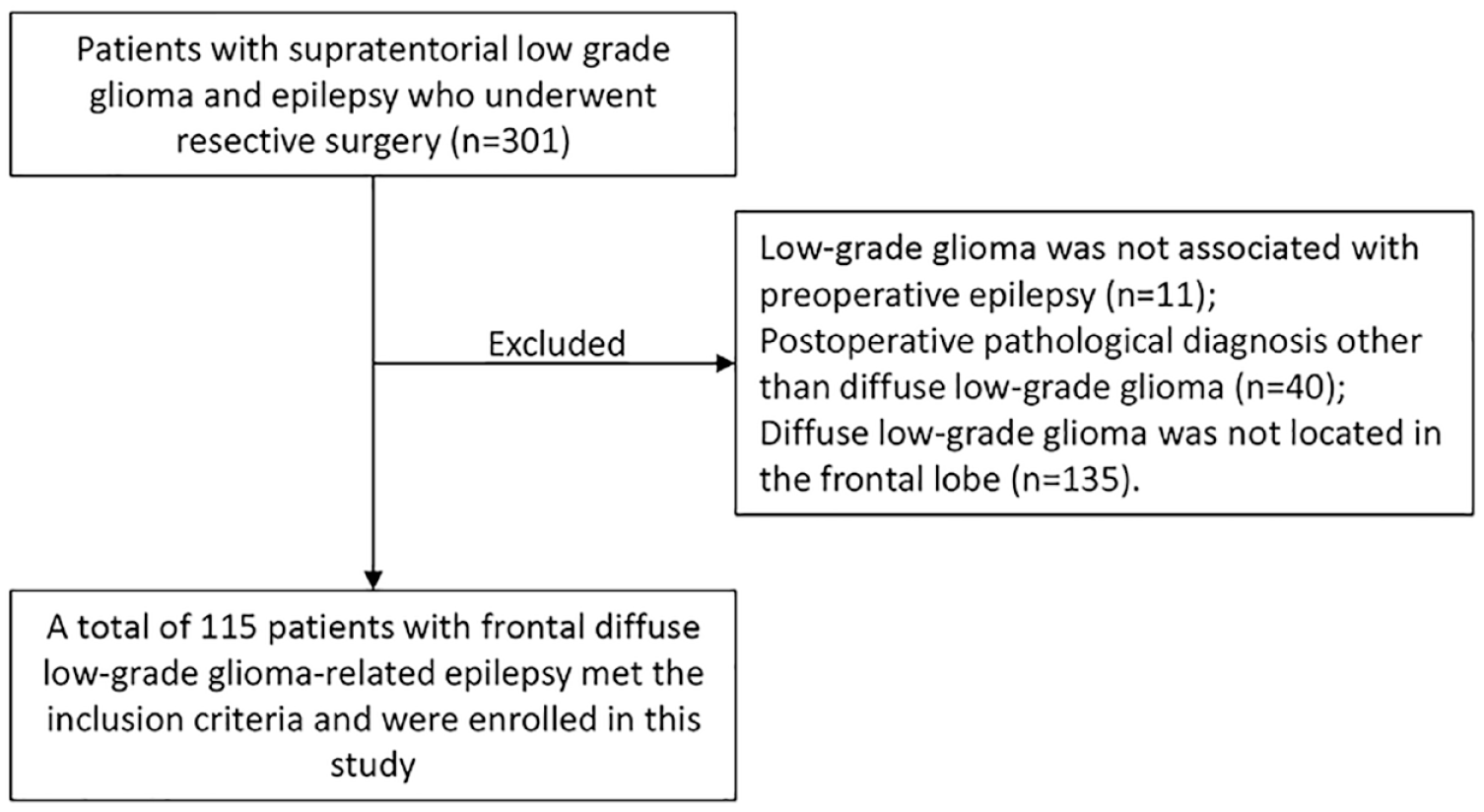
Flow diagram of patients.
Preoperative evaluation and surgical strategy
Routine preoperative evaluations, including semiology, detailed history, neurological examination, brain magnetic resonance imaging (MRI) on 1.5 T or 3.0 T scanners, and a 2-h scalp electroencephalogram (EEG), were performed in all patients. If needed, long-term video EEG was performed to evaluate the epileptogenic zone, and functional MRI was performed to assess the relationship between the eloquent areas and tumors. The size of tumors (maximal diameter) was measured by independent neurosurgeons according to preoperative T2-weighted fluid-attenuated inversion recovery (FLAIR) magnetic resonance (MR) images. With the aim of maximal safe resection of tumors, surgery was performed mainly according to the preoperative MRI and was additionally guided by intraoperative electrocorticography (ECoG) in selected patients.
Follow-up and surgical outcomes
Patients were evaluated by independent neurosurgeons in the outpatient clinics or by telephonically at 3 and 6 months postoperatively and yearly thereafter. Seizure outcomes were evaluated according to the International League Against Epilepsy (ILAE) classification. 16 Patients with ILAE class 1 and 2 outcomes were classified as having favorable seizure outcomes, whereas patients in ILAE classes 3–6 were defined as having unfavorable seizure outcomes.
Statistical analysis
Patients’ clinical characteristics were described using range (median) or mean ± standard deviation for continuous variables and frequencies for categorical variables. To evaluate the relationship between seizure outcome and clinical variables, patients were first categorized into two groups (favorable seizure outcome group corresponding to ILAE class 1 and 2 outcomes and unfavorable seizure outcome group corresponding to ILAE class 3–6 outcomes). In the univariate analysis, the Mann–Whitney U test was used to compare the differences among continuous variables, and Pearson’s chi-square test or Fisher’s exact test was used to compare differences among categorical variables. Variables showing a p value less than 0.1 were then entered into a backward binary logistic regression model to identify independent predictors of seizure outcomes. All statistical analyses in the present study were performed using SPSS version 22 (SPSS Inc., USA). Statistical significance was set at p < 0.05.
The present study was designed as a retrospective study. We tried our best to collect cases that may meet our inclusion criteria as many as possible, and the power analysis before the study was not carried out. Based on the principle that 5–20 events per variables (4 variables in this study) are needed for reliable results in prediction models developed using logistic regression model, 17 and the fact that seizure recurrence rate was 22.6% in this study, we estimated that 90 patients or more are needed. That means the sample size in our study is enough to provide reliable results.
Results
Patient characteristics
In total, 115 patients who met the inclusion criteria were enrolled in this study. Sixty-four (55.7%) patients were men, and 51 (44.3%) were women. The age at surgery ranged from 5.50 to 70.00 years (median, 37.00 years), age at seizure onset ranged from 3.00 to 69.99 years (median, 35.00 years), and seizure duration ranged from 0.03 to 30.00 years (median, 0.08 years). Seizure was the first clinical symptom in 95 (82.6%) patients and a unique symptom in 87 (75.7%) patients. Twenty-nine (25.2%) patients had focal seizures only, and the remaining 86 (74.8%) patients had focal to bilateral tonic-chronic seizures preoperatively. Other clinical characteristics of the patients are summarized in Table 1.
The relationships between clinical characteristics and postoperative seizure outcomes in patients with frontal DLGG-related epilepsy (n = 115).

DLGG, diffuse low-grade gliomas; ECoG, electrocorticography; fMRI, functional magnetic resonance imaging; SD, standard deviation; SE, status epilepticus; VEEG, video electroencephalogram.
For comparisons of binary variables, chi-square test with continuity correction was used.
Seizures occurred during the first week after surgery.
p < 0.05.
Preoperative evaluation, surgical treatment, and pathological diagnosis
Preoperative brain MRI on a 1.5-T or 3.0-T scanner with T1-weighted sequences, T2-weighted sequences, FLAIR sequences, and contrast-enhanced MRI was performed in all patients. Tumors were located in the left frontal lobe in 54 patients (47.0%) and in the right frontal lobe in 61 patients (53.0%). Contrast enhancement within the tumor was observed in 40 (34.8%) patients. The mean tumor size was 4.53 ± 1.42 cm. Tumors of 28 (24.3%) patients were adjacent to eloquent areas, and functional MRI was performed to evaluate the relationship between tumors and eloquent areas. A routine scalp EEG was performed in all patients to evaluate the association between tumors and seizures, and long-term video EEG was additionally performed in 41 (35.7%) cases. Among 41 (35.7%) patients who received long-term video EEG monitor, interictal epileptiform discharges were observed in all these patients with 24 (20.9%) in unilateral and 17 (14.8%) in bilateral; seizures were monitored in nine (7.8%) patients, five (4.3%) had unilateral ictal epileptiform discharges, and three (2.6%) had bilateral ictal epileptiform discharges. Among the 74 (64.3%) patients who only received 2-h scalp EEG, seizures were monitored in none, interictal abnormal discharges were observed in 35 (30.4%) patients, and normal EEG results were observed in 39 (33.9%) patients. Preoperative antiepileptic drug (AED) treatment was performed in 66 (57.4%) patients, and detailed information of preoperative AED treatment is listed in Supplemental Table S1.
Resective surgery was performed based on the findings of the preoperative assessment, and 24 (20.9%) patients underwent intraoperative ECoG. Pathological examination revealed WHO grade II astrocytoma in 76 patients (66.1%) and WHO grade II oligodendroglioma in 39 patients (33.9%). Isocitrate dehydrogenase 1 (IDH1) mutation tests were performed in 111 (96.5%) patients [29 (25.2%) tested by only immunohistochemistry, and 82 (72.3%) tested by both immunohistochemistry and genetic sequencing], and IDH1 mutations were noted in 90 patients (78.3%). Detailed tumor molecular data and its relationship with postoperative seizure outcomes are summarized in Table 2. The extent of resection was assessed according to brain MRI at 72 h or 3 months postoperatively, and 68 (59.1%) patients underwent complete resection.
The relationships between tumor molecular data and postoperative seizure outcomes (n = 115).

IDH1, isocitrate dehydrogenase 1; ATRX, alpha thalassemia/mental retradation syndrome, X-linked; MGMT, O6-methylguanine DNA methyltransferase; NOS, not otherwise specified (absence of diagnostic molecular testing).
Postoperatively, all patients received AEDs for at least 6 months, and detailed information of postoperative AED treatment is listed in Supplemental Table S2.
Seizure outcomes and complications
With a mean follow-up of 4.11 ± 2.06 years, 89 (77.4%) patients were seizure-free, and all were in ILAE class I outcome. Among the remaining 26 patients (22.6%), 7 (6.1%) had ILAE class III outcomes, 11 (9.6%) had ILAE class IV outcomes, and 8 (7.0%) had ILAE class V outcomes. At last follow-up, AEDs were discontinued in 72 (62.6%) patients.
Surgical complications were observed in 16 patients (13.9%). Eight patients (7.0%) recovered completely during follow-up, and the remaining eight patients (7.0%) had permanent neurological deficits (seven had mild hemiparesis, and one had aphasia).
Predictors of seizure outcomes
Patients were divided into a favorable seizure outcome group and an unfavorable seizure outcome group according to the ILAE classification to analyze the predictors of seizure outcomes. In univariate analysis, the extent of resection was significantly associated with postoperative seizure outcomes (p < 0.05), and variables, including age at seizure onset, monthly seizure frequency, and seizure types, had a p value of less than 0.1. All factors with p < 0.1 in the univariate analysis were entered into a logistic regression model in a binary backward manner for multivariate analysis. Multivariate analysis revealed that total tumor removal [odds ratio (OR), 0.31; 95% confidence interval (CI), 0.12–0.82; p = 0.018] and older age at seizure onset (OR, 0.96; 95% CI, 0.93–0.99; p = 0.042) were independent predictors of favorable seizure outcomes (Table 3).
Predictors of postoperative seizure outcomes in patients with frontal DLGG-related epilepsy (n = 115).

CI, confidence interval; DLGG, diffuse low-grade gliomas; OR, odds ratio.
p < 0.05.
Discussion
DLGGs are prone to invade the frontal lobes. 4 Seizure control, which may markedly influence postoperative quality of life, is now considered an important goal in DLGG treatment. 18 Herein, we report the surgical outcomes and analyze the prognostic factors of frontal DLGG-related epilepsy in a relatively large case series of 115 patients.
In previous studies, the seizure-free rate was reported to be 64.5–82.0% in patients with DLGG-related epilepsy.19–22 Consistent with these results, we found that 77.4% of the patients were seizure-free, with a mean follow-up of 4 years. Surgical complications were observed in 13.9% of patients, and permanent neurological deficits were present in 7.0% of patients, which was higher than the surgical complication rate in a meta-analysis reported by De Witt Hamer et al. 23 This may be due to the fact that the case series in our study only recruited patients with frontal DLGG-related epilepsy whose eloquent areas were more prone to be involved.
Factors including the extent of resection, seizure duration, age at surgery, and tumor location have been reported as predictors of seizure outcomes in patients with DLGG-related epilepsy.24–27 In the present study, only patients with frontal DLGG-related epilepsy were included, and we found that total tumor removal and older age at seizure onset were independent predictors of favorable seizure outcomes.
Total tumor resection is a well-known predictor of favorable postoperative seizure outcomes in patients with DLGG-related epilepsy.25,26,28 In accordance with these study results, we also found such associations in this study, despite the differences in the patient cohort. Patients with residual tumors after surgery tend to have unfavorable seizure outcomes. This may be partly attributed to the neurotransmitters and modulators released by residual tumors. 29 Some studies additionally analyzed the optimum extent of the resection threshold that may affect postoperative seizure outcomes, although with discrepant results. Optimum thresholds of >91%, >85%, and >80% of tumor removal, which were associated with favorable seizure outcomes, have been reported by Still et al., 27 Ius et al., 30 and Xu et al., 21 respectively. Such analysis was not performed in our study because of the limited sample size.
Another independent predictor of postoperative seizure outcomes identified by multivariate analysis was age at seizure onset. We found that older age at seizure onset was increasingly associated with postoperative seizure freedom, a result that has not been reported in previous studies on surgical treatment for DLGG-related epilepsy. However, we cannot explain this result based on the selection criteria of the present study. Age at the time of surgery is seldom considered an independent predictor of seizure outcome, and it is associated with age at surgery and seizure duration. Patients with a younger age at seizure onset were more likely to have a younger age at the time of surgery or longer seizure duration, which have been reported as predictors of seizure outcomes in some previous studies.8,27,31 However, we could not confirm the associations between seizure outcomes and age at surgery or seizure duration in the present study. We presume that a more widespread epileptogenic network or the formation of a secondary epileptogenic focus may play a role in some patients, partly due to the higher plasticity of neural networks in patients with younger age at seizure onset.29,32,33
This study had some limitations. First, this was a single-center retrospective study, and the inherent biases of its retrospective nature cannot be ruled out. Second, most patients in this study were treated at the neuro-oncology center with the aim of maximum safe tumor removal, and the assessment of epileptogenic focus may have been neglected to some extent during preoperative evaluations. Further studies in which all patients underwent detailed preoperative evaluations of epileptogenic foci may provide additional useful information. Finally, changes in quality of life and cognitive function were not evaluated in this study, which may also be important in the surgical treatment of DLGG-related epilepsy. Despite these limitations, our results may be useful for preoperative counseling and postoperative medical management in patients with frontal DLGG-related epilepsy.
Conclusion
Surgical resection is effective in patients with frontal DLGG-related epilepsy. Favorable seizure outcomes are more likely to be achieved in patients with complete tumor removal and those who were older at seizure onset.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221141856 – Supplemental material for Resective surgery for patients with frontal lobe diffuse low-grade glioma-related epilepsy: predictors of seizure outcomes
Supplemental material, sj-docx-1-taj-10.1177_20406223221141856 for Resective surgery for patients with frontal lobe diffuse low-grade glioma-related epilepsy: predictors of seizure outcomes by Kai Zhang, Dingyang Liu, Zhuanyi Yang, Xuejun Li, Zhiquan Yang and Xinghui He in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
