Abstract
Extraintestinal manifestations occur rather frequently in ulcerative colitis (UC) and Crohn’s disease patients and are usually related to an exacerbation of the underlying intestinal bowel disease but sometimes may run a course independent of the inflammatory bowel diseases (IBD). About one-third of patients with IBD develop extraintestinal manifestations, such as pyoderma gangrenosum (PG). PG is an uncommon inflammatory skin disorder of unknown pathogenesis. There are no specific serological or histological markers, and diagnosis is predominantly clinical. Topical and systemic therapies are both vital aspects of treatment and immune modulators have been used with increasing success in recent years, although immunosuppressive drugs raise some concerns due to an increased risk of serious and opportunistic infections and cancer, particularly in elderly and comorbid patients, underlining the unmet need for safer alternative therapies. Thus, in this case report, we highlighted an adsorptive granulocyte/monocyte apheresis (GMA) as a new therapeutic possibility in IBD patients with extraintestinal manifestations. We report a case of a 60-year woman with a history of UC with a Mayo grade 3 score which was associated with a PG. Given that the patients maintained clinical remission with vedolizumab, we preferred not to perform a combined treatment with other antitumor necrosis factor-alpha or ciclosporin, thus avoiding an increased risk of serious infections in the patient. Therefore, we performed the extracorporeal leukocyte apheresis. The patient progressed favorably, with progressive improvement of skin and bowel disease. Therefore, adsorptive GMA has a very favorable safety profile and has been confirmed in numerous studies. In this study, we underlined that an intensive regimen of GMA paves the way to an ideal option for patients with severe and refractory PG complicated with UC.
Introduction
Extraintestinal manifestations (EMs) occur rather frequently in ulcerative colitis (UC) and Crohn’s disease (CD) patients with significant morbidity in inflammatory bowel disease (IBD) patients even more so than the intestinal disease itself.1,2 EMs are usually related to an exacerbation of the underlying intestinal disease but sometimes may run a course independent of the IBD. 3 About one-third of patients with IBD develop EMs, such as pyoderma gangrenosum (PG).2,4 The annual incidence of PG is 3–10 per million persons 5 and has been reported to occur in about 2% of patients with IBD, while up to 30% of patients with PG have an underlying IBD. 6 PG is a rare inflammatory skin disease often associated with underlying systemic diseases such as UC. PG is characterized by painful erythematous and necrotic areas or nodules that rapidly develop into destructive, necrotic, sterile ulcers with rough violet margins. 7 The pathogenesis is unclear but involves immune pathways, and a biopsy of the lesion shows predominantly neutrophil infiltration, large sterile abscess formation with venous and capillary thrombosis, and hemorrhage. There are no specific serological or histological markers, and diagnosis is largely clinical. Both local and systemic therapies are important aspects of treatment, and in recent years immunomodulators have been used with increasing success, and antitumor necrosis factor (TNF)-alpha drugs such as the monoclonal antibody infliximab (IFX) have played an increasing role.8,9 Therefore, the neutrophil-rich inflammatory infiltrate suggests a central role of neutrophils 7 and immunosuppressive drugs raise concerns about the increased risk of serious and opportunistic infections and cancer, particularly in elderly and comorbid patients, underlining the unmet need for safer alternative therapies. Therefore, adsorptive granulocyte/monocyte apheresis (GMA) was an extracorporeal leukocyte apheresis unit and a nonpharmacological therapeutic intervention created to remove activated leukocytes, filled with cellulose acetate beads and selective apheresis device adsorbs granulocytes and monocytes from peripheral blood.10,11 GMA is typically performed once or twice a week for 5–10 sessions, with each session lasting 60 min at a flow rate of 30 mL/min. 12 GMA efficacy and safety have been confirmed for inflammatory disorders of the skin such as PG13,14 and also in some PG and IBD patients.15,16 Thus, in this case report, we highlighted an adsorptive GMA as a new therapeutic possibility in IBD patients with manifestations EMs.
Case report
A 60-year woman was admitted to our the Department of Gastroenterology in August 2016 for severe abdominal pain, bloody stool, and diarrhea (up to 10 discharges in 24 h, which is three at night). A laboratory examination revealed an elevated C-reactive protein (CRP) and fecal calprotectin (FCP) (1120 mg/kg), normal leukocytes and mild anemia (Hb 11.9 g%). A colonoscopy revealed moderately active chronic colitis with Paneth cell metaplasia in the left colon and rectum (Mayo UC score 2). So, the patient started oral and topical mesalazine [5-aminosalicylic acid (5-ASA)] therapy (oral administration of 5-ASA of 3.2 g/day two daily plus 4 g enemas for 8 weeks. Significant and rapid clinical improvement was observed up to week 24 and this medication was used as maintenance treatment. In August 2018, the patient returned to our hospital for a severely active UC flare-up, refractory to oral corticosteroids. At this time, the patient had more than 10 bowel movements a day of liquid bloody mucus stools with abdominal pain. Therefore, the patient underwent to colonoscopy. An endoscopic picture of UC with a Mayo grade 3 score with significant edema, absent vessel markings, and spontaneously bleeding ulcers were observed (Figure 1). Histological examination confirmed a severe disease with multiple ulcers in the absence of cytomegalovirus (CMV). At once, a large painful hemorrhagic ulcer of her left lower abdomen appeared (Figure 2), pending histological examination the lesion was treated by the dermatologist with topical steroids and antibiotic therapy without any response. Histological examination highlighted a neutrophil-rich inflammatory infiltrate and a pustule that develops into necrotizing ulcers, compatible with PG. Bacterial and fungal infections in swabs were negative. The patient was treated with intravenous 5 mg/kg IFX induction therapy (weeks 0, 2, and 6) and subsequently the maintenance phase with treatment every 8 weeks for the treatment of exacerbation UC and PG. The patient achieved a clinical remission of RCU and PG at 23 weeks (Figure 2), after that the patient showed a clinical relapse of IBD with significantly high CRP and FCP levels. It was confirmed by proctosigmoidoscopy (Mayo UC score 3). Therefore, in September 2020, the patient swapped to anti-integrin alpha-4-beta-7 (Vedolizumab) (VDZ) (300 mg) at T0, T2, T4, and T8. The gastrointestinal symptoms improved visibly and in February 2021 the patient underwent to control colonoscopy that revealed a mild focal descending colon hyperemia and the mucosal healing of the remainder colon (Mayo UC score 0–1). At the end of February, a new painful pustular ulcer on her leg appeared proving that convection therapy does not work. In Figure 3, an increase in the ulcerated area was evident over time [Figure 3(a)–(c)]. Histological examination showed a dense infiltrate of neutrophils and necrosis confirming the association of PG with IBD. Given that the patients with UC maintained clinical remission with VDZ (intravenous infusion of 300 mg every 8 weeks), the remission was highlighted by negative fecal lactoferrin and slightly elevated serum CRP (0.89/0.75). Probably the slight increase in CRP was ascribable to inflammatory skin conditions with Clostridium difficile, Yersinia, and campylobacter-negative cultures. In particular, we preferred not to perform a combined treatment with other anti-TNF-alpha (Adalimumab) or ciclosporin A (CyA), thus avoiding an increased risk of serious infections in patients. Therefore, we performed the extracorporeal leukocyte apheresis using Leukocyte Adsorber, LA25 (Apheretica srl, Bologna, Italy).

Endoscopic findings. At relapse before the VED treatment colonoscopy confirms ulcerative colitis with a Mayo grade 3 score with significant edema, absent vessel markings, and spontaneously bleeding ulcers.
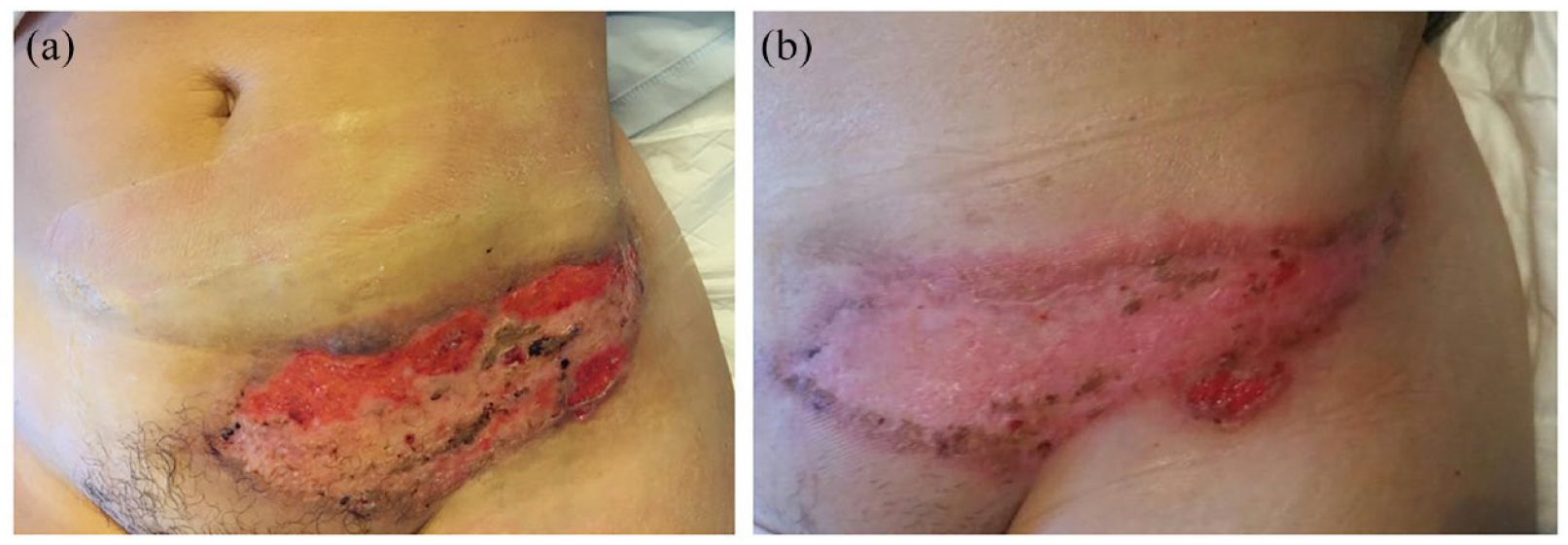
(a) Painful hemorrhagic ulcer of her left lower abdomen and (b) after Infliximab, left lower abdomen ulcer improved.

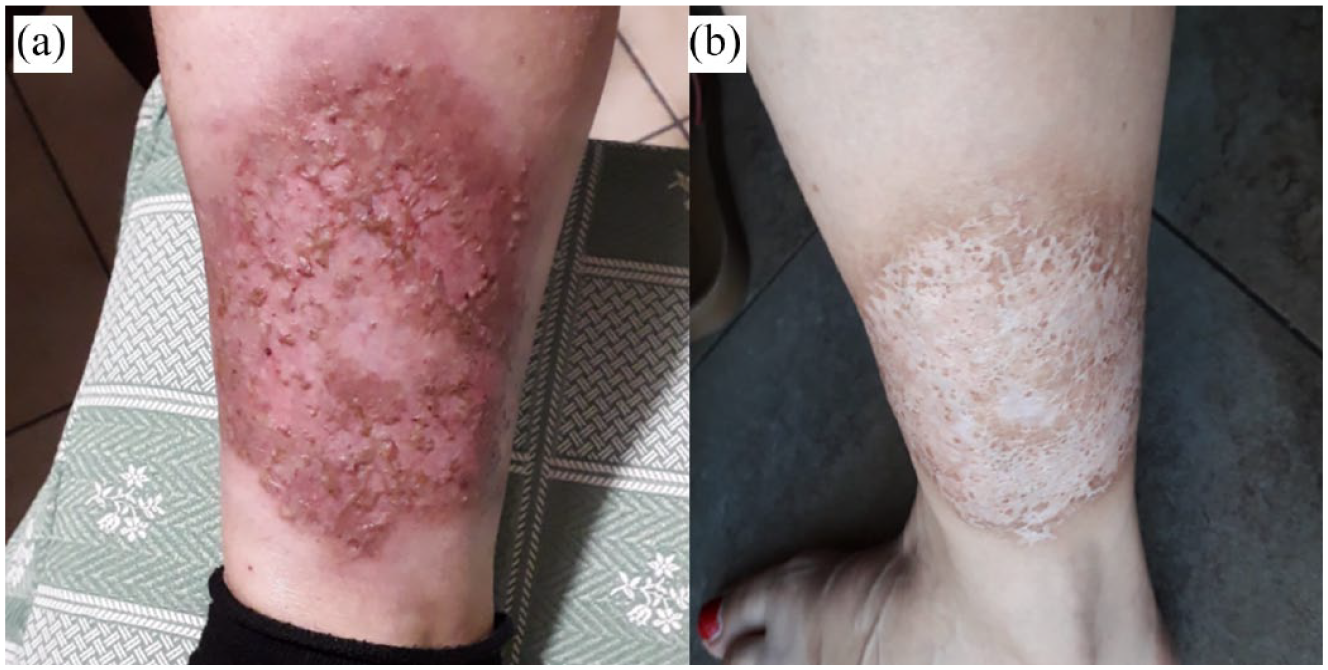
Clinical images of pyoderma gangrenosum. (a) At the time of onset. Tenderness and erythema with infiltration in the right lower leg and right knee, (b) 7 weeks later, the eruption ulcerated and (c) rash worsened together with increasing pain and discomfort.
During VDZ treatment, treatments were given once a week for 5 weeks. During each GMA session, blood drains from the cubital vein in one arm circulates through the column, and returns to the cubital vein in the other arm. The flow rate was 30 mL/min and the duration of each session was 60 min. Heparin is used as an anticoagulant (5000 units by intravenous injection). After GMA treatment, a great improvement was observed: the patient’s skin lesions also improved dramatically, and pain was markedly alleviated after the third session of GMA. After the last session of GMA, the ulcers became crusted and dried-up [Figure 4(a)]. Discharge of the ulcer disappeared and swelling of the purulent lesion was lost allowing her to talk again without pain, non-changed alterations in bioumoral and infectious parameters [CMV, Epstein Barr virus, varicella-zoster virus, in the vital parameters (arterial pressure, heart rate)], demonstrating the efficacy of GMA treatment. The patient progressed favorably, with progressive improvement of skin and bowel disease, and is currently under maintenance therapy with VDZ after completing the induction with good response. At present, no recurrences of ulcers were found during a follow-up period of 12 months [Figure 4(b)].
(a) After the last session of granulocyte/monocyte apheresis, the ulcers became crusted and dried-up and (b) after 12 months, the skin ulcer improved, and at present, the skin eruptions have receded.
Discussion
PG is a severe extraintestinal manifestation of IBD with rare skin lesions having significant pain, pustules, and ulcers in the patients. 6
The pathogenesis is unclear, but the most widely accepted theory is the immunological theory, and biopsy of the lesion shows mainly neutrophil infiltration 7 . A neutrophil-rich inflammatory infiltrate without infection suggests a central role for neutrophils.17,18 Nonetheless, the ultimate mechanism of neutrophil activation and tissue damage has not been elucidated but appears to be an immune-mediated injury similar to IBD. In particular, GMA has immunomodulatory effects, including downregulation of inflammatory cytokine profiles, changes in leukocyte surface receptors, and induction of regulatory T cells. 19
The diagnosis of PG is clinically suspected and other causes of ulceration excluded by histological and microbiological examination. There are no laboratory parameters available for PG, and the histopathology of PG is nonspecific and varies with the stage of the lesion. Especially in UC patients, the occurrence of PG is associated with the onset of the disease.
PG treatment includes wound care, antibiotics, corticosteroids, and immunomodulators. Patients with IBD require long-term medical therapy and may experience severe side effects from steroid or monoclonal antibody therapy. 20 However, response to treatment varies, and many patients with PG have a disease that does not respond to these drugs. Therefore, in this case report, we chose GMA as a new treatment option for patients with PG IBD. GMA is an in vitro immunomodulatory therapeutic strategy that effectively removes activated leukocytes from peripheral blood to correct imbalances in immune regulatory mechanisms. 21 The short-term effectiveness of GMA in IBD (mainly UC) has been demonstrated. The treatment appeared to be well tolerated, resulting in short-term remission with a sparing effect. In addition, it can be used in diseases of other entities that produce similar immune conditions. 22 To date, nine cases of PG treated with GMA (including the present case) have been reported in the literature (Table 1). PG is more common in young patients, 73% of whom are under 60 and the mean age is 37. Most patients are female (male:female = 2:7). Our patient has a history similar to that presented by Shibuya et al. 23 in which GMA was initiated after treatment with VDZ. Although VDZ immediately improved bowel symptoms, it had no effect on PG 24 ; in particular, the absence of over-expression of MAdCAM-1 in the human skin would explain the lack of effectiveness of the VDZ in most of the EIMs, excluding EiMs a progression parallel to the inflammatory bowel state. Therefore, in this case, we believe that GMA was effective for PG associated with UC during VDZ treatment. In all cases, the introduction of GMA, on the other hand, was decisive for the course of the skin lesion, showing its effectiveness. Today, the management of PG is based on clinical experience. No clinical studies with high-level evidence or guidelines or standard therapeutic approach for the improvement of both skin and intestinal lesions have been published. 25 Systemic corticosteroids have been recommended as first-line treatment for patients with IBD-associated PG with or without immunomodulators, but with poor efficacy. 26 Anti-TNF agents such as IFX may also be available as new treatment options, although these treatments are associated with an increased risk of opportunistic infections. 27 GMA has multiple effects, including depletion of activated neutrophils expressing Mac-1, reducing serum levels of proinflammatory cytokines, such as TNF-α and IL-1β, and stimulating specific cells with immunomodulatory roles. In particular, GMA charges regulatory T cells that suppress inflammatory responses because TNFα and IL-1β regulate the activation of inflammasomes, thus GMA may be effective in patients with PG.
Literature review of PG cases in UC patients.

5-ASA, 5-aminosalicylic acid; GMA, granulocyte/monocyte apheresis; LCAP, leukocytapheresis; PG, pyoderma gangrenosum; PSC, primary sclerosing cholangitis; UC, ulcerative colitis.
This case study highlighted that in patients with UC and PG GMA can be associated with VDZ treatment for intestinal disease resulting evident in efficacy and absence of adverse events. Indeed, GMA has a very favorable safety profile that has been confirmed in numerous studies.31–33 In this study, we underlined that an intensive regimen of GMA paves the way to an ideal option for patients with severe and refractory PG complicated with UC.
