Abstract
Periodontitis is a persistent inflammation of the soft tissue around the teeth that affects 60% of the population in the globe. The self-maintenance of the inflammatory process can cause periodontal damage from the alveolar bone resorption to tooth loss in order to contrast the effects of periodontitis, the main therapy used is scaling and root planing (SRP). At the same time, studying the physiopathology of periodontitis has shown the possibility of using a local drug delivery system as an adjunctive therapy. Using local drug delivery devices in conjunction with SRP therapy for periodontitis is a potential tool since it increases drug efficacy and minimizes negative effects by managing drug release. This review emphasized how the use of local drug delivery agents and natural agents could be promising adjuvants for the treatment of periodontitis patients affected or not by cardiovascular disease, diabetes, and other system problems. Moreover, the review evidences the current issues and new ideas that can inspire potential later study for both basic research and clinical practice for a tailored approach.
Keywords
Introduction
Periodontitis is an inflammatory chronic disease with multifactorial etiology. Biofilm is fundamental for the beginning of the disease, but it is not the only factor involved. The host immune response, genetics, and some systemic diseases take part in the pathogenesis of periodontitis. Moreover, lifestyle has a role in the pathophysiology of periodontitis.1,2
In the last years, the treatment of periodontitis has changed. Today scaling and root planing (SRP) is the main procedure that allows the removal of pathogenic biofilm, which has an important role in the etiology of periodontal disease.3,4 The lack of response occurs more frequency in sites with deep periodontal pockets and in teeth with complex anatomy where the ultrasonic instruments cannot totally remove biofilm. 5 Using only antimicrobial therapy does not achieve the outcomes in all periodontal patients because the host immune response plays an important role in periodontitis. Another type of adjunctive therapy that can be used is based on host modulatory agents. 6 Immune senescence is another factor that actively participates in the increase of inflammation in periodontitis. This is why there is a high prevalence of old patients affected by periodontitis. Aging causes an increase in sensitivity to infection in patients; additionally, the co-existing of systemic diseases contributes to immune response alteration. 7 Local drug administration can be an alternative strategy to treat old patients with and without comorbidity for better clinic results. Another therapeutical strategy against periodontitis is the use of natural agents that, in the last years, have shown special attention. Chlorhexidine, triclosan, desiccant agents, vitamins, and probiotic compounds, cetylpyridinium chloride, and others have been analyzed to understand their propriety and the possibility to be used in periodontal disease.8–13 Herbals medicine have different properties such as anti-inflammatory, antimicrobial and antioxidant.
Materials and methods
The articles included in this critical review were identified using the major search engines: PubMed, Google Scholar, and Scopus. The keywords used in all search engines were ‘local drug delivery and periodontitis’ and ‘natural agent and periodontitis’. When searching for the keywords ‘local drug delivery and periodontitis’, 88 articles were found on PubMed, 19,800 results on Google Scholar, and 618 on Scopus. When the keywords ‘natural agent and periodontitis’ were searched, 75 articles were found on PubMed, 19,100 articles were found on Google Scholar, and 379 articles were found on Scopus. The searches covered the following time frame 1980–2023. Of these articles, 201 articles were included in the present review after the exclusion of duplicate papers and those that did not fit the criteria for article selection. At least two independent researchers reviewed titles and abstracts for inclusion. For all articles that passed the initial screening, full articles were requested. Each full article was evaluated by two researchers for final inclusion/exclusion. In case of disagreement, a third researcher was consulted, and the decision was made by consensus. The initial selection was based on the following criteria: Randomized Clinical Trials (RCTs), cohort studies, case–control studies, and case series that included at least a sample size of 15, meta-analysis and systematic review. No studies were excluded based on the language of publication.
Local drug delivery systems
The use of systemic drug administration for treating oral infection in the last 50 years has shown positive results.14,15 Nevertheless, the use of systemic administration for drugs gives many disadvantages, such as dysbiosis and low quantities of the drug in the site where it is desired. However, this may cause gastrointestinal problems, drug resistance, and toxicity.16–18
Therefore, over the past 30 years, local drug delivery systems (LDDSs) have obtained an interest in studying how to use drugs in specific site where it is needed and how to control their release. For this purpose, polymers as drug carriers were discovered. They can defend the bioactive agents during the delivery into the body and have the quality to regulate the release kinetics. The bioactive principle can be filled with the polymeric matrix or linked in the polymeric chain. 19 Drug delivery systems allow to control and prolong the release of drugs into a specific site, and it can also be contained within different target agents simultaneously. In this way, reducing the dose and the number of intakes of drugs20,21 is possible. The use of an LDDS can give more benefits than systemic administration. It can bypass gastrointestinal problems and the systemic metabolism of the drugs before reaching the site of interest, giving LDDS a higher efficiency.20,21 Moreover, LDDS allows for administering in a noninvasive way the drug in the subgingival pockets. 22 Additionally, this type of drug administration allows loading two or more drugs of different categories in the same moment into the periodontal pockets. 23 Fibers, irrigations, membranes, films, nanoparticles (NPs), and microparticles are some of the forms of LDDS created 24 (Figure 1). The LDDS, which gives curative effects for periodontal issues, includes three main categories of drugs: antibacterial, inflammation-modulating, and alveolar and bone repairing agents.

The disadvantages of the systemic drug administration and the advantages of the local drug administration in treating periodontitis.
Antibacterial LDDS
The oral bacteria that live in the periodontal pocket and create the biofilm are one factor contributing to the initiation and progression of periodontal disease. Consequently, using antibiotics to treat periodontitis is fundamental to root up their presence. 25 The antibiotics used in periodontal treatment can be classified based on their capability against bacteria: in the first group, there are antibiotics with bacteriostatic properties that inhibited the protein’s synthesis [tetracycline, doxycycline, and minocycline (MCL)]; in the second case, there are antibiotics with bactericidal ability that inhibited the DNA’s synthesis (metronidazole, ornidazole, and quinolone); and in the third one, there are antibiotics with bacteriostatic properties that inhibited cell’s wall synthesis. 26
In the last years, the research focused on using LDDS instead of using systemic administration of antibiotics for the disadvantages they cause. At the beginning of 1983, a type of delivery system based on fibers with antibacterial agents was suggested. Different polymers were tested to find one for the delivery system.27–29 US FDA-approved Actisite composted by ethyl vinyl acetate fiber with tetracycline. Although clinical trials are indicated to have good effects, there are disadvantages, such as discomfort for the patient during placement, gingival redness, and the necessity to be removed because it is non-biodegradable.27,30,31 The property of not being biodegradable is a problem for some delivery systems. Then some types of biodegradable polymers were evaluated as carriers for the delivery of tetracycline: poly-hydroxybutyric acid and poly lactic-co-glycolic acid (PLGA). PLGA can control drug release. Moreover, it is possible to change the polymeric structure in the base of the kinetic of the drug.32,33 From a pilot study, Maze et al. 34 found that using a PLGA strips and films with 25% tetracycline can release the antibiotic for 10 days. In this study, 10 patients were enrolled affected by periodontitis that, after the SRP, continued to possess five sites with bleeding and probing depth (PD) of 5 mm. In these patients, four sites have been randomly selected. One site was used as control, and in the other sites, treatment was performed with PLGA strips and films of 25% of tetracycline, strips without PLGA or SRP. At the end of the study, the group tested with strips and films of PLGA and tetracycline showed a decrease in clinical parameters such as bleeding on probing (BoP) (p ⩽ 0.01) and probing PDs (p ⩽ 0.01). Additionally, a significative (p ⩽ 0.05) reduction of the spirochete and motile rods concentration was seen, respectively, after 4 weeks and after 8 weeks. There was only the PD reduction in the group where SRP were performed (p ⩽ 0.01). A marketed preparation, approved by US FDA, called Periochip, is based on a gelatin biopolymer matrix in which chlorhexidine gluconate is embedded. This delivery system is able to release the drug for 7 days. 35
In the first period of research, the main delivery system used was based on fibers, strips, and films. As a result of technological science development, the possibility of using nanosized and microsized particles as delivery systems that include microspheres and nanosystems has emerged. Microspheres are globular polymer structures that can have a diameter from 1 to 1000 μm in which the drug is spread inside the polymer matrix. 36 These microparticles have the capability to stabilize and release the drug contained inside of their matrix. In an in vivo study, Kong et al. 37 investigated the possibility of using a different combination of PLGA and poly-ε-caprolactone to control the delivery of doxycycline. This type of delivery system was tested in 30 patients affected by moderate periodontitis with periodontal pockets depths between 4 and 6 mm. These patients were divided into three groups: one group received only SRP, the other group received SRP and doxycycline microspheres, and the last group was treated with SRP and commercial doxycycline gel. As a result, the doxycycline was released for 11 days using this delivery system. Additionally, in the group tested with this delivery system, it was seen an enhance of PD (mean reduction between 0.5–1.2 mm). In another study, Ali et al. 38 showed that doxycycline loaded on microspheres released the drug for 21 days. Moreover, it was seen a significant inhibition of Porphyromonas gingivalis (p < 0.05) using a sphere formulated at pH 9.0, whereas no significative difference was noted using spheres formulated between pH 7.0 and 9.0 in the inhibition of Fusobacterium nucleatum (p > 0.05). A delivery system called Arestin based on PLGA microspheres loaded with MCL hydrochloride was approved by the US FDA. 39
Over the last few years, nanosystems such as liposomes, micelles metallic, and polymeric NPs have received wide attention in medicine. Thanks to their dimensions, lower than 100 nm, they can reach particular sites where other delivery systems cannot. 37 NPs have a specific targeting action because their physical and chemical characteristics increase therapeutic efficiency. At the same time, NPs reduce the side effects present in conventional systems. In the periodontic field, polymeric micelles, metallic and polymeric NPs, nanofibers, and liposomes were studied as new delivery systems. 40 Due to their anti-microbial activity and bone regeneration, metallic NPs, such as silver, gold, and titanium dioxide, are common in the dental field.40–42 Metallic particles possess bioactive properties, even if they are also cytotoxic and not biodegradable.43,44 For these reasons, polymeric NPs have been introduced as a delivery system in periodontal treatment. Polymeric NPs are biocompatible, stable, and biodegradable. Their size allows them to arrive at the target site where other LDDS cannot. 45 Yao et al. 46 in 2014 testing a MCL loaded on polymeric NPs in beagle dogs with periodontitis. In this study 12 beagle dogs were enrolled that were randomly divided into four groups, which it had been administered saline, free MCL, MCL-NPs, and Periocline. This study showed that using MCL-NP significantly reduces periodontal symptoms through the ability of NP to penetrate the junctional epithelium and release the drug for 12 days. After 6 days, in the group tested with this new delivery system and in the group with Periocline, it was seen a significative improvement in the periodontal parameters, including plaque index (PI) (p < 0.05), gingival index (GI) (p < 0.01), and periodontal pocket depth (PPD) (p < 0.01) compared to the group in which was administered free MCL. After 12 days in the group tested with NPs and MCL, PI (p < 0.05) and PPD (p < 0.01) remain lower than the other groups. 46 NPs can make from chitosan, a natural material with biodegradability, toxicity and antimicrobial properties. 47 In addition, different reports have shown that chitosan has higher drug permeability thanks to is capability to adhere in mucous membranes.48,49 In 2020, Xu et al. 50 formulated a doxycycline loaded on chitosan NPs. It was tested on cellular fibroblast culture in presence of P. gingivalis. It was used as a control group, a fibroblast cell culture with P. gingivalis untreated. At the end of the study, the group tested was seen a decrease in the cytokines level (SD n = 3) compared to the control group. This type of delivery system showed a higher antibacterial activity against P. gingivalis, and it had the capability to downregulate the inflammatory activity of NLRP3 inflammasome (p < 0.05) and Interleukin (IL)-1β (p < 0.001).
Liposomes are another type of nanosystem recently developed. Liposomes possess site-specific targeting and tissue permeability and have the capability to release drugs slowly. The amphiphilic nature of this nanosystem allows them to load both hydrophilic and hydrophobic drugs. Additionally, this chemical characteristic allows them to easily enter the cells and reach the target site. For these reasons, liposomes are used in the medical field as carriers for various types of drugs, from anti-cancer to antimicrobials. 51 A trial study conducted by Hu et al. 52 in 2019 showed that using doxycycline loaded on liposome as the delivery system can reduce inflammation and stimulate bone reconstruction (Figure 2). This study was conducted on 33 adult rats in which surgically deep pockets were created and were administered a mixed of saline and bacterial to induce periodontitis. It was used as a control site for the first upper molar of each rat. This type of delivery system performed a 75% inhibitor antibacterial capacity, whereas a traditional antibacterial performed 40%. From the histological analysis, a significant number of osteoclasts in the untreated control sites emerged compared to the tested group (p < 0.05). Additionally, in the tested group, there are fewer inflammation cells and smaller pockets depth than in the control group. The authors assumed that in the tested group, there is less inflammation because of the decrease of bacterial made by doxycycline loaded on liposomes. 52 Liposomes possess a wide range of advantages, but on the other hand, there are disadvantages that must be considered. There is the expensive liposome fabrication, the instability of their formulation, and the occasional oxidation and hydrolysis-like reactions of the phospholipids. 53
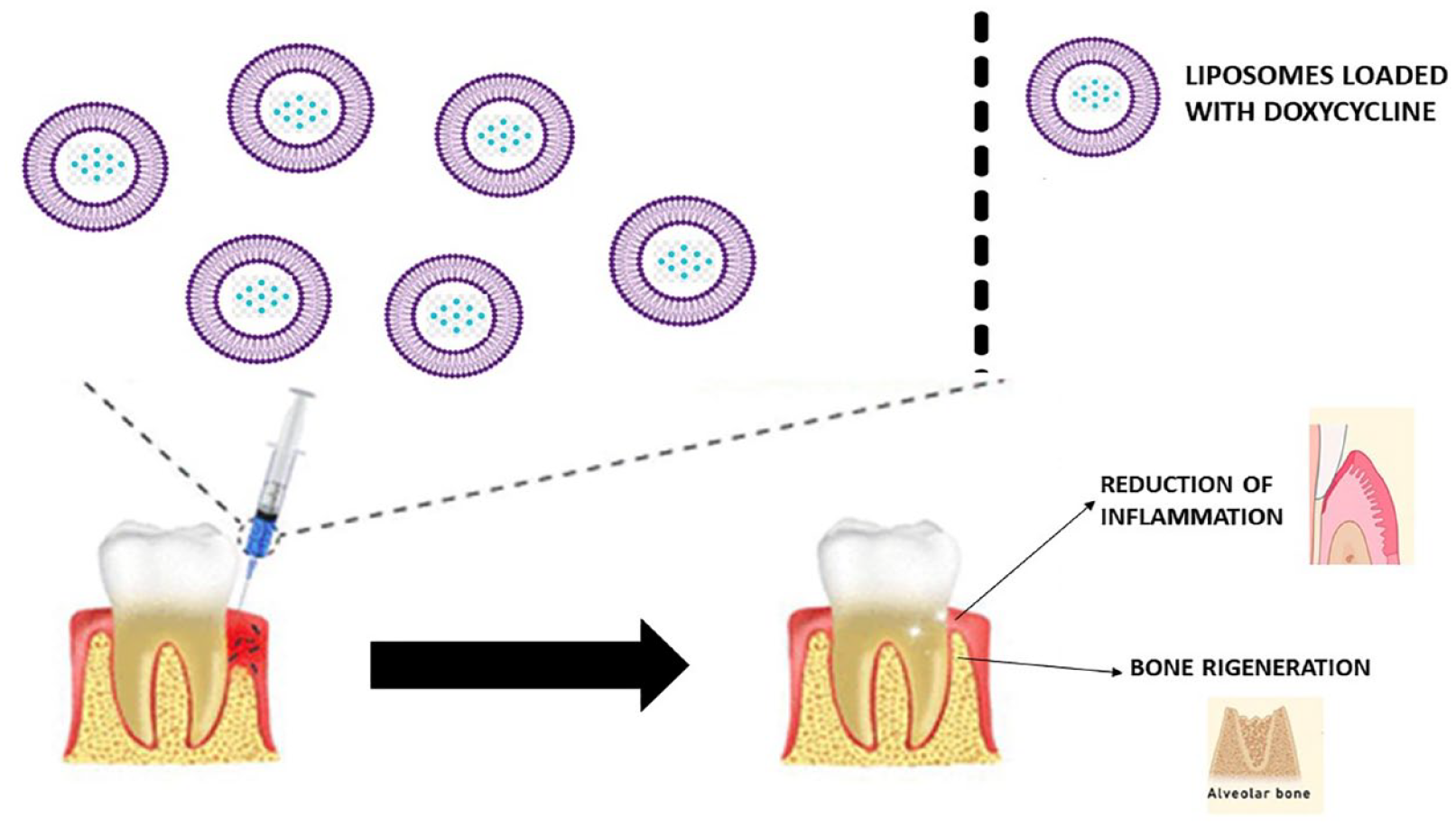
The image describes what happened in Hu et al.’s study, which used liposomes loaded with doxycycline.
Another type of delivery system is gel formulations that can be made of many polymers for instance, chitosan, carboxy methyl cellulose, and carbopol. A gel formulation made up of chitosan and 15% of metronidazole has been reported as efficient in the treatment of chronic periodontitis. In this study, 15 patients having moderate-to-severe periodontitis there were enrolled and were divided into three groups. Patients of the first group were treated with SRP and administered chitosan gel; the second group performed SRP and administered chitosan gel with 15% of metronidazole; and in the last group, only SRP was performed. The group in which chitosan and metronidazole were tested showed a lower level of PD (p < 0.01) after 6, 12, and 24 weeks compared to the other two groups. Additionally, PI (p < 0.05) and GI (p < 0.05) improved significantly in the groups treated with chitosan and metronidazole gel. There were no differences in clinical attachment level (CAL) and gingival recession (p > 0.01) between the three groups at all times of the measurement (6, 12, and 24 weeks). 54
Scientific progress has allowed the formulation of different types of polymeric gel preparations such as a particular type, called thermoresponsive gel, that possesses a transition state from liquid to semi-solid depending on the different stimuli. In 2016 Ruan et al. 55 formulated a thermoresponsive gel based on chitosan/β-glycerophosphate that was used to deliver 2% of MCL. For the study, 72 rats were enrolled in which periodontal disease was induced. The rats were divided into three groups based on the PI and GI value: group A received MCL ointment, group B received MCL gel, and group C received saline. After 14 days, group B showed a lower PD (p < 0.05) level compared to the A and C groups. Group B had a lower significant decrease (p < 0.05) in PD value compared to group A. From these results, the authors deduced that this delivery formulation effectively released the drug in the periodontitis model animal. 55 Some gels are classified as hydrogels, which means that they can swell and deform. In 2018 Wang et al. 56 prepared and tested an MCL loaded on an in situ hydrogel composed of PLGA and N-methylpyrrolidone. From the preliminary studies, this type of hydrogel showed to be an encouraging clinical delivery system for the treatment of periodontitis. Even if MCL was released for a period of 4 days, almost of it was liberated during the first day of administration. 56 Although this type of gel has been demonstrated to be excellent vectors of intraoral delivery, it had the disadvantage of releasing the medicines very quickly.57–60 Garrett et al. 61 have formulated a 407 poloxamer double barrier thermosensitive gel system loaded with moxifloxacin and PLGA NPs to solve this disadvantage. This study showed that this type of gel prolonged the release of the drug for a duration of 7 days. It was tested in ligature-induced models in rats. They were divided into seven groups: (1) control group, (2) untreated group, (3) group treated with gel in situ without drug, (4) group treated with NPs, (5) group treated with conventional metronidazole gel, and (6) group treated with gel of moxifloxacin and group treated with NPs loaded moxifloxacin gel. Each group received the specific drug type once a day for 3 weeks. After 2 weeks of treatment, the groups treated with moxifloxacin and NP in situ gel showed better results (p < 0.05) than those treated with conventional metronidazole gel. After 3 weeks, the group treated with NPs gel ultimately recovered from periodontitis. This result was seen from the histologic tissue evaluation where there was no inflammation tissue, bone apposition, and intact cementum. The delivery system with NPs loaded moxifloxacin results more effectiveness (p < 0.005 for each case) compared to the in situ gel with moxifloxacin and the conventional metronidazole gel. Moreover, once a week application of this gel had showed to give a superior efficacy compared to the commercial gel that had to be apply twice a day.
Two types of gel formulations with specific tetracycline are commercially available. One is Atridox approved by US FDA, which contains doxycycline and consists of a two-syringe system released for 7 days. 61 The second one is Periocline, marketed in Japan, based on MCL, released only during the first day of administration. 62
A systematic review evaluated how the use of local antimicrobial administration can enhance the periodontal clinical parameters in patients affected by periodontitis. In this systematic review, 50 different studies were evaluated. In the control groups of 43 studies SRP were performed. In eight studies, the control groups were administered a placebo, while in three studies, the control groups were administered SRP and a placebo. In the test groups were administered commercially local antimicrobials, including tetracycline, MCL, doxycycline, chlorhexidine, and piperacillin. Some of these studies evaluated other substances: chitosan, chitosan with metronidazole, MCL powder, and tetracycline strips. The results of this systematic meta-analysis showed that in most of the studies analyzed, the association of SRP with local antimicrobials allowed for improved clinical parameters such as PD [Weighted mean difference (WMD) = 0.365] and CAL (WMD = 0.263) in the tested groups compared to the control groups after 6–9 months of control. In the long-term studies evaluated, there was a significant difference for PPD (WMD = 0.190) but not for CAL. Although these interesting results have been considered, the studies analyzed in the systematic review used different antimicrobials with different doses and applications. This aspect may have influenced the results obtained. 63 The heterogeneity of the evaluated studies underline the necessity to do further investigation regarding the enhancement that the use of local delivery system with antimicrobial can add to non-surgical therapy.
An important aspect that must be evaluated is the possible antibiotic resistance against this delivery system. For this reason, different studies have been conducted. NPs enhance the antibiotics activity and can overcome bacterial drug resistance. 64 Liposomes and dendrimers are two types of NPs able to overcome bacterial resistance mechanisms. Through their basic constituents, liposomes can increase the efflux of antibiotics from the bacterial wall and decrease the uptake of the drugs. These properties allow liposomes to kill the microbes quickly before they can develop mutation.65,66 Additionally, a new target against bacteria is under investigation that contrasts quorum sensing, a type of cross-talk between pathogens. 23 More studies are necessary to find if these NPs could give the same results using antibiotics for the treatment of periodontitis.
Anti-inflammation LDDS
Periodontitis is an inflammatory disease where there is an interdependency between oral biofilm and host response. The pathogenic biofilm is a fundamental cause but not the only one. Indeed, host inflammation gives a crucial contribution to the development of the disease. 67 For this reason, in the past few years, the host immune response has become an interesting target for developing new therapeutic approaches. 68
Due to the role that immune responses have in this disease, some old drugs (rapamycin, MCL, lovastatin) and new drugs (resolvin E1, GW501516, GSK805 and more) have been considered for the immunomodulation treatment. Normally, these drugs are administered parenterally or orally in different forms such as capsules, solutions, gels, and lozenges. Unfortunately, oral administration reduces efficacy due to the poor water solubility, the low cell permeability of the drugs, and the difficulty transition in the digestive tract. 69 Moreover, many small molecules used for their immunomodulation activities possess periodontal pockets as target sites, and if they are administered systemically could cause adverse effects. 70 The bigger challenge is to induce immunomodulation and prolong retention in the periodontal pocket. 71 Nanodelivery systems represent a solution for administering small molecules as periodontitis treatment. NPs and nanofibers are biocompatible and can also ensure drug release, increase drug solubility, and deliver drugs to the target sites.72,73 Here some nanodelivery systems have been studied with their benefits. Statins are a group of drugs used to treat diseases related to the increase of cholesterol, such as cardiovascular disease. 74 Moreover, statins possess antioxidant, anti-inflammatory, new bone formation, and new angiogenesis properties. 75 In the last decades, statins have been an effective strategy against inflammatory in periodontitis. As a local delivery system, use of statins in a gel formulation of chitosan has been shown to enhance the pharmacodynamic efficacy. 76 Donos et al. 77 conducted a systematic review to evaluate the results of various studies related to the use of different host-immune regulating agents for the treatment of periodontitis. This study has emerged that using 1.2% of statin gels after SRP in the bone and furcation (class II) defects allows for improved PD (reduction of 1.83 mm) and CAL. The treatment with this gel was followed in the tested group after the SRP, and the clinical parameter was measured before the treatment, after 6 and 9 months. Although the encouraging results, it is impossible to say which type of statin is more effective than the others. 77 For this reason, further studies are necessary in the future. Another study conducted by Garg and Pradeep. 78 investigated the efficacy of two types of statin gel for treating Class II furcation defects. In the study, 90 patients were enrolled with Class II furcation defects in the mandibula. They were randomly divided into three groups: group 1 treated with SRP and placebo gel, group 2 was treated with SRP and 1.2% rosuvastatin gel, and group 3 was treated with 1.2% of atorvastatin gel. The gel administration was performed, after SRP, at the baseline and after 6 months, and the patients’ clinical parameters were recorded at the baseline and after 6 and 9 months. The result of this study showed that the group treated with rosuvastatin had a greater reduction of the PD (mean value 3.03 ± 1.18 after 9 months) and an improvement of the vertical CAL (4.06 ± 1.48) compared to the group treated with atorvastatin. From the radiographic analysis emerged, a superior reduction of the bone defects depth (measured from the furcation to the base of the defects) in the groups tested with rosuvastatin (1.31 ± 0.42 after 6 months, 1.77 ± 0.39 after 9 months) and atorvastatin (1.06 ± 0.43 after 6 months, 1.42 ± 0.44 after 9 months) than to the placebo group. Additionally, there was a superior improvement of the defect depth reduction in the rosuvastatin group (30.80% ± 8.35% after 6 months, 41.86% ± 6.76% after 9 months) compared to the atorvastatin group (25.54% ± 8.89% after 6 months, 34.31% ± 8.04% after 9 months). 78
In two animal trials using NPs of lovastatin loaded on chitosan and N-phenacylthialzolium bromide loaded on PLGA, inflammation was reduced in periodontal pockets and improved periodontal tissues.79,80 This new NP system was tested by Lee et al. 79 on beagle dogs with three wall defects created on the maxillary bilateral canines. The canines were randomly divided into two groups; one was the control group, and the other was the tested group. Dogs were sacrificed 8 weeks after surgery, and their tissue was analyzed. The histological analysis emerged that there were new cement materials apportion in the tested group compared to the control group, where there was only fibrous tissue. Additionally, from the micro-CT imagine, a statistical difference was seen (p = 0.04) in the new bone formation between the tested (41, 32%) group and the control (34, 01%). The use of this NP system showed biocompatibility and antibacterial activity and allowed the new bone formation in the three wall defects of beagle dogs. Lin et al. 80 tested this NP system on 12 rats. After the induction of periodontitis, the rats were divided into four groups: group A control group, group B treated with nanospheres only, group C treated with nanospheres loaded with metronidazole, and group D treated with N-phenacylthiazolium bromide. After 21 days of the treatment, rats were sacrificed. From the histological analysis conducted on the animals, results have emerged that the bone loss was lower in group D (p < 0.05). Groups C and D showed lower inflammation tissue (p < 0.05) and more collagen apposition (p < 0.05) compared to the control group. This NP system has been shown to reduce periodontitis inflammation in rats.
Moisei et al. 81 formulated liposomes packed with atorvastatin and studied the efficacy after their local administration in human macrophages cells culture. The results showed a larger decrease of lipopolysaccharide-induced-proinflammatory cytokines concentration in the cells culture treated with atorvastatin loaded on liposomes [74% inhibition of IL-6, 48% inhibition of Tumor Necrosis Factor (TNF), and 56% inhibition of IL-8] compared to the control group in which atorvastatin alone was administered (54% inhibition of IL-6, 36% inhibition of TNF, and 26% inhibition of IL-8). This study indicates that atorvastatin liposomal preparation might be applicable as a local therapy against periodontitis inflammation. Ciprofloxacin 82 and MCL 83 are two different antimicrobials that also possess anti-inflammatory properties. An in vitro study on human gingival fibroblasts promoted anti-inflammatory effects thanks to the use of chitosan NPs loaded with MCL that has target sites on these cells. 84 Ciprofloxacin showed antimicrobial properties and even immunomodulatory properties.85–87 In another in vitro study, ciprofloxacin was incorporated in a double-layer nanofiber mat and was tested on a culture of Escherichia coli and Aggregatibacter actinomycetemcomitans. From the statistical analysis, it was seen that this delivery system (double-layer loaded with metronidazole, chitosan, and ciprofloxacin) showed better results (p < 0.01) against A. actinomycetemcomitans compared to the double-layer loaded only with metronidazole. This system was able to control release for over 3 weeks. 88
NPs possess inborn properties that can improve to reduce and root out periodontal disease. 89 For instance, chitosan has antibacterial and anti-inflammatory properties, 90 but some cases were reported where it showed proinflammatory reactions. 91 NPs can incorporate immunomodulatory drugs. Through the bio-adhesive property of the polymers, this system is a promising way to treat periodontitis.92–97 Biopharmaceuticals are a class of drugs that can be used for their immunomodulation property in periodontal treatment. 98 The macromolecular structures that characterize biopharmaceuticals give them their specific properties, but on the other hand, these parts hinder the transition into the target sites.99–101 The elevated molecular mass of biopharmaceuticals causes restricted storing of them in the human body, their phagocytosis and their renal clearance. Moreover, biopharmaceuticals could cause an exacerbation of the immune response. 102
Biopharmaceuticals’ current study for periodontitis treatment (ustekinumab, denosumab, AMY-101) are developed as conventional injection solutions and demonstrated modest curative effectiveness. These NPs can incorporate and protect the drugs from premature degradation and can cargo them to the target site.103–106 At the same time, it is important to know that the nanodelivery system used to add biopharmaceuticals must be formulated following particular terms for protecting the biopharmaceuticals properties, for example, the protein susceptibility to low pH during the PLGA deterioration.107,108 In the following paragraphs, some studies are conducted to investigate applying nanosystems as delivery for biopharmaceuticals. Capurso et al. 109 developed PLGA NPs as an antigen delivery system that exhibit antigens to T and B lymphocyte cell cultures that showed to reduce inflammation (p < 0.05). 109 In a study made by Xu et al. 110 in 2017 was used an injectable thermosensitive hydrogel packed with aspirin and erythropoietin. It was tested on 48 rats that were divided into six groups: control group (healthy mice), mice affected by periodontitis untreated, mice treated with hydrogel, mice treated with hydrogel and aspirin, mice treated with hydrogel and erythropoietin, and mice treated with hydrogel, aspirin, and erythropoietin. To evaluate periodontal regeneration, the distance between CEJ and the alveolar bone crest in each group was measured using the micro-CT of the rat’s collected tissues. The distances between CEJ and alveolar bone crest in the groups tested with hydrogel + aspirin and hydrogel + erithropoietin are included to 25.1% and 34.8% of contraction (p < 0.01). This distance measured in the group tested with hydrogel + aspirin + erithropoietin was no different (p > 0.05) from the control group; this showed how erythropoietin allowed bone regeneration. Additionally, it was seen that in the group tested with hydrogel + aspirin + erythropoietin, the bone volume fraction (bone volume/tissue volume) (23.8%) (p < 0.01) and the tissue mineralize value (21.9%) (p < 0.01) improved compared to the other groups. This hydrogel was biocompatible and capable of releasing the drugs for 21 days. The in vivo experiment revealed that the hydrogel was capable of controlling inflammation [COX-2 decrease 1.5 ± 1.6 and matrix metalloproteinase (MMP)-9 decrease 15 ± 3.6] (p < 0.01) and stimulating the periodontium regeneration in rats affected by periodontitis. 110 Chitosan and hyaluronic acid NPs were applied to carry anti-IL6 antibodies as a bioadhesive delivery system for the treatment of arthritis. This study showed that these two nanocarriers successfully captured and immobilized IL6, reducing inflammation in human cell cultures. 111 Nanofibers have been investigated for incorporated and delivery of immunomodulatory such as Cp40 and C-C motif chemokine ligand 22 (CCL22) in the periodontal pockets. 112 Cp40 is a host immune modulatory drug that inhibits the C3 complement. 113 However, CCL22 is an immunomodulatory drug that controls T-cell recruitment. 114 In their study, Glowacki et al. 114 used CCL22 loaded on PLGA microparticles to treat periodontitis in murine and dogs affected by periodontitis. There were three groups: the test group (SRP + CCL22), the untreated group (only SRP), and the group treated only with the vehicle (after SRP). At the end of the experiment, the maxilla of the animals was resected and analyzed using micro-CT and histological testes. The results showed how this immunomodulatory drug (CCL22) loaded on PLGA was able to reduce the inflammation (p < 0.05) and the alveolar bone resorption (in the test group, the distance between cementoenamel junction (CEJ) and crestal alveolar bone was 0.5–1.0 mm lower than controls). In fact, there were a reduction in physiologic values of the BoP and the PD. 114 In present days, studies have underlined the new immunotherapy approach in periodontitis. From in vitro and in vivo studies, different drugs have been shown their effective, in particular, AMY-101, an analog of compstatin 40 (Cp40). The administration of Cp40 in animals affected by periodontitis showed a decrease in bone resorption and inflammation. 113 Hasturk et al. 115 have tested the efficacy of AMY-101 in adults affected by periodontitis. AMY-101 is able to inhibit C3, a type of immune complement that plays an important role in the physiopathology of periodontitis. 116 In Hasturk et al.’s study, 32 patients affected by periodontitis (PD ⩾ 5 mm) were enrolled and randomly divided into two groups. One group was treated with a placebo (saline), whereas the other group was treated with AMY-101. The drug and the placebo were administrated in the two groups three times: on days 0, 7, and 14. The study ended after 90 days, and the group tested with AMY-101 showed an enhance of the clinical parameters compared to the placebo and the control group. The group treated with AMY-101 showed an improvement in the PD (−0.5 ± 0.79 mm) (p < 0.162), BoP (−25% ± −0.07%) (p < 0.001) and modified GI (−0.29 ± 0.026) (p < 0.001). Moreover, it was seen the decrease of MMP (metalloprotease) 8 and 9 in the chemotactic factor for granulocytes (CFG) from the baseline (p < 0.05). The authors concluded that AMY-101 allowed decreased periodontal inflammation and did not show adverse effects during its administration. 115 However, the main question remains about their real delivery efficiency to the periodontal pocket. As a result, new advanced delivery systems are being developed. Moreover, nowadays, we do not know if these systems can cause adverse effects; for this reason, future studies will need to investigate their possible adverse effects. 117
Another class of drugs used for host modulation are bisphosphonates. These drugs possess antiresorptive and osteostimulative properties. A study conducted by Ipshita et al. 118 investigated the efficacy of 1% of alendronate and aloe vera in treating class II furcation defects. A total of 90 patients were enrolled in the present study and randomly divided into three groups. One group was treated with SRP and placebo gel, the second group was treated with SRP, and 1% of alendronate gel and the third group was treated with SRP and aloe vera gel. Clinical parameters were recorded at the baseline and after 6 and 12 months. At the end of the study, the group treated with alendronate gel showed a reduction of PD (4.10 ± 0.56 after 12 months) and an improvement of clinical vertical CAL (3.77 ± 0.29 after 12 months) and horizontal CAL gain (3.71 ± 0.31 after 12 months) compared to the other two groups. From the radiographic analysis emerged that in the group treated with alendronate, there was a greater bone depth reduction (38.09 ± 9.53 after 6 months, 44.86 ± 6.29 after 12 months) compared to the group treated with aloe vera (11.94 ± 15.10 after 6 months, 14.59 ± 25.49 after 12 months). 118 Similar results were obtained in the study conducted by Sharma and Pradeep. 119 Sharma and Pradeep’s study researched the efficacy of 1% of alendronate after SRP compared to a placebo gel. In the study, 73 patients were enrolled, and a total of 66 infrabony defects were treated with 1% alendronate gel or placebo gel. The clinical parameters were recorded at the baseline and after 2 and 6 months. From the radiographic images emerged that in the group treated with 1% alendronate gel, the bone fill percentage (40.4% ± 11.71%) was higher compared to the placebo group (2.5% ± 1.02%). 119 Despite the promising results obtained in the previous studies, it is well known that the use of bisphosphonates is related to osteonecrosis of the jaws. For this reason, further studies are necessary to evaluate the possible adverse effects that this type of drug administered locally in the periodontal pockets can cause.
Pro-resolving lipid mediators such as lipoxin and resolving are another target of the drug against inflammation. These molecules are normally present in the human body and participate in the resolution of inflammatory processes. Through the development of technology, nano-pro-resolving medicines (NPRM) were created in the medical field, microparticles able to load the drug and aim at a specific tissue. Van Dyke et al. 120 created and tested in animals a new type of microparticle associated with lipoxin called nano proresolving medicines mean bezol-lipoxin A4 (NRPM-bLXA4). This drug system was tested in animals in which were created surgically bone defects. After the induction of periodontal defects randomly, the four quadrants of the animals were treated with NRPM alone with bLXA4 alone or NRPM-bLXA4. At the end of the study, it was seen that elements treated with NPRM-bLXA4 showed an improvement in periodontal tissue regeneration. Presurgical PD range from 6 to 8 mm, while at the end of the experiment PD range from 1 to 3 mm. The histological evaluation showed a periodontal regeneration in the tested sites (p = 0.012). Moreover, the concentration of inflammation and resolving mediators in blood was evaluated. The blood sample results showed that using this drug system has raised the blood level of pro-resolving endogenous mediators (RvD1 2.4, RvD2 5.8, RvD3 0.7, RvD5 0.7, RvD6 19.6) and endogenous lipoxins (LXA4 1.3, LXB4 69.5, LTB4 4.1), whereas it has decreased the level of proinflammatory prostaglandins-2 (PGE2) 3.4 and PGD2 (9.2). This study showed that using this new target of drug system can allow obtaining the periodontal regeneration and the resolution of the periodontal inflammation. 120
It was found that using anti-inflammatory and other medication increases medical efficacy. For instance, an animal trial study used a dual nanogel of triclosan and flurbiprofen for periodontal treatment. The inhibitory zones where the nanogel were administered were twice larger than those where the mixture of triclosan and flurbiprofen was administered. This showed how drugs used together possess superior capabilities. 121 Also, the combination of chlorhexidine dihydrochloride and ibuprofen showed to give better periodontal treatment outcomes than using drugs individually. This result comes from a study where these drugs were used with PLGA contained within an in situ forming implant (ISFI). The study was conducted on 24 mice, which induced periodontitis. Infrabony defects were randomly divided into three groups: the test group (treated with ISFI and chlorhexidine dihydrochloride and ibuprofen), the control group (untreated), and a group treated with ISFI without drugs (placebo). After 7 days post-administration, it was seen that there was a decrease of inflammation in the tester group compared to the other two groups (59% versus untreated and 56% versus placebo, p < 0.05). Additionally, histomorphometry analysis has shown that in the test group, there was an improvement in periodontal wound healing after 7 days (41% versus untreated and 22% versus placebo p < 0.05) and 15 days (50% versus untreated and 47% versus placebo, p < 0.05). Moreover, this study showed the potential use of ISFI as delivery system in in vivo lesion sites 122 (Table 1).
Some of the anti-inflammation LDDS for the treatment of periodontitis.

CCL22, C-C motif chemokine ligand 22; LDDS, local drug delivery system; PLGA, poly lactic-co-glycolic acid.
It is possible to conclude saying that, from a systematic review, it was seen that the local use of statins gel in defects with PPD ⩾ 5 mm and vertical bone loss ⩾3 mm associated with non-surgery treatment showed a reduction of PPD (1.83 mm) after 6 months. Furthermore, the systemic administration of sub-antimicrobial doses of doxycycline associated with non-surgery treatment improved the clinical results of patients affected by stages III and IV of periodontitis. 77 Further studies are fundamental to discovering new host modulator drugs that can be used with LDDS for the treatment of periodontitis.
Regeneration LDDS
In periodontitis, the endurance of inflammation has resulted in the demolition of the softer and hard tissue around the elements. 4 For this reason, the correction of alveolar bone defects and periodontal tissue regeneration are parts of the periodontal treatment.123,124 The main aims of contemporary periodontal therapies are the regeneration of periodontal tissues. These therapies consist of the association of frameworks, cells, and molecules that guide bone and tissue regeneration. 125 Various drugs combined with a delivery system were developed in the periodontal regeneration field, such as osteogenesis drugs and growth factors. 126
Membranes are the most often used devices for periodontal regeneration. In the beginning, there were only non-biodegradable membranes. Nowadays, there are also biodegradable membranes; some can be loaded with growth factors and osteogenesis drugs. 127 Park et al. 128 showed that using poly (L-lactide) membranes loaded with growth factor-BB could stimulate the apposition of new bone in rats. After 14 days, it was seen from the histological imagine the new bone apposition in the tissue of the rats treated. Liu et al. 129 created a dual-drug core–shell nanofiber membrane loaded with an inhibitor (SP600125) and a human bone morphogenetic protein-2 (BMP-2). This drug system was tested in dogs with class II function defects. The sample dogs were divided into six groups: group A control, group B untreated, group C SP-PMs, group D BMP-2, group E SP600125-loaded polymeric micelles (SP-PMs)/BMP-2, and group F single. Every month in each group was evaluated the bone volume using a micro-computerized tomography (TC). After 3 months in the dogs tested with membrane loaded with drugs, bone apposition increased (p < 0.01) compared to the group that used the membrane without drugs. The results obtained showed that using the dual-core–shell system allows prolonging the release of the drugs to reduce inflammation and promote bone apposition. Another study using this type of membrane demonstrated to be able to: inhibit inflammation, avoid the defect’s development, and stimulate and accelerate bone healing.
Despite the promising results obtained by using the membranes, these systems have the disadvantage of exposing themselves to external force. 123 For this reason, another scaffold regenerative device has been developed. Iqbal et al. 130 formulated a spongy scaffold made of chitosan and hydroxyapatite loaded with doxycycline. Five types of this scaffold with different weight % were formulated and tested in mouse pre-osteoblasts cells culture. All five scaffolds were shown to promote pro-osteoblast cell viability, but the 10% and 25% weight scaffold formulations have shown greater promotion in the pre-osteoblast adhesion, viability, and differentiation. 130 Scaffolds can be used as a carrier to delivery stem cells. Baba et al. developed a resorbable framework loaded with mesenchymal stem cells and platelet-rich plasma (PRP). This scaffold was used in a group of 10 patients affected by periodontitis with PD ⩾ 4 mm. The intraoral radiographs did before the treatment were compared to the new radiographs taken after 1, 2, 3, 4, 6, 12, 24, and 36 months. All the participants received the non-surgery periodontal therapy. Three sites with one-wall defects, six with two-wall defects, and one with three-wall defects were treated with the resorbable framework loaded with mesenchymal cells and PRP. Two healthy teeth for each patient were considered as a control group. From the radiographs and the clinical parameters, this treatment showed to improve CAL (gain 3.24 mm) (p < 0.0001), reduce PD (−3.6 mm) (p < 0.001), and bone defects (increase of 4.7 mm) (p = 0.0039) after 36 months. The use of this scaffold showed an improvement in hard and soft tissue regeneration in the teeth of patients affected by periodontitis. Moreover, the study showed how the use of stem cells loaded in a scaffold could be an alternative therapy in the periodontal regenerative field. 131
It was seen that natural biomaterials, such as heparin and heparan sulfate glycosaminoglycans, are able to protect and prolong the release of bioactive molecules. Based on this discovery, a nanosphere-encapsulated microsphere system was developed recently as a new delivery device for bone regeneration. In the in vivo study, this delivery system loaded with CCL22 was shown to stimulate the increase of bone growth factors such as BMPs 4 and 7. The presence of these growth factors allows the regeneration of the alveolar bone in the animal model. 107 Zhang et al.108,109 developed a core/shell-structure microsphere where growth factors stimulate the defects healing, and simvastatin reduces the inflammation. The use of this delivery system showed good biocompatibility, a decrease of apoptosis and inflammation of this delivery system.
Gel-based is another type of delivery system. A biological hydrogel was studied based on hyaluronic acid and fibroblast growth factor type 2 (rhFGF-2/HA) for the periodontal regeneration treatment. In this study, 60 periodontopathic patients with infrabony defects were enrolled. They were randomly divided into two groups, the test group and the control group. The control group received the open flap debridement, whereas the tester group received open debridement and the application of rhFGF-2/HA. After 1-year the group of patients in which this hydrogel was administered showed a reduction of PD (4 mm) and the probing attachment level 132 (>4 mm) compared to the control group. This result suggests that this new approach could offer an alternative way to treat periodontal defects. 133 LDDS allows drugs to be delivered into bone defect sites and controls their release. Applying hydrogel loaded with osteogenesis drugs can improve the efficiency of bone regeneration. For this reason, Gunjigaur Vemanaradhya et al. 134 formulated a methylcellulose carrier loaded with 1.2% of rosuvastatin gel. The study enrolled 46 patients affected by periodontitis and divided them into two groups: patients of group I were treated with SRP, whereas those of group II were treated with SRP associated with 1.2% of simvastatin gel. The clinical parameters were recorded before and after 45 days of the treatment. At the end of the study, in both of the groups, there was an enhancement of the clinical parameters, but in group II, there was a greater improvement of PI, GI, and Sulcus Bleeding Index (SBI) (p < 0.001). It was seen that the administration of this hydrogel decreases the inflammation (p < 0.001).
Periodontal regeneration is a complex process involving different aspects, such as controlling the infection, reducing inflammation, and stimulating the stem/progenitor cells to heal periodontal tissue. 135 To achieve these aspects is fundamental to deliver multiple drugs together. 136 The release of different drugs nowadays is possible due to the development of LDDS. For example, Lee et al. developed a controlled-release system where PLGA microspheres loaded with lovastatin were encapsulated into chitosan loaded with tetracycline. The structure allows to release at first tetracycline to manage the infection followed by the liberation of lovastatin to reduce inflammation and stimulate bone formation. This system was administered in dogs with different wall defects to investigate its efficacy. The results showed that this system was biocompatible and able to stimulate bone growth and control infection and inflammation. 79
LDDS periodontitis and systemic diseases
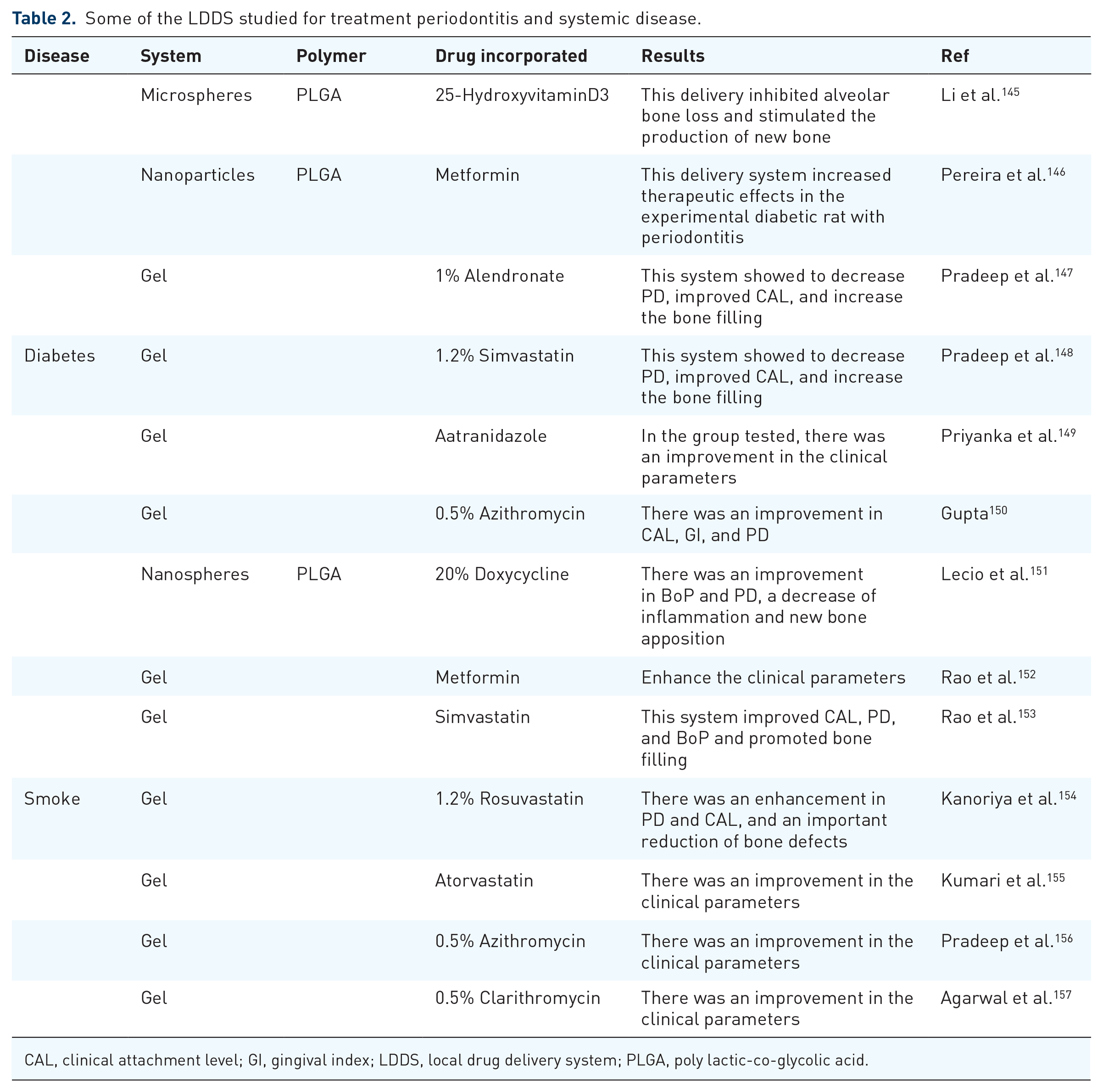
Many studies have shown that periodontal disease is linked with systemic diseases, for instance, diabetes, cardiovascular disease, pregnancy complications, and some inflammatory disease. Recently, other diseases were discovered to have a correlation with periodontitis, such as obesity, metabolic disease, respiratory diseases, cognitive disorders, and rheumatoid arthritis137–144 (Table 2).
Some of the LDDS studied for treatment periodontitis and systemic disease.
CAL, clinical attachment level; GI, gingival index; LDDS, local drug delivery system; PLGA, poly lactic-co-glycolic acid.
Both types of diabetes mellitus are two of systemic diseases that can induce and increase periodontitis. Different LDDSs were studied to introduce a new periodontal treatment for diabetic patients. Lie et al. 145 created a polylactic acid 69 microsphere loaded with 25-hydroxyvitamin D3 (25OHD). 25 OHD, also called vitamin D, controls the cell’s apoptosis and proliferation. 158 Polylactic acid (PLA) is a biodegradable biocompatibility polymer that creates microspheres as drug carriers. 159 In Lie’s study, 40 rats were enrolled that were divided into four groups based on the type of treatment received: a control group (healthy), an untreated group, the group treated with drug-free microspheres (DPF), and a group treated with 25OHD-loaded microspheres. Diabetes and periodontitis were inducted in rats; in particular periodontitis was prompted in the second maxillary molars. In the DPF and DPV groups, 1 mg/mm of the different types of drugs were administered into the periodontal pockets. From the histologic analyses emerged that in the group treated with DPV, there was an important bone formation (p < 0.05) in the inter-proximal area between the first and second molar compared to the other three groups. At the end of the experiment, it was seen that this delivery system had prolonged the half-life of the 25OHD, avoiding the loss of alveolar bone and promoting the growth of new bone. 145 Metformin is a common agent used for its anti-hyperglycemic activity in the therapy against diabetes. Moreover, it possesses anti-inflammatory and anti-absorption activities. Pereira et al. 146 2018 tested the efficacy of a delivery system composed of PLGA NPs loaded with metformin. The experiment was conducted on rats divided into: groups treated with PLGA drug-free, another control group affected by both the pathologies and untreated, experimental group 1 treated only with metformin and experimental group 2 treated with PLGA loaded with metformin. The histological analyses showed a significant reduction of bone loss (p < 0.05) and inflammation (p < 0.05) in the group treated with PLGA and metformin compared to the other groups. From the radiographic analysis, we saw a decrease in bone loss (0.81 ± 0.28 mm) in the group treated with the delivery system. The authors concluded saying that using PLGA NPs loaded with metformin can increase the therapeutic effects in the experimental diabetic rat with periodontitis. 146 Bisphosphonates are a class of drugs that were studied as an adjunctive therapy in periodontitis. Pradeep et al. 147 tested a gel with 1% of alendronate in 41 patients affected by type 2 diabetes and periodontitis. The patients enrolled presented PD ⩾ 5 mm or CAL ⩾ 4 mm and vertical bone loss ⩾3 mm. The recruited patients were randomly divided into a control (treated with a placebo) and a test group (treated with alendronate gel). All patients received the SRP therapy before the drug administration. BoP, PD, CAL, and PI were recorded at the baseline after 2 and 6 months. The group treated with this gel showed a significant reduction (2.44 ± 0.92 mm) of the infrabony defects depth compared to the control (0.14 ± 0.07 mm). Moreover, there was an improvement of PI (p < 0.05) in the tester group compare the placebo group after 6 months. However, there was no significant difference (p > 0.05) about PD, CAL, and BoP after 2 and 6 months between the two groups. Furthermore, Pradeep et al. 156 had tested another gel loaded with 1.2% of simvastatin in patients affected by periodontitis and type 2 of diabetes. In the study, 38 patients with PD ⩾ 5 mm or CAL ⩾ 4 mm and vertical bone loss ⩾3 mm were enrolled. They were divided into two groups: placebo and tester. All the patients received the SRP at the baseline before the drugs administration. Clinical parameters including PD, CAL, PI, and BoP were recorded at the baseline, after 3, 6 and 9 months. At the end of the study, there were a significant enhance of BoP (p < 0.05), PD, and CAL (p < 0.001), the reduction of infrabone defects depth (1.38 ± 0.73 and 1.62 ± 0.71 mm at 6 and 9 months) and the new bone formation (27.63% ± 13.14% at 6 months and 32.64% ± 12.90% at 9 months) in the tester group compared the control ones. 148 Different types of antibiotics were tested to treat patients with diabetes and periodontitis. Priyanka et al. 149 administered a gel with 3% of satranidazole in diabetic patients affected by periodontitis. In the study, 64 diabetic (type 2) patients with PD ⩾ 5 mm were enrolled. They were divided into two groups: placebo and tester groups. In both groups, patients performed SRP at the baseline. The subgingival administration of this antimicrobial, as adjunctive therapy, enhances the periodontal clinical parameters, including PD (reduction of 4.73 mm) and CAL (3.92 mm gain) of the tested group. 149 In another study, it was administered, after SRP, 0.5% of azithromycin gel in diabetic patients affected by periodontitis. In the study, 25 patients treated 50 pockets depth (greater than 5 mm) were enrolled. The patients were divided into two groups: a control (treated with SRP) and a test group (treated with SPR + gel of azithromycin). Each site was evaluated with clinical parameters including GI, PD, and CAL at baseline, after 6 and 12 weeks. At the end of the study, it was seen a significant improvement in the CAL (p < 0.001) (4.40 ± 1.04 mm gain at 12 weeks), the GI (1.02 ± 0.048 reduction after 12 weeks), and the PD (4.84 ± 0.59 mm decrease after 12 weeks) in patients tested compared to the control group. 150 Lecio et al. 151 administered nanospheres of PLGA containing 20% of doxycycline to treat diabetic patients affected by periodontitis. A total of 40 patients with PD ⩾ 5 mm and diabetes type 2 were enrolled in the study. They were randomly divided into two groups. One group received SRP and PLGA alone, whereas the other group (tester) received SRP and PLGA loaded with 20% doxycycline. After the SRP, in the subgingival pockets these LDDSs were administered. Clinical parameters were measured at the baseline after the treatment at 1, 3, 6, and 9 months. At the end of the study, it was seen an improvement of the periodontal clinical parameters (p < 0.05) such as BoP (p < 0.05), CAL (gain ⩾ 2 mm), and PD (reduction of 0.71 mm). Moreover, doxycycline allows to decrease the inflammation (p < 0.05) and stimulate the production of anti-inflammatory mediators in the group tested. 151
Many studies have revealed that smokers have an elevated risk of suffering from periodontitis. 160 Rao et al. 152 prepared a gel loaded with 1% of metformin and tested it in smokers with vertical defects caused by periodontitis. The patients included in this study had PD ⩾ 5 mm, CAL ⩾ 4 mm, vertical bone loss ⩾3 mm and smoked more than 10 cigarettes/day for at least 5 years. A total of 50 patients were enrolled and divided into control (SRP + placebo) and tester (SRP + 1% of metformin gel). The results showed that this type of gel enhances clinical parameters. In particular, after 3 and 6 months, PD and CAL gain (p < 0.05) significantly reduced in the tester group compared to the control. Radiographically a greater new bone apposition (26.17% ± 6.66%) was seen in the tester group after 6 months compared to the control ones (155). They also studied and tested the efficacy of a gel loaded with 1.2% of simvastatin in smokers with periodontitis. The patients enrolled had vertical bone loss ⩾3 mm, PD ⩾ 5 mm, or CAL ⩾ 4 mm and smoked more than 10 cigarettes/day for more than 5 years. In the study, 40 patients were divided into control (SRP + placebo) and tester groups (SRP + simvastatin). From these clinical trials, it was seen that this gel improved CAL (p < 0.001), PD (p < 0.001), bleeding index (p < 0.001), and promoted bone filling (reduction of 1.17 ± 0.45 mm of the infrabony depth). 153 Another type of statin called rosuvastatin was evaluated by Kanoriya et al. 154 In their study, they administered 1.2% of rosuvastatin local gel delivered in the pockets of smoker patients with periodontitis. The study was conducted on 60 patients divided into a control group (SPR + placebo gel) and a test group (SRP + rosuvastatin gel). Clinical parameters were evaluated at the baseline and after 6 and 9 months of the treatments. The results showed an enhance of clinical parameters, for instance, PD (3.66 ± 0.47 mm reduction after 9 months) and CAL (3.26 ± 0.69 mm after 9 months) in the smoker and periodontopathic patients compared to the control group. Additionally, it was seen an important reduction of the bone defects (1.39 ± 0.05 mm after 9 months). 154 Similar results were obtained in the study conducted by Kumari et al., 155 in which atorvastatin was used. The previous studies’ results underline how statins possess a great anti-inflammatory properties and the capability to stimulate bone apposition. Many LDDSs with antimicrobials were evaluated in these years in smokers periodontopathic patients. In a study conducted by Pradeep et al., 156 it was investigated that the use of a gel with 0.5% of azithromycin in smoker patients was affected by periodontitis. In the study, 54 patients were enrolled and divided into control (SRP + placebo gel) and tester groups (SRP + azithromycin gel). The patients enrolled had PD ⩾ 5 mm or (CAL) ⩾4–6 mm, vertical bone loss ⩾3 mm, and smoked more than 10 cigarettes/day for at least 5 years. The results of the study showed that this drug delivery system determined a CAL gain of 2.44 ± 0.64 mm after 9 months. Similar results were obtained in a study in which was locally administered gel with 0.5% of clarithromycin in 61 smoker patients affected by periodontitis. The patients enrolled were divided into control (SRP + placebo gel) and tester group (gel with 0.5% of clarithromycin). At the end of 6 months, the group tested showed an enhance of clinical parameters (p < 0.05) compared to the control group. In particular, in the group tester, there were the reduction of PD (4.64 ± 0.63 mm), GI (1.06 ± 0.28), PI (2.82 ± 0.64), and an improvement of CAL (gain 4.90 ± 0.46 mm). 157 The authors of the both studies justify these results underlining the better efficacy of subgingival administration of the antimicrobials. 156 More research is needed to investigate about the possible side effects of them. Moreover, there are other drugs for the treatment of these systemic diseases that could be tested with the use of the delivery system.
Natural agents
As reported by Worth Health Organization 80% of the world population relies on traditional medicine (herbal) as their primary healthcare. In developing nations, herbs and their derivatives are present in 25% of medical drugs used.161,162 China and India are two of the most populated countries where for over 2000 years oral infections, including periodontal disease, have been treated using herbal medicine. 163
A new trend is emerging regarding the use of herbal medicine as a nutritional supplement and as a treatment in dentistry. Many types of medical herbs own antibacterial, antimycotic, and antiviral properties. Herbal drugs used in proper concentration do not modify natural flora. Herbal derivatives that possess antibacterial properties are used as a component of toothpaste to hinder bacterial adherence and plaque formation. Some other plant parts are used for their anti-inflammatory and antibacterial properties in other dental treatment modalities in the form of gel, mouth rinsing solution, decoction, and infusions164,165 (Figure 3).

Some of the properties of the herbal medicine discussed in this review.
The use of modern chemotherapeutics has given positive effects for the treatment of periodontic disease but brings with it different side effects such as discoloration of the tooth, antimicrobial resistance, taste alteration, and price. Recently, medicinal plant utilization has risen and could be a new additional therapy for all country the lowest and highest socioeconomic population to the higher one. 166
Turmeric (Curcuma longa) is an Indian spice widely used. In 1910, curcumin, the main bioactive component of turmeric, was identified by Lampe and Milobedzka. Curcumin has a large range of biological properties, including anti-inflammatory, antioxidant, anticarcinogenic, antiviral, and antimicrobial activities. The anti-inflammatory power of curcumin is related to the capability to downregulate the activity of cyclooxygenase 2, lipoxygenase and inhibits the production of inflammatory cytokines. Many studies have been made to discover the efficiency of curcumin as an additional therapy for periodontal disease. A study conducted in 2015 used curcumin gel (10 mg/g) associated with and without photoactivation, add-on SRP as a periodontal treatment. This study demonstrated that adding curcumin gel in SRP instead of only SRP treatment gave a substantial decrease in parameters including SBI, PPD, and CAL. Also, there was a statistically significant decrease of microbial parameters, particularly about A. actinomycetemcomitans and black pigment-producing microorganism after 3 months where curcumin gel was enforced. Moreover, where curcumin gel was associated with multiple photoactivation applications, were obtained better effectiveness. 167
In another study, it was confronted that the efficacy of curcumin gel (2%) and chlorhexidine gel (0.2%) was associated with SRP in a group of 30 cases with periodontitis. The clinical parameters were studied during the day of the treatment, after 30 days and after 45 days. The results of this study showed by comparing the two modalities of treatment that the efficiency of curcumin gel was considerably greater than the chlorhexidine gel in decreasing clinical parameters of periodontitis. The researchers ended up reporting that curcumin gel has superior efficacy in treating mild and moderate periodontal pockets than chlorhexidine. 168
Green tea is contained in the foliage of Camellina sinensis, and it is consumed in the form of beverage in the world. Green tea has polyphenols as bioactive ingredients, and most of them are catechins. 169 These compounds possess antibacterial, antioxidant, anti-inflammatory, and anticarcinogenic properties.170–173 Moreover, green tea catechins possess antioxidant and antibacterial properties against periodontopathogen, particularly P. gingivalis an Prevotella intermedia.174,175 In 2009, an epidemiologic study showed a moderate reverse correlation between the routine supply of green tea and periodontal disease. 176
Many clinical studies have investigated the possibility of using green tea as an adjective treatment against periodontitis. In a recent clinical trial, 30 patients were divided into two groups. In both groups, participants received the procedure of SRP, but in one of the two groups, the participants were invited to drink commercial green tea two times a day for 6 weeks. After this period, the participants of the group that consumed the commercial green tea showed to have significantly reduced probing depth and BoP in relation to the control group. However, there was no significant decrease of PI in the green tea group compared to the control group. 177
Hrishi et al. 178 in 2014 clinical research to study whether using a dentifrice with green tea after SRP could give better results instead of using a fluoride and triclosan dentifrice. Thirty patients were separated in two groups after the initial SRP, the control group was given the fluoride and triclosan dentifrice and the test group the dentifrice with green tea. After 4 weeks, the parameters were recorded, and the test group revealed a statistical decrease in GI, BoP, and CAL. Moreover, some biochemical parameters were recorded in the gingival crevicular fluid: total antioxidant capacity and glutathione-S-transferase activity. Also, this biochemicals parameters showed a reduction after 4 weeks in the test group in comparison to the control group. These results prove the beneficial anti-inflammatory that can be received using green tea as an additional treatment after SRP for periodontitis patients.
Gadagi et al. 179 2013 decided to do a clinical trial testing green tea strips in patients with and without diabetes with periodontitis. They were randomly placed in strips with a placebo and with green tea in periodontal pockets. At the end of the study, which was conducted for 4 weeks, clinical indices (GI, CAL, and PPD) were investigated. The result showed that in the site there were placed strips of green tea in patients with and without diabetes, there was a decrease in the clinical indices in comparison to the control group. Moreover, in healthy patients, the prevalence of P. gingivalis in their pockets has been reduced from 75% at initially to 25% after the trial. Nonetheless, this result has not been found in the periodontal pockets of patients affected by diabetes.
Resveratrol is another polyphenol compound used as herbal medicine from Japan and China (3,5,4-rihydroxystilbene). It is present in red wine, apples, peanuts, and several vegetables. Many preclinical and in vitro studies were performed to analyze the properties of resveratrol, and these showed that resveratrol possesses anti-inflammatory, anti-carcinogenic, and antimicrobial effects. The anti-inflammatory property of resveratrol could reduce pro-inflammatory cytokines, including IL6, IL-1B, IL-8, IL-12, and TNF.180,181 Animal trials were conducted to study the effects of resveratrol as a treatment for periodontitis, and these showed encouraging results. Nevertheless, the effects are not totally studied, making it difficult to introduce resveratrol as a therapeutic agent for clinical trials.182,183
Propolis produced by honeybees is a very elaborate combination of 230 components and more, including flavonoids, cinnamic acids, and their esters (caffeic acid and caffeic acid phenethyl). Propolis has numerous natural properties like anti-inflammatory, antioxidant, antibacterial, antiviral, fungicidal, hepatoprotective, free radical scavenging, immunomodulatory, and anti-glycemic effects.184–186 Most of these therapeutical properties, like anti-inflammatory and antimicrobial, are associated with caffeic acid phenethyl ester, a component of propolis.187–190 Borgnakke 191 in 2017 did a clinical trial in a group of 50 patients with type 2 diabetes and chronic periodontitis distributed in test and control groups. After the initial SRP, one group received one propolis capsule (400 mg/day) for 6 months, and the second group received a placebo. The results demonstrated that in the propolis group, hemoglobin A1C (HbA1C) significantly decreased, and periodontal health improved, although there were no modifications in the control group.
Sanghani et al. 192 in 2014 made a clinical study where 5 mg of propolis was administered as adding SRP in 20 sites of the intervention group. After 1 month, clinical periodontal parameters were improved in the intervention group. In addition, there was an improvement in microbiological parameters, including P. gingivalis, P. Intermedia, and F. nucleatum. The researchers observed these results ended by saying that the use of subgingival delivery of propolis as an addition of SRP has given superior results in clinical and microbiological parameters in patients with periodontitis. In a different study conducted by Pundir 193 in 2017, 30 patients were distributed in two groups and in the intervention group was administered 20% of propolis hydroalcoholic solution twice a week after initial stage mouths disinfection, whereas the control group received only SRP. After 3 months in the intervention group, there was a gain of attachment and a decrease in the numbers of periodontopathogens, and clinical parameters compared to the control group.
Aloe vera (Aloe barbadensis), which belongs to Liliacea family, possess many minerals and vitamins that give immunomodulatory, antiviral, antitumor, anti-inflammatory, anti-aging, and antioxidant properties.132,194,195 For these properties, aloe vera was considered in dentistry to treat many oral diseases such as lichen planus, oral submucous fibrosis, stomatitis, alveolar osteitis, and periodontitis.132,194–201
As a consequence of aloe vera’s properties, various studies were made to evaluate the efficacy of treatment in periodontitis. Kurian et al. 202 2018 did a randomized clinical trial of 90 volunteers distributed in three groups, one group was given SRP and placebo gel, the second one was given SRP + 1% metformin gel, and the third one was given SRP + aloe vera gel. At the end of the study, 1 year, the results demonstrated a significant decrease of GI, BoP, PPD, and CAL in all groups, but in the metformin and aloe vera groups, the decrease of PPD, CAL, and bone fill was higher.
A different study conducted on 20 patients with chronic periodontitis tested the effectiveness of aloe vera gel combined with SRP. After 2 months, GI and PD had a significant decrease in the intervenor group compared to the control group. 203
Conclusion
Currently, in vitro and in vivo studies demonstrate encouraging outcomes on the application of LDDS in the therapy of periodontal disease, inducing an improvement in periodontal clinical parameters. This is due to the refinement this administration technique has undergone over the years and to fewer side effects than systemic administration of different drugs. However, further studies must be conducted to better test these delivery systems in humans and to investigate their possible side effects in patients, especially those with systemic diseases. Significant potential in the treatment of periodontal disease is also induced by medicinal herbs, which are able to improve the clinical parameters of periodontitis with a reduced number of side effects after their utilization in humans. In conclusion, both natural and LDDS agents could be used as a new approach against periodontitis.
