Abstract
Background:
Evidence on whether long-term exposure to air pollution increases the mortality risk in patients with chronic obstructive pulmonary disease (COPD) is limited.
Objectives:
We aimed to investigate the associations of long-term exposure to particulate matter with diameter <10 µm (PM10) and nitrogen dioxide (NO2) with overall and disease-specific mortality in COPD patients.
Design:
We conducted a nationwide retrospective cohort study of 121,423 adults ⩾40 years diagnosed with COPD during 1 January to 31 December 2009.
Methods:
Exposure to PM10 and NO2 was estimated for residential location using the ordinary kriging method. We estimated the risk of overall mortality associated with 1-, 3-, and 5-years average concentrations of PM10 and NO2 using Cox proportional hazards models and disease-specific mortality using the Fine and Gray method adjusted for age, sex, income, body mass index, smoking, comorbidities, and exacerbation history.
Results:
The adjusted hazard ratios (HRs) for overall mortality associated with a 10 µg/m3 increase in 1-year PM10 and NO2 exposures were 1.004 [95% confidence interval (CI) = 0.985, 1.023] and 0.993 (95% CI = 0.984, 1.002), respectively. The results were similar for 3- and 5-year exposures. For a 10-µg/m3 increase in 1-year PM10 and NO2 exposures, the adjusted HRs for chronic lower airway disease mortality were 1.068 (95% CI = 1.024, 1.113) and 1.029 (95% CI = 1.009, 1.050), respectively. In stratified analyses, exposures to PM10 and NO2 were associated with overall mortality in patients who were underweight and had a history of severe exacerbation.
Conclusion:
In this large population-based study of patients with COPD, long-term PM10 and NO2 exposures were not associated with overall mortality but were associated with chronic lower airway disease mortality. PM10 and NO2 exposures were both associated with an increased risk of overall mortality in underweight individuals and those with a history of severe exacerbation.
Introduction
Chronic obstructive pulmonary disease (COPD) is a major global health burden, contributing to 3.2 million deaths worldwide each year. 1 The number of annual COPD-related deaths is estimated to reach 5.4 million by 20602 and to continuously increase due to population aging. 3 Increasing air pollution will also contribute to a higher prevalence and incidence of COPD,4–6 as exposure to air pollutants damages the lung epithelial cells directly and triggers pulmonary and systemic inflammation.7–10
In COPD patients, short-term air pollution exposure increases the risk of COPD-related outcomes.11–13 In meta-analysis studies, short-term exposure (single-day lags or multiday averages) to particulate matter with diameter <10 µm (PM10) increased the risk of COPD-related emergency department visits, hospital admissions, and mortality.11,12 In addition, compared with those without COPD, patients with COPD had a higher risk of all-cause and respiratory mortality after short-term exposure to PM10 and nitrogen dioxide (NO2). 13
However, evidence on whether long-term air pollution exposure increases the mortality risk in COPD patients is limited. In a study of older adults (⩾65 years) with a discharge diagnosis of COPD from emergency admissions in the United States, annual PM10, averaged over 1–4 years (same year and 3 preceding years), was associated with higher all-cause mortality. 14 This study, however, only included older adults who had severe symptoms requiring an emergency department admission, and smoking status, a key risk factor for both COPD and mortality, was not considered. Furthermore, the health effects of long-term exposure to NO2 have not been evaluated in COPD patients. Thus, we used national health insurance data to evaluate the associations of long-term exposure (up to 5 years) to PM10 and NO2 with all-cause and disease-specific mortality in COPD patients.
Patients and methods
Study population
We conducted a retrospective cohort study using data from the South Korean National Health Insurance Service (NHIS). South Korea has a single-payer national health insurance system that covers more than 97% of all citizens. The NHIS maintains records of all inpatients and outpatient visits, prescriptions, and procedures in the National Insurance Database (NHID), including information on free-of-charge health screening exams provided by the NHIS annually or biennially. Details of the NHIS-NHID are published elsewhere.15,16
All patients ⩾40 years who met the definition of COPD (see definition below) during 1 January to 31 December 2009, the earliest year available for air pollutants levels, were included (N = 269,595) (Figure 1). We excluded individuals who died in 2009 (N = 11,522) or did not undergo a health screening exam between 1 January 2007 and 31 December 2009 (N = 126,043). We then excluded participants with missing information on smoking status (N = 8,593), body mass index (BMI, N = 120), or area of residence (N = 11) and participants whose air pollutant levels in their area of residence could not be estimated (N = 9,222). The final sample size was 121,423 participants (76,843 men and 44,580 women). We used retrospective data from a nationwide study of COPD cases in Korea. We used all data available from eligible participants and did not perform power calculations to select the sample size.

Flow chart of study participants.
Data sources
The NHIS data comprise four databases on insurance eligibility (demographics, area of residence, and insurance status), medical treatments (claims of hospital admissions, outpatient visits, treatments and procedures reimbursed by the NHIS and details of the associated diseases, and prescriptions), medical institutions, and health screening exams. Disease diagnosis was coded using the International Classification of Diseases–Tenth Revision (ICD-10),15–17 and prescriptions were coded using the Korean Drug and Anatomical Therapeutic Chemical codes for all reimbursed claims. The claims data are routinely audited by the NHIS to improve reliability.
COPD was defined as the presence of ICD-10 J43.X (except J43.0) or J44.X and a prescription for any single or combination of COPD drugs at least twice in 2009. COPD drugs were defined as long-acting muscarinic antagonists, long-acting beta-2 agonists (LABA), inhaled corticosteroids with LABA, short-acting muscarinic antagonists (SAMA), short-acting beta-2 agonists (SABA), a combination of SAMA and SABA, methylxanthines, and systemic beta agonists.18,19
The presence of exacerbation was defined in the same year as COPD diagnosis. Moderate exacerbation of COPD was defined as an outpatient visit with an ICD-10 J43.X (except J43.0) or J44.X and a prescription for systemic steroids or antibiotics on the same visit. Severe exacerbation of COPD was defined as an inpatient or emergency room visit with one of the following ICD-10 codes: COPD [J43.X (except J43.0) or J44.X], or COPD-related disease [pneumonia (J12.X–J17.X), pulmonary thromboembolism (I26, I26.0, or I26.9), dyspnea (R06.0), or acute respiratory distress syndrome (J80)], and a prescription for systemic steroids or antibiotics at the same visit. 20
Age was categorized as 40–49, 50–59, 60–69, 70–79, 80–89, or ⩾ 90 years. Income levels were categorized as ⩽30th, 30–70th, or >70th percentiles. Residential area was categorized as Seoul, metropolitan cities (Busan, Daegu, Daejeon, Gwangju, Incheon, and Ulsan), and others. BMI was categorized as underweight (<18.5 kg/m2), normal (18.5–<23 kg/m2), overweight (23–<25 kg/m2), or obese (⩾25 kg/m2) based on the Asian-Pacific criteria. 21 Smoking status was categorized as never, former, or current based on the self-administered questionnaire at the health screening exams. The presence of COPD exacerbation was none, moderate, and severe. The presence of comorbidities was identified using ICD-10 codes for any hospital visit in the same year as the COPD diagnosis and was summarized using the Charlson Comorbidity Index (CCI). 22
Air pollution measurements
We collected hourly concentrations of PM10 and NO2 measured at 429 monitoring stations in South Korea between 1 January 2009 and 31 December 2013 from Korea’s Ministry of the Environment. 23 PM10 was measured using the β-ray absorption method and NO2 using the chemiluminescence method. 23 NO2 was measured in ppm and was transformed to µg/m3 using conversion factor 1.88. 24 For each station, the daily average concentrations of each air pollutant were calculated using only the days when ⩾75% of the hourly measurements were available and were considered missing otherwise. The numbers (%) of missing observations for daily average PM10 and NO2 between 2009 and 2013 were 2,832 (2.8%) and 2,134 (2.1%), respectively. 23
Based on the average measured daily concentrations, we estimated the daily average exposure concentrations of PM10 and NO2 at the neighborhood level (for urban regions) or a town/township level (for rural regions) using ordinary kriging (R-package ‘Automap’) 25 with a spatial resolution of 0.01° × 0.01°. The performance of each kriging model was evaluated using 10-fold cross-validation. The R 2 of PM10 and NO2 in 2009 was 0.35 and 0.50, respectively. To estimate long-term exposure, we generated 1-year (1 January 2009 to 31 December 2009), 3-year (1 January 2009 to 31 December 2011), and 5-year (1 January 2009 to 31 December 2013) average concentrations of each air pollutant (Supplementary Figure 1). In addition, we calculated the number of days that exceeded the World Health Organization (WHO) standards for PM10 (24-h mean concentration = 50 µg/m3) during the 1-, 3-, and 5-year period, as the WHO standards for 24-h mean NO2 were unavailable. 26
Study outcomes
Vital status, date of death, and cause of death information in the NHIS-NHID are linked to the death certificate information from the National Statistical Office. The primary outcome was overall mortality. Secondary outcome was disease-specific death, which was classified as lung cancer (C34), other cancers (C00–D48, except C34), pneumonia (J12–J18), chronic lower airway disease including bronchitis, emphysema, COPD, asthma, and bronchiectasis (J40–J47), cardiovascular disease (CVD, I20–I25, I30–52), cerebrovascular disease (I60–69), diabetes mellitus (E10–14), hypertension (I10–15), neurologic disorders (G00–G99), injuries (S00–T98), and others. 18
Statistical analysis
Each participant was followed up from 1 January 2010, until death or the end of the study period (31 December 2018), whichever was first. We estimated the hazard ratios (HRs) and 95% confidence interval (CI) for overall mortality associated with a 10-µg/m3 increase in the 1-year average concentrations of PM10 and NO2 using Cox proportional hazards models stratified by residential area (Seoul, metropolitan cities, and others) and adjusted for baseline age, sex, income level, BMI (continuous), smoking status, CCI at the time of COPD diagnosis, and the presence of exacerbation (none, moderate, and severe). In addition, we estimated the HRs using the 3-year and 5-year average concentrations of each air pollutant as exposure. For these analyses, follow-up started on 1 January 2012 (N = 141,081) and 1 January 2014 (N = 137,089), respectively, and those who died in the year used to define COPD diagnosis were excluded. We checked the proportional hazards assumption by including interaction terms of all predictors with time and found no evidence of departure from model assumptions. In addition, we evaluated nonlinear relationship between air pollution concentrations and overall mortality using restricted cubic splines for PM10 and NO2 with three knots at 10th, 50th, and 90th percentiles.
For secondary outcomes, we estimated subdistribution HRs (95% CIs) for each cause of death per 10 µg/m3 increase in 1-year average concentrations of PM10 and NO2 using the Fine and Gray method to account for competing risks. 27 For each cause of death, all the other causes of death were considered as competing events. We also estimated the HRs (95% CIs) for overall mortality and each cause of death associated with a 10-day increase in the number of days exceeding the PM10 WHO standard. The models were adjusted for the same confounders as above.
In addition, we performed stratified analyses by pre-defined subgroups: age (<60, 60–69, and ⩾70 years), sex, BMI category, smoking status, CCI category (1, 2, and ⩾ 3), and the presence of exacerbation. As a sensitivity analysis, we repeated the analyses for overall mortality excluding potential intermediate variables, such as BMI and the presence of exacerbation. We adjusted for the same confounders used in overall mortality. All analyses were performed using Statistical Analysis System (SAS) version 9.4 (SAS Institute Inc., Cary, NC, USA) and R statistical software, version 3.6.2.
Results
Participants and air pollutant concentrations
Among the 121,423 participants (76,843 men and 44,580 women), 84% were ⩾60 years, 60% were never smokers, nearly 16% had experienced severe exacerbation, and 77% had at least one additional comorbid condition (Table 1). Compared with women, men were more likely to be former or current smokers and have experienced severe exacerbation.
Baseline characteristics of study participants in 2009.
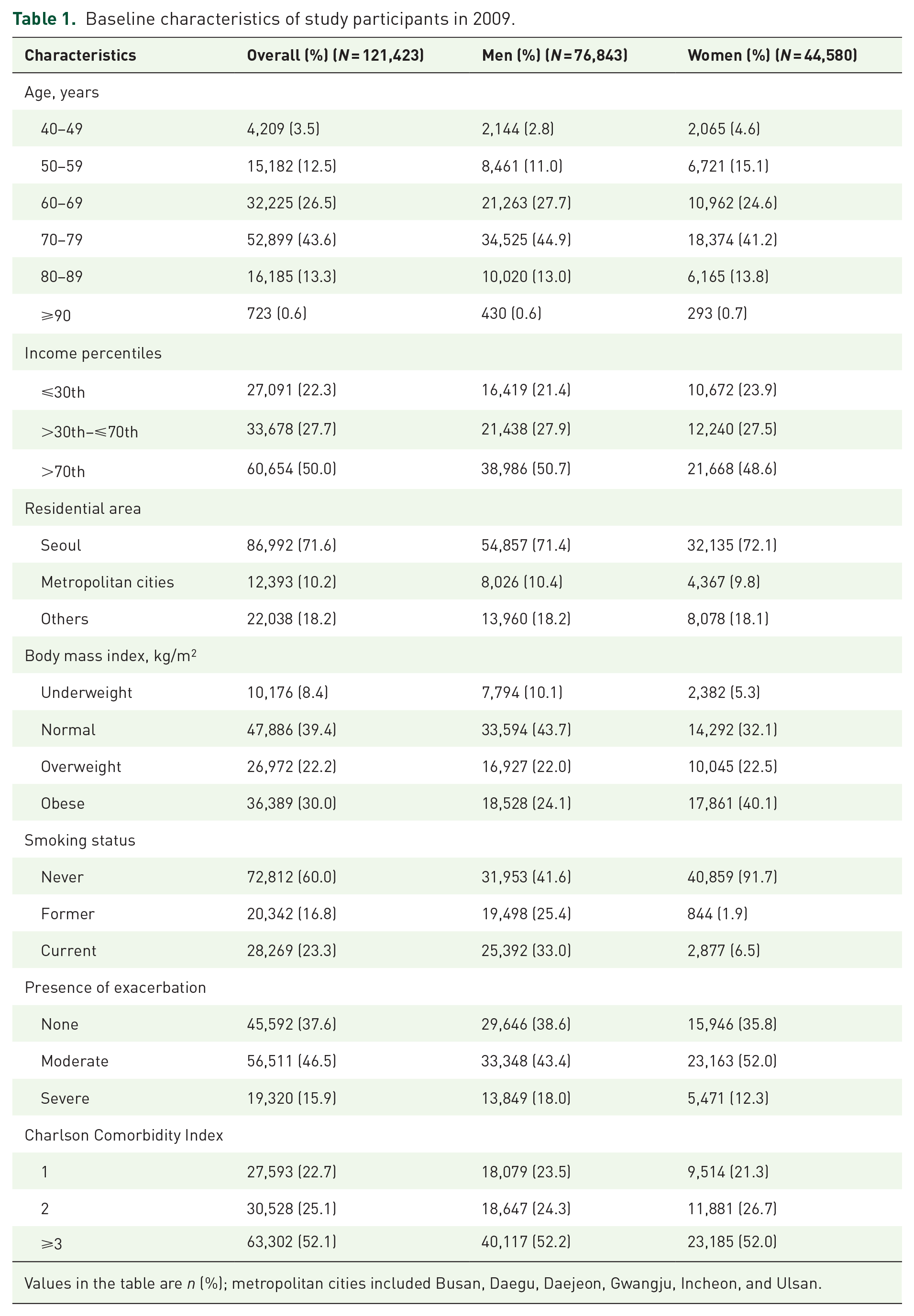
Values in the table are n (%); metropolitan cities included Busan, Daegu, Daejeon, Gwangju, Incheon, and Ulsan.
During the median follow-up of 4.4 years (889,079.0 person-years), 44,266 participants died [incidence rate (IR) = 49.8 per 1000 person-years]. Participants who died were more likely to be male, underweight or normal weight, former or current smokers, have experienced severe exacerbation, and have CCI ⩾3 (Supplementary Table 1). Of the 141,081 participants followed up since 2012, 29.3% died, and of the 137,089 participants followed up since 2014, 21.7% died during the study period (Supplementary Tables 2 and 3).
The median [interquartile range (IQR)] 1-year average PM10 and NO2 concentrations were 49.9 (47.7–56.6) µg/m3 and 36.9 (30.0–48.2) µg/m3 in 2009 (Supplementary Table 4). Air pollution concentrations in this study population differed between the regions with the highest PM10 concentrations in the Metropolitan cities and the highest NO2 concentrations in Seoul (Figure 2 and Supplementary Figure 2). During the study period, the annual mean PM10 concentrations declined in all regions, whereas the NO2 concentrations remained relatively constant (Supplementary Figure 2). In 2009, the median number of days of above the WHO standards of PM10 was 140 (125–180) (Supplementary Table 5).

Distribution of 1-, 3-, and 5-years average concentrations of (a) PM10 and (b) NO2 by region.
Air pollution and mortality risk
The adjusted HRs for overall mortality per 10 µg/m3 increase in 1-, 3-, and 5-year PM10 exposure were 1.004 (95% CI = 0.985, 1.023), 1.006 (95% CI = 0.985, 1.027), and 1.019 (95% CI = 0.994, 1.046), respectively (Table 2). The adjusted HRs for overall mortality per 10 µg/m3 increase in 1-, 3-, and 5-year NO2 exposure were 0.993 (95% CI = 0.984, 1.002), 0.995 (95% CI = 0.985, 1.004), and 0.990 (95% CI = 0.979, 1.001), respectively. There was no evidence for a nonlinear association between PM10 and overall mortality (p value for nonlinearity = 0.10), whereas NO2 was nonlinearly associated with overall mortality (p value for nonlinearity = 0.003; Supplementary Figure 3).
Adjusted hazard ratios (95% confidence intervals) for overall and disease-specific mortality per 10 µg/m3 increase in PM10 and NO2 by exposure periods.
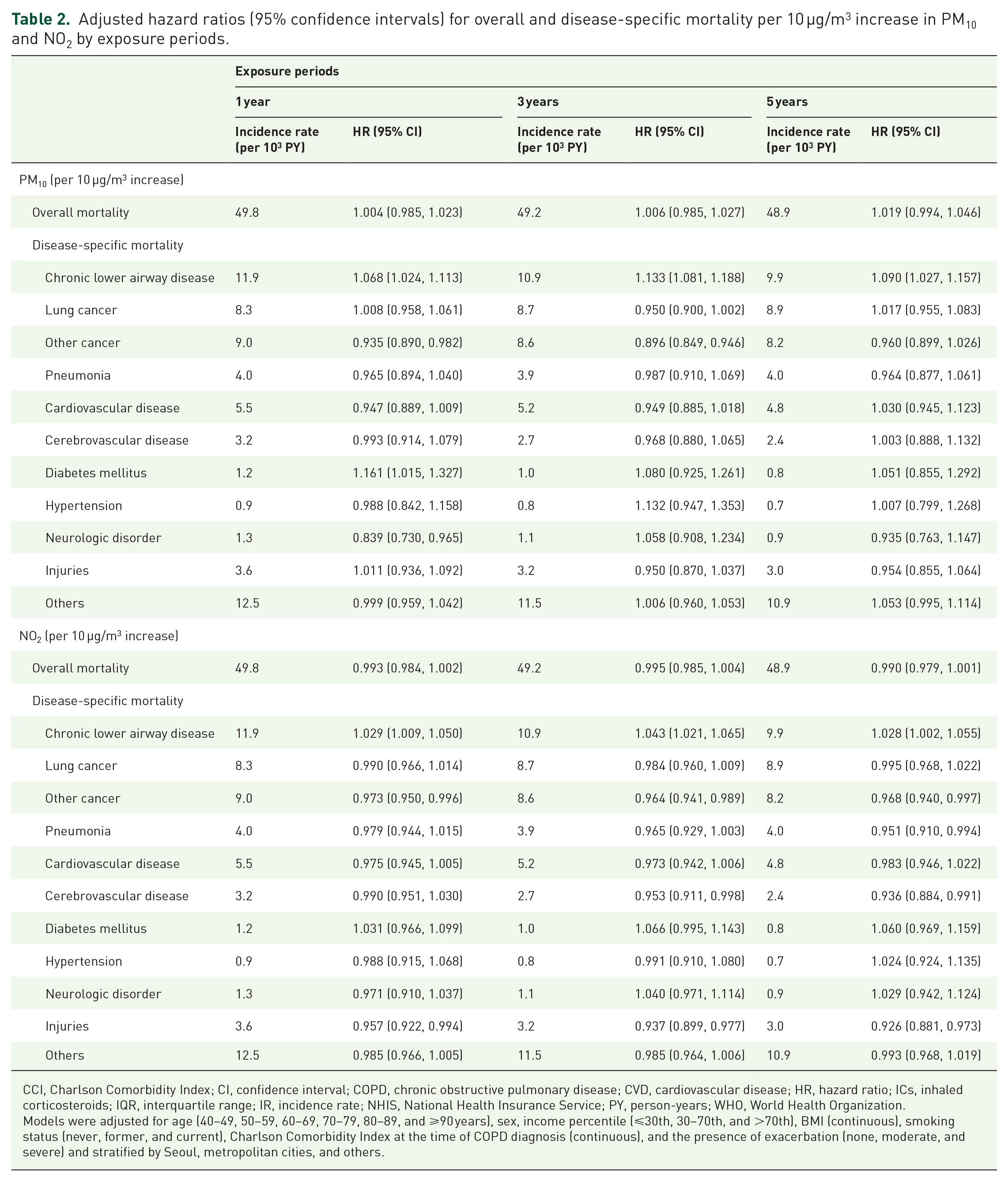
CCI, Charlson Comorbidity Index; CI, confidence interval; COPD, chronic obstructive pulmonary disease; CVD, cardiovascular disease; HR, hazard ratio; ICs, inhaled corticosteroids; IQR, interquartile range; IR, incidence rate; NHIS, National Health Insurance Service; PY, person-years; WHO, World Health Organization.
Models were adjusted for age (40–49, 50–59, 60–69, 70–79, 80–89, and ⩾90 years), sex, income percentile (⩽30th, 30–70th, and >70th), BMI (continuous), smoking status (never, former, and current), Charlson Comorbidity Index at the time of COPD diagnosis (continuous), and the presence of exacerbation (none, moderate, and severe) and stratified by Seoul, metropolitan cities, and others.
Among disease-specific mortalities, chronic lower airway disease (19.6%, N = 8,679) was the most common cause of death, with an IR of 11.9 per 1000 person-years (Table 2). For a 10-µg/m3 increase in 1-year PM10 and NO2 exposures, the adjusted HRs for chronic lower airway disease mortality were 1.068 (95% CI = 1.024, 1.113) and 1.029 (95% CI = 1.009, 1.050), respectively. In addition, 1-year exposure to PM10 was associated with an increased risk of diabetes-related mortality. We did not find any statistically significant associations of PM10 and NO2 with other causes of death (Table 2). The associations were similar for the 3- and 5-year exposures, and when we used the number of days exceeding the PM10 WHO standards (Supplementary Table 6).
In the subgroup analysis, long-term exposure to PM10 was associated with overall mortality in adults aged 60–69 years, women, underweight individuals, current smokers, and those with a history of severe exacerbation (Figure 3). For NO2, long-term exposure was associated with overall mortality in underweight individuals and those with a history of severe exacerbation. Both air pollutants, however, were associated with a lower risk of overall mortality in individuals who had no additional comorbidity other than COPD. Excluding BMI and the presence of exacerbation from the final model did not change the results materially (Supplementary Table 7). The results were similar for the number of days exceeding the WHO standards for PM10 (Supplementary Figure 4).

Hazard ratios (95% confidence intervals) for overall mortality by subgroups. Hazard ratios for the associations of (a) PM10 and (b) NO2 with overall mortality are adjusted for age (40–49, 50–59, 60–69, 70–79, 80–89, and ⩾90 years), sex, income percentile (⩽30th, 30–70th, and >70th), BMI (continuous), smoking status (never, former, and current), Charlson Comorbidity Index at the time of COPD diagnosis (continuous), and the presence of exacerbation (none, moderate, and severe) and stratified by Seoul, metropolitan cities, and others.
Discussion
In this nationwide study of COPD patients, long-term exposures of 1–5 years to PM10 and NO2 were not associated with overall mortality; however, they were associated with mortality due to chronic lower airway disease. In stratified analyses, exposures to PM10 and NO2 were associated with overall mortality in patients who were underweight and had a history of severe exacerbation.
In this study, long-term exposure to PM10 and NO2 was not associated with overall mortality, which differs from previous studies. In a meta-analysis, short-term daily exposure to PM10 was associated with a higher risk of acute exacerbation [pooled odds ratio (OR) = 1.03; 1.02, 1.04] and mortality (pooled OR = 1.01; 1.008, 1.014) in patients with COPD, 11 and an increase of 50 µg/m3 in daily average NO2 with a 2-day lag increased overall mortality (relative risk = 1.15; 1.01, 1.30) in patients who attended emergency room for COPD. 28 Long-term PM10 exposure up to 4 years was associated with higher overall mortality (HR = 1.22; 1.17, 1.27) in a cohort of older adults (⩾65 years) who had been discharged from an emergency department for COPD in the United States between 1985 and 1999. 14
The differences between these results and our findings may be due to different adjustment factors, selection of study populations, and different exposure periods. First, most studies did not account for smoking status, which is a major risk factor for COPD and mortality,11,14,28 or the presence of exacerbation, which increases the risk of mortality in patients with COPD.11,14,28,29 Indeed, we found that current smokers had a higher risk of overall mortality with PM10 exposure and patients with a history of severe exacerbation had a higher risk with PM10 exposure. Second, this study included adults ⩾40 years who were diagnosed with COPD, whereas other studies included participants who had not been diagnosed with COPD or were restricted to older adults with COPD.11,30,31 This study showed a significant association between long-term air pollution exposure and overall mortality in patients with a history of severe COPD exacerbation. Our finding was consistent to previous study by observing that long-term PM10 exposure was associated with higher overall mortality in older adults who had been discharged from an emergency department for COPD indicating severe exacerbation. 14 These findings would suggest that patients with more severe COPD are particularly susceptible to systemic and pulmonary inflammation upon exposure to noxious particles than those with mild to moderate COPD. 32 Finally, the biological mechanisms leading to mortality may differ between short- and long-term exposure to air pollution in COPD patients. Short-term exposure to air pollution may trigger acute inflammation of the airways and exacerbation, 33 resulting in increased mortality,33,34 whereas long-term exposure leads to airway remodeling, such as fibrosis and smooth muscle hyperplasia, 33 therefore, COPD progression. 35 Further studies are needed to elucidate the different mechanisms of short- and long-term exposure.
The most common cause of death was chronic lower airway disease, and its risk was higher with exposure to PM10 or NO2. PM increases the permeability of lung epithelial cells by direct damage and accumulation in the airways. 7 Long-term exposure to air pollution increases oxidative stress due to lung inflammation and triggers pro-inflammatory signaling pathways locally and systematically.8,10 Air pollution exposure also induces the regulation of genes involved in inflammation, such as toll-like receptor 4, tumor necrosis factor-α, and transforming growth factor-β. 9 It can also sensitize the airways and impair the immune system.36,37 Thus, air pollution exposure may increase the risk of chronic lower airway disease mortality.
In this study, cancer including lung cancer and CVD were the next most common causes of death. Long-term exposure to PM and NO2 has been associated with an increased risk of lung cancer mortality and cardiovascular mortality in other populations.30,38,39 We, however, did not find these association in COPD patients. Given the main cause of death in mild or moderate COPD patients is lung cancer and that COPD itself is a strong risk factor of lung cancer development, 40 the additional effect of air pollution on lung cancer mortality might not be significant in COPD patients. Moreover, in the Asian population, COPD patients do not have an increased risk of CVD mortality compared with those without COPD. 18 The association of air pollution with lung cancer and CVD mortality in patients with COPD may be complex and depend on factors such as the duration and severity of COPD and smoking behaviors.
In the stratified analysis, PM10 was associated with overall mortality in women but not in men. This may be due to biological differences,41,42 smoking behaviors, outdoor activity patterns, or occupational exposures. 43 For example, women may have a higher susceptibility to lung damage by exogenous toxicants, such as smoking. 41 Also, as middle-aged and older women tend to spend more time close to their homes than men, residence-based air pollution models may be more accurate for women than men.43,44
This study has several limitations. First, we used claims data to define COPD, as spirometry data were not available. This definition, however, has been validated in previous studies.18,19 In addition, smoking status was assessed using self-reported questionnaire, and, therefore, is subject to potential misclassification and residual confounding. Second, air pollution concentrations were estimated based on the individual’s area of residence. Outdoor air pollution concentrations do not reflect actual exposure 45 or the indoor air quality. In addition, residential information was available only at the district level, further limiting the accuracy of exposure concentration. The measurement error in the exposure, however, is likely to be nondifferential by mortality status; therefore, it would underestimate the association between air pollution and mortality. Considering the direct impact of air pollution on airways, further studies using more individualized measurements, such as wearable air pollution trackers, are needed to better understand the risk of morbidity and mortality in COPD patients. Finally, this study was conducted using data from South Korea; the results might not be generalizable to other ethnic groups, to other countries with different levels of air pollution, or to patients with different risk factor for COPD. For example, in South Korea, female smoking rate is extremely lower compared with those in other countries;46,47 thus, the findings may be different in studies in women with a higher prevalence of smoking.
A major strength is the availability of important covariates, including smoking status, and the presence of exacerbation. We also evaluated the association between air pollution and disease-specific mortalities. Furthermore, we used three (1-, 3-, and 5-year) exposure periods to better understand the relevant exposure windows in the association between air pollution and mortality in COPD patients and the 5-year average exposure is the longest exposure period that has been used in these studies.
Conclusion
In conclusion, in this large population-based cohort of patients with COPD, long-term to PM10 and NO2 exposures were not associated with overall mortality, but they were associated with an increased risk of chronic lower airway disease mortality. Furthermore, air pollution was associated with overall mortality in those with a history of severe exacerbation, suggesting that more attention on air pollution exposure is necessary in severe COPD patients.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231176175 – Supplemental material for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea
Supplemental material, sj-docx-1-taj-10.1177_20406223231176175 for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea by Suna Kang, Yun Soo Hong, Jihwan Park, Danbee Kang, Hyunsoo Kim, Jin Lee, Woojin Kim, Sung-Won Kang, Eliseo Guallar, Juhee Cho and Hye Yun Park in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-2-taj-10.1177_20406223231176175 – Supplemental material for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea
Supplemental material, sj-tif-2-taj-10.1177_20406223231176175 for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea by Suna Kang, Yun Soo Hong, Jihwan Park, Danbee Kang, Hyunsoo Kim, Jin Lee, Woojin Kim, Sung-Won Kang, Eliseo Guallar, Juhee Cho and Hye Yun Park in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-3-taj-10.1177_20406223231176175 – Supplemental material for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea
Supplemental material, sj-tif-3-taj-10.1177_20406223231176175 for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea by Suna Kang, Yun Soo Hong, Jihwan Park, Danbee Kang, Hyunsoo Kim, Jin Lee, Woojin Kim, Sung-Won Kang, Eliseo Guallar, Juhee Cho and Hye Yun Park in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-4-taj-10.1177_20406223231176175 – Supplemental material for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea
Supplemental material, sj-tif-4-taj-10.1177_20406223231176175 for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea by Suna Kang, Yun Soo Hong, Jihwan Park, Danbee Kang, Hyunsoo Kim, Jin Lee, Woojin Kim, Sung-Won Kang, Eliseo Guallar, Juhee Cho and Hye Yun Park in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-5-taj-10.1177_20406223231176175 – Supplemental material for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea
Supplemental material, sj-tif-5-taj-10.1177_20406223231176175 for Air pollution and mortality in patients with chronic obstructive pulmonary disease: a cohort study in South Korea by Suna Kang, Yun Soo Hong, Jihwan Park, Danbee Kang, Hyunsoo Kim, Jin Lee, Woojin Kim, Sung-Won Kang, Eliseo Guallar, Juhee Cho and Hye Yun Park in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
