Abstract
The optimal method for decontamination of implant surfaces for peri-implantitis treatment remains controversial. In recent years, erbium-doped yttrium aluminum garnet (Er:YAG) laser irradiation and implantoplasty (IP) (i.e. mechanical modification of the implant) have been reported to be effective in decontaminating implant surfaces during the surgical treatment. Also, a lack of adequate keratinized mucosa (KM) around the implant is known to be associated with more plaque accumulation, tissue inflammation, attachment loss, and mucosal recession, increasing the risk of peri-implantitis. Therefore, free gingival graft (FGG) has been recommended for gaining adequate KM around the implant. However, the necessity of acquiring KM for the treatment of peri-implantitis using FGG remains unclear. In this report, we applied the apically positioned flap (APF) as resective surgery for peri-implantitis treatment in conjunction with IP and Er:YAG laser irradiation to polish/clean the implant surface. Furthermore, FGG was conducted simultaneously to create additional KM, which increased the tissue stability and contributed to the positive results. The two patients were 64 and 63 years old with a history of periodontitis. The removal of granulation tissue and debridement of contaminated implant surfaces were performed with Er:YAG laser irradiation post flap elevation and then modified smooth surfaces mechanically using IP. Er:YAG laser irradiation was also utilized to remove the titanium particles. In addition, we performed FGG to increase the width of KM as a vestibuloplasty. Peri-implant tissue inflammation and progressive bone resorption were not observed, and both patients maintained good oral hygiene conditions until the 1-year follow-up appointment. Bacterial analysis via high-throughput sequencing revealed proportional decreases in bacteria associated with periodontitis (Porphyromonas, Treponema, and Fusobacterium). To the best of our knowledge, this study is the first to describe peri-implantitis management and bacterial change before and after procedures by resective surgery combined with IP and Er:YAG laser irradiation for peri-implantitis treatment, accompanied by FGG for increasing KM around the implants.
Background
Periodontitis and peri-implantitis are caused by plaque accumulation1,2 and present with similar symptoms. 3 Progressive peri-implantitis may lead to implant loss due to bone resorption around the implant. 4 However, further disease progression of peri-implantitis has been reported even after surgical treatment.5,6 A possible explanation for this is the difficulty in decontamination of the roughened and threaded surfaces of the dental implant. 7 Furthermore, there is no standard method of implant surface decontamination that is superior to other methods of decontamination.7–9 Thus, the simultaneous use of multiple decontamination methods has been recommended for implant surface decontamination. 9
Inadequate width of the keratinized mucosa (KM) is an important factor that affects peri-implant tissue inflammation; insufficient oral hygiene due to limited access for plaque control, and inadequate KM width may lead to more severe peri-implant tissue inflammation. 10 Free gingival graft (FGG) has been reported as an effective strategy to prevent gingival inflammation, mucosal recession, and crestal bone loss.11,12 In addition, FGG increases the KM width and reduces probing depth (PD) and bleeding on probing (BOP). 13 Therefore, establishing stable peri-implant tissues resistant to inflammation is essential even after the treatment of peri-implantitis.
Several studies have reported that the predominant bacterial species for periodontitis such as Porphyromonas gingivalis, Tannerella forsythia, and Treponema denticola were also increased in peri-implantitis site.2,14 Bacterial diagnosis is regarded as a beneficial method for clinicians to estimate the effect of periodontal disease. Periodontitis and peri-implantitis have been described as polymicrobial diseases in which various bacteria are present.15–17 Thus, detecting only limited bacterial taxa may not be sufficient for a bacteriological evaluation of the pathology and the treatment effect. High-throughput sequencing technology is ideal for the comprehensive evaluation of changes in bacterial composition in polymicrobial diseases prior to and post-treatment. 18
Here, we report two cases that had an apically positioned flap (APF) combined with IP and erbium-doped yttrium aluminum garnet (Er:YAG) laser irradiation, before and after implantoplasty (IP), for peri-implantitis treatment; and with FGG to increase the KM and oral vestibule. The clinical and bacteriological effects of the treatments were evaluated longitudinally via high-throughput sequencing.
Case presentation
Case 1
A 64-year-old female patient presented to Tokyo Medical and Dental University (TMDU) hospital with signs of inflammation around the maxillary left second premolar (Figure 1(a)–(d)). The patient had no smoking history and good general health, with no financial issues or cultural beliefs that prevented her from receiving the reported treatments. In another clinic, she had implants (Straumann AG, Basel, Switzerland) placed 16 years ago in the maxillary left second premolar (implant 25, tissue level, sandblasted and acid-etched sandblasted, large grit, acid-etched (SLA) surface, 4.8 mm × 10 mm), first molar (implant 26, tissue level, SLA surface, 4.8 mm × 12 mm), and third molar (implant 28, tissue level, SLA surface 4.1 mm × 8 mm). Clinical and radiographic examinations revealed bone resorption, which mainly comprised suprabony defect with 7-mm PD and BOP at the mesial-buccal and middle-buccal sites of the implant 25 (Figure 1(b)–(d)). In addition, slight bone resorption was noted around the implant 26 with 4-mm PD and BOP at the mesial-buccal site. The KM width and vestibular depth (VD) of both implant areas were 1.5 and 2 mm, respectively (Table 1). Implant 28 was left submerged and unloaded. The patient was diagnosed with peri-implantitis and generalized periodontitis of stage IV with grade B based on the latest classification of periodontal and peri-implant diseases and conditions.19,20 Treatment options with nonsurgical and surgical procedures were discussed with the patient, and written informed consent was obtained.
Baseline presentation in case 1. (a) Intraoral view of the maxillary left second premolar on the buccal side. (b) Periapical radiograph of maxillary left second premolar. (c) Pretreatment intraoral view of the maxillary left second premolar on the buccal side after removing the superstructure. Bleeding and pus are observed around the maxillary left second premolar implant. (d) Cone-beam computerized tomography (CBCT) image of maxillary left second premolar on the buccal area. Bone resorption defect is noted on the buccal side.
Clinical parameters of patients.

BOP, bleeding on probing; KM, keratinized mucosa; PD, probing depth; VD, vestibular depth.
The cement-retained and connected superstructure of implants 25 and 26 was removed by a crown and bridge remover. First, nonsurgical treatment was applied once at implants 25 and 26. Under local anesthesia, the surface of the subgingival area was debrided using Er:YAG laser (Erwin AdvErL EVO, J. Morita, Tokyo, Japan) with a crystal contact tip (600 μm in diameter, C600 F, J. Morita). The irradiation condition of the laser was 60 mJ/pulse (10.6 J/cm2) and 20 Hz frequency with water spray in a semicircular motion in a near contact mode. The expected pulse energy at the chip was approximately 30 mJ/pulse. Irrigation was then performed with 0.2% benzethonium chloride (BTC) solution (Neostelin Green, Nippon Shika Yakuhin KK, Shimonoseki, Japan) (Figure 2(a) and (b)).

Intraoral view of the maxillary left second premolar on the buccal side that received nonsurgical therapy in case 1. (a) Intraoral view of using Er:YAG laser irradiation, on the buccal side. Local anesthesia was administered. An Er:YAG laser device (Erwin AdvErL, J. Morita) with a crystal contact tip (600 μm in diameter, C600 F, J. Morita) was used with the following parameters: 60 mJ/pulse (10.6 J/cm2) and 20 Hz frequency, with water spray. The expected pulse energy at the chip was approximately 30 mJ/pulse. (b) Intraoral view during irrigation with 0.2% BTC on the buccal side.
The PD in relation to implant 25 at the mesial-buccal site remained 6 mm with BOP 1 month post nonsurgical treatment, whereas implant 26 showed a PD of 3 mm and no BOP. In addition, the patient complained of mucosal pain during brushing, which we attributed to the lack of KM and VD around the implants (Figure 3(a), Table 1). After re-evaluation, APF and FGG were performed for surgical therapy, and the combination of IP and Er:YAG laser irradiation to ensure debridement of the implant surface and remove the granulation tissue and the titanium particles were conducted at implant 25. Under local anesthesia, a vertical incision was made distal to the left first premolar, and a combination flap (full-thickness in the coronal area and partial-thickness in the apical area) and full-thickness flap were elevated on the buccal and palatal sides from the distal site of the left first premolar to distal site of the first molar, respectively. The removal of granulation tissue and debridement of the contaminated implant surface in the suprabony and infrabony defects were performed with Er:YAG laser irradiation (Figure 3(b)), with the same conditions as mentioned above. The implant threads in the suprabony defect around implant 25 were removed and polished with diamond (144, SHOFU, Kyoto, Japan) and Arkansas bur (FG57, SHOFU, Kyoto, Japan) (Figure 3(c)–3(e)). The titanium particles were removed using Er:YAG laser using the same conditions as mentioned above. A gingival graft (size 22 mm × 7 mm, thickness; 1 mm) was harvested from the palate, applied to the recipient site, and secured with silk 4-0 sutures (E21-40B2, ELP Akiyama Co., Tokyo, Japan) and a polyglactin absorbable 4-0 simple suture (Vicryl rapide, Johnson & Johnson Medical S.p.A., Rome, Italy) (Figure 3(f)). A periodontal pack (COE-PAK; GC America INC, Alsip, IL, USA) entirely covered the grafted area, and the bilayer artificial dermis (Terudermis, Olympus Terumo Biomaterials Corp, Tokyo, Japan) was sutured with silk 4-0 simple sutures at the donor site such that the silicone membrane was on the outside after removing the excess collagen matrix. Amoxicillin (750 mg, three times a day) and loxoprofen (60 mg, three times a day) were prescribed for the next 5 days. The sutures, periodontal pack, and the silicone membrane of the bilayer artificial dermis were removed 2 weeks after the surgery.

Intraoral view of the maxillary left second premolar that received surgical therapy in case 1. (a) Intraoral view of the maxillary left second premolar on the buccal side at 4 months post nonsurgical therapy on the buccal side. The reduction in inflammation was confirmed, and no KM was observed and gingival recession is confirmed. (b) Intraoral view of using Er:YAG laser irradiation for decontamination and degranulation around dental implants on the buccal side. (c) Intraoral view of IP using diamond bur for decontamination of dental implant surface on the buccal side. IP was performed on the exposed thread of implant surfaces at buccal and supracrestal area. (d) Intraoral view of IP using Arkansas bur for decontamination of dental implant surface on the buccal side. (e) Intraoral view immediately after IP on the buccal side. (f) The FGG sutured by periosteal sutures.
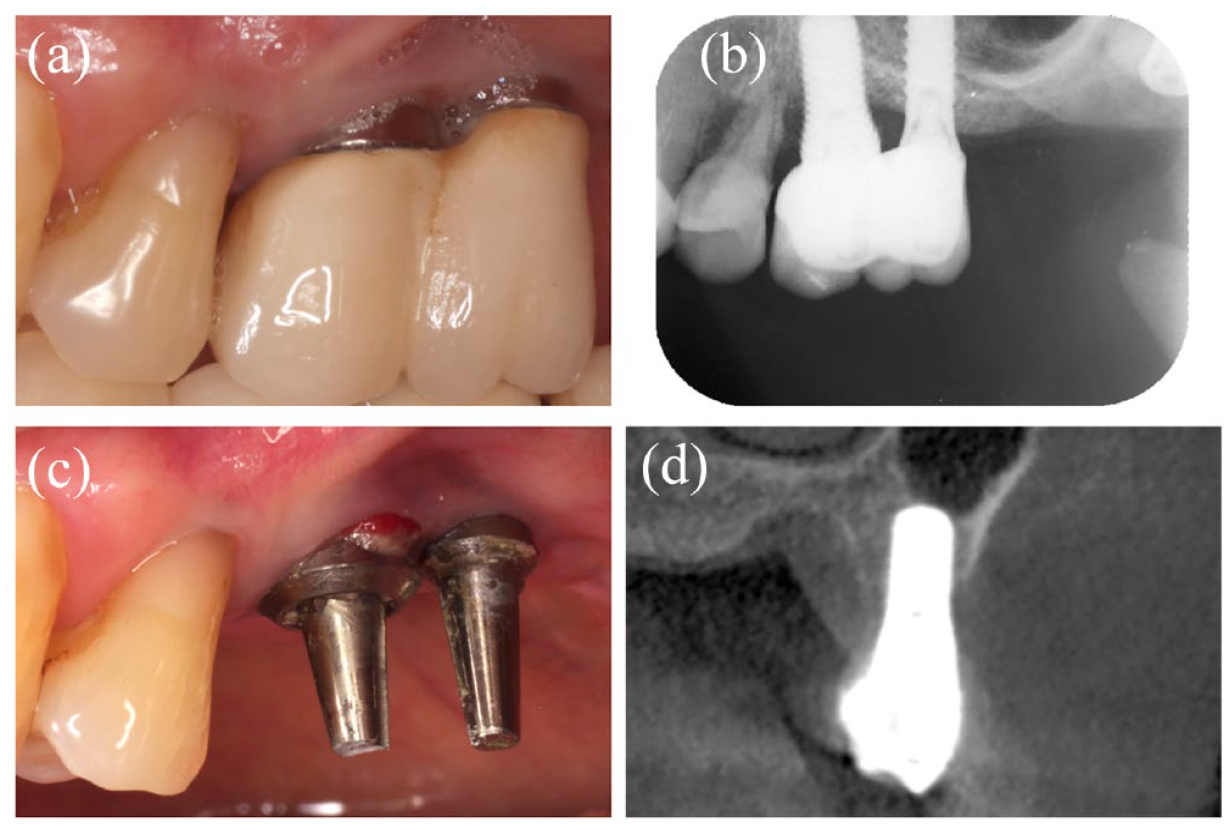
Approximately 1 year postsurgery, the PD decreased to 3 mm at the implants 25 and 26, and BOP was not observed at any site. KM width increased to 3 and 2.5 mm at the implants 25 and 26, respectively (Table 1). The patient was satisfied with the results of the treatment. Patient adherence was high throughout the treatment period. The patient followed up every 3 months for oral prophylaxis (Figure 4(a)–(c)). Subgingival plaque samples were collected from the deepest pocket at six sites in peri-implantitis and healthy implant sites by placing some sterile paper points for the 30 s at baseline, 1 month after nonsurgical treatment, and 1 year after surgery.

Intraoral view and radiograph of the maxillary left second premolar taken 1-year postsurgical therapy in case 1. (a) Intraoral view of the maxillary left second premolar on the buccal side taken 1-year postresective therapy on the buccal side. (b) Periapical radiograph of the maxillary left second premolar. (c) CBCT of the maxillary left second premolar.
Case 2
A 63-year-old female patient with no significant medical history presented to TMDU hospital with discomfort around her maxillary right molars after brushing (Figure 5(a) and (b)). She had implants (Nobel Biocare Services AG, Zürich, Switzerland) placed 10 years ago in the maxillary right second premolar (implant 15, Brånemark System Mk IVTiU RP 4.0 × 11.5 mm) and maxillary right first molar (implant 16, Brånemark System Mk IV TiU RP 4.0 × 10 mm) from the same hospital.

Baseline presentation in case 2. (a) Intraoral view of the maxillary right molar area on the buccal side. (b) Periapical radiograph of maxillary right molar area.
Clinical and radiographic examinations revealed signs of peri-implantitis with 6 mm PD with BOP and bone resorption, mainly comprising suprabony defects at both implant sites, and pus at the middle-buccal site of implant 15 and at the mesial-buccal site of implant 16 (Figure 5(b)). The KM width was 1.5 and 1.0 mm, and the VD was 4.0 and 3.0 mm at the sites of implants 15 and 16, respectively (Table 1). The patient was diagnosed with peri-implantitis and generalized periodontitis of stage IV with grade B according to the latest classification of periodontal and peri-implant diseases and conditions.19,20 Treatment options with nonsurgical and surgical procedures were discussed with the patient, and written informed consent was obtained. She did not have any financial issues or cultural beliefs that prevented her from receiving the reported treatments. Although the patient underwent the same nonsurgical procedures as in case 1, there were no satisfactory improvements with 5 mm of PD, BOP, and pus at the two implant sites, even though slight improvements in PD were observed. Even though PD was 5 mm, these implants were diagnosed with peri-implantitis because the implants showed BOP, increased PDs when compared with previous examinations, and the existence of bone loss beyond crestal bone level changes resulting from initial bone remodeling. 19 Furthermore, the KM width and VD decreased to 0 and 2.5 mm at the site of implant 15 and 0.5 mm and 2.5 mm at the site of implant 16, respectively (Table 1). The patient complained of difficulty in brushing at the areas of implants 15 and 16 due to inadequate width of KM and VD. Tooth 14 was replaced with a temporary crown for periodontal treatment (Figure 6(a)). Thus, the same procedures as in case 1 were performed (Figure 6(b) and (c)).

Intraoral view of the maxillary right molar area that received surgical therapy in case 2. (a) Intraoral view of the maxillary right molar area on the buccal side at 1 month after nonsurgical therapy on the buccal side. The continuing inflammation and gingival recession were confirmed. (b) Intraoral view of post-IP on the buccal side. (c) The FGG sutured by periosteal sutures.
Four months postsurgery, an abnormality of the buccal frenum was noted between teeth 14 and 15, and frenotomy was performed to improve the environment for plaque control using scalpel number 15. A porcelain-fused-to-metal crown was attached to tooth 14. The peri-implant problems were not observed at the 1-year follow-up (Figure 7(a) and (b)). Approximately 1-year after surgery, the PD decreased to 3 mm at the sites of implants 15 and 16, respectively. No BOP was observed at either site. KM width increased to 7 and 8 mm at the sites of implants 15 and 16, respectively (Table 1). The patient was satisfied with the result, and the patient was recalled for follow-up once every 3 months. Patient adherence was high throughout the treatment period. Subgingival plaque samples were obtained in the same way as case 1.

Intraoral view and radiograph of the maxillary left molar area taken 1-year postsurgical therapy in case 2. (a) Intraoral view of the maxillary right molar area on the buccal side taken 1-year postresective therapy on the buccal side. (b) Periapical radiograph of the maxillary right molar area.
Result of bacteriological examination
A total of 16S rRNA gene amplicon sequencing was conducted to evaluate the bacterial changes in the healthy implant and peri-implantitis sites at baseline, 1 month after nonsurgical treatment, and 1 year after surgery. Deoxyribonucleic acid (DNA) was extracted from the plaque using a DNA extraction kit (Mora-extract AMR Inc., Tokyo, Japan), and the V3-V4 region of the 16S rRNA gene (341F and 805R) was amplified using the Q5 Hot Start High-Fidelity DNA Polymerase (New England Biolabs, MA, USA). The libraries were then sequenced on the MiSeq platform (Illumina, CA, USA) to obtain 2 × 300 bp paired-end reads.17,18 The read data were submitted to the DNA Data Bank of Japan (http://www.ddbj.nig.ac.jp/) under accession number DRA012487. The obtained reads were taxonomically assigned to the Human Oral Microbial Database v15.1 at 97% sequence identity by the Illinois Mayo Taxon Organization from the RNA Dataset Operations (IM-TORNADO) pipeline. 21
Nine phyla were assigned, whereas 0.80–4.4% of taxa were unclassifiable at the phylum level (Figure 8). In case 1, Bacteroidetes was the most predominant at the peri-implantitis site (implant 25) at baseline and 1 month after nonsurgical treatment (42.3% and 37.5%, respectively); however, its proportion decreased at the 1-year follow-up (1.7%). At the healthy implant site (implant 36) with 2 mm of PD, no BOP, and 0 mm of KM width, Fusobacteria was the most predominant at baseline, but showed a substantial decrease at follow-up (34.4–0.17%); the decrease was also observed at the peri-implantitis site. The healthy implant site showed an increased proportion of Firmicutes at follow-up compared with that at baseline (22.9–40.2%). In contrast, the proportion of Firmicutes remained relatively constant from baseline to follow-up at the peri-implantitis site (14.4%, 28.4%, and 20.0%). The proportion of Proteobacteria at the peri-implantitis site increased through from baseline to follow-up (2.0%, 19.7%, and 26.7%). That of Actinobacteria increased at both sites through from baseline to follow-up (11.2–42.7% at the healthy implant site; 1.9%, 3.1%, and 46.2% at the peri-implantitis site).

Bacterial composition at the phylum level.
In case 2, a decrease in the proportion of Bacteroidetes was observed in both sites at follow-up (28.8% and 0.025% at the healthy implant site 32; 34.1%, 42.3%, and 13.8% at the peri-implantitis site 15). In addition, the decreases in the proportion of Fusobacteria (22.7% and 0.037% at the healthy implant site; 30.1%, 9.3%, and 5.7% at the peri-implantitis site) were seen in both sites. On the contrary, the increases in Firmicutes (20.2% and 61.9% at the healthy implant site; 25.5%, 24.4%, and 39.7% at the peri-implantitis site) and Proteobacteria (0.062% and 26.3% at the healthy implant site; 1.0%, 1.3%, and 14.9% at the peri-implantitis site) were observed in both sites. The proportion of Actinobacteria decreased at follow-up compared with baseline in the healthy implant site (25.7–9.1%) but increased in the peri-implantitis site from baseline to follow-up (4.2%, 21.5%, and 21.4%).
A total of 69 genera were assigned at the genus level (Figure 9). In case 1, the proportion of Porphyromonas (in the phylum Bacteroidetes), Fusobacterium (in the phylum Fusobacteria), and Treponema (in the phylum Spirochaetes) substantially decreased at the peri-implantitis site through from baseline to follow-up: 37.86%, 20.10%, and 0.02% in Porphyromonas, 15.16%, 4.90%, and 0.74% in Fusobacterium, and 14.24%, 2.54%, and 0.01% in Treponema. Moreover, the proportion of Fusobacterium showed a substantial decrease at the healthy implant site (34.41–0.10%). In contrast, substantial increases were observed as follows: Actinomyces (in the phylum Actinobacteria; 4.51–22.12% at the healthy implant site; 1.24%, 2.57%, and 13.31% at the peri-implantitis site), Rothia (in the phylum Actinobacteria; 0.089–17.38% at the healthy implant site; 0.0075%, 0.35%, and 25.74% at the peri-implantitis site), Enterococcus (in the phylum Firmicutes; 0–23.79% at the healthy implant site; 0% at the peri-implantitis site through from baseline to follow-up), and Streptococcus (in the phylum Firmicutes; 6.79–11.80% at the healthy implant site; 0.070%, 12.53%, and 16.71% at the peri-implantitis site).

Bacterial composition at the genus level.
In case 2, a decrease in the proportion of Fusobacterium and an increase in the proportion of Streptococcus were common at the healthy implant site and peri-implantitis site, respectively; in particular, the healthy implant sites showed an obvious increase in the proportion of Streptococcus (0.38–61.08%). An increase in the proportion of Haemophilus (in the phylum Proteobacteria) was observed only at the healthy site (0.012–24.49%). The proportion of Actinomyces decreased at the healthy implant site (21.83–2.91%) but increased at the site of peri-implantitis from baseline to follow-up (1.81%, 15.25%, and 16.98%).
Discussion
Management of inflammation caused by peri-implantitis is demanding due to the difficulty of implant decontamination owing to its uneven shape and rough surface. For establishing an easy-to-maintain implant surface and a favorable soft tissue condition in cases of peri-implantitis with predominantly exhibit horizontal bone resorption, APF combined with IP and Er:YAG laser irradiation for peri-implantitis treatment and FGG to improve KM around the implants led to good results in this report.
There are limitations in nonsurgical approaches for the treatment of peri-implantitis.19,22 However, nonsurgical treatment has been recommended to prepare healthier peri-implant soft tissue prior to surgical treatment.20,23 The debridement of the implant surface with adjunctive local delivery of antibiotics, glycine powder air polishing, or Er:YAG laser irradiation may aid in more effective reduction in the clinical signs of peri-implant mucosal inflammation, compared with the use of curettes with adjunctive irrigation with chlorhexidine. 24 In addition, 0.2% BTC solution is useful as an antimicrobial agent in the oral cavity. 25 Therefore, Er:YAG laser irradiation and irrigation with 0.2% BTC solution were performed as a nonsurgical treatment.
Although the clinical conditions of these two cases did not fully recover by nonsurgical treatments, the proposed surgical procedure provided good clinical outcomes in both cases. Recently, the regenerative therapy for the treatment of peri-implantitis has been reported to be effective. 26 However, bone defect morphology is an important factor in the application of regenerative therapy for peri-implantitis. 27 Since the bone defects in these cases had a predominantly horizontal morphology, resective therapy rather than regenerative therapy was selected for the treatment of these cases.
Various debridement methods (ultrasonic scalers, stainless steel curettes, titanium curettes, plastic curettes, Er:YAG laser, diode laser, air-powder, and IP) have been proposed over the past two decades. IP is effective in decontamination as it establishes a more favorable surface that decreases adherence of bacteria, prevents subsequent formation of the plaque biofilm, 28 and enhances fibroblast migration. 29 A systematic review stated that IP does not cause any severe mechanical or biological complications. 30 On the contrary, another review discussed the potential effect of titanium particles released during surgical treatment of peri-implantitis on the pathogenesis of peri-implant bone loss. 31 To avoid this anticipated trouble, we applied the Er:YAG laser, with the same conditions and motion for the debridement of the implant surface, to minimize the infection caused by titanium particles before IP and remove the residual titanium particles attached to the internal surface of the gingival flap, periosteum, and bone surface after IP. Recently, Er:YAG laser has been utilized for removing metal tattoos in the gingiva by tissue ablation. 32 The tissue ablation by Er:YAG laser is based on the absorption of light energy mainly by water. 33 Therefore, we also applied the Er:YAG laser to remove the residual titanium particles and found it an effective method that is easier and less time consuming than other methods, such as removal with a scalpel or scissors. Furthermore, laser irradiation ushers the inflamed tissues rapidly into the healing and regenerative phases by thoroughly decontaminating of diseased tissues. 34 Although IP alone shortens the operation time, the combination of IP and laser may not only eliminate the source of infection but also accelerate healing.
APF is one of the resective therapy for peri-implantitis treatment, 35 and APF combined with IP is an effective and reliable strategy for the treatment of peri-implantitis.36,37 In our study, the mean PD reduction was 2.0 mm, 12 months after treatment. In contrast, Romeo et al. 36 and Englezos et al. 37 reported that mean PD reductions were 2.2 and 5.4 mm, respectively, 24 months after APF combined with IP. The mean PD reduction in this study was similar to that reported by Romeo et al. 36 but lesser than that by Englezos et al. 37 The reason for this may be that the PD at the baseline in the report of Englezos et al. 37 was larger than that in our study. However, APF combined with IP alone cannot increase KM width. The presence of at least 2 mm of KM is essential as it has a protective effect on the peri-implant health,36,38 and implants with <2 mm of KM width were more prone to develop peri-implant complications. 39 We compensated for the limitations of APF by harvesting an FGG, which we applied in these cases. It has been reported that the combination of IP and APF with FGG can improve KM and PD for treatment of peri-implantitis. 35 The rate of FGG shrinkage post-FGG therapy was approximately 30%.40,41 In these cases, the rate of FGG shrinkage in case 1 appeared to be greater than in case 2. The VD in case 1 was considered narrower than in case 2 prior to treatment, leading to this difference in the rate of FGG shrinkage.
The treatment resulted in an overall decrease in the phyla Bacteroidetes and Fusobacteria and maintained or increased the phyla Firmicutes and Proteobacteria according to the results of the comprehensive bacteriological analysis. In addition, particular subordinate genera seemed to be responsible for the phylum-level changes. Although the pattern of changes was different among different genera, a decrease in the genera Porphyromonas, Treponema, and Fusobacterium and an increase in the genus Streptococcus seemed like a change from pathological bacterial flora to a healthy state. This was because Porphyromonas, Treponema, and Fusobacterium are considered strongly associated with not only periodontitis but also peri-implantitis, 42 whereas Streptococcus is an early colonizer on the surface of healthy teeth. 43 In healthy implant sites, decreases of the genus Fusobacterium were observed in both cases without specific procedures. Bacteria can easily be transmitted from site to site in the same oral cavity via gingival crevicular fluid and saliva. 44 Both patients in our study had periodontitis, and periodontal pathogens were detected in peri-implantitis site at the baseline. Studies have shown that residual periodontal pockets are risk indicators for peri-implantitis. 45 The cause for peri-implantitis in our patients may be the presence of periodontitis in the same oral cavity. Therefore, comprehensive periodontal treatment should be performed before implant treatment. High-throughput sequencing can deliver mass data on the bacterial composition, allowing for a better understanding of the subtle changes of each bacterial taxon with high resolution. One of the limitations of this study was the small sample size, which resulted mainly from difficulty in recruiting patients who fulfilled the enrollment criteria for this report. Further studies with large sample sizes, including clinical trials, are desired to establish a new therapeutic protocol for peri-implantitis and soft tissue improvement as an integrated application of APF with FGG, IP, and Er:YAG laser irradiation.
Conclusion
Our case report suggest that peri-implantitis may be successfully treated by performing APF as resective surgery for peri-implantitis treatment, combined with IP and Er:YAG laser irradiation for cleaning the implant surface and peri-implant tissue, accompanied by FGG for increasing KM. We have further demonstrated that a patient with a history of peri-implantitis may show a good prognosis even after a year when the appropriate treatment strategy is selected and oral hygiene, including supportive periodontal therapy, is maintained.
Footnotes
Acknowledgements
Supercomputing resource was provided by the Human Genome Center at the Institute of Medical Science (University of Tokyo, Tokyo, Japan; http://sc.hgc.jp/shirokane.html). We thank Hugo Song of HUGO LS (![]() ) for editing the draft of this manuscript.
) for editing the draft of this manuscript.
