Abstract
Background:
Patient-reported outcomes (PROs) are increasingly mandated in kidney care to incorporate patients’ perspectives.
Objectives:
We assessed whether educational support for clinicians using electronic (e)PROs could enhance person-centered care.
Design:
A process evaluation, using a mixed methods longitudinal comparative concurrent design was undertaken of educational support to clinicians on routine use of ePROs. In two urban home dialysis clinics in Alberta, Canada, patients completed ePROs. At the implementation site, clinicians were provided with ePROs and clinician-oriented education via voluntary workshops. At the non-implementation site, neither were provided. Person-centered care was measured using the Patient Assessment of Chronic Illness Care-20 (PACIC-20).
Methods:
Longitudinal structural equation models (SEMs) compared change in overall PACIC scores. The interpretive description approach, using thematic analysis of qualitative data, further evaluated processes of implementation.
Results:
Data were collected from questionnaires completed by 543 patients, 4 workshops, 15 focus groups, and 37 interviews. There was no overall difference in person-centered care throughout the study, including after delivery of workshops. The longitudinal SEMs revealed substantial individual-level variability in overall PACIC trajectories. However, there was no improvement at the implementation site and no difference between the sites during both the pre- and post-workshop periods. Similar results were obtained for each PACIC domain. Qualitative analysis provided insights into why there was no substantial difference between sites: (1) clinicians wanted to see kidney symptoms, not quality of life, (2) workshops were tailored to clinicians’ educational needs, not patients’ needs, and (3) variable use of ePRO data by clinicians.
Conclusion:
Training clinicians on use of ePROs is complex and likely only part of what is required to enhance person-centered care.
Registration:
NCT03149328. https://clinicaltrials.gov/ct2/show/NCT03149328
Keywords
Introduction
Internationally, patient-reported outcomes (PROs) are being collected, at times mandated, in kidney care.1,2 PRO measures are instruments that patients complete to self-report health outcomes relevant to their quality of life (QOL). 3 They are systematically collected with the hope that patients’ responses will inform and guide their care,1,4–8 and in turn support person-centered kidney care.4,9–13 The global kidney community acknowledges the urgent need for evidence-informed strategies to improve both the QOL and healthcare experiences of people living with dialysis. 14 Optimal use of PROs at point of care may help address this need.
Use of PRO data to enhance person-centered dialysis care remains largely unexplored, 1 perhaps because routine collection, use, and integration is a complex intervention. Person-centered dialysis care upholds attributes of centeredness, namely that a patient is a person who is unique, and has the right to be heard and to have shared responsibility of their care. 15 Use of PROs is one way of drawing attention to the patients’ perspectives of their health outcomes. Decades of research has previously focused on efficiency or effectiveness of PRO data usage to support monitoring symptoms or communication in various populations, 16 including kidney care settings.1,17,18 However, international evidence-based sources and syntheses have identified that clinicians need training and education to support PRO use in their clinical practice at point of care2,19–23 (i.e. at the individual level between a person and clinician/s) to enhance person-centered care.
While multidisciplinary kidney practitioners have expressed some support for use of PRO responses at point of care,6,17,24 they have also identified barriers. For example, they expressed skepticism about the benefits to routine patient care, 24 confused/conflated clinician assessment with PRO self-report,24,25 perceived responses to address concerns are beyond their scope of practice,7,26 lacked effective interventions to address concerns,7,26 worried that PRO collection might increase their workload6,8,24 and psychological referrals, 24 and feared litigation of negligence if PRO responses were not addressed. 24 These uncertainties have resulted in poor uptake, further identifying the need for a clinician-oriented intervention.
Understanding how to provide educational support to clinicians to optimize use and integration of kidney patients’ PRO responses is crucial. The purpose of the study was to assess, from clinicians’ and patients’ points of view, how person-centered care could be enhanced through clinician-oriented educational support for multidisciplinary kidney clinicians in routinely utilizing electronic PROs (ePROs). The research question was: To what extent do clinicians and patients report improved person-centered kidney care associated with educational support to clinicians on use of ePROs?
Methods
Design, complex intervention, sites
This mixed methods study was a process evaluation of educational support to clinicians on routine use of ePROs, which is a complex intervention. 27 We used a longitudinal comparative concurrent design, 28 with clinician and patient focus groups and interviews. The rationale for this approach was to provide contextual understandings and practice implications of the impact of the complex intervention: clinician-oriented educational support for routine use of ePROs. The study was registered. 29
There were two phases over 2 years at two sites. See Figure 1 – Project timeline. The sites included two home dialysis clinics (peritoneal dialysis and home hemodialysis) providing care to all of Alberta, Canada: Northern (implementation site) and Southern (non-implementation site). The two sites were comparable in geographic service to Albertans, dialysis staffing, facilities, and number of home hemodialysis patients (<100); however, the implementation site had more peritoneal dialysis patients overall (>300) than the non-implementation site (>200). At the implementation site, ePRO responses were provided to clinicians; they had collected PROs on paper for 8 years prior to the study. In the non-implementation site, ePRO responses were not provided to clinicians; patients had not previously been invited to complete PROs. In both the sites, patients had online access to their ePRO data, to track responses overtime, share with any of their healthcare team, or use in self-care.

Project timeline.
The Knowledge-To-Action Framework 30 guided both phases of this patient-oriented research. 31 See Figure 2 – Adapted Knowledge-to-Action Framework. Phase I included clinician and patient consultation at the implementation site to develop the clinician-oriented educational support. Following the knowledge-to-action steps, in consultation to determine knowledge gaps of routine use of ePROs, the education support that clinicians endorsed were tailored, co-designed workshops. Workshops included knowledge adapted to the local context, and content that addressed barriers and supports to use of ePROs in kidney care. In phase II, the implementation site was supported via clinician workshops.

Adapted knowledge-to-action framework.
Our research team encompassed multi-disciplinary clinicians, decision-makers, researchers, patient organizations, and patients. The Patient Advisory Committee was co-chaired by LL, a Patient Partner, and Advisory members were trained in patient-oriented research and involved throughout the project.
Participant recruitment
Through purposive sampling, 32 all clinicians (nurses, nephrologists, dietitians, social workers) from the implementation site were invited via information sessions, posters, and emails from the department between July 2017 and July 2019. Patients attending their regularly scheduled clinic appointments at both sites were initially screened and invited to participate by a unit clerk or a nurse, and then followed up by study staff present in waiting rooms. Patients were eligible if they were age ⩾18, could read/write in English, and provide written consent; and ineligible if they had visual impairment, severe cognitive impairment, or were in health crisis. At the implementation site, patients were invited to participate in focus groups or interviews. Although caregivers were not recruited, they were admitted if required to support the patient. Recruitment of both clinicians and patients was ongoing throughout study through purposive and snowball sampling. 32 Ethical approval was granted by University of Alberta/Alberta Health Services (#Pro00068365) and the University of Calgary (#REB17-0506). Informed consent was obtained at the first point of data collection, re-confirmed at subsequent interactions.
Data collection and analysis
Quantitative measures
Throughout phases I and II, patients at both sites completed all questionnaires by accessing a digital health platform (Cambian). 33 Research staff phoned/emailed/mailed patients 7–14 days prior to appointments (scheduled approximately every 3 months), inviting them to complete questionnaires online, or via phone or mail if needed. The Patient Assessment of Care for Chronic Conditions-20 questionnaire (PACIC-20) 34 was used to assess person-centered care. Other ePRO questionnaires were as follows: (1) Edmonton Symptom Assessment System-renal (ESAS-r:Renal), 35 (2) Kidney Disease Quality of Life-36 (KDQOL-36), 36 (3) EQ-5D-5L 37 (see Table 1). Internal consistency reliability (based on the ordinal Cronbach’s alpha) 38 in this sample was 0.97 for the overall PACIC score, and ranged from 0.82 (delivery system design) to 0.91 (problem-solving) for the domain scores.
ePRO data collection.
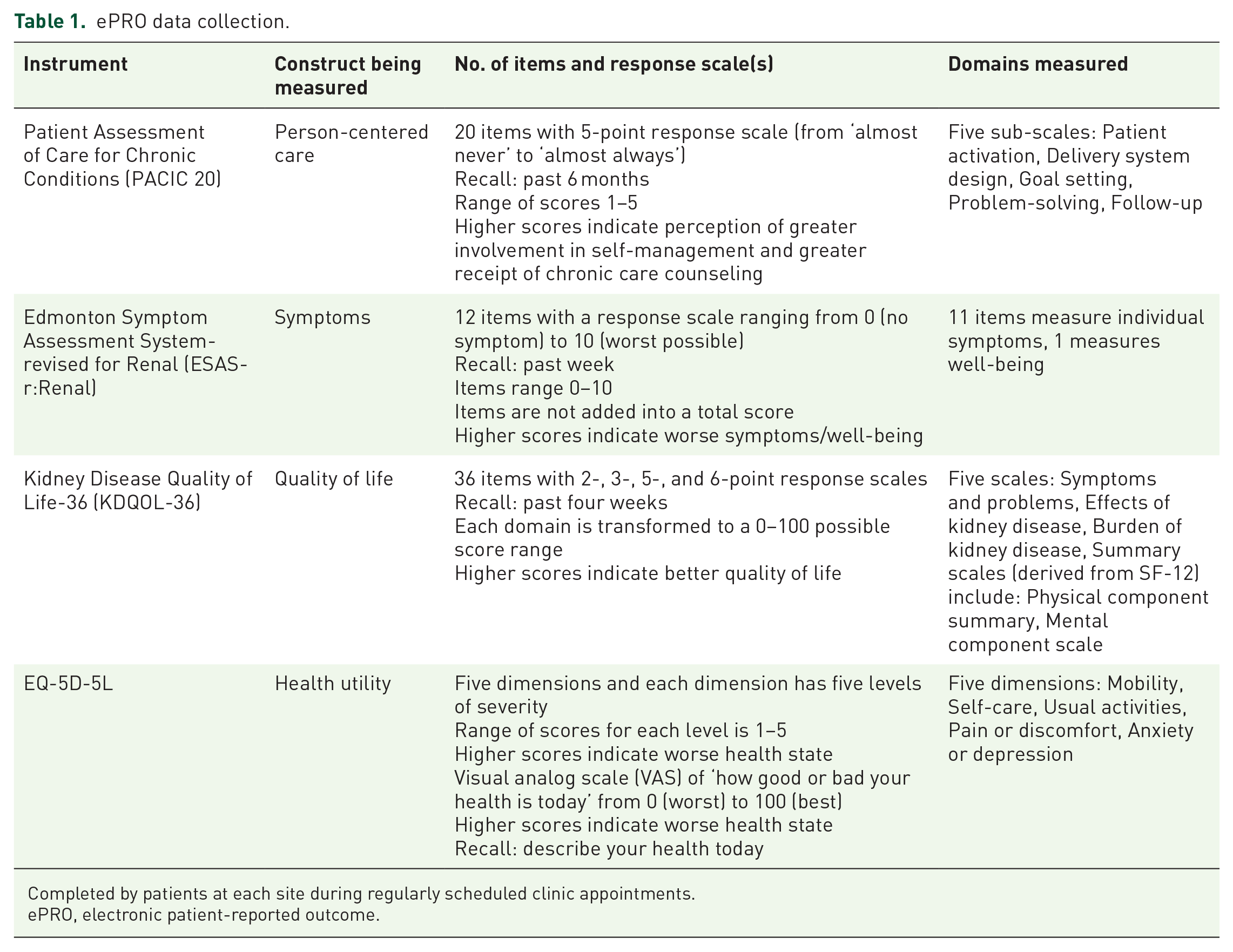
Completed by patients at each site during regularly scheduled clinic appointments.
ePRO, electronic patient-reported outcome.
In phase II, clinicians in the implementation site were asked to anonymously answer two rating-scale questions asking how often they reviewed and used ePRO data to inform their care. Clinicians were asked to respond to eight questions about their knowledge and beliefs regarding the use of ePROs prior to the first workshop and after all workshops were completed.
Quantitative analysis
We used descriptive statistics to compare the samples at the two sites at the start of the study (time 0) using the Chi-square test for categorical variables and t-test for continuous variables.
To address the research question, we first graphically compared the average linear changes over time for the overall PACIC score and domain scores between the two sites over the 24-month period. We subsequently fit longitudinal structural equation models (SEMs), using Mplus software (version 8.6), 39 to statistically compare average change over time between the two sites. Separate SEMs were fit before and after the workshop start date. The SEMs were specified with two latent factors representing the intercept (outcome score at start of study) and the slope (linear change in outcome scores) over eight repeated measures that occurred at the time of each visit, which were regressed on the grouping variable (site) and the following covariates to control for statistically significant differences between sites: education, ethnic group, and diabetes. Individually varying times of observations relative to the start of study were specified to account for variations in study entry and the timing of each visit. Multilevel multiple imputation with covariates 40 was applied to created 20 data files with imputed item response data for participants who had missing data for one or more of the PACIC items (5.0% imputed data). Full information maximum likelihood was applied to accommodate unequal numbers of measurement occasions for each person.
Qualitative data
Qualitative data were collected across both phases in the implementation site. Data were collected via 12 focus groups (6 clinician, 6 patient) and 7 interviews (1 clinician, 6 patients) during phase I (July 2017–May 2018), and via 30 interviews (16 clinicians, 11 patients, 3 research assistants) and 3 patient focus groups during phase II (February 2019–August 2019). Interviews or focus groups were conducted by the study lead (KS-M), project coordinator (JC), and two trainees (nursing students) in private rooms at the clinic, at the university, or by phone, lasting 30–90 min. There were no prior relationships between interviewers and participants. Regular debriefing between interviewers and study leads enhanced data quality and reflexivity. Semi-structured interview or focus group guides (Supplementary File 1) were iteratively revised with the research team and Patient Advisory Committee, informed by ongoing data analysis. Interviews or focus groups were audio-recorded and transcribed verbatim.
Qualitative analysis
Guided by the interpretive description approach, 41 data were analyzed in phase I to guide ePRO provision and inform workshop creation, and in phase II to assess how person-centered care was enhanced (or not) through routine ePRO use. Transcripts from initial interviews or focus groups were listened to and read to create a codebook, iteratively refined during analysis. The data were coded by JC and a trainee under the supervision of KS-M; questions about categorization were resolved through consensus. Managed with N-VivoTM, data were thematically analyzed for meaning saturation42,43 to understand diversity of perspectives. Patient and clinician data were analyzed separately, then compared and contrasted. We ensured trustworthiness and rigor by addressing credibility (iterative discussions, triangulation, and negative/alternative case analysis), confirmability (audit trail, field notes), and transferability (context). 41 The Consolidated Criteria for Reporting Qualitative Research guidelines 44 in Supplementary File 2 provides additional details.
Results
Participant demographics
Of the 693 approached patients, 543 (78%) participated and routinely provided ePROs (Table 2). Patients had a mean age of 56, identified primarily as Caucasian (65.4%), were predominantly male (65.8%), received peritoneal dialysis (71.3%); 56 withdrawals; 41 loss-to-follow-up. In total, 2252 ePROs were completed over 2 years. At the implementation site, 198 patients chose to complete online; 86 by phone or mail. At the non-implementation site, 111 chose online; 148 by phone or mail. Interviews and focus groups were attended by 42 patients, 3 caregivers, and 34 clinicians. Forty-one clinicians attended between one and four workshops, and 38 completed questionnaires before and after workshops (Table 3).
Characteristics of patients by site at baseline.

ePRO, electronic patient-reported outcome; eQ-5D-5L, health equity; MCS, mental component summary; PACIC, Patient Assessment of Chronic Illness Care; PCS, physical component summary; SD, standard deviation.
Each variable treated as a separate variable.
Demographics of participants in qualitative focus groups and interviews, and clinician workshops.

N/A, not applicable.
Clinicians that attended one or more workshops and responded to eight questions about their knowledge and beliefs regarding the use of patient-reported outcomes (PROs) prior to the first workshop and after all workshops were completed.
Phase II is denoted in round brackets. Note that there were eight participants in phase II who were not a part of phase I (eight patients), and 30 participants who were in phase I but not a part of phase II (16 patients and 14 healthcare providers).
For clinicians, type of dialysis in their practice.
Caregiver to a person with this modality.
Person-centered care
Figure 3 depicts substantial individual-level variability in overall PACIC trajectories, with greater improvement over time in the non-implementation site relative to the implementation site during the pre-workshop period, whereas the trajectories appear more similar during the workshop period. The results of the SEMs (see Table 4) confirmed that the overall PACIC score was lower at the start of the study for the non-implementation site (intercept of non-implementation = 3.10, p = 0.00; difference in intercepts = 0.32 for the implementation site, p = 0.02) with greater improvement over time during the pre-workshop period (difference in slopes = −0.41, p = 0.02, for the implementation site). However, there was no detectable change over time in overall PACIC scores during the post-workshop periods in both sites (slope of non-implementation = −0.08, p = 0.27; difference in slopes = −0.03, p = 0.80). The SEM results for PACIC domains suggest that the intercepts and slopes were very similar between the two sites pre- and post-workshops for most of the domains; the only detected differences were for the pre-workshop intercepts and slopes for goal setting (difference in intercepts = 0.40, p = 0.03; difference in slopes = −0.46, p = 0.05) and for follow-up (difference in intercepts = 0.52, p = 0.01; difference in slopes = −0.64, p = 0.01).

Trajectories of PACIC by site pre- and post-workshops.
Structural equation modeling results.

BIC, Bayesian Information Criterion; LL, log-likelihood; PACIC, Patient Assessment of Chronic Illness Care.
Summary scores were calculated after multiple item-level imputation to accommodate missing data.
Pre-workshop sample size is 435, whereas post-workshop sample size is 455.
Regression coefficient for site (referent = non-implementation) controlling for covariates. All covariates were centered on the grand mean.
Following process evaluation, we analyzed clinicians’ and patients’ perspectives at the implementation site for insights into why there was no substantial difference in person-centered care between sites. Insights include: (1) clinicians wanted to see kidney symptoms, not QOL; (2) workshops were tailored to clinicians’ educational needs, not patients’ needs; and (3) variable use of ePRO data. Illustrative quotes are provided in Table 5, along with exemplars of negative/alternative cases (when applicable) to represent a diversity of perspectives.
Illustrative quotes by insight.

ePRO, electronic patient-reported outcome; GP, General Practitioner; PD, peritoneal dialysis; RN, Registered Nurse.
Clinicians wanted to see kidney symptoms, not QOL
For 8 years prior to the study, the implementation site had collected the ESAS-r:Renal on laminated paper during appointments, and the KDQOL-36 annually. Both were entered into an electronic health record; clinicians did not see KDQOL-36 responses, but sporadically saw the ESAS-r:Renal. During initial focus groups, clinicians discussed what additional health or QOL information they might like patients to provide. Clinicians chose to see the questionnaire they were most familiar with, the ESAS-r:Renal, not the KDQOL-36 or EQ-5D-5L. The ESAS-r:Renal items address 11 symptoms, plus one on wellbeing. Clinicians requested usual clinic questionnaires [ESAS-r:Renal, visual analog scale (0–100) from EQ-5D-5L, and four questions about activities of daily living (bathing, dressing, walking, transferring bed to chair)] be completed simultaneously online to aid workflow. Kidney symptoms were the priority, perceived as something clinicians could ‘control’ [quote(q)1–3], whereas QOL was ‘vague’ (q4). Patients were asked what other information would be important. They emphasized their desire for a relational connection, to be asked ‘How are you doing?’ They wanted clinicians to acknowledge the ePRO responses they were already providing (q5).
Prior to each appointment, patients completed the ESAS-r:Renal. Although clinicians had online access via Cambian, they asked for ePROs to be printed. They did not have computers in all the clinic rooms, shared computers in the hemodialysis clinic, and used a hybrid paper/electronic chart. Thus, the ESAS-r:Renal was printed and handed to the nurses. Nurses entered the ESAS-r:Renal responses into electronic charts after appointments. Clinicians later asked to see ESAS-r:Renal trends over time, printed in color (q6). All patients had online access to their data, and many commented on the benefits of seeing their responses over time (q7).
Workshops were tailored to clinicians’ educational needs, not patients’ needs
Workshops were tailored to multi-disciplinary clinicians’ educational needs. Workshop objectives were guided by Bloom’s taxonomy to address knowledge, skills, and attitudes, 45 further informed by extant literature on clinician training to use PROs, and through consultation with team experts. (See Table 5, column 1, for workshop objectives.) Workshop locations, format, length, timing (for shifts), frequency, learner-activities, and door-prize-incentives were co-created with clinicians.
Workshop 1 addressed ePRO use and integration into practice. Patients and clinicians alike did not understand that after completion, clinicians should acknowledge, review, and discuss responses with patients (q8–10). Neither group understood why patients were completing ePROs; clinicians did not know how to invite patients to complete ePROs nor that patients should answer them by themselves (q11–14). An invitation script, informed by the data (q15), was provided and clinicians said a personalized version to each other. Attendees reviewed strategies to use patients’ responses to inform patient-clinician interactions (q16–17).
Patients co-presented Workshop 2 focusing on patients’ valuing of, and relationship to, use of ePROs in their care. Patients read quotes about actual and envisioned use in shared decision-making (q18–22). Attendees discussed case scenarios when ePROs identified significant symptom concerns (q23–25), especially when clinicians felt ‘not-equipped to actually attend to it’ (q24). ePROs identified needs outside of traditional dialysis care, such as pain, depression, anxiety; clinicians debated whether they may attend to the entirety of a patient’s needs (q26–30).
Strategies were discussed in Workshop 3 for use of ePROs to support within-team communication or coordination of care. While nurses typically looked at ePROs first, attendees discussed how ePROs could support communication within home dialysis (q31–33). Clinicians brainstormed follow-up when numerous challenges were identified for one patient through longitudinal ePRO responses (q34), and what fell outside scope of dialysis care (q26, 27, and 35). Clinicians considered and challenged ePRO use to support communication beyond the home dialysis team (q35–38).
Routine integration of ePROs as a fundamental change to practice was discussed in Workshop 4. Given ePROs were often not seen as important or useful (q39–41), attendees discussed whether this information could provide a ‘missing part in our practice’ (q41), and optimal ways to integrate ePROs into workflow (q39, 42–44). ‘Contradictions’ between ePROs and clinicians’ assessments were debated alongside strategies to use ePROs as complementary to clinicians’ assessments (q45–47).
Variable use of ePRO data
Despite tailored educational support, use of ePROs was variable. In phase II, clinicians were invited every 2 weeks to anonymously answer two rating-scale questions. They did so a total of 244 times; 47% of the time they looked at ePRO results most or all the time, and 42% of the time they used ePRO information most or all the time (Figure 4). In summary, ePRO data were viewed or used less than half of the time.

Clinicians’ reports of viewing and using ePRO data.
Clinicians’ knowledge and beliefs about use of ePROs, compared before and after workshops, showed disparate uptake: 35% of clinicians reported an increase in looking at ePROs, 57% improvement in skills explaining ePRO completion to a patient, 38% greater competence in follow-up, and 51% greater understanding that patients’ self-reports differed from proxy reports. However, 33% found less importance for ePROs being used by kidney programs or practitioners, 25% less enhancement of person-centered care with routine ePRO use (64% no change), and a 35% decrease in clinician responsibility to respond to ePROs (Figure 5).

Change in clinicians’ knowledge and beliefs about use of ePROs.
After the workshops, clinicians’ narratives about how they used ePROs was clinician dependent, spanning the spectrum of person-centered kidney care. On one end, clinicians did not change how they used ePROs (q48), did not consider them useful (q49), and no longer invited patients to complete ePROs if they were not participating in the study (q50). The majority of clinicians expressed increased awareness (q51), and looked at responses prior to a patient leaving the clinic to ‘address it’ (q52). On the other end of the spectrum, some felt their practice had changed and that ePROs facilitated their engagement with a person ‘as a whole’ (q53 and 54).
Patients did not know who saw their ePRO responses (q55), which were frequently not acknowledged (q56). Patients were left wondering if they could or should bring them up, or if that was the clinicians’ responsibility (q57 and 58). Patients saw value in completing ePROs, appreciated seeing their trends over time, and envisioned ePROs supporting ‘a mindful part of the patient and clinician experience’ (q59) or self-care (q61 and 62).
Discussion
We found that person-centered care was not enhanced through educational support for multidisciplinary kidney clinicians to routinely use ePROs. Overall PACIC score and domain scores were similar at implementation and non-implementation sites throughout the study, including after clinician workshops. While the extant PRO literature emphasizes training clinicians,2,19–23 our findings show that this alone seems insufficient to show enhancement in person-centered care. The qualitative findings confirm the PACIC analysis, and offer explanatory insights for this departure; each of the three insights are discussed in relation to literature in the field.
First, clinicians wanted self-reports on kidney symptoms, not QOL. Similar to many chronic illnesses, treatment of symptoms and use of technology has reified ‘dialysis-centered care’. 46 Health systems reinforce this by care being organized by body parts; clinicians specialize accordingly. Understandably, clinicians wanted to see what they were familiar with. They may also have wanted to see which kidney symptoms lowered QOL, so they could be addressed (if within perceived scope of practice). Reports about non-dialysis concerns (i.e. pain, depression, anxiety) left clinicians feeling ill-equipped, a finding previously reported. 1 In a similar study on use of ePROs in home dialysis in Canada, nurses also chose to look at symptoms (ESAS-r:Renal) over QOL (KDQOL-36) even when both were provided.5,6 While clinicians were symptom-focused, patients wanted their responses to be acknowledged and personalized. They wanted to be asked ‘How are you?’ and really listened to. Improving patient–clinician communication was also identified as a top challenge in in-center hemodialysis in Canada. 47 But dialysis patients’ perspectives on use of PROs to facilitate communication or in their care has rarely been explored.1,4,8 Clinicians’ focus on symptoms, regardless of wider training of routine integration of ePROs into care, may have truncated movement toward person-centered care.
Second, workshops were tailored to clinicians’, not patients’, educational needs. While patients’ perspectives and the extant literature informed this design, clinician’s knowledge, skills, and attitudes were targeted. While clinicians are a part of person-centered care, they are one part, not the whole. A focus on person-centered kidney care necessitates a culture shift from a clinician or system orientation, toward shared emphasis with patients’ values and priorities. 48 But this is a fundamental change to kidney practice, re-orienting delivery from symptom-centered or dialysis-centered to person-centered.9,49–51 Future patient-oriented research may focus on empowering the person receiving dialysis to routinely use ePROs, an approach not previously explored.
Third, clinicians’ use of ePRO data was variable, only looking at and using it less than half of the time. To address ongoing questions about use of ePROs in routine kidney care, we created two animated videos, one for clinicians and one for patients, which were made available after the study was completed (https://www.healthyqol.com/kidney). In the global movement toward person-centered kidney care,1,48–51 use of PROs by clinicians may ultimately be less important than addressing systemic challenges in dialysis care that hinder person-centered practices, such as traditional provision of healthcare where clinicians’ knowledge or biomedical information is privileged over patient knowledge, and healthcare organized in silos according to body parts (i.e. kidney, cardiac, mental health). 26
The strengths of our study include longitudinal ePRO collection in a large sample alongside a mixed methods approach for data triangulation with insights illuminated through strategic approaches to rigor, further informed by the Knowledge-to-Action Framework, 30 including credibility (i.e. iterative, co-development of an educational support with clinicians, use of multiple data sources, attention to negative case analysis); confirmability (i.e. reflexive field notes, audit trails during data collection and analysis, interviews with research assistants who collected data at the two sites); and transferability (tailoring workshops to local context while leveraging extant literature). Nevertheless, our study is not without weaknesses. Although clinicians received training on integration of ePROs into care, they elected to only receive an ePRO focused on symptoms. In addition, clinicians at the implementation site had previously collected this information for 8 years prior to the study, and it is unknown whether this impacted findings. An ePRO focused on symptoms may not have been the optimal information for clinicians to see, and it may have limited a person-centered approach to kidney care, which is intended to encompass the whole of a person.9–13 The PACIC-20 showed that care was already very good in both sites, thus improvement may have been untenable. Furthermore, the lack of integration of the ePRO digital health platform with the electronic medical record system used by the clinicians at the implementation site may have impacted their receptiveness to routine use of ePROs. Finally, this study was conducted prior to the COVID pandemic. With the wide-scale adoption of telehealth options during the pandemic, future studies may investigate whether in/acceptability of telehealth innovations in kidney care impacts routine use of ePROs.
Conclusion
The results of this study reveal that training clinicians on the use of PROs is complex and likely only part of what is required to enhance person-centered care. While this is counter-culture to the extant literature that encourages clinician training for routine collection, use, and integration of ePROs, person-centered kidney care may necessitate a wider strategy that addresses the complexity of such a culture shift. Our findings provide an important contribution to the literature because we identified three insights to explain why this might have occurred. First, clinicians wanted self-reports on symptoms, not QOL. Second, workshops were tailored to clinicians’ educational needs, not patients. Third, use of ePRO data by clinicians was variable. Our findings emphasize the need for research that emphasizes patients’ priorities in using their self-report to inform their care. While ePRO use has the potential to improve patient outcomes, it seems to require intervention beyond clinician education.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231173624 – Supplemental material for Electronic patient-reported outcomes in clinical kidney practice (ePRO Kidney): a process evaluation of educational support for clinicians
Supplemental material, sj-docx-1-taj-10.1177_20406223231173624 for Electronic patient-reported outcomes in clinical kidney practice (ePRO Kidney): a process evaluation of educational support for clinicians by Kara Schick-Makaroff, Scott Klarenbach, Jae-Yung Kwon, S. Robin Cohen, Joanna Czupryn, Loretta Lee, Robert Pauly, Jennifer M. MacRae, Bruce Forde and Richard Sawatzky in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We wish to thank the Patient Advisory Committee (people with lived experiences of kidney failure) for their insights and guidance on all aspects from study conception and design, to wording of recruitment posters, to data analysis, and reporting of results.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
