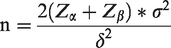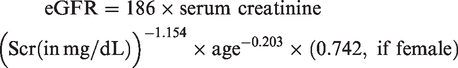Abstract
Background and objective
Icodextrin is a novel, potentially advantageous peritoneal dialysis solution characterized by low glucose content and high molecular weight. This study aimed to compare the impact of icodextrin and glucose solutions on peritoneal dialysis patients.
Methods
This multicenter, prospective, randomized controlled trial included 40 peritoneal dialysis patients. Participants were randomized in a 1:1 ratio into an experimental group (icodextrin group) and a control group (glucose group). Both groups used 1.5% or 2.5% glucose dialysis solutions for three daytime exchanges. At night, the icodextrin group used 7.5% icodextrin, while the glucose group used 2.5% glucose solution. Patients were followed up bimonthly, assessing nighttime ultrafiltration, electrolytes, lipid and glucose profiles, albumin levels, and quality of life using the short form-36 health survey scale at 4 months.
Results
After 2 and 4 months, the icodextrin group demonstrated a considerably greater nighttime net ultrafiltration than the glucose group (P < 0.05). After 4 months, the icodextrin group exhibited lower serum sodium, chloride, and systolic blood pressure levels but higher calcium levels, albumin levels, and short form-36 health survey scores than the glucose group (P < 0.05).
Conclusions
Icodextrin enhances ultrafiltration, maintains electrolyte balance, and improves nutritional status in peritoneal dialysis patients. These effects contribute toward improvement in the patients’ quality of life. The protocol was registered at chictr.org.cn (Registration Number: ChiCTR2200062379).
Introduction
Peritoneal dialysis (PD) is a treatment option for end-stage kidney disease that uses the peritoneum as a semipermeable membrane to facilitate fluid as well as solute exchange. 1 Conventional glucose-based dialysis solutions, although commonly used, are readily absorbed into the bloodstream, reducing the osmotic gradient and potentially impairing long-term ultrafiltration (UF). Additionally, prolonged exposure to high glucose levels may damage the peritoneal membrane, leading to reduced UF capacity. 2 Icodextrin, composed of glucose polymers with high molecular weights, provides sustained UF and reduces glucose exposure, thereby minimizing metabolic disturbances and preserving peritoneal membrane integrity.3,4 We conducted a multicenter randomized trial to evaluate the differential impacts of icodextrin and glucose solutions on UF, metabolic parameters, electrolyte balance, nutritional and inflammatory status, and quality of life (QoL) in PD patients.
Methods
Participants
We selected patients who had received at least 90 days of PD treatment at six hospitals in China between 1 June 2022 and 1 January 2024. Eligibility criteria included the following: (a) age 18–65 years; (b) undergoing three daily exchanges of 2.5% glucose solution (2.0 L) and a single long nighttime dwell (8–10 h); and (c) classified as high, high-average, or low-average transporters using the peritoneal equilibrium test (PET). Exclusion criteria included the following: (a) allergies to starch/icodextrin or glycogen storage disorders; (b) serious conditions (e.g. liver cirrhosis and heart failure); (c) pregnancy or lactation; and (d) participation in other interventional studies within 3 months.
Sample size calculation
The primary outcome of this study was an increase in nighttime net UF compared with the baseline value. According to a literature review, the average increase in nighttime net UF compared with the baseline value was 342.53 ± 25.70 mL in the icodextrin group, while that in the glucose group was 73.59 ± 24.09 mL. With a two-sided α of 0.05 and a power of 90%, using the following sample size calculation formula, the calculation result was n = 1. Considering a 1:1 randomization ratio, each group should have at least one participant. To ensure the feasibility and scientific rigor of the trial, each group was ultimately set to include 20 participants.
Study design
In total, 40 patients were included in this study. The patients were randomly assigned in a 1:1 ratio to either the icodextrin group or the glucose group using an online system. The specific dialysis regimen was as follows: both groups of patients underwent three exchanges of 2.0 L of 1.5% or 2.5% glucose PD solution (Baxter Healthcare Ltd., China) during the day. At night, the icodextrin group received 2.0 L of 7.5% icodextrin (Baxter Healthcare Ltd., China), while the glucose group received 2.0 L of 2.5% glucose dialysis solution (Baxter Healthcare Ltd., China) for 10–12 h nighttime dwell. Each patient underwent a total fluid exchange of 8.0 L per day.
Outcome measures
Follow-ups were conducted on a bimonthly basis to assess nighttime net UF, biochemical markers, inflammatory indicators, blood pressure (BP), and body weight. The estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease formula, as follows:
QoL was evaluated using the short form-36 health survey (SF-36) scale at baseline and 4 months of treatment. Adverse events, including hypotension, peritonitis, electrolyte disturbances, rash, and gastrointestinal bleeding, were recorded.
Statistical analyses
Data analyses were conducted using the Statistical Package for Social Sciences software, version 25.0. Quantitative data with a normal distribution were analyzed using the t-test and presented as mean ± standard deviation. For non-normally distributed data, values were presented as median (interquartile range) and analyzed using nonparametric tests. For the comparison of indicators before and after treatment in each group, we used a paired test. Categorical data were evaluated using Pearson’s χ2 test and Fisher’s exact test. A P-value <0.05 was considered to indicate statistical significance.
Ethical statement
The study was conducted in accordance with the 1975 Declaration of Helsinki, as revised in 2024. The Ethics Committee of The First Affiliated Hospital of Kunming Medical University approved the study (approval no. 2022-L52. approval date: 1 June 2022), and informed consent was obtained from all participants. The protocol was registered at chictr.org.cn (Registration Number: ChiCTR2200062379). The reporting of this study conforms to the Consolidated Standards of Reporting Trials (CONSORT) statements. 5
Results
Patient characteristics
In total, 40 continuous ambulatory PD patients, including 29 men and 11 women with a mean age of 46.88 ± 10.57 years and mean dialysis duration of 24.07 ± 10.31 months, were enrolled. The most common type of primary kidney disease was chronic glomerulonephritis. Based on PET results, patients were classified as high transporters (n = 15), high-average transporters (n = 21), or low-average transporters (n = 4). There were no notable differences between the two groups regarding baseline demographics, clinical characteristics, and SF-36 scores (P > 0.05). A summary of the baseline data is provided in Table 1.
Baseline demographics and clinical features of patients (

CAPD: continuous ambulatory peritoneal dialysis; SBP: systolic blood pressure; DBP: diastolic blood pressure; PET: peritoneal equilibrium test; UF: ultrafiltration; TC: total cholesterol; TG: triglycerides; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; eGFR: estimated glomerular filtration rate; ALB: albumin; CRP: C-reactive protein.
Comparison of post-treatment indicators between the two groups
Net UF
After 2 months of therapy, the nocturnal net UF in the icodextrin group was 653.68 ± 178.62 mL, while that in the glucose group was 147.50 ± 54.40 mL. There was a marked difference in the mean UF between the two groups (t = 12.12, P < 0.001, 95% confidence interval (CI): 419.69–592.65). After 4 months of treatment, the mean nocturnal net UF in the icodextrin group increased to 681.85 ± 168.23 mL, while that in the glucose group was 133.50 ± 56.50 mL, representing a statistically significant difference (t = 13.18, P < 0.001, 95% CI: 461.66–635.04). Changes in the nocturnal net UF before and after treatment in both groups are shown in Figure 1. In the icodextrin group, the nocturnal net UF was obviously higher in all types of transport than that in the glucose group. The most notable increase in nocturnal net UF was observed in the high-transport subgroup, as shown in Table 2 and Figure 2(a) and (b).

Significant difference in the nighttime net UF between both groups after treatment. UF: ultrafiltration; ICO: icodextrin group; GLU: glucose group.
Changes in UF categorized by PET groups (

M: months; UF: ultrafiltration; PET: peritoneal equilibrium test; ΔUF: Nighttime net ultrafiltration after 2 months of treatment or after 4 months of treatment minus baseline nighttime net ultrafiltration.

(a) Change in the net UF based on different PET categories after 2 months of treatment in the two groups and (b) change in the net UF based on different PET categories after 4 months of treatment in the two groups. UF: ultrafiltration; PET: peritoneal equilibrium test; ICO: icodextrin group; GLU: glucose group; H: high; HA + LA: high-average + low-average.
Electrolyte and glucose–lipid metabolism
After 2 months of treatment, the calcium level was higher, while blood sodium and phosphorus levels were lower in the icodextrin group (P < 0.05). After 4 months of treatment, the blood calcium level remained higher, whereas the blood sodium and chloride levels were lower in the icodextrin group compared with those in the glucose group; these differences were statistically significant (P < 0.05). After 2 and 4 months of treatment, the fasting blood glucose, blood potassium, and blood lipid levels were similar between the two groups (P > 0.05), as shown in Table 3.
Comparison of electrolyte and glucose–lipid metabolism indicators after treatment (

TC: total cholesterol; TG: triglycerides; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; M: months.
All indicators are measured in mmol/L.
Renal function, inflammation, and nutritional indicators
As shown in Table 4, the Scr level and urine volume were similar between the two groups after 2 and 4 months of treatment (P > 0.05). After 2 months of treatment, the albumin levels were initially similar; however, after 4 months of treatment, this level was higher in the icodextrin group (37.43 ± 5.65 g/L) than in the glucose group (33.52 ± 3.79 g/L) (t = 2.53, P = 0.016, 95% CI: 0.78–7.05). No differences were observed in the C-reactive protein (CRP) levels between the two groups after treatment (P > 0.05).
Renal function, inflammation, and nutritional indicators after treatment (

eGFR: estimated glomerular filtration rate; ALB: albumin; CRP: C-reactive protein; M: months.
Weight and BP
After 2 months of treatment, BP and weight were similar between the two groups. After 4 months of treatment, the mean systolic BP (SBP) in the icodextrin group was lower (137.78 ± 6.63 mmHg) than that in the glucose group (145.55 ± 10.35 mmHg) (t = 2.72, P = 0.010, 95% CI: 1.98–13.57). After 4 months of treatment, in the icodextrin group, both weight and BP had decreased compared with those at baseline, while in the glucose group, weight had significantly increased compared with that at baseline (P < 0.05). Table 5 provides detailed information about the BP and weight changes after treatment.
Comparison of BP and weight after treatment (

BP: blood pressure; SBP: systolic blood pressure; DBP: diastolic blood pressure; M: months.
P1: comparison between the two groups after treatment.
P2: comparison between each group’s treatment after 4 months and baseline.
*and # represent the comparison of BP and weight between the two groups after 2 months and 4 months of treatment, respectively.
SF-36 scale
After 4 months of treatment, there was a significant difference in the physiological function scores between the icodextrin and glucose groups (80.50 ± 7.27 vs. 67.50 ± 8.35) (t = 5.25, P < 0.001, 95% CI: 7.99–18.01). We also noted a difference in the mental health scores between the two groups (75.80 ± 8.58 in the icodextrin group and 68.60 ± 8.24 in the glucose group) (t = 2.71, P = 0.010, 95% CI: 1.82–12.58). As shown in Table 6, the icodextrin group performed significantly better in all parameters (P < 0.05).
SF-36 scale scores after treatment (

SF-36: short form-36 health survey; M: months.
Adverse events
During the treatment period, one patient in the icodextrin group developed refractory peritonitis. The condition improved following antibiotic therapy but necessitated a shift to hemodialysis. No other adverse events were observed. It remains unclear whether the occurrence of adverse events such as cardiovascular events and peritonitis differs between the icodextrin and glucose groups.
Discussion
Icodextrin PD solution is crucial for improving UF, regulating fluid and electrolyte balance, boosting nutritional status, and enhancing the overall QoL. Long-term use of glucose-based dialysis solutions can lead to changes in peritoneal function. 6 Over time, some patients may experience a decline in peritoneal function, reduction in UF capacity, and an increased incidence of complications such as heart failure. Research has indicated that approximately 44% of dialysis patients suffer from heart failure, with 50% of them exhibiting reduced ejection fraction. 7 The prognosis for patients with heart failure combined with chronic kidney disease (CKD) is poor, 8 making volume management particularly crucial for PD patients. Icodextrin provides stable UF and reduces total body water as well as extracellular water, resulting in better volume control. In addition, it lowers the left ventricular mass index, 9 thereby reducing the risk of hypertension and heart failure in PD patients and improving technique survival. We found that after 4 months of treatment, the nighttime net UF was higher, while SBP was lower in the icodextrin group, suggesting that icodextrin improves volume status by enhancing UF and removing excess body fluids, resulting in better BP control.
Sodium plays an essential role in fluid balance, and sodium overload often leads to fluid overload. Recent studies have indicated that sodium accumulation in the interstitial tissue can cause local inflammation, leading to peritoneal fibrosis, altered peritoneal permeability, and a vicious cycle of fluid and sodium overload.10,11 Icodextrin aids in the elimination of small solutes, including sodium and chloride, alleviating the damage caused by sodium overload. Hypokalemia is a common electrolyte imbalance in PD patients and is linked to higher incidences of peritonitis as well as higher mortality. Yi et al. 12 suggested that icodextrin enhances the nutritional status of PD patients, raising serum potassium levels and reducing the incidence of hypokalemia. In CKD patients with calcium–phosphorus metabolism disorders, the incidence of cardiovascular events is often higher. 13 Although limited research has examined the effect of icodextrin on calcium–phosphorus metabolism, our study found that after 4 months of treatment, individuals in the icodextrin group had lower serum sodium and chloride levels but higher calcium levels. At the 2-month interval, serum phosphorus levels in the icodextrin group were lower than those in the glucose group; however, the levels became similar at 4 months of treatment. This suggests that icodextrin improves water and sodium clearance and may alleviate inflammation and regulate calcium–phosphorus metabolism in CKD patients, potentially by increasing dietary intake. However, the long-term effects on phosphorus metabolism require further investigation. The serum potassium levels were similar between the two groups at 2 and 4 months, highlighting the need for larger sample sizes and extended follow-up durations to further explore the role of icodextrin in regulating potassium levels.
Previous research has been inconclusive regarding the benefits of icodextrin on metabolic and renal functions. Studies have shown that glucose-based PD solutions increase the risk of hyperglycemia. 14 Paniagua et al. 15 reported that diabetic patients using icodextrin required less insulin and had improved fasting blood glucose levels. However, a meta-analysis indicated no difference between icodextrin and glucose-based PD solutions in their ability to improve triglyceride, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, and very low-density lipoprotein cholesterol levels. 16 In the present study, no statistically significant differences were observed between the two groups with respect to glucose metabolism, lipid profiles, or residual renal function. These findings suggest that within the study duration and sample size, icodextrin may exert limited impact on these parameters, and further studies with larger cohorts and longer follow-up are required to clarify its potential effects.
Malnutrition and inflammation are predictors of mortality in PD patients. Recent studies indicate that excess fluid can elevate the levels of inflammatory cytokines such as tumor necrosis factor (TNF) and interleukin-6 (IL-6), suppressing appetite. Inflammation also activates the ubiquitin–proteasome pathway, leading to muscle loss and malnutrition in PD patients.17,18 Albumin, a liver-synthesized protein, is a marker of nutritional status, and hypoalbuminemia often leads to poor outcomes for PD patients. 19 The study revealed that after 4 months of therapy, the icodextrin group had higher albumin levels, although no such difference in CRP levels was observed. This suggests that icodextrin may significantly improve serum albumin levels by enhancing UF and electrolyte balance, thereby reducing gastrointestinal adverse effects, improving appetite, and enhancing nutritional status. The absence of a CRP-lowering effect warrants further exploration of other inflammatory markers (e.g. ferritin, TNF, and IL-6) to confirm the role of icodextrin in alleviating inflammation.
An innovative feature of our study is the use of the SF-36 scale. To the best of our knowledge, no previous study has used this scale to assess the QoL of PD patients using icodextrin. Health-related QoL (HRQoL) is a complex concept that includes physical, psychological, and social functioning.20–22 Chronic illnesses often diminish an individual’s HRQoL, and dialysis patients typically experience reduced physical function, depression, and muscle weakness, with a QoL far below that of healthy individuals.23,24 The SF-36 scale evaluates eight dimensions and is often used to assess treatment interventions or track changes in health status. Our study revealed that after 4 months of treatment, the icodextrin group showed higher scores in all eight dimensions of the SF-36 scale, indicating that icodextrin improves the emotional well-being, social functioning, and overall health status of patients. This may be attributed to icodextrin’s ability to reduce sodium and fluid retention, increase small-solute clearance, minimize gastrointestinal reactions, improve dietary intake, enhance albumin levels, and control BP.
Our study has certain limitations. First, the relatively small sample size may have introduced bias and limited the subgroup analyses of diabetic and nondiabetic patients regarding glucose and lipid metabolism. Second, echocardiography was not performed to assess cardiac function. Third, studies with a larger sample size and longer treatment duration are needed to assess the effect of icodextrin on residual kidney function.
Conclusion
Compared with glucose-based PD solutions, icodextrin offers benefits in enhancing UF, maintaining electrolyte balance, improving nutritional status, and enhancing QoL, supporting the use of icodextrin as an effective and widely adopted option in the management of PD patients.
Footnotes
Acknowledgement
We thank the staff of the Department of Nephrology of the First Affiliated Hospital of Kunming Medical University, the main research hospital, for their support. We also thank the staff at the other five centers who participated in the study for their efforts.
Author contributions
ZZ conceptualized the study; LY and YCX developed the protocol; YCX collected data; LY prepared and submitted the manuscript; and ZZ performed an overview of the study. All authors were involved in the revision of the manuscript and have approved of the final version of the manuscript.
Data availability statement
Data requests should be addressed to the corresponding author, Zhou Zhu, followed by the completion of a data sharing application form (contact the corresponding author to obtain the application form), according to the requirements of the Ethics Committee of the First Affiliated Hospital of Kunming Medical University and the relevant hospital leaders. After review and approval, the complete anonymous dataset will be provided within 12 months. Before data sharing, the research purpose, research plan, and data analysis method will be reviewed as part of the data request, and a contract must be signed before sharing the data with a third party.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Funding
This work was supported by Yunnan Major Scientific and Technological Project, (grant no.202505AJ310033) and Yunnan Province Clinical Collaborative Project on Major and Complex Diseases Integrating Chinese and Western Medicine (grant no. 300072).
Public and patient involvement
No patients or members of the public were directly involved in the design, implementation, or reporting of this trial. However, patient perspectives were indirectly incorporated through the use of the SF-36 scale to assess their health-related QoL, ensuring that the evaluation extended beyond laboratory and clinical parameters to include patient-reported outcomes.