Abstract
Background:
Bimekizumab is a monoclonal IgG1 antibody that selectively inhibits both interleukin (IL)-17A and IL-17F, and is a promising drug for patients with moderate-to-severe plaque psoriasis.
Objectives:
This study aimed to assess the efficacy and safety of bimekizumab in treating patients with psoriasis and to determine the optimal maintenance dosing schedules of bimekizumab.
Methods and design:
Eligible trials were identified from PubMed, Cochrane Controlled Register of Trials, Embase, ClinicalTrials.gov, and Chinese medical databases. Only double-blind, randomized, active comparator, or placebo-controlled trials of bimekizumab treatment on patients with psoriasis were included in this study.
Results:
Five studies were identified, which included 2473 patients with moderate-to-severe plaque psoriasis. The results indicated that bimekizumab had better efficacy than placebo or active comparator for Psoriasis Area and Severity Index (PASI) 90 [risk ratio (RR) = 29.29, 1.52; 95% confidence interval (CI) = 10.30–83.30, 1.06–2.19], PASI 100 (RR = 59.87, 2.06; 95% CI = 15.06–237.99, 1.12–3.79), and Investigator’s Global Assessment scores of 0 or 1 (IGA 0/1) (RR = 21.55, 1.36; 95% CI = 9.25–50.19, 1.02–1.81). Faster onset of clinically meaningful responses was observed with bimekizumab compared with both active comparators (RR = 2.59; 95% CI = 1.32–5.10) and placebo (RR = 40.46; 95% CI = 13.19–124.13), with PASI 75 response observed at week 4 after one dose. Subgroup analysis showed no significant difference in the reduction of PASI scores between 320 mg q4w dosage and q8w dosage (RR = 1.00; 95% CI = 0.96–1.03). Rates of patients with adverse events (AEs) were comparable in the bimekizumab and active comparator groups (RR = 1.13; 95% CI = 1.01–1.26), and oral candidiasis was one of the most common treatment-emergent AEs.
Conclusion:
The results of this meta-analysis suggest that bimekizumab is more efficacious and has a rapid onset of action than active comparators and placebo in the treatment of moderate-to-severe plaque psoriasis. After 16 weeks of initial maintenance treatment, both bimekizumab maintenance dosing schedules (320 mg every 4 and 8 weeks) had similar efficacy.
Introduction
Psoriasis is a common, chronic, immune-mediated inflammatory skin disease that affects approximately 2–3% of the global population and leads to a substantial burden for individuals and society.1–3 Psoriasis might be associated with several severe comorbidities, such as depression, psoriatic arthritis, and cardiometabolic syndrome.4,5
Accumulating studies have identified interleukin (IL)-17A, along with its family member IL-17F, as important drivers of psoriasis pathogenesis. 6 Both IL-17A and IL-17F are overexpressed in lesional and nonlesional skin of patients with psoriasis6,7 and have been shown to work with tumor necrosis factor (TNF) to stimulate the production of key proinflammatory cytokines in preclinical models. 8 Compared with other biologics (e.g. those targeting TNF, IL-23, and IL-12/23), IL-17A inhibitors have a rapid onset of action.9,10
Bimekizumab is a humanized monoclonal IgG1 antibody that selectively inhibits both IL-17A and IL-17F. 11 Available evidence suggests that IL-17F is independently involved in the pathobiology of plaque psoriasis. IL-17F enables keratinocytes to induce the expression and release of many psoriasis-related proteins 12 and produces a feed-forward inflammatory response by activating STAT1, and NF-kB. 13 The dual inhibition of IL-17A and IL-17F resulted in a greater reduction in the production of proinflammatory cytokines, expression of inflammation-linked genes, and disease-relevant immune cell migration, compared with IL-17A blockade alone. 14 Currently, RG7624 (Genentech, San Francisco, CA, USA) and NI-1401 (NovImmune, Switzerland), targeting IL-17A and IL-17F simultaneously in psoriasis, are both in clinical phase I trials. 15
Several early phase and phase III randomized clinical studies have shown convincing efficacy in patients with moderate-to-severe plaque psoriasis. The evidence, however, is inadequate for drawing reliable conclusions, as their control groups and maintenance dosing schedules varied. Therefore, this meta-analysis aimed to assess the efficacy and safety of bimekizumab in treating patients with psoriasis and determine the optimal maintenance dosing schedules of bimekizumab.
Methods
Search strategy and inclusion criteria
On 1 June 2022, we conducted a systematic search of PubMed, Cochrane Central Register of Controlled Trials, Embase, ClinicalTrials.gov, and Chinese medical databases (Wanfang and Chinese National Knowledge Infrastructure), using the search terms ‘psoriasis’ and ‘bimekizumab’ or ‘UCB4940’. The details are listed in Table S1 (Supplemental Table 1). All the studies identified using this strategy were reviewed independently by two authors, and eligible reports that met all inclusion criteria were included in our meta-analysis. Trials were selected based on the following inclusion criteria: (1) patients given a diagnosis of psoriasis and treated with bimekizumab, (2) double-blinded, active comparator, or placebo-controlled trial, and (3) outcome measures included Psoriasis Area and Severity Index 75 (PASI 75), PASI 90, PASI 100, Investigator’s Global Assessment scores of 0 or 1 (IGA 0/1), and rates of patients with adverse events (AEs).
Assessment of the risk of bias
We analyzed the risk of bias independently using the Cochrane collaboration tool, which included adequate sequence generation, allocation concealment, blinding of participants and personnel, incomplete outcome data, selective reporting, and other bias. 16 Disagreement was resolved by consensus. The 2020 PRISMA checklist is attached in Supplemental Figure 1.
Data extraction and outcome measures
Four independent reviewers evaluated the literature and extracted information including authors, year of publication, country, study phase, clinicaltrials.gov number, sample size, treatment parameters (frequency of each injection and dose of each injection), study duration, and outcome measures. Our analysis focused on comparing the treatment effects of bimekizumab and placebo or active comparator across studies. Efficacy outcomes included PASI 75, PASI 90, PASI 100, and IGA 0/1, and rates of patients with AEs.
Statistical analysis
From the studies included in this analysis, we extracted for bimekizumab, placebo, and active comparator, PASI 75, PASI 90, PASI 100, IGA, and the risk ratio (RR) for the occurrence of at least one AE. Dichotomous data were analyzed using RR corresponding to the 95% confidence interval (CI). Heterogeneity between studies was assessed by the I2 test. I2 > 50% indicated the evidence of heterogeneity 17 , and random-effects models were applied. 18 Otherwise, the fixed-effects model was adopted. 19 Sensitivity analysis was performed by subgroup analysis of the placebo and active comparator, as the difference in the response in outcomes in subgroups was the cause of heterogeneity. p-value < 0.05 was considered significant. All meta-analyses were performed using RevMan version 5.3.
Results
Search results
We initially identified 233 records from all electronic databases. After removing 98 duplicates, 126 records were excluded based on titles and abstracts. The eligibility of nine full-text papers was assessed, and four were excluded (duplicated trials, n = 2; insufficient data, n = 2). Finally, five randomized control trials (RCTs) met the selection criteria and were included.20–24 The flowchart for screening studies is shown in Figure 1.

PRISMA flowchart.
Study characteristics
The five trials included a total of 2473 enrolled patients, which consisted of one phase II 21 and four phase III trials.20,22–24 The psoriasis clinical subtype in all trials was plaque psoriasis. A single intervention group bimekizumab 320 mg q4w was presented in one study, 22 whereas three trials20,23,24 presented two interventions (bimekizumab 320 mg q4w and 320 mg q8w). The duration from treatment to outcome assessment ranged from 12 to 56 weeks. Detailed characteristics of the included studies are shown in Table 1.
Characteristics of randomized controlled trials included.
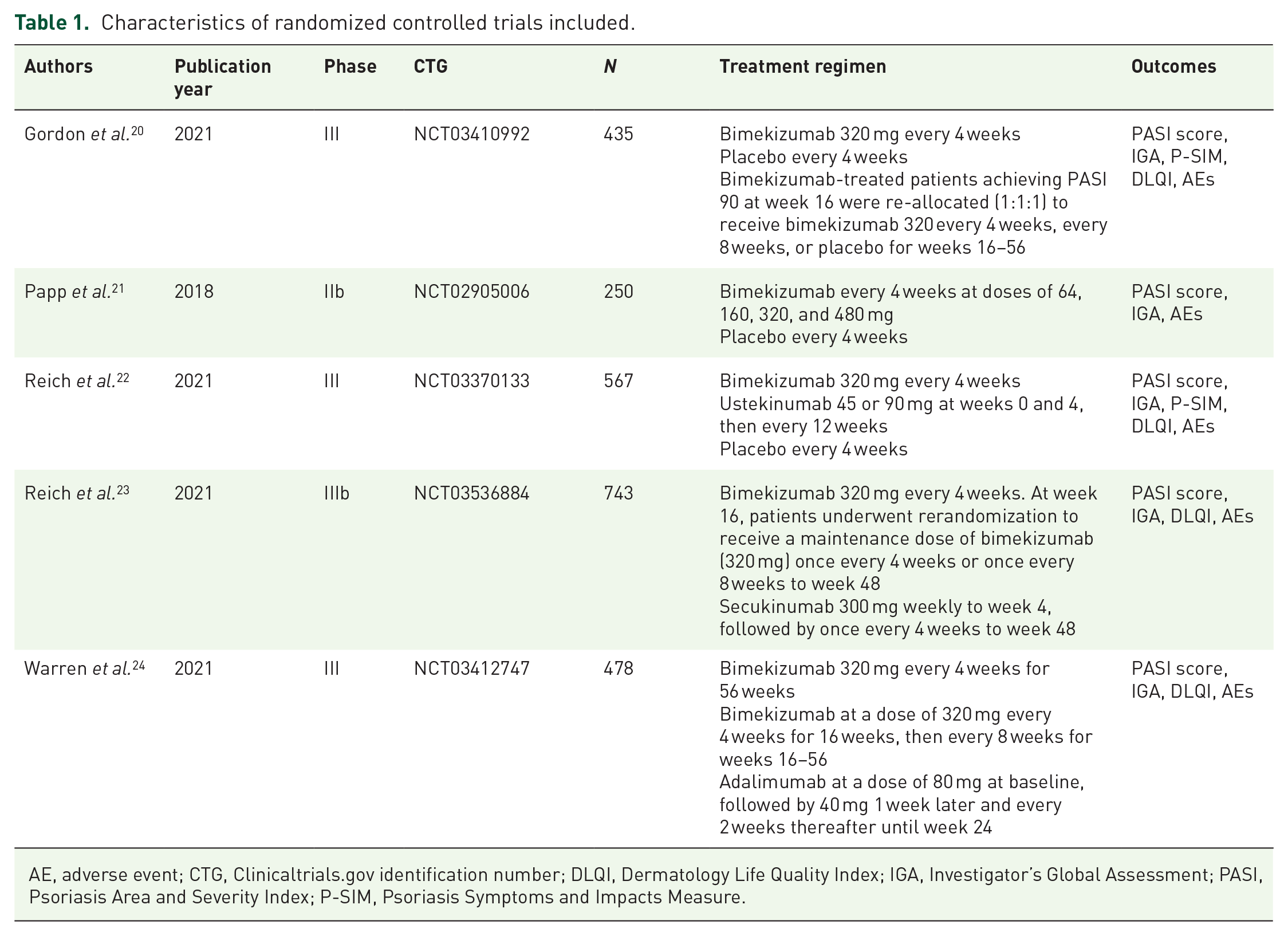
AE, adverse event; CTG, Clinicaltrials.gov identification number; DLQI, Dermatology Life Quality Index; IGA, Investigator’s Global Assessment; PASI, Psoriasis Area and Severity Index; P-SIM, Psoriasis Symptoms and Impacts Measure.
Risk of bias assessment
In this meta-analysis, all trials were assessed as having a low risk of bias (Figure 2). They all were randomized trials, and almost all followed a double-blind protocol. Besides, all trials used allocation concealment.

Risk of bias summary for each included study.
Efficacy outcomes
In all the included studies, including 1570 randomized bimekizumab-treated patients, the effects of bimekizumab treatment and two maintenance dosing schedules of bimekizumab on various clinical indexes were analyzed. Overall, significant improvement in the efficacy of bimekizumab for the treatment of plaque psoriasis was observed in various clinical indexes. The subgroup analysis showed no significant difference in the reduction of PASI score between 320 mg q4w dosage and q8w dosage.
PASI 75 at week 4
The pooled analysis showed that bimekizumab treatment significantly increased the proportion of participants achieving PASI 75 at week 4 compared with controls (RR = 5.58; 95% CI = 2.29–13.58; I2 = 97%; p-value < 0.00001). The subgroup analysis showed a higher proportion of patients achieving PASI 75 at week 4 in the bimekizumab group than in the placebo group (RR = 40.46; 95% CI = 13.19–124.13; I2 = 0%; p-value = 0.55) as well as in the active comparator group (RR = 2.59; 95% CI = 1.32–5.10; I2 = 96%; p-value < 0.00001) (Figure 3).

Meta-analysis of efficacy of bimekizumab for patients achieving at least 75% reduction in the PASI score at week 4.
PASI 90
A meta-analysis was performed on five studies that reported PASI 90.20–24 The pooled effect of bimekizumab on PASI 90 was significantly better than that of the placebo (RR = 29.29; 95% CI = 10.30–83.30; I2 = 20%; p-value = 0.29). Similarly, the response rate in PASI 90 was greater for bimekizumab than for the active comparator (RR = 1.52; 95% CI = 1.06–2.19; I2 = 96%; p-value < 0.00001) (Figure 4).

Meta-analysis of efficacy of bimekizumab for patients achieving at least 90% reduction in the PASI score.
PASI 100
All studies20–24 have compared bimekizumab with placebo or an active comparator regarding the PASI 100. The subgroup analysis showed that a higher proportion of patients achieving PASI 100 response was found in the bimekizumab group than in the placebo group (RR = 59.87; 95% CI = 15.06–237.99; I2 = 0%; p-value = 0.89) as well as in the active comparator group (RR = 2.06; 95% CI = 1.12–3.79; I2 = 95%; p-value < 0.00001) (Figure 5).

Meta-analysis of efficacy of bimekizumab for patients achieving at least 100% reduction in the PASI score.
IGA 0 or 1
Similar efficacy was observed in IGA. The random-effects model meta-analysis revealed a significant increase in achieving IGA response (IGA 0/1) in the bimekizumab group as compared with the placebo group (RR = 21.55; 95% CI = 9.25–50.19; I2 = 20%; p-value = 0.29) as well as in the active comparator group (RR = 1.36; 95% CI = 1.02–1.81; I2 = 94%; p-value < 0.00001) (Figure 6).

Efficacy of bimekizumab in achieving IGA 0 or 1.
Clinical efficacy according to bimekizumab maintenance dosing schedules
The analyses of bimekizumab maintenance dosing schedules (every 4 weeks and every 8 weeks) showed no significant difference in the reduction of clinical indexes (PASI 90 response, PASI 100 response, and IGA 0/1) between 320 mg q4w dosage and q8w dosage (RR = 1.00; 95% CI = 0.96–1.03; I2 = 9%; p-value = 0.36) (Figure 7). No significant heterogeneity was detected.

The effect of bimekizumab 320 mg q4w versus 320 mg q8w on PASI 90, PASI 100, and IGA 0 or 1.
AEs
All trials reported AEs, and bimekizumab was well tolerated. The meta-analysis revealed that bimekizumab was associated with a slightly higher incidence of AEs than the placebo (RR = 1.39; 95% CI = 1.15–1.69; I2 = 22%; p-value = 0.29). Across all studies, the overall incidence of AEs was comparable in the bimekizumab group (76.8%) and active comparator group (71.5%) (RR = 1.13; 95% CI = 1.01–1.26; I2 = 67%; p-value = 0.05) (Figure 8). The most frequently reported AEs in the bimekizumab group were infections and skin or subcutaneous tissue disorders, including nasopharyngitis (occurred in 7–10% of the patients), upper respiratory tract infection (occurred in 4–38.9% of the patients), and oral candidiasis (occurred in 2–19.3% of the patients).

Forest plot of the effect of bimekizumab treatment on adverse events.
Discussion
This meta-analysis investigated the efficacy and safety of bimekizumab in the treatment of patients with plaque psoriasis across the five RCTs. Bimekizumab showed greater efficacy than both ustekinumab and placebo in all the evaluated measures, namely, PASI 75, PASI 90, PASI 100, and IGA response. A faster onset of clinically meaningful responses was observed with bimekizumab than with both active comparators and placebo, with PASI 75 response observed at week 4 after one dose. Our findings also suggested that after 16 weeks, the 320 mg q4w and 320 mg q8w dosage nearly provided similar benefits for patients. To the best of our knowledge, this is the first systematic analysis and comparison of RCT data on two bimekizumab maintenance dosing schedules (every 4 weeks and every 8 weeks) for the treatment of plaque psoriasis.
The greater efficacy of bimekizumab may be attributed to its dual inhibition of IL-17A and IL-17F.8,11 Available evidence indicates that blockade of both IL-17A and IL-17F may lead to a greater reduction in the production of proinflammatory cytokines, expression of psoriasis-linked genes, and inflammatory cell migration than blocking IL-17A alone. 14
In an experimental colitis model, dual blockade of both ligands has been reported as more effective than the blockade of either cytokine alone. 25 Sbidian et al. 26 compared the efficacy and safety data of systemic pharmacological treatments in psoriasis and conducted a network meta-analysis of 167 randomized controlled trials with a total of 58,912 patients. They found that bimekizumab showed a higher proportion of patients reaching PASI 90 than other anti-IL-17 drugs (i.e. secukinumab and brodalumab). Another network meta-analysis showed that bimekizumab 320 mg was superior in achieving PASI 90 and PASI 100 compared with other biologics, including risankizumab, ixekizumab, brodalumab, and secukinumab. 27 An alternative explanation for these results is that IL-17A inhibitors have a rapid onset of action compared with other biologics9,10,28 (e.g. those targeting IL-23, IL-12/23, and TNF). This is crucial because the speed of treatment response is very important for patients and remains an unmet need. 29 Moreover, bimekizumab had favorable effects on active psoriatic arthritis, 28 ankylosing spondylitis, 30 and hidradenitis suppurativa. 31 Overall, the results of this study on patients with moderate-to-severe psoriasis support the superior efficacy of bimekizumab.
We performed a subgroup analysis of two maintenance dosing schedules of bimekizumab and found that both bimekizumab maintenance dosing schedules (every 4 and 8 weeks) resulted in similarly high rates of clinical improvement and durable skin clearance. Previous studies have suggested that less frequent treatment schedules could increase the adherence to treatment regimens.32,33 Besides, considering the cost, receiving maintenance dosing every 8 weeks might be more acceptable than every 4 weeks.
It is essential to understand AEs and safety profile of psoriasis treatments. The results of our meta-analysis show that the frequency of AEs was higher in the bimekizumab group than in the placebo group. Oral candidiasis was one of the most common treatment-emergent AEs, which is consistent with the contributions of the IL-17 pathway to host protection against candida infections at the oral mucosa.33,34 A retrospective study contained 779 patients affected by moderate-to-severe psoriasis receiving ixekizumab and refractory Candida infection, pneumonia, urethral, and prostatic carcinoma were the most common AEs occurred during the treatment period. 35 A case of complicated coronavirus disease 2019 in a psoriatic patient treated with ixekizumab was also reported. 36 In addition, common AEs observed under brodalumab treatment are similar to those observed with the other IL-17 inhibitors: nasopharyngitis, upper respiratory tract infections, and Candida infections. 37 These types of studies that compared AEs between bimekizumab and other IL-17 inhibitors are not common, however. Therefore, the safety of bimekizumab remains to be further examined.
This study has some limitations. First, the analysis included a small number of studies. Fortunately, all studies are well-designed randomized controlled trials, and we performed sensitivity analyses to confirm the robustness of our findings. Second, the sample size is not large enough, and subgroup analyses were limited. Third, all the included studies had short study period; thus, the long-term efficacy of bimekizumab on psoriasis could not be determined.
Conclusion
This meta-analysis showed that bimekizumab was a promising and emerging biologic for the treatment of moderate-to-severe plaque psoriasis owing to its proven efficacy in clinical improvements (PASI 75, PASI 90, PASI 100, and IGA), with acceptable safety. Faster onset of clinically meaningful responses was observed with bimekizumab than with both active comparators and placebo, with PASI 75 response observed at week 4 after one dose. Nasopharyngitis, upper respiratory tract infection, and oral candidiasis were the most frequently reported AEs. In addition, after 16 weeks of initial maintenance treatment (320 mg q4w), both bimekizumab maintenance dosing schedules (every 4 and 8 weeks) had similar efficacy.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231163110 – Supplemental material for Bimekizumab for the treatment of moderate-to-severe plaque psoriasis: a meta-analysis of randomized clinical trials
Supplemental material, sj-docx-1-taj-10.1177_20406223231163110 for Bimekizumab for the treatment of moderate-to-severe plaque psoriasis: a meta-analysis of randomized clinical trials by Yuqian Wang, Sheng Li, Juan Bai, Xiaoxuan Cai, Shunli Tang, Peiyi Lin, Qingmiao Sun, Jianjun Qiao and Hong Fang in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
