Abstract
Background:
Inflammatory bowel disease (IBD) is a chronic inflammatory disease of the digestive tract. The aetiology and pathogenesis of IBD are complex, which may lead to metabolic disorders. As a kind of metabolite, polyunsaturated fatty acid (PUFA) is closely related to IBD.
Objectives:
The aim of this study was to explore the correlation between the serum PUFAs and the pathogenesis of IBD.
Design:
The study is a hospital-based case-control study.
Methods:
The serum free PUFAs of all participants, including 104 patients with IBD and 101 normal controls, were detected by liquid chromatography–mass spectrometry (LC-MS).
Results:
Compared with the normal control, the levels of C18:2, α-C18:3 (ALA), ɤ-C18:3, C20:4 (AA), C20:5 (EPA), ω-3 C22:5, ω-6 C22:5 and C22:6 (DHA) PUFAs in patients with Crohn’s disease (CD) were obviously decreased. However, in patients with ulcerative colitis (UC), the levels of AA, EPA, ω-3 C22:5, ω-6 C22:5 and DHA were downregulated. The concentrations of seven PUFAs were significantly downregulated in the active CD group. In addition, four PUFAs had comparatively higher levels in the remission UC group.
Conclusion:
The present study revealed substantial differences in the levels of serum fatty acids between normal controls and patients with IBD. In detail, patients with CD were deficient in PUFAs, including the essential fatty acids. Moreover, as the disease activity aggravated, some PUFAs decreased dramatically.
Keywords
Introduction
Inflammatory bowel disease (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), is a chronic bowel-recurrent inflammation that impairs patients’ quality of life and is associated with lifelong high medical costs.1–4 Thus, continuous monitoring of the activity to assess patients’ condition and adjusting therapeutic strategy timely is critical. Gastrointestinal endoscopy is the most conclusive method to assess the damage to intestinal mucosa. However, as an invasive examination with risks of bleeding and perforation, it may also lead to poor compliance. 5 In recent years, the employment of non-invasive laboratory parameters has become more attractive in assessing the activity of IBD.
Polyunsaturated fatty acids (PUFAs) are recognized to be vital in regulating and contributing to human health.6–8 Based on the location of the first double bond on the carbon chain, PUFAs are divided into two major classes: ω-3 and ω-6. 9 ω-3 fatty acids mainly include α-linolenic acid (α-C18:3, ALA), ω-3 C22:5, docosapentaenoic acid (DPA), eicosapentaenoic acid (C20:5, EPA) and docosahexaenoic acid (C22:6, DHA). ω-6 mainly consists of linoleic acid (C18:2, LA), ɤ-octadecatrienoic acid (ɤ-C18:3), arachidonic acid (C20:4, AA) and ω-6-docosaentaenoic acid (ω-6 C22:5). 10
Studies have shown that diets rich in ω-3 or low in ω-6 could significantly decrease the risk of IBD.11,12 A high intake of ω-3 could lower the risk of UC and prevent the incidence in people over 45 years old.13,14 Inflammatory factors play crucial roles in the progression of IBD, which is elevated in the intestinal mucosa of patients with active IBD.12,15–17 ω-3 has ever been reported to decrease their expressions, such as tumour necrosis factor-α (TNF-α) and interleukin-6 (IL-6).16,17 ω-3 reduces the inflammatory reaction through multiple aspects, including chemotaxis of leukocytes, expression of adhesion molecules and leukocyte-endothelial adhesion.12,18 AA, a vital component of ω-6, is a key precursor of pro-inflammatory mediators and participates in the metabolism of prostaglandin, leukotriene, thromboxane and other metabolites, all of which promote the production of inflammatory factors. 12 Various researches have ever attempted to clarify the relationship between PUFAs and IBD. However, whether serum PUFAs can be applied as a novel laboratory parameter to assess IBD activity needs to be further investigated.
The present study involved liquid chromatography–mass spectrometry (LC-MS) technology to delineate the concentrations of serum free PUFAs of 205 individuals and compared the PUFA profile of IBD patients with that of healthy patients. Then, the levels of PUFAs were compared between IBD patients in disease activity and remission. This study aimed to explore the relationship between the PUFA profile and IBD disease activity.
Material and methods
Study population
All participants were hospitalized in the Department of Gastroenterology, Zhongda Hospital, Southeast University, from June 2019 to December 2020. A total of 205 people were enrolled in this hospital-based case-control study. Of all subjects, 101 represented the normal controls who were recruited by routine examinations, while 104 were diagnosed with IBD. In addition, among these 104 people, 64 suffered from CD, and 40 had been confirmed as UC (Figure 1). The study assessed the disease activity of all IBD patients by Crohn’s disease activity index (CDAI) and Mayo score.

Flow chart of the procedure of subjects’ registration.
The inclusion criteria of the IBD patients for this study were as follows: (1) patients hospitalized at Zhongda Hospital Southeast University; and (2) comprehensive analysis of clinical manifestations, laboratory tests, image diagnostics, gastrointestinal endoscopy and pathology leading to IBD diagnosis. The diagnostic basis of all the IBD patients referred to the ‘2018 Consensus Opinions on the Diagnosis and Treatment of Inflammatory Bowel Disease’ released by the Inflammatory Bowel Disease Group of the Chinese Medical Association Gastroenterology Branch. 19 In addition, the study excluded individuals who suffered from cerebrocardiovascular diseases, severe dysfunction in the liver and kidney, other autoimmune diseases, severe infectious diseases or other gastrointestinal diseases at the same time.
The project has already been approved by the Clinical Research Ethics Committee of Zhongda Hospital affiliated with Southeast University (Approval Number: 2021ZDSYLL297-P01). All the procedures of the present study followed the Declaration of Helsinki.
Sample collection
The clinical characteristics and personal information of all the participants were documented at the inception of the study. The team synchronously collected their venous blood (3 ml) by using vacuum tubes without coagulant while inside an empty stomach for at least 8 h. All blood samples were transported at 2–8°C and then stored away from light and timely centrifuged at 1000–1200 r/min for 15 min within 2 h. The cleared supernatants were sent to the Laboratory of Precision Medicine Centre, Zhongda Hospital Southeast University, within 8 h. If the test could not be completed within 8 h, then the serum samples were stored as numbered in a −80°C ultralow temperature freezer in a dark environment.
Sample preparation and LC-MS technique
One hundred microlitres of the internal standard (IS) working solution was added to each participant’s 100 μl thawed serum, followed by the addition of 600 μl hydrochloric acid/methanol before being mixed using a vortex device. The 400 μl retrieved supernatant was blow-dried under a pure nitrogen atmosphere at 40°C. Upon the addition of 300 μl derivating agent, the samples were incubated for 45 min before detection by liquid chromatography–mass spectrometry (LC-MS).20,21
LC-MS analysis in the present study used a Derivatization of Various Fatty Acids Quantitative Assay Kit (Health Bio Co., Ltd, Beijing, China) and was carried out on a Triple Quad 3200MD (AB Sciex, USA) in multiple reaction monitoring (MRM) mode with negative electrospray ionization (ESI). The ion-spray voltage of the equipment was set at 5500 V, and the gas temperature was 550°C. The curtain gas was 20 lbf/in2, and the collision gas was set at 6 lbf/in2. The chromatographic column was equipped with Agilent ZORBAX ECLIPSE plus C18 (100 mm × 3 mm, 3.5 μm, Agilent, USA), and the column temperature was maintained at 45°C. In addition, 50% acetonitrile was used as the probe rinse solution. The injector temperature of the equipment was set at 15°C, and each injection volume was accurate to 5 μl. Each sample size per kit was controlled at 0.5–1.0 ml.
Fatty acid quantification
Each serum free fatty acid level was first normalized by internal standard and then quantified to absolute concentration using the calibration curve of the corresponding chemical standard. Taking the concentration of the calibrator as the independent variable xi and the peak area ratio of the corresponding concentration calibrator to the internal standard as the dependent variable yi, the linear regression equation y = ax + b and the correlation coefficient r were calculated. When r ⩾ 0.990 of the calibration curve, the signal intensity of low-value and high-value quality control products was put into the regression equation to obtain the concentration of the quality control products. When the relative deviation of high-value quality control products was ⩽±15% and that of low-value quality control products was ⩽±20%, the signal intensity of serum samples was put/entered into the regression equation to obtain the target concentration of serum samples.
Statistical analysis
Statistical analysis, as well as a part of graph representation, was accomplished using SPSS 25.0 software. The preparation of metabolomic analysis and relative charts was performed with the R software package (v3.6). Normality distribution and variance homogeneity tests were carried out for measurement data. An independent-sample t-test was applied between the two groups of data. Data from three or more groups were analysed using one-way analysis of variance (ANOVA) and are expressed as the mean ± standard error of the mean (SEM). Mann–Whitney and Kruskal–Wallis tests were used in cases of nonnormally distributed data, expressed as medians (lower quartile, upper quartile). The significance of classified data was used with the chi-square test. The correlation between the concentrations of the free fatty acids and IBD activity was assessed by the Spearman rank correlation test. A Mayo score ⩾ 3 and CDAI ⩾ 150 were used as reference variables. The sample size was estimated by PASS 15.0 software with power calculations based on an alpha value of 0.05 and a beta value of 0.9. All p < 0.05 were considered to have statistical significance.
Results
Clinical characteristics of subjects
A total of 205 individuals were included and classified as normal controls (n = 101, 49.3%), CD (n = 64, 31.2%) or UC (n = 40, 19.5%).
Table 1 displays the clinical characteristics of all participants. No significant differences in sex, height, smoking history, alcohol consumption, chronic diseases, alanine transaminase (ALT) or aspartate aminotransferase (AST) between the normal control group and Group CD were documented. No obvious statistical differences between the normal control group and Group UC in age, sex, height, weight, body mass index (BMI), smoking history, alcohol consumption, chronic diseases, ALT or AST (p > 0.05) were documented (Table 1). As CD often occurs in children and adolescents, the overall age of the disease is relatively low,22–24 and the nutritional status of IBD patients is poor.25–30 In the study, it was found that the age, weight and BMI of the CD patients involved in this study were lower than those of individuals belonging to the normal controls (p < 0.05). Significant differences in serum albumin (ALB) and lipid levels between the normal control and the two IBD subgroups were found. The ALB and lipid levels of IBD patients were lower than those of normal controls (p < 0.05). Correlation analysis was conducted between BMI, ALB, TC and TG and the concentration of total free fatty acids, and there was no statistical difference (Supplementary Table S1).
Baseline clinical characteristics of the 205 subjects involved in the study.

ALB, albumin; ALT, alanine transaminase; AST, aspartate aminotransferase; CD, Crohn’s disease; CDAI, Crohn’s disease activity index; CNS, central nervous system; N/A, not applicable; TC, total cholesterol; TG, triglyceride; UC, ulcerative colitis.
p < 0.05, compared with the normal control group.
Levels of serum free PUFAs of all the participants
First, the concentrations of all subjects’ serum samples were examined to delineate specific PUFAs by LC-MS. PUFA levels significantly differed between the normal controls and IBD patients.
Table 2 shows the concentrations and composition (percentage of total fatty acids) of ω-3 and ω-6 in all participants. The overall level of these PUFAs in CD patients was obviously lower than that in the normal controls (p < 0.05). Similarly, the level of ω-3 in UC patients was obviously lower than that in the normal control group, while there was no significant statistical difference in the levels of ω-6 between Group UC and the normal group (p > 0.05) (Table 2). While the percentages of ω-3 and ω-6 in total fatty acids were compared, both the CD patients and UC patients were significantly lower than the normal control group.
Concentrations and composition (percentage of total fatty acids) of serum free fatty acids in normal controls and IBD patients.

Mean ± SE.
CD, Crohn’s disease; IBD, inflammatory bowel disease; PUFA, polyunsaturated fatty acid; SE, standard error; UC, ulcerative colitis.
p < 0.05 for comparison between normal controls and CD patients.
p < 0.05 for comparison between normal controls and UC patients.
When comparing the normal and CD groups, we found the presence of eight species of PUFAs with significant differences, such as C18:2, ALA, ɤ-C18:3, AA, EPA, DHA, ω-3 C22:5 and ω-6 C22:5, with p values less than 0.05 (Figure 2). In addition, we compared the normal group with Group UC. Figure 2 shows that the values of AA, EPA, DHA, ω-3 C22:5 and ω-6 C22:5 were significantly different between the normal controls and UC patients, with p values of 0.001, 0.002, <0.001, 0.019 and <0.001, respectively.

The concentrations of serum free unsaturated fatty acids from the normal group, Group CD and Group UC.
Furthermore, based on the concentrations measured by LC-MS, the present study plotted heatmaps to further demonstrate the differences in the overall PUFA profile among the IBD samples with different disease activity levels (Figure 3).

Analysis of differential PUFAs of all the serum samples between CD (a) and UC (b) through LC-MS with heatmaps, in which the Z-value was used to normalize the level of the same fatty acid.
The study next investigated the delta 6-desaturase (D6D) activity based on the measured PUFA composition, including the ratio of ɤ-C18:3 to C18:2. This method for estimating D6D activity is rather straight and more commonly used than invasive assays. 31 No significant difference in D6D activity between IBD patients and normal controls (p > 0.05) could be documented (Table 3).
Concentrations of D6D activity in normal controls and IBD patients.

D6D activity = ɤ-C18:3/C18:2. Mean ± SE.
CD, Crohn’s disease; DD, delta 6-desaturase; IBD, inflammatory bowel disease; SE, standard error; UC, ulcerative colitis.
Correlation analysis between the disease activity of IBD and the concentrations of serum PUFAs
This study further generated a violin plot to explore the existing correlation between disease activity and the level of serum PUFA profile in patients with CD and UC (Figure 4). First, we divided Group CD into two subgroups representing the active CD group and the remission CD group, according to CDAI < 150. A rank-sum test was applied to compare the levels of PUFAs and revealed that the concentrations of seven species of fatty acids were significantly downregulated in the active CD group, namely, C16:1, C18:1, LA, AA, EPA, ω-3 C22:5 and ω-6 C22:5, with p values less than 0.05. Second, using a Mayo score of less than 3, we divided Group UC into two subgroups, representing the active UC group and the remission UC group. Similarly, through the rank-sum test, we identified significant differences in LA, α-C18:3, ɤ-C18:3 and ω-6 C22:5, with p values of 0.04, 0.022, 0.003 and 0.034, respectively. Compared with the active UC group, the above four fatty acids had comparatively higher levels in the remission UC group. When comparing the active CD group with the active UC group, the levels of all measured PUFAs were not significantly different, with p > 0.05.
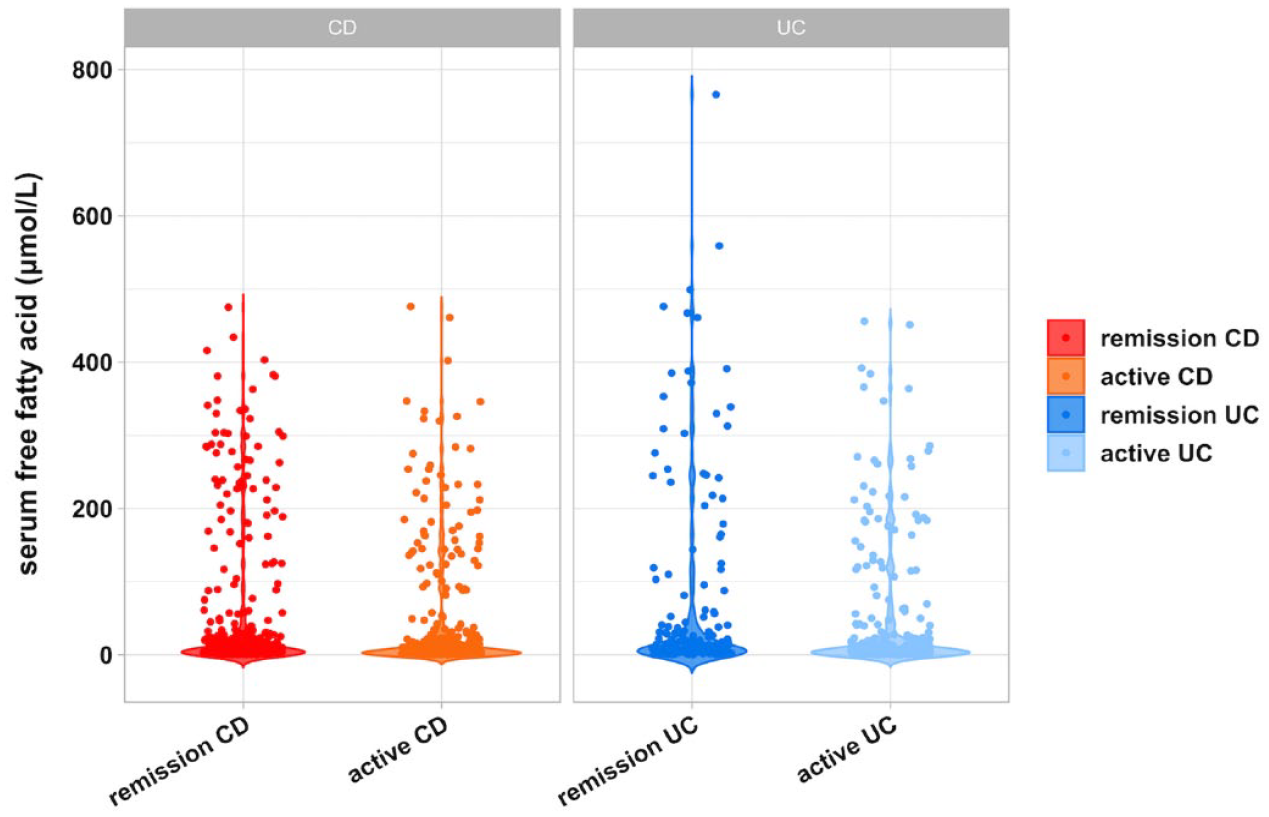
Violin plot of the levels of all detected fatty acids in CD and UC.
The present study performed PCA on the overall changes in the serum PUFA profile of the disease remission state and the activity state, aiming to further clarify the differences. As shown in Figure 5(a), a significant separation between the serum PUFA spectrum of the remission CD group and the active CD group (p = 0.004) was revealed. This indicates that a change in the activity of CD disease is associated with an obvious change in the PUFAs. Furthermore, the research shows that both PCA1 (97.02%) and PCA2 (2.56%) account for 99.58% of the difference. The calculated R value of 0.1303 indicates that the difference between the groups is greater than the difference within the same group. According to Figure 5(b), no obviously statistical differences were observed with respect to the overall changes in PUFAs between the remission UC group and the active UC group (p = 0.065).
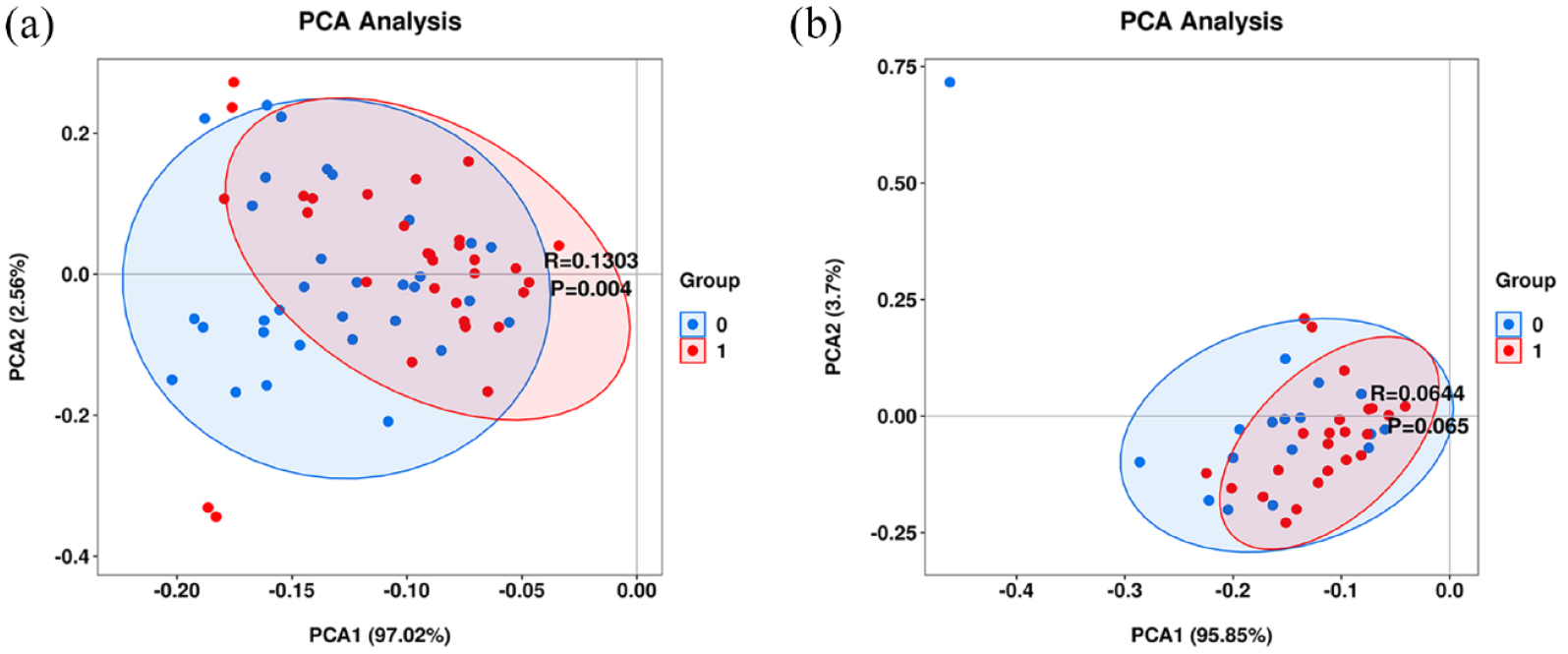
(a) PCA between the remission CD group and the active CD group. Group 0 represents the remission CD group with red colour coding, while Group 1 is the active CD group with blue colour coding. (b) PCA between the remission UC group and the active UC group. Group 0 represents the remission UC group with red colour coding, while Group 1 is the active UC group with blue colour coding.
Discussion
IBD is closely related to genetic factors and environmental and dietary changes, as well as to the disorders of the immune system and metabolism.32,33 The current in-depth exploration of its mechanism has gradually focused on metabolomics, while PUFA represents one of the hotspots of IBD metabolomics research. 34 Therefore, a deep exploration of the metabolic changes of the PUFA profile in IBD patients helps to find potential pharmacological targets in the future. 35
The study showed the significant differences between IBD patients and the normal controls with respect to several specific ω-3 and ω-6 PUFAs. The concentrations in the normal control group were relatively higher. As the essential fatty acids, LA and ALA were poor in CD patients. It suggested that proper dietary supplementation of essential fatty acids may help to improve or even prevent the occurrence and development of CD. Surely, our team needs further prospective studies to verify this conjecture.
Disease activity can cause increased vascular permeability at which moment the PUFAs could be transferred to the damaged parts, causing lipid metabolic disorder. The composition of the peripheral PUFA could generate a secondary change.36,37 Under certain conditions, PUFAs can be released from the phospholipid pool on the cell membrane and reach a free state to participate in the metabolism, thereby producing hundreds of lipid regulatory substances and contributing to various inflammatory reactions. 38 The metabolism of ω-3 was associated with the secretion of some anti-inflammatory mediators.11,39–41 ω-6 fatty acids are the synthetic precursors of endocannabinoids, producing pro-inflammatory substances. 42 The two are metabolized through the common enzyme reaction system, and ω-3 can competitively inhibit the reaction of ω-6 with a corresponding enzyme and reduce the production of pro-inflammatory factors through an antagonistic mechanism. 40 Interestingly, while these two types of fatty acids trigger opposite effects on inflammation, their levels were decreased synchronously in the IBD group in this study. This is rather different from the researches conducted by Grimstad et al. 43 and Mayr et al. 44 We speculated that alleviation of inflammation can be achieved by compensatory consumption of ω-3, leading to a synchronous decrease in ω-3 and ω-6. It was consistent with the results of some other studies.45,46
D6D activity participates in the transformation of LA into ɤ-C18:3 and C20:4, the precursors of some inflammatory mediators like prostaglandin and leukotriene, which is consistent with the potential association between D6D activity and inflammation. 47 The estimation of specific delta desaturase activity is helpful to evaluate the activity of this pathway, suggesting that inhibitors targeting D6D may have therapeutic potential. 48 However, no significant difference was found in D6D activity among the participants in this study. We will increase the sample size for further exploration in the future.
In addition, we also found that the level of PUFAs decreased with disease activity of IBD. In particular, C18:2 and ω-6 C22:5 PUFAs were associated in both CD and UC. During the active period, the concentration of unsaturated fatty acids was significantly reduced. The present study displayed that increased consumption of ω-6 leads to an increased production of inflammatory factors, thus promoting the progression of IBD. In addition, increased consumption of ω-3 is relevant to increased production of anti-inflammatory factors to maintain homeostasis. Just consistent with the research by Veselinovic et al., 49 a defined daily dose of ω-3 supplementation can lead to a clinical improvement in patients with chronic inflammatory diseases, suggesting that specific PUFAs do have potential power for improving the symptom severity of IBD patients. A small sample study by Bugajska et al. 50 not only found a reduction in serum n-3 levels in IBD patients, but also found the same trend when they examined the colonic mucosa. Contrary to us, Scoville et al. 51 found that serum n-3 levels in CD patients were positively correlated with inflammation and cytokine levels. They thought that the utilization of n-3 fatty acids might result in an accumulation in the serum and the production of pro-inflammatory cytokines. Such differences in results are quite normal and need to be further verified with more large samples and excavation of the mechanism. At the mechanism level, current opinions suggest that the PUFAs might promote endoplasmic reticulum stress, leading to the inflammation of intestinal epithelium cells and fibroblasts, or it may be due to the specific deletion of Xbp1 or Gpx4 in intestinal epithelium cells. 52 However, all the speculations need to be confirmed by more lab studies.
Certainly, the findings need to be validated in future studies, especially in combination with other new metabolite levels and special intestinal bacteria. According to current opinion, gut microbes may be involved in mediating the saturation and transformation of PUFAs, resulting in anti-inflammatory effects on the body. Equally, dietary intake of essential fatty acids may increase beneficial intestinal bacteria to help maintain body balance. Our team will also conduct more exploration of IBD based on this view.34,53,54 While several studies on clinically used biomarkers for IBD detection indicate rather different results, they also feature limitations. Therefore, it is of excellent clinical significance to look for a new serological indicator with high sensitivity and high specificity for the accurate evaluation of disease activity in IBD. The team will continue to explore this direction later.
Inevitably, the research has a few limitations, such as the single-centre retrospective study. The fact that the disease activity score of IBD patients was affected by the subjective factors of patients and the evaluation of physicians may also lead to bias. Therefore, further multicenter prospective studies with large samples to acquire more evidence will be better. Furthermore, the dietary fatty acid intake of the participants was not investigated. This may translate to a rather altered snapshot of the levels of ω-3 and ω-6 in the participants’ blood. Further studies on the molecular mechanisms by which PUFAs regulate IBD activity need to be substantiated by in vitro experiments and in vivo animal models.
Conclusion
The study found that the PUFA profile and the level of each fatty acid were different between normal subjects and IBD patients. IBD patients had lower levels of PUFAs as well as CD patients had poor essential fatty acids. There is a nonlinear relationship between the concentrations of PUFAs and IBD patients’ pathogenetic condition. The concentrations of ω-3 and ω-6 were both inversely correlated with disease activity.
Supplemental Material
sj-docx-1-taj-10.1177_20406223231156826 – Supplemental material for Characterization of serum polyunsaturated fatty acid profile in patients with inflammatory bowel disease
Supplemental material, sj-docx-1-taj-10.1177_20406223231156826 for Characterization of serum polyunsaturated fatty acid profile in patients with inflammatory bowel disease by Jingjing Jiang, Lu Chen, Rui Sun, Ting Yu, Shuyu Jiang and Hong Chen in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
All authors are grateful for the excellent technical support from the Laboratory of Precision Medicine Center, Zhongda Hospital Southeast University. We thank Doctor Hailu Wu for a critical reading of the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
