Abstract
Hypertrophic cardiomyopathy (HCM) is a common genetic disorder characterised by unexplained left ventricular hypertrophy. Left ventricular outflow tract obstruction is an integral component of the disease, often resulting in significant symptoms, but also carrying a risk of progression to heart failure and death. Advancements in our understanding of the pathophysiology of HCM have led to the development of new therapies targeting the molecular basis of the disease at the level of the cardiac sarcomere, the basic contractile apparatus of the myocardium. Myosin modulators are a novel class of small molecules which target cardiac myosins directly to modulate cardiac contractility. The myosin inhibitors present the first advancement in pharmacological management of obstructive HCM in almost 35 years, with a growing body of evidence for the safety, tolerability and efficacy of mavacamten, and to a lesser extent aficamten. The aim of this review is to summarise the current management of patients with obstructive HCM and review the most recent available data from clinical trials pertaining to myosin inhibition.
Keywords
Introduction
Hypertrophic cardiomyopathy (HCM) is a primary disorder of the myocardium characterised by unexplained hypertrophy of the left ventricle in the absence of other abnormal loading conditions. It affects between 1:200 and 1:500 of the general population.1,2 It is a disease of tremendous phenotypic diversity and clinical presentation. Left ventricular outflow tract obstruction (LVOTO), defined as a peak instantaneous left ventricular outflow tract (LVOT) pressure gradient of ⩾30 mmHg, is an integral component of the disease, occurring in around 25–30% of patients under resting conditions, but up to 70% of patients either under resting conditions or with provocation with physiologic manoeuvres such as Valsalva or exercise. 3 While some patients with significant LVOTO are asymptomatic, breathlessness, exertional chest discomfort, presyncope or exertional syncope may be present. The presence of LVOTO confers a risk of progression to New York Heart Association (NYHA) class III/IV symptoms or death (heart failure or stroke-related), particularly in patients above the age of 40 years. 4 In addition, LVOTO also appears as a marker or modifier of risk for sudden cardiac death in the HCM risk-SCD calculator advocated by the European Society of Cardiology in their 2014 HCM Guideline document.5,6 A comprehensive assessment of LVOTO with both resting and exercise echocardiography is now strongly advocated in clinical guidelines, particularly in symptomatic patients, to identify patients at risk of progression to heart failure and institute timely and appropriate medical therapy or invasive septal reduction strategies. 7
LVOTO arises due to the interplay of several anatomical factors that are present in varying degrees in individual patients. Hypertrophy of the basal interventricular septum and hypertrophy and anterior displacement of the papillary muscles serve to reduce the cross-sectional area of the LVOT. In addition, the mitral valve apparatus is frequently abnormal in HCM patients, 8 with elongated or redundant leaflet tissue, and anterior displacement of the coaptation line of the anterior and posterior leaflets towards the basal septum. Rapid ventricular ejection through a narrowed outflow tract exposes the anterior mitral valve leaflet to Venturi or drag forces, resulting in the development of systolic anterior motion of the mitral valve (SAM). The contact between the anterior mitral valve leaflet and basal septum in mid-to-late systole creates a further barrier to left ventricular outflow, but also malcoaptation of the mitral valve leaflets, giving rise to posteriorly directed mid-to-late systolic mitral regurgitation.
In some patients, the level of obstruction is in the mid-ventricle (MVO), as opposed to the outflow tract, and results from systolic cavity obliteration due to apposition of the papillary muscles with the mid-interventricular septum. SAM and mitral regurgitation are not typical features in this form of obstruction, but around 25% of patients are prone to developing apical infarction and aneurysm formation, 9 which is an independent predictor of adverse outcomes such as sudden cardiac death, lethal arrhythmia, stroke and progression to end-stage heart failure.9,10 The presence/absence of an apical aneurysm is incorporated into the risk stratification model of the 2020 American Heart Association (AHA)/American College of Cardiology (ACC) guideline document. 7
The aim of this review is to summarise the current management of patients with obstructive HCM and review the most recent available data from clinical trials pertaining to myosin inhibition.
Current treatment options for obstructive HCM
While some individuals are asymptomatic, most patients with either LVOTO or MVO will present with symptoms requiring therapy. The general approach to managing obstruction is that of a stepwise escalation from pharmacological therapy to invasive septal reduction therapies (SRTs) in those individuals who remain symptomatic despite maximally tolerated medical therapy.
Pharmacological therapy
Non-vasodilating beta-blockers (titrated to maximum tolerated dose) remain the cornerstone of therapy and act via a negative inotropic and chronotropic effect to reduce obstruction and improve diastolic filling. In the event of intolerance to beta-blocker, then a non-dihydropyridine calcium-channel blocker such as verapamil may result in symptomatic improvement. However, caution should be exercised in patients with severe obstruction (gradient >100 mmHg) and raised pulmonary artery pressures due to the risk of precipitating pulmonary oedema. If these agents prove ineffective in symptom control, then disopyramide (a class IA anti-arrhythmic agent with negative inotropic effects) can be added as a second-line therapy. Monitoring of the QTc interval on 12-lead electrocardiogram (ECG) is required after institution of therapy, and uptitration of dosage may be limited by anticholinergic side effects. Consideration should be given to optimising drug therapy for comorbidities, particularly the coexistent prescription of antihypertensive agents and diuretics. Vasodilatation and hypovolaemia should be avoided as far as possible, and therefore any medications that cause these effects should be avoided where possible. In a few cases, low-dose loop or thiazide diuretics can be used cautiously tailored to patients’ needs, in those who have dyspnoea secondary to an element of fluid overload.
Approximately two-thirds of patients can be managed with medical therapy alone, and be rendered asymptomatic or mildly symptomatic, often with a >50% reduction seen in LVOT gradients on echocardiography.11,12 In addition, the Toronto group demonstrated (in a cohort of 649 patients studied in their centre) that long-term survival of patients treated conservatively with medical therapy who demonstrate both a significant symptomatic improvement and reduction in resting LVOTO was similar to patients treated with invasive SRT. 13
Septal reduction therapies (SRT)
Despite the efficacy and safety of medical therapy, a considerable number of patients remain symptomatic, necessitating further strategies for relief of LVOTO. Consideration of SRT requires a detailed assessment of clinical, anatomic and haemodynamic factors in the individual patient to determine suitability for surgical myectomy (SM) or alcohol septal ablation (ASA). These therapies require a necessary degree of expertise and the involvement of a multidisciplinary team, and should only be performed by experienced operators in the setting of a comprehensive HCM clinical programme. Patients who remain symptomatic despite maximally tolerated medical therapy and in whom an LVOT gradient of ⩾50 mmHg has been demonstrated (either at rest or with provocation) are candidates, provided they have sufficient septal hypertrophy to allow safe and effective treatment.5,7
SM and ASA have been demonstrated to be the most effective and definitive therapeutic strategies for relieving symptoms and relieving LVOTO in HCM.14–17 ASA, via injection of alcohol with subsequent infarction of a target area in the hypertrophied basal septal adjacent to the SAM-septal contact point, leads to regression of hypertrophy with scar formation over a 6- to 12-month period, with associated reduction in LVOT gradients and improved functional class. In experienced centres, ASA offers a suitable alternative for patients of advanced age or higher surgical risk due to comorbid medical conditions. Data from the multinational European ASA registry (Euro-ASA), which included 1275 highly symptomatic patients, demonstrated survival rates at 1, 5 and 10 years of 98%, 89% and 77% respectively. LVOT gradients were reduced from 67 ± 36 to 16 ± 21 mmHg and symptoms significantly improved, with 89% of patients reporting symptoms of NYHA class <II at follow-up. 17 ASA is associated with the risk of requirement of permanent pacemaker therapy and the need for repeated procedures, although outcomes thereafter are still favourable.18,19
SM represents the gold standard of treatment for symptomatic obstructive HCM in high-volume dedicated HCM centres of excellence, with a post-operative 30-day mortality of 0.5%. Long-term improvement in symptomatic status is reported in more than 90% of patients. Long-term survival at 1, 5 and 10 years is 98%, 96%, and 83%, respectively, for all-cause mortality, and 99%, 98% and 95% for HCM-related mortality. 16 In a recent comparison of ASA versus SM, a cohort of 585 patients undergoing ASA and 3274 patients undergoing SM at three large, specialised centres were retrospectively analysed with regard to all-cause mortality. 20 Patients undergoing ASA were older and had more comorbidities. Over a median follow-up of 6.4 years, the 10-year all-cause mortality rate was 26.1% in the ASA group and 8.2% in the SM group, with mortality remaining higher in the ASA group after correction for age, sex and comorbidities (hazard ratio, 1.68). Myectomy surgery should be the treatment of choice in patients with excessive septal hypertrophy or when concomitant surgery on the coronary arteries or mitral valve apparatus is required.
Dual-chamber short atrioventricular delay pacing, although a potential therapeutic option, has shown only a modest benefit in randomised controlled trials and has a limited role in the management of LVOTO. Its primary use is in older patients, those with an independent indication for device therapy, or patients in whom surgical risk is prohibitive and anatomy is unsuitable for ASA.
Myosin modulators and inhibitors
Myosin modulators are a novel class of small molecules which target cardiac myosins directly to modulate cardiac contractility. Advancements in our understanding of the pathophysiology of HCM have led to the development of new therapies targeting the molecular basis of the disease at the level of the cardiac sarcomere, the basic contractile apparatus of the myocardium. HCM-associated mutations affecting the genes encoding sarcomeric proteins result in a state of myocardial hypercontractility. Around 70% of identifiable mutations involve the β-myosin heavy chain (MYH7) and myosin-binding protein C (MYBPC3).21,22 Myosin, which contains the ATPase involved in actin–myosin cross-bridge formation, serves as the molecular motor for myocardial contraction. Mutations in MYH7 affect the activity of myosin ATPase to increase myocardial force generation, while MYBPC acts as a brake to myofibril contraction. These mutations serve to increase the availability of myosin heads able to form cross-bridges with actin, with a reduced percentage remaining in a super-relaxed energy-sparing state. While in health 40–50% of the myosin heads are in this super-relaxed state, in HCM this ratio shifts to as low as 15–20%.23,24 This excess of sarcomeric myosin–actin cross-bridge formation during systole, and more importantly diastole, leads to activation of hypertrophic, inflammatory and profibrotic signalling pathways.21,25 The result is one of progressive adverse myocardial remodelling, fibrosis, and impaired myocardial relaxation, serving to promote myocardial inefficiency and increased energy consumption. 26
Selective allosteric inhibitors of cardiac myosin such as the small molecules mavacamten (previously designated MYK-461; Bristol Myers Squibb) 27 and aficamten (designated CK-274; Cytokinetics), 28 via a reduction in the number of myosin heads available for cross-bridge formation, lead to a transition of the overall myosin head population towards an energy-sparing state. 23 In a murine model, Green et al. demonstrated a reduction in contractility via a decrease in ATPase activity of the myosin heavy chain. With chronic administration of MYK-461, the development of ventricular hypertrophy, myocyte disarray and myocardial fibrosis was suppressed, and hypertrophic and profibrotic gene expression was attenuated. 29
Clinical trials of myosin inhibitors in hypertrophic cardiomyopathy
Mavacamten, the first in-class myosin inhibitor, was first evaluated in an open-label, non-randomised, phase II trial which studied 21 patients with obstructive HCM in the PIONEER-HCM study. 30 Two cohorts of patients were studied: those on background therapy with beta-blockade (dosage of mavacamten 2–5 mg/day) and those without (dosage of mavacamten 10–20 mg/day). In the higher dosed cohort, a significant reduction in LVOT gradients was demonstrated (103 mmHg at baseline to 19 mmHg at 12 weeks). In addition, resting left ventricular ejection fraction (LVEF) was reduced by a mean of 15%, and peak VO2 was increased by a mean of 3.5 ml/kg/min.
Following on from these initial positive results, the EXPLORER-HCM [Clinical Study to Evaluate Mavacamten (MYK-461) in Adults With Symptomatic Obstructive Hypertrophic Cardiomyopathy] was designed to evaluate the efficacy and safety of mavacamten in symptomatic obstructive hypertrophic cardiomyopathy, and includes the largest study population to date of trials of myosin inhibitors. 31 Patients with obstructive HCM and an LVOT gradient ⩾50 mmHg and with NYHA Class II–III symptoms were randomised on a 1:1 basis to receive either mavacamten or placebo for a 30-week duration. The primary endpoint was a ⩾1.5ml/kg/min increase in peak VO2 and at least one NYHA class reduction, or a ⩾3.0 ml/kg/min increase in VO2 without NYHA class worsening. The study cohort compromised 251 patients, of whom 37% on mavacamten versus 17% on placebo met the primary endpoint. Mavacamten resulted in improvements in exercise capacity, with a mean increase in peak VO2 of 1.4 ml/kg/min. NYHA functional class improved by at least one class in 80 of the 123 patients on mavacamten, and improved health status, as assessed by the Kansas City Cardiomyopathy Questionnaire–Clinical Score Summary (KCCQ-CSS). Patients on mavacamten had greater reductions than those on placebo in post-exercise LVOT gradient (with a mean reduction of 36 mmHg from baseline). Safety and tolerability were similar in both treatment arms. No serious events of congestive heart failure occurred in the mavacamten group. Seven patients in the mavacamten group (compared with two in the placebo group) had a transient decrease in LVEF to <50%. Three of these patients had protocol-driven temporary discontinuation of treatment, with subsequent normalisation of LVEF and resumption of therapy to study completion. In the other four patients with an LVEF <50% at the week 30/end-of-treatment visit, confirmation of LVEF recovery to baseline after the 8-week washout period occurred in three patients. EXPLORER-HCM provided the first data from a randomised phase III trial of clinical benefit, safety and tolerability of disease-specific treatment in HCM. Despite these promising and positive results, it is important to note that 63% of patients failed to meet the primary endpoint of the study.
Several substudies of the main EXPLORER-HCM studied have assessed the effects of mavacamten on cardiac structure and function. Key echocardiographic parameters were assessed in 251 symptomatic obstructive HCM patients (n = 123 on mavacamten and n = 128 on placebo therapy), derived from serial echocardiograms performed over a 30-week period. 32 Mavacamten therapy resulted in significant improvements in diastolic function, LVOT obstruction, left atrial size (as measured by left atrial volume index) and filling pressures (assessed from the lateral E/E′ ratio). These findings were mirrored by a reduction in NT-proBNP (N-terminal pro hormone B-type natriuretic peptide – a biomarker of myocardial wall stress). In addition, 81% of patients had complete resolution of systolic anterior motion of the mitral valve, and patients had significant reductions in LVOT obstruction (measured under resting, Valsalva and post-exercise conditions). In the EXPLORER-HCM Cardiac Magnetic Resonance substudy analysis, which included 35 patients (n = 17 on mavacamten and n = 18 on placebo), changes in left ventricular mass index (LVMI), wall thickness, and LVEF were assessed over the 30-week treatment period. 33 A reduction in LVMI, wall thickness and LAVI was seen in the patients treated with mavacamten, along with a reduction in LVEF (although maintained within the normal range, with no patients experiencing a drop in LVEF to <50%). No changes were noted in late gadolinium enhancement or extracellular volume fraction. Results of the landmark EXPLORER-HCM trial are summarised in Figure 1.
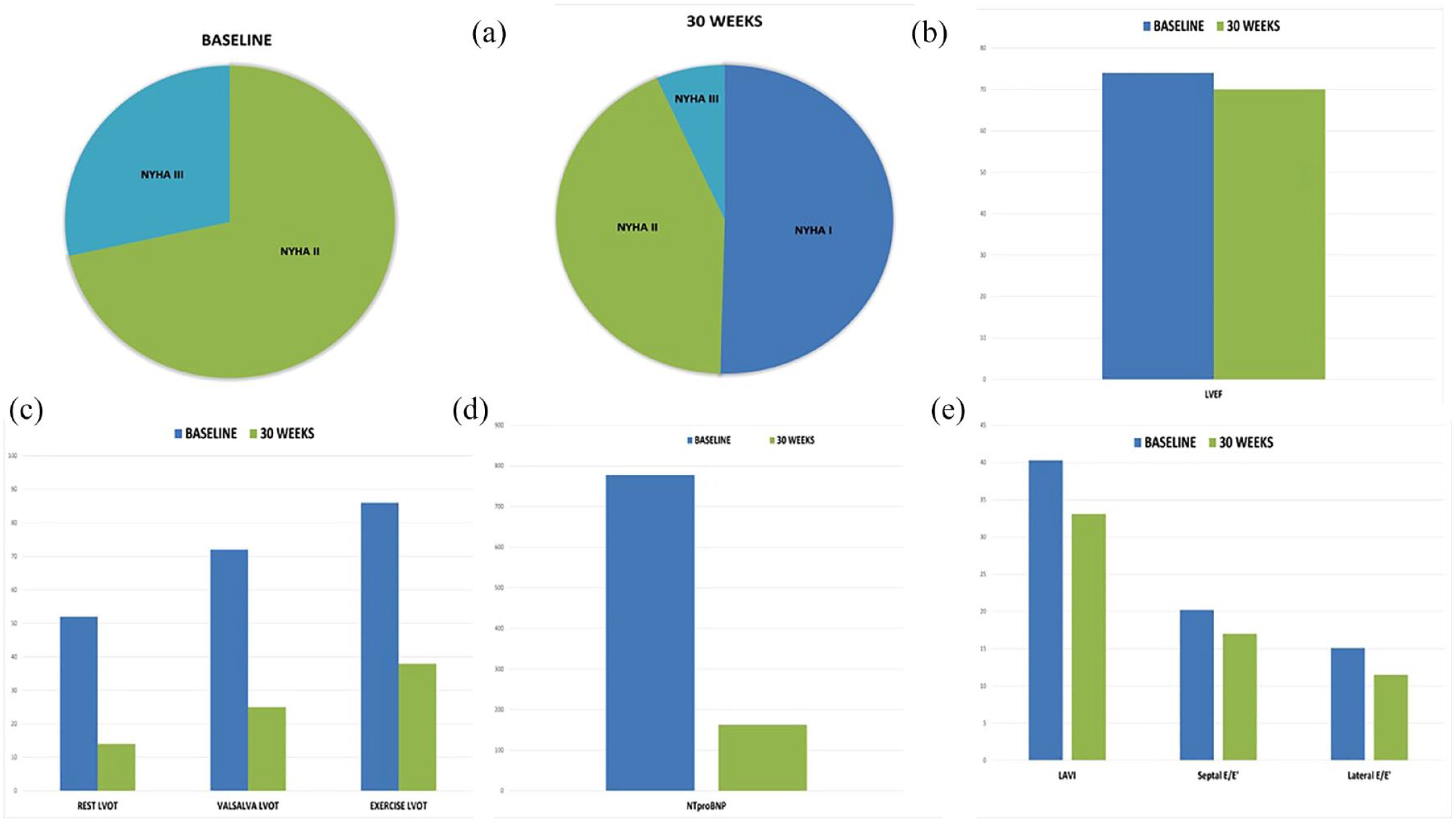
Results of the EXPLORER-HCM trial of mavacamten in symptomatic obstructive HCM. Key results of the EXPLORER-HCM trial are shown, demonstrating the effect of mavacamten on symptoms, left ventricular systolic and diastolic function, left ventricular outflow tract obstruction, and cardiac biomarkers. (a) NYHA class at baseline and 30 weeks for the study cohort. (b) The non-significant change in LVEF over the study period in the mavacamten group. (c) The significant reduction in LVOT gradients assessed at rest, with Valsalva and post-exercise. (d and e) Effects of mavacamten on diastolic dysfunction, with significant reductions in NT-proBNP and left ventricular filling pressures (as assessed by septal and lateral E/E′ ratios) over the study period.
VALOR-HCM is a multicentre phase III, double-blind, placebo-controlled, randomised study of mavacamten versus placebo in a cohort of symptomatic patients with obstructive HCM who were on maximal-tolerated medical therapy and eligible for SRT. Patients were randomised in a 1:1 fashion to mavacamten (n = 56) or placebo (n = 56), with dose titration over a 16-week period guided by LVEF and recorded LVOT gradients at rest and with Valsalva. 34 The primary endpoint (decision to proceed to SRT or guideline-eligible to proceed) was reached in 17.9% of patients in the mavacamten group versus 76.8% in the placebo group. LVOT gradients were significantly reduced in the mavacamten group, and a significant improvement in NYHA class and KCCQ-CSS was also demonstrated, with the findings providing complementary evidence of efficacy, safety and tolerability to EXPLORER-HCM. Results of the VALOR-HCM trial are summarised in Figure 2.

Results of the VALOR-HCM trial of mavacamten in symptomatic obstructive HCM. Key results of the VALOR-HCM trial are shown, demonstrating the effect of mavacamten on symptoms, left ventricular outflow tact obstruction, and eligibility for septal reduction therapy (SRT) based on guideline criteria. (s) The percentage of patients eligible for SRT at baseline and after 16 weeks of treatment for the study cohort. (b) The change in NYHA class symptoms after treatment. (c) The significant reduction in LVOT gradients (in mmHg) assessed at rest and Valsalva in the mavacamten group compared with the placebo group at study completion.
While the initial studies have focused on patients with obstructive HCM, a patient group in whom negative inotropic agents are typically used, MAVERICK-HCM assessed the effect of mavacamten in patients with non-obstructive HCM. MAVERICK-HCM was a double-blind, placebo-controlled, phase II study in adult patients with symptomatic non-obstructive HCM. 35 The study enrolled 59 patients with NYHA class II/III symptoms, an LVEF ⩾55% and NT-proBNP ⩾300 pg/ml. Patients were randomised in a 1:1:1 fashion to either mavacamten (two groups defined using pharmacokinetic-adjusted dose targeting a plasma level of either 200 ng/ml or 500 ng/ml) or placebo. Treatment with mavacamten resulted in significant reductions in NT-proBNP, suggesting improvements in myocardial wall stress. Significant reductions in LVEF to ⩽45% were seen in five patients on mavacamten but were reversible in all cases.
Aficamten is a next-generation cardiac myosin modulator with a similar mechanism of action to mavacamten, but with a shorter half-life and a shallower concentration-response profile. 28 After demonstrating promising results in preclinical trials, the REDWOOD-HCM (Randomised Evaluation of Dosing With CK-274 in Obstructive Outflow Disease in HCM) trial was designed to further evaluate the safety, tolerability and efficacy of aficamten at various doses. Designed as a multicentre, randomised, placebo-controlled, double-blind, dose-finding trial, REDWOOD-HCM randomised patients to receive one of two aficamten dosing regimens (which included the use of echocardiography to guide dosing levels) or placebo therapy over a study period of 10 weeks. Preliminary results from a cohort of 41 symptomatic obstructive HCM patients with a baseline LVEF >60% and on stable background therapy were presented at the Heart Failure Society of America 2021 Annual Scientific Meeting. Administration of aficamten resulted in reductions in LVOT gradients and symptomatic improvement within 2 weeks of drug initiation. Ninety-three percent of patients in the high-dose aficamten group had resting LVOT gradients of <30 mmHg and a Valsalva LVOT gradient of <50 mmHg at 10 weeks, compared with 78% in the low-dosed cohort and 8% in the placebo group. In addition, significant reductions in NT-proBNP levels were seen in both aficamten dosing cohorts, and an improvement in symptoms by at least one NYHA class was seen in 31% of the placebo group, 43% of the low-dosed aficamten group and 64% of the high-dosed aficamten group.
An overview of the completed and ongoing trials of myosin inhibitors in HCM is provided in Tables 1 and 2.
Completed trials of myosin inhibitors in hypertrophic cardiomyopathy.
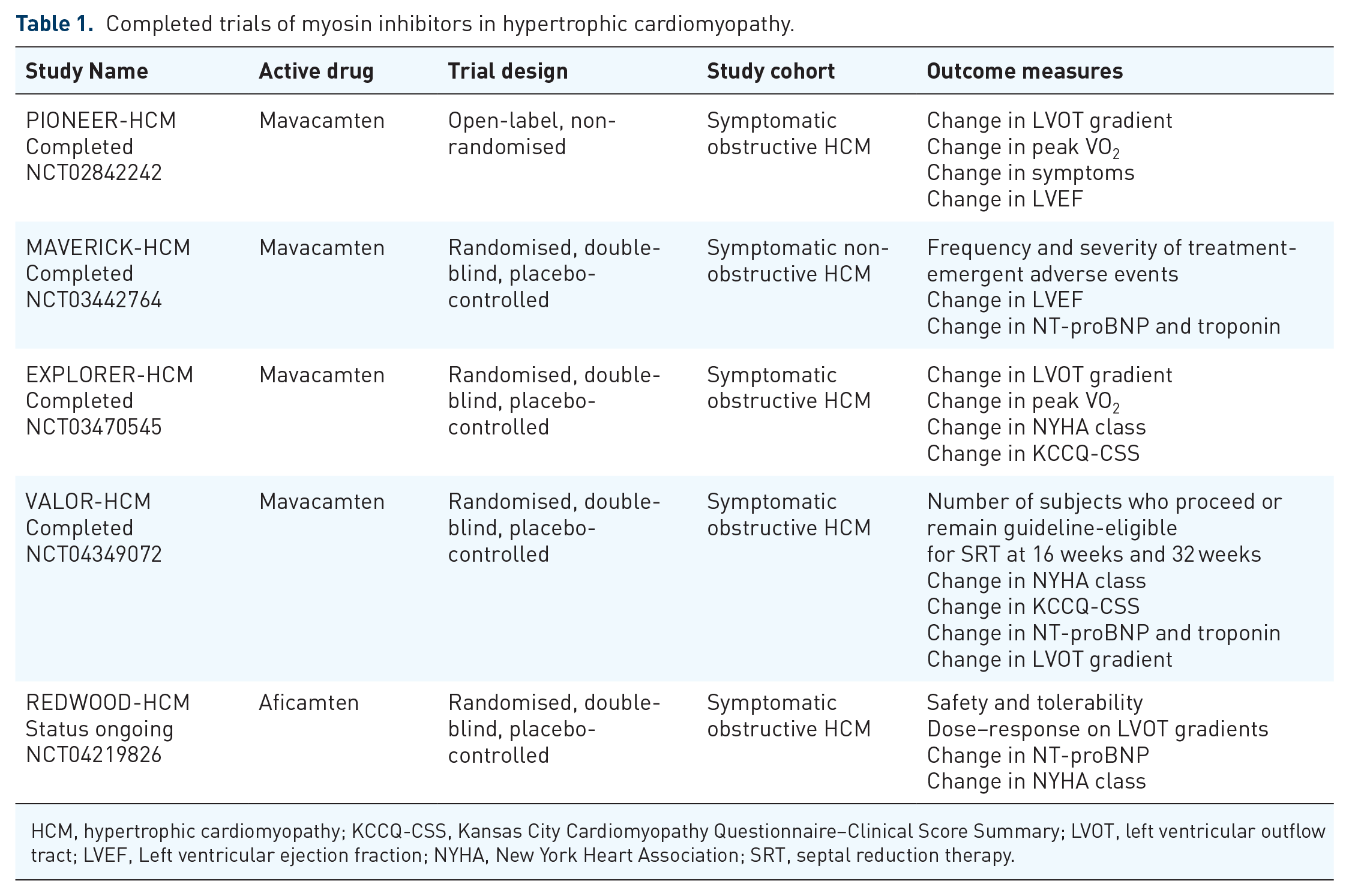
HCM, hypertrophic cardiomyopathy; KCCQ-CSS, Kansas City Cardiomyopathy Questionnaire–Clinical Score Summary; LVOT, left ventricular outflow tract; LVEF, Left ventricular ejection fraction; NYHA, New York Heart Association; SRT, septal reduction therapy.
Ongoing trials of myosin inhibitors in hypertrophic cardiomyopathy.

CTnT, cardiac troponin T; HCM, hypertrophic cardiomyopathy; KCCQ-CSS, Kansas City Cardiomyopathy Questionnaire–Clinical Score Summary; LVOT, left ventricular outflow tract; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association.
Clinical perspectives
The myosin inhibitors present the first advancement in the pharmacological management of obstructive HCM in almost 35 years, with a growing body of evidence for the safety, tolerability and efficacy of mavacamten, and to a lesser extent aficamten. This has recently led to the Food and Drug Administration (FDA) granting approval for the use of mavacamten in symptomatic obstructive HCM patients with class II/III symptoms, although at present it is only available through the Camzyos restricted Risk Evaluation and Mitigation strategy (REMS) programme (which requires prescribers and pharmacies to be enrolled in the programme and patients to enrol and comply with monitoring requirements, given the boxed warning for the risk of heart failure). In addition, in the United Kingdom, the National Institute for Health and Care Excellence (NICE) are currently appraising the clinical and cost-effectiveness of mavacamten with an expected decision next year.
While the initial enthusiasm surrounding the role of myosin inhibitors is warranted, it is important to put the results of these recent trials into clinical perspective. Almost half of the patients enrolled in EXPLORER-HCM remained in NYHA class II/III after 30 weeks of treatment. Residual LVOT obstruction (⩾30 mmHg), either at rest or with provocation, was seen in up to half of patients, with up to 25% of the study population having a residual gradient of ⩾ 50 mmHg and thereby remaining eligible for invasive SRT. Indeed, in VALOR-HCM up to 18% of patients either proceeded to or remained guideline-eligible for SRT at the end of the study period.
Data from experienced centres performing invasive SRTs in the setting of a comprehensive HCM programme consistently show excellent outcomes with regard to both improvement in symptomatic status and more durable relief of LVOT obstruction than existing pharmacological therapies, and as such will remain the gold-standard treatment of choice for definitive treatment of obstruction in high-volume interventional and surgical centres. However, the addition of mavacamten to our treatment armamentarium is to be strongly welcomed, and pharmacological therapies represent a complementary rather than a competitive approach to the management of symptomatic LVOT obstruction, allowing a stepwise escalation of treatment options on an individualised basis.
Limitations
There are some limitations to this review. Some studies included and discussed in this review were open-label, single-group assignment, non-randomised studies, although the most robust data presented and discussed pertain to the large, randomised trials. The study design of these trials required frequent monitoring for side effects and changes in left ventricular function. This intensity of follow-up and monitoring will prove problematic in routine clinical practice in many health systems, and therefore further data are needed regarding the long-term safety and real-world applicability of using myosin inhibitors in routine clinical practice.
Conclusion
LVOT obstruction is an integral component of the hypertrophic cardiomyopathy disease phenotype, playing a major role in symptomatology and progression to heart failure. Advancements in our understanding of the pathophysiology of HCM have led to the development of new therapies targeting the cardiac sarcomere. Myosin modulators and inhibitors present the first advancement in pharmacological management of obstructive HCM in almost 35 years, with a growing body of evidence for their safety, tolerability and efficacy. While further trials are required to determine the long-term safety and real-world applicability of these agents, these early results represent a promising new development in the individualised management of patients with obstructive HCM.
