Abstract
Accessory mitral valve tissue is a rare congenital cardiac anomaly that can lead to left ventricular outflow tract obstruction. We present the case of an older female patient with accessory mitral valve tissue, a subaortic membrane, an unruptured aneurysm of the aortic sinus, and left ventricular outflow tract obstruction. Successful excision of the accessory mitral valve tissue and repair of the aortic sinus were performed. Postoperative echocardiography showed complete removal of the subaortic membrane, with a residual accessory mitral valve tissue (5 mm). Color Doppler imaging revealed a significant reduction in mosaic flow signals in the left ventricular outflow tract, with the peak blood flow velocity decreasing to 1.6 m/s. The postoperative course was uneventful, and the patient was followed up with echocardiography at 3 days, 5 days, 1 month, 3 months, and 6 months after the surgery. Accessory mitral valve tissue is a rare congenital defect frequently associated with other cardiovascular congenital malformations. This report also provides a comprehensive clinical review of accessory mitral valve tissue, covering anatomical classification, associated cardiac anomalies, pathophysiology, diagnostic approaches, and treatment strategies to offer an improved clinical understanding of the condition.
Keywords
Introduction
Accessory mitral valve tissue (AMVT) is an additional valvular structure attached to the normal mitral valve apparatus within the left-sided cardiac chambers. It is a rare congenital anomaly that was first described in 1842; the first surgical intervention was reported in 1963. 1 AMVT results from incomplete separation of the mitral valve from the endocardial cushion tissue during embryonic development. 2 Typically originating from the anterior mitral leaflet, AMVT protrudes into the left ventricular outflow tract (LVOT), potentially causing LVOT obstruction. It is frequently associated with other congenital cardiovascular malformations. Here, we present the case of an elderly female patient diagnosed with AMVT, LVOT obstruction, a subaortic membrane, and an unruptured aneurysm of the aortic sinus.
Timeline
1 May 2024: The patient was referred to the Department of Emergency
1 and 16 May 2024: Transthoracic echocardiography (TTE) was performed
17 May 2024: Surgical treatment was carried out (The operation time was 09:30–13:10). Pre-operative and postoperative transesophageal echocardiography (TEE) were performed to evaluate surgical curative effects.
20 May 2024: The first TTE was performed after the operation.
22 May 2024: The second TTE was performed after the operation.
26 May 2024: The patient was discharged from our hospital.
Case presentation
A female patient in her early 60s was referred to the Emergency Department of Mianyang Central Hospital on 1 May 2024 due to exertional shortness of breath that had persisted for 1 month. Her symptoms had worsened over the past week, leading to increasing dyspnea, orthopnea, and atypical chest pain, which hindered her daily activities.
An electrocardiogram showed third-degree atrioventricular block. The patient, with an 8-year history of hypertension, had elevated blood pressure (164/69 mmHg) and a grade 3/6 systolic murmur along the left sternal border. Chest auscultation revealed normal breath sounds. No peripheral edema or jugular venous distention was observed. Laboratory tests showed leukocytosis with a white blood cell count of 11.12 × 109 cells/L (normal value: 3.5–9.5 × 109 cells/L) as well as elevated levels of procalcitonin (2.59 µg/L; normal value: 0–0.046 µg/L), C-reactive protein (28.73 mg/L; normal value: 1–10 mg/L), and N-terminal pro-B-type natriuretic peptide (1909 ng/L; normal value: <125 ng/L).
TTE revealed a parachute-like anomaly arising from the left ventricular side of the anterior mitral leaflet, protruding into the LVOT during systole. The parasternal long-axis view showed the relationship between the normal mitral valve and the abnormal structure (Figure 1). The peak blood flow velocity across the LVOT was elevated at 3.0 m/s, with a corresponding peak gradient of 36 mmHg (Figure 2). A fixed, membrane-like structure was also observed, which was attached to the basal portion of the interventricular septum. Additionally, an unruptured aneurysm of the aortic sinus was identified, measuring 16 mm × 6 mm (Figure 3). Further TTE findings included mild-to-moderate mitral regurgitation, mild aortic regurgitation, and mild dilation of the left atrium (3.9 cm) and left ventricle (end-diastolic diameter = 5.1 cm). Left ventricular systolic function was preserved, with an ejection fraction of 63%. The dimensions and morphology of the aorta were normal. Based on these finding, a diagnosis of AMVT associated with a subaortic membrane and an unruptured aneurysm of the aortic sinus was established.

The parasternal long-axis view revealing a fixed accessory mitral valve tissue attached to the anterior mitral leaflet causing left ventricular outflow tract obstruction.

Apical five-chamber view: continuous-wave Doppler examination showing a peak blood flow velocity across left ventricular outflow tract of 3.0 m/s.
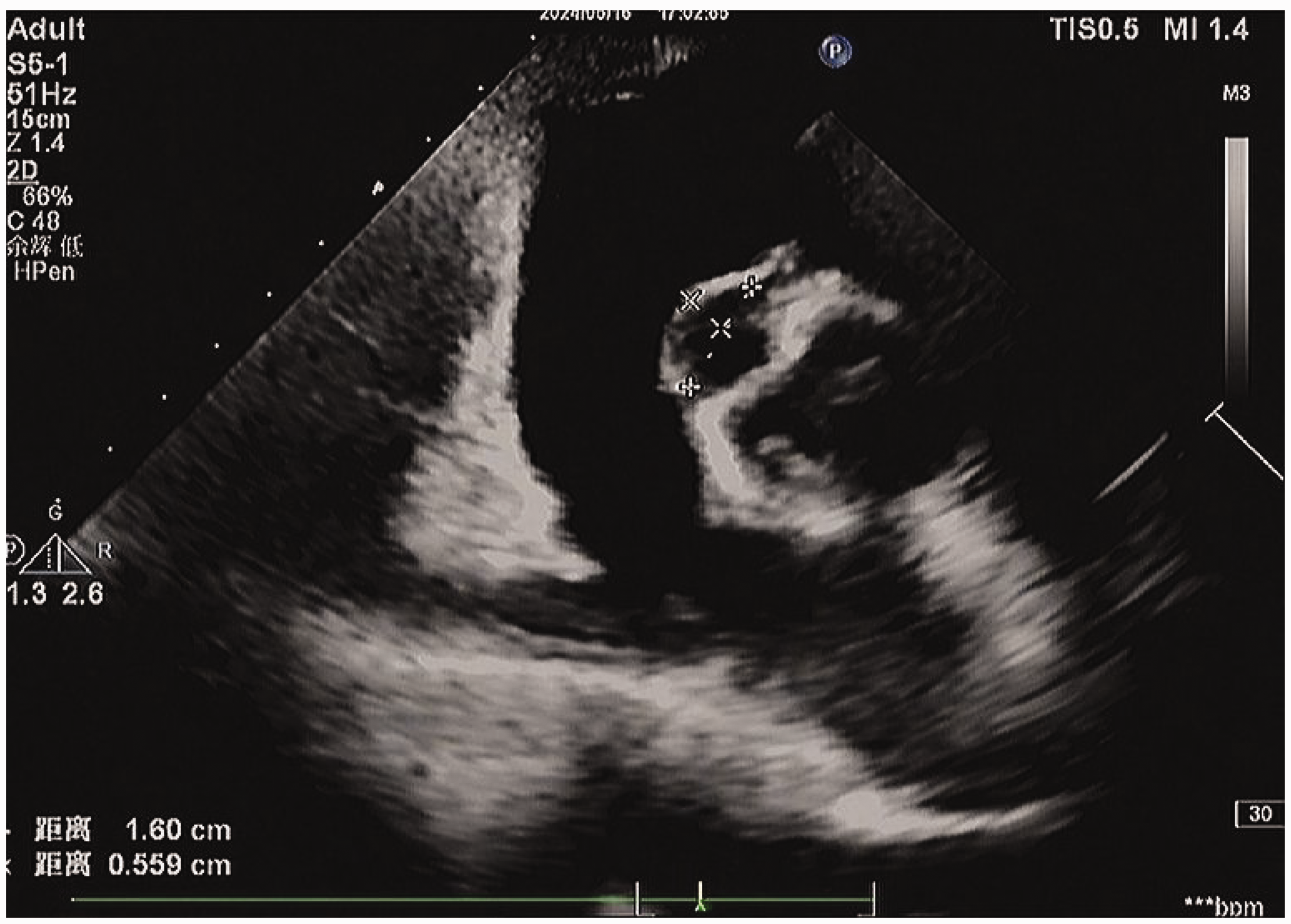
The short-axis view revealing an unruptured aneurysm of the aortic sinus measuring 16 mm × 6 mm.
Following a comprehensive clinical evaluation, surgical intervention was deemed necessary due to the presence of LVOT obstruction and the patient’s progressively worsening symptoms. The patient consented to the procedure and signed the informed consent form. Intraoperative findings were consistent with the TTE imaging diagnosis. Resection of the AMVT and subaortic membrane along with repair of the aortic sinus were successfully performed. Immediate postoperative TEE confirmed complete excision of the subaortic membrane, with a residual AMVT measuring 5 mm. Color Doppler imaging showed a marked reduction in mosaic flow signals in the LVOT, with peak blood flow velocity decreasing to 1.6 m/s. Histological examination revealed accessory valvular tissue with mucoid degeneration and inflammatory cell infiltration. The membranous structure demonstrated proliferation of fibrous connective tissue (Figures 4 and 5).

Low-magnification histological examination revealing accessory valvular tissue with mucoid degeneration and inflammatory cell infiltration.

High-magnification histological examination revealing accessory valvular tissue characterized by mucoid degeneration and inflammatory cell infiltration.
The postoperative course was uneventful. Upon discharge, the patient was prescribed a long-term regimen of furosemide (20 mg), spironolactone (20 mg), and rivaroxaban (15 mg) each to be taken orally once daily. Follow-up echocardiography was conducted at 3 days, 5 days, 1 month, 3 months, and 6 months after the surgery. At the final follow-up, the patient reported complete resolution of shortness of breath and no recurrence of chest pain. This case report adheres to the Case Report (CARE) guidelines. 3
Discussion
Epidemiological data
AMVT is a rare congenital cardiovascular malformation. Its embryological origin remains unclear; however, it is believed to be related to abnormal or incomplete separation of the mitral valve from the endocardial cushions.4,5 Although the exact prevalence of AMVT is unclear, some studies estimate it to occur in approximately 1 in 26,000 adults.6,7 AMVT is more commonly identified in males, with a reported male-to-female ratio of 1.75:1. 8 Historically, AMVT was considered a cardiac anomaly predominantly affecting children. However, increasing evidence has shown that it can also be diagnosed in adults and older adults, with reported in patients aged 6 days to 96 years.9–12 AMVT can present in various morphological forms, including membrane-like, flap-like, sheet-like, balloon-like, string-like structures, parachute-like masses, nodular gelatinous masses, structures, and pedunculated structures. These descriptive terms reflect the wide range of its anatomical appearances. 13 Histopathological examination of typically reveals mucoid degeneration and infiltration by inflammatory cells. 14
Anatomical classification
Currently, two primary classification systems are used to categorize AMVT. Prifti et al. classified AMVT based on morphological characteristics into fixed type (Type I) and mobile type (Type II) forms. Type I is further divided into nodular (Type IA) and membranous (Type IB) subtypes. Type II is subdivided into pedunculated (Type IIA) and membrane-like (Type IIB) forms. Type IIB can be further classified based on chordal development as follows: rudimentary chordae tendineae (Type IIB1) and well-developed chordae tendineae (Type IIB2). 15 An alternative classification proposed by Yetkin et al. is based on the anatomical relationship of the AMVT to the mitral valve. 13 In this system, Type I refers to tissue located above the mitral valve, Type II to the tissue on the mitral valve, and Type III to the tissue below the mitral valve.
Cardiac malformations and pathophysiology
AMVT is frequently associated with various congenital cardiovascular anomalies, including ventricular septal defect (VSD), subaortic membrane, atrial septal defect, patent ductus arteriosus, mitral cleft, transposition of the great arteries, aortic coarctation, Ebstein’s anomaly, coronary artery malformations, and double-outlet right ventricle.7,11,13 Li et al. reported a case of AMVT coexisting with a parachute-like mitral valve, VSD, bicuspid aortic valve, and aortic sinus aneurysm. 16
From a pathophysiological standpoint, AMVT is recognized as a potential cause of LVOT obstruction. The following two primary mechanisms contribute to this obstruction: (1) the mass effect of the accessory tissue itself and (2) progressive fibrous tissue deposition caused by turbulent blood flow induced by the AMVT.17–19 Notably, AMVT may cause latent LVOT obstruction, making stress echocardiography essential in patients presenting with exertional dyspnea or exercise-induced syncope. 14 When AMVT coexists with a VSD, the peak LVOT velocity and pressure gradients may be underestimated. This underestimation arises from the following two factors: (a) part of the AMVT may protrude into the VSD during systole, reducing the degree of LVOT obstruction; (b) the VSD may serve as an alternative outflow tract, resulting in lower left ventricular pressure. 20
Mitral valve function is usually preserved in AMVT; however, mild mitral regurgitation can occur. In Yetkin’s classification, Type I AMVT patients often have distorted or tethered mitral valves from the accessory tissue, leading to significant mitral regurgitation, atrial enlargement, and progression to atrial fibrillation. The latter further worsens mitral regurgitation and leaflet coaptation, ultimately leading to heart failure.13,21,22 In addition to mitral valve involvement, aortic regurgitation is not uncommon in AMVT. The aortic valve may be affected either by the Venturi effect associated with LVOT obstruction or by direct attachment of AMVT to the subaortic region of the interventricular septum or aortic cusps. 23 Finally, AMVT is considered a potential risk factor for cardiac embolism due to its mobility, fragility, and the presence of mucoid degeneration.8,24,25
Diagnosis
The anatomical attachment sites of AMVT, its involvement in the LVOT, and associated cardiac malformations determine the clinical presentation and symptoms. Approximately one-third of patients with AMVT are asymptomatic, while others—particularly those with LVOT obstruction—may present with exertional dyspnea, fatigue, and dizziness.12,26,27 When AMVT is associated with significant valvular regurgitation, patients may develop atrial fibrillation, congestive heart failure, or malignant arrhythmias.28,29 AMVT is also a potential source of cardiac embolism; therefore, some individuals may experience thromboembolic events such as transient ischemic attacks30,31 or retinal artery occlusion.25,32
Two-dimensional TTE is the gold standard for diagnosing AMVT. It evaluates AMVT morphology and identifies various associated cardiac anomalies, such as left ventricular hypertrophy, LVOT obstruction, valvular regurgitation, and congenital malformations. Color and continuous-wave Doppler echocardiography further aid in assessing blood flow velocity across the LVOT. TEE provides a more detailed visualization of the morphology and attachment sites of AMVT. Intraoperative TEE is particularly valuable for assessing the spatial relationships among AMVT, the mitral valve, and the LVOT and for guiding complete resection while preserving mitral valve integrity. Three-dimensional echocardiography offers comprehensive imaging of the mitral valve and the surrounding structures. It is useful for determining anatomical orientation and informing surgical approaches aimed at preserving mitral valve anatomy.33,34
Treatment
Surgical intervention is not indicated in all cases of AMVT. Treatment decisions should be based on a comprehensive clinical evaluation, considering the patient’s symptoms, presentation, and the presence of associated cardiac malformations. Currently, cardiac surgery is recommended only for patients with a significant LVOT gradient—defined as a peak gradient of ≥50 mmHg or a mean gradient of ≥25 mmHg—or for those undergoing correction of other congenital anomalies or evaluation of an intracardiac mass. 17 Surgical management involves complete resection of the AMVT to relieve LVOT obstruction while carefully avoiding injury to surrounding structures, such as the mitral valve, mitral curtain, aortic valve, and the basal portion of the interventricular septum. In asymptomatic patients without associated cardiac anomalies, conservative management with routine follow-up is preferred. However, due to the risk of thromboembolic complications, antiplatelet therapy is recommended even in asymptomatic and uncomplicated cases. Additionally, lifestyle modifications, such as avoiding cigarette smoking and discontinuing oral contraceptive use, are advised to minimize the risk of cardioembolic events, even in patients with isolated, asymptomatic AMVT.35,36
Conclusion
AMVT presents with a range of symptoms; patients may be asymptomatic or have severe complications such as thromboembolic events, heart failure, LVOT obstruction, and dangerous arrhythmias. It is often linked to other congenital cardiovascular anomalies. Echocardiography is crucial for diagnosing and monitoring AMVT, providing detailed insights into the lesion’s morphology, location, and relationship with surrounding structures as well as assessing LVOT hemodynamics and identifying coexisting malformations.
Footnotes
Acknowledgements
We would like to thank Lin Shi for his technical support and writing suggestions.
Author contributions
J.X. conceptualized and designed the study as well as drafted the initial manuscript. N.Z. and M.Z. were responsible for data collection and literature review. DN.G. and YF.W. critically reviewed and revised the manuscript and approved the final version for submission. All authors have read and approved the final manuscript. All authors have agreed to be accountable for all aspects of the work.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Ethical statement
The authors have de-identified all patient details in the Case report section, and this report has been granted approval for publication by the review committee.
Funding
The authors received no financial support for the research, authorship, or publication of this article.
