Abstract
Purpose:
Takayasu’s arteritis (TAK) is a large-vessel vasculitis. Glucocorticoids (GCs) combined with cyclophosphamide (CTX) is a common treatment option, but their efficacy is limited. The aim of this retrospective study was to study the efficacy and safety of tocilizumab (TCZ) in the treatment of TAK.
Patients and methods:
A total of 63 patients with TAK were divided into two groups according to different treatment strategies: 31 patients in the TCZ group and 32 patients in the CTX group. After 6 months of treatment, disease activity and side effects were evaluated and were compared between the two groups.
Results:
After 3 months of treatment, the National Institutes of Health (NIH) score (p = 0.005), Indian Takayasu Clinical Activity Score (ITAS) 2010 (p = 0.043), and the Indian Takayasu Activity Score with the Acute-Phase Response (ITAS.A; p = 0.036) were lower in patients treated with TCZ compared with those treated with CTX. In the TCZ group, the proportion of patients with NIH scores ⩽1 reached 50% after 1 month, 90% after 3 months, and 96% after 6 months of treatment, whereas in the CTX group, these proportion were 36%, 30%, and 78%, respectively. Thus, TCZ treatment of TAK is accompanied with a faster remission rate than CTX treatment. Moreover, the total cumulative incidence of adverse events was lower in the TCZ group than in the CTX group (p = 0.035).
Conclusion:
TCZ was faster and safer in alleviating TAK.
Introduction
Takayasu’s arteritis (TAK) is a chronic granulomatous systemic vasculitis and primarily involves large vessels.1,2 The disease attacks the aorta and its main branches, resulting in stenosis, occlusion, and aneurysm formation.1,2 TAK requires long-term immunosuppressive therapy. Traditionally, glucocorticoids (GCs; with the same dose as prednisone) play an important role in the treatment of TAK. 3 GC monotherapy, however, has difficulty in effectively controlling the activity and progression of the disease, and the side effects are also considerable. 4 According to European League Against Rheumatism (EULAR) recommendations, 4 the combination of immunosuppressive agents is particularly important in disease control and GC dose reduction. A questionnaire survey of Chinese experts showed that the combination of GC and cyclophosphamide (CTX) was the most common regimen (63–78%) for inducing remission in TAK patients. 5 Nevertheless, given the fact that 38% of Chinese and 44% of Japanese TAK patients are women in their 20s and 30s, 6 CTX cannot be considered as an ideal choice for the treatment of TAK, due to its potential risk of amenorrhea and infertility. 7
Recently, a number of studies have shown that the serum interleukin-6 (IL-6) level in patients with TAK is significantly higher compared with healthy controls8,9 and is positively correlated with disease activity.8,9 Moreover, immunohistochemistry has confirmed high expression of IL-6 in the vascular wall of patients with TAK. 10 Therefore, IL-6 is a key factor involved in the immune system and inflammatory response in TAK. Tocilizumab (TCZ) is a recombinant humanized monoclonal antibody that binds to the IL-6 receptor and blocks its biological effects, inhibiting the activity and progression of TAK. 11 According to the EULAR recommendations, 4 TCZ can be considered in cases of relapsing or refractory TAK.
Nevertheless, the numbers of TAK patients who received TCZ in the only randomized clinical trial 12 (and its follow-up extension), 13 retrospective studies,14–18 and prospective studies19–22 were relatively small, except for some very recent studies,23–25 including two retrospective ones23,24 and one prospective study. 25 Furthermore, the results of the small randomized clinical trial by Nakaoka et al. 12 and its extension study 13 only showed a statistically borderline significance (p = 0.0596) for a reduced hazard ratio (HR) for the time to the first relapse and demonstrated nonsignificant improvements in a number of secondary end points in patients treated with TCZ. For these two reasons, further support is required for demonstrating the efficiency of TCZ in TAK treatment. The aim of this study was to evaluate the efficacy and side effects of TCZ compared with CTX in the treatment of TAK.
Subjects and methods
Ethics
Informed consent was obtained in the written form from all participants and their legal guardians. This retrospective study was conducted in accordance with the ethical principles of the Declaration of Helsinki and was approved by the Ethics Committee of Beijing Anzhen Hospital, Capital Medical University (approval no. 2021041X).
Patients
This retrospective study enrolled a total of 63 patients diagnosed with TAK who were admitted to Beijing Anzhen Hospital affiliated with Capital Medical University from January 2015 to December 2019. The patients were divided into two groups according to their treatment plans. The inclusion criterion was the diagnosis of TAK in accordance with the American College of Rheumatology 1990 criteria for TAK classification. 26 The exclusion criteria were as follows: (1) TAK-related treatment in the last 6 months; (2) TAK combined with other rheumatic diseases; (3) neutrophil count <1.0 × 109/l or platelet (PLT) count <100 × 109/l; (4) alanine aminotransferase (ALT) >2 times the upper normal limit or creatinine (Cr) >1.5 times the upper normal limit; (5) history of primary or secondary immunodeficiency or malignant disease; (6) TAK combined with infection, tumor, or trauma. The patients selected for inclusion in the study were divided into two groups according to the treatment received. Thirty-one patients were treated with TCZ (TCZ group), and 32 patients were treated with CTX (CTX group). In TCZ group, 14 patients had new diagnosis, but they refused CTX due to fertility requirements, so their treatment was conducted with TCZ. Seventeen patients were relapsing or refractory. Thus, in order to improve the status of the disease control, treatment with TCZ was chosen. In CTX group, there were 22 newly diagnosed patients. Ten patients were relapsing: among them, eight patients had never used CTX before; two patients who used to receive oral CTX were treated intravenously (for information about the previous treatment, see Table 1).
Comparison of basic conditions between the TCZ and CTX groups before treatment.
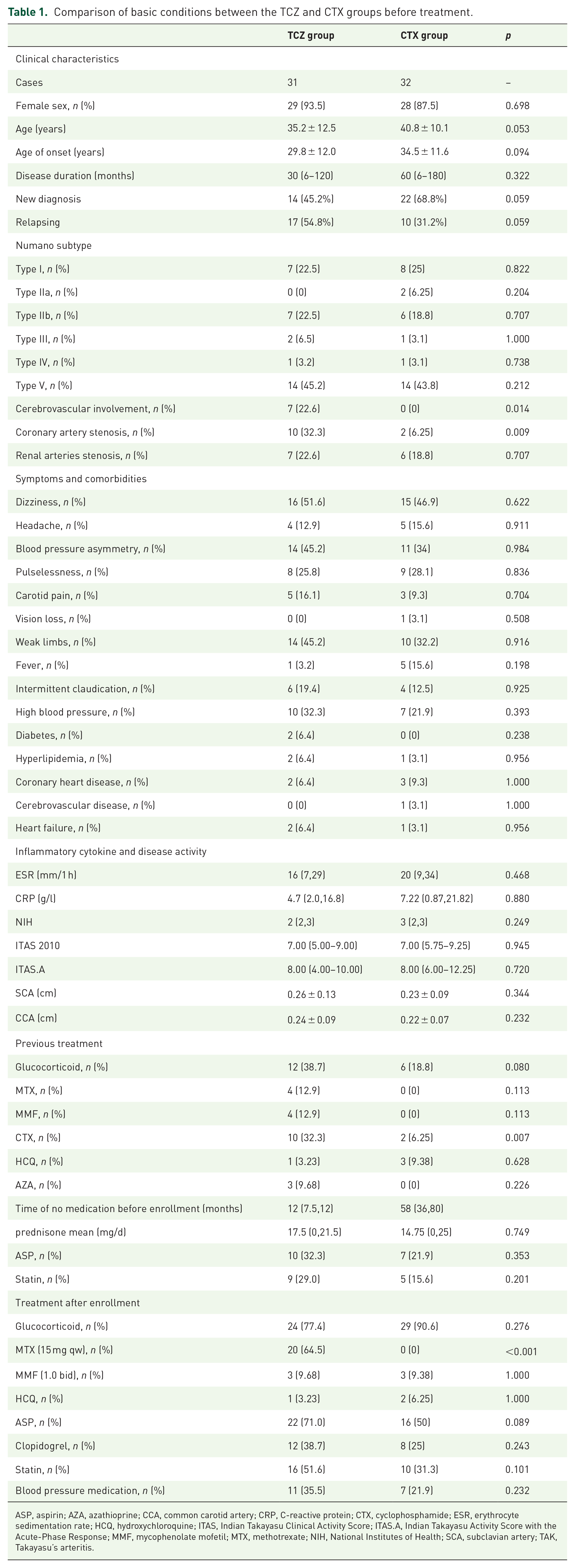
ASP, aspirin; AZA, azathioprine; CCA, common carotid artery; CRP, C-reactive protein; CTX, cyclophosphamide; ESR, erythrocyte sedimentation rate; HCQ, hydroxychloroquine; ITAS, Indian Takayasu Clinical Activity Score; ITAS.A, Indian Takayasu Activity Score with the Acute-Phase Response; MMF, mycophenolate mofetil; MTX, methotrexate; NIH, National Institutes of Health; SCA, subclavian artery; TAK, Takayasu’s arteritis.
The TCZ group was treated with TCZ 8 mg kg−1 intravenously every 4 weeks. Among these patients, treatment of 20 patients was combined with oral administration of methotrexate (MTX) 15 mg once a week. Another three patients were treated with mycophenolate mofetil (MMF) 1.0 g administered twice daily and one patient received hydroxychloroquine (HCQ) 0.2 g administered twice daily. Twenty-two patients were treated with aspirin 0.1 g once a day. Twelve patients were treated with clopidogrel 0.75 g once a day. Sixteen patients were treated with statin 20 mg once a day. For 24 patients, treatment was combined with oral prednisone. In the CTX group, 27 patients were treated with CTX 1.0 g administered intravenously once every 4 weeks. Another five patients were administered oral CTX 0.1 g once every other day. Among these, there was one patient who received CTX intravenously for 3 months who was switched to oral CTX for another 3 months. Among these patients, treatment of three patients was combined with oral MMF 1.0 g administered twice daily and two patients received HCQ 0.2 g administered twice daily. Sixteen patients were treated with aspirin 0.1 g once a day. Eight patients were treated with clopidogrel 0.75 g once a day. Ten patients were treated with statin 20 mg once a day. Twenty-nine patients had their treatment combined with oral prednisone (please see Table 1 for further information).
Collection of clinical data
The demographic data of the two groups before the treatments were collected. The activity of TAK was scored according to the National Institutes of Health (NIH) criteria, 2 the Indian Takayasu Clinical Activity Score 2010 (ITAS 2010) and the Indian Takayasu Activity Score with the Acute-Phase Response (ITAS.A). 27 The baseline data before treatment and the clinical manifestations, experimental indicators, NIH score, ITAS 2010, and ITAS.A were recorded. The scores after 6 months of treatment were also recorded. According to the level of vascular involvement, TAK was classified by the Numano classification standard as I, IIa, IIb, III, IV, and V. 28 GC doses and infection incidents were also observed.
Laboratory parameters
Laboratory data, including indicators of liver and kidney function, blood lipid level, acid uric, routine blood test results, the erythrocyte sedimentation rate (ESR), the C-reactive protein (CRP), and IL-6 levels were obtained from laboratory examination reports at the participating hospital.
Imaging
For evaluating the disease activity score after 6 months of treatment, before- and after-treatment thoracic aortic MRA, abdominal aortic MRA, or aortic CTA, together with peripheral arterial ultrasound, were conducted for all the patients. We measured the mural thickness of the common carotid artery (CCA) and subclavian artery (SCA) for assessing reactive vascular changes.
Peripheral vascular Doppler examinations were performed by the same vascular Doppler specialist who always blinded to the treatment. CTA and MRA were interpreted in consensus by two radiologists. The mural thickness at the thickest part of the vessel, as well as the narrowest diameters of the lumen of the SCA and CCA, was measured by the Doppler ultrasonography at baseline and after 6 months.
Follow-up and remission
Because the administration of TCZ and CTX was intravenous, the patients were required to be hospitalized every month, in which data regarding their symptoms, laboratory parameters, inflammatory factors, GC dose and imaging changes were collected. After 6 months of treatment, imaging was performed to assess vascular changes. The NIH score, ITAS 2010, and ITAS.A were also recorded after 1, 3, and 6 months of treatment. An NIH score ⩽1 was defined as disease remission.
Statistical analysis
For a significance level of 0.05, using a two-sided two-sample t test, group sample sizes of 31 (TCZ group) and 32 (CTX group) achieve 99% power in detecting the difference in ESR between TCZ group 59.3 ± 37.3 and CTX group 23.5 ± 29.8. The expected mean values and standard deviations of changes in ESR were taken from the study of Kong et al. 19 Statistical analyses were performed using SPSS statistical software, version 23.0 (Chicago, IL, USA). Continuous data were expressed as the mean value ± standard deviation (mean ± SD), and the independent-samples t test was used to compare differences in data with a normal distribution between the two groups. The paired-samples t test was used to compare differences in data with a normal distribution before and after treatment. The median and interquartile ranges were used to describe skewed continuous variables, and the rank-sum test was used to compare differences in these variables between the two groups. The paired-samples rank-sum test was also used to compare differences before and after treatment. The cumulative incidence of adverse events (AEs) was calculated by the Kaplan–Meier method. Categorical variables were analyzed using the Chi-square test. p < 0.05 was considered statistically significant.
Results
No statistically significant differences in baseline patient data
A comparison was carried out between the baseline conditions of the patients in the TCZ group and the CTX group before treatment. There were 29 women and 2 men, aged 18–58 years, in the TCZ group. The course of disease was 1–300 months, with a median course of 30 months. In the CTX group, there were 28 women and 4 men, aged 24–57 years. The course of disease was 1–240 months, with a median course of 60 months.
Age, sex, disease duration, Numano type, symptoms, liver and kidney function, uric acid, blood sugar, blood lipid, ESR, CRP, IL-6 levels, and other indicators were compared between the two groups. There were no statistically significant differences in the baseline conditions, clinical manifestations, Numano subtype, symptoms, comorbidities, laboratory parameters, or disease activity scores between the two groups before treatment (p > 0.05; Tables 1 and 2).
Comparison of inflammatory markers in the two groups before and after treatment.

ALT, alanine aminotransferase; Cr, creatinine; GLU, glucose; HDL-C, high-density lipoprotein; HGB, hemoglobin; LDL-C, low-density lipoprotein; IL-6, interleukin-6; NE, neutrophil count; PLT, platelet count; TG, triacylglycerol; THO, cholesterol; WBC, white blood cell.
Patients were treated with combined oral administration of twice-daily MMF 1.0 g, twice-daily HCQ 0.2 g, and prednisone. The number of patients treated with the combined oral administration of once-a-week MTX 15 mg was higher in the TCZ group than those in the CTX group. There was no other difference in the medication proportion between the two groups (Tables 1 and 2).
TAK patients with TCZ treatment demonstrated lower disease activity indices as compared with those with CTX treatment
After treatment, the PLT counts in the two groups were lower (p < 0.05) than those before treatment, but the values were still within the normal ranges (Table 2). In the TCZ group, the level of low-density lipoprotein (LDL-C) was also lower than that before treatment (2.09 ± 0.72 mmol/l versus 2.53 ± 0.87 mmol/l; p = 0.044), in which both values were within the normal range (Table 2). In the CTX group, the white blood cell (WBC) count was lower than that before treatment (6.56 ± 1.89 × 109/l versus 7.78 ± 2.32 × 109/l; p = 0.025). The same holds true for lymphocyte count (1.54 ± 0.45 × 109/l versus 2.13 ± 0.98 × 109/l, p = 0.003).
After 6 months of treatment, the ESR [TCZ group, 2 (2,7) mm/1 h versus 16 (7,29) mm/1 h, p = 0.000; CTX group, 9 (6.25,16) mm/1 h versus 20 (9,34) mm/1 h, p = 0.004] in both groups was lower than that before treatment. After 1 month of treatment, the ESR [TCZ group, 5 (2,10) mm/1 h versus 16 (7,29) mm/1 h, p = 0.000; CTX group, 9.5 (5,14.5) mm/1 h versus 20 (9,34) mm/1 h, p = 0.005] in the two groups was lower than that before treatment, but there was no difference between the two groups (p = 0.13). After 3 and 6 months of treatment, the decreases in ESR in the TCZ group were significantly larger than those in the CTX group [3 months: 3 (2,6.5) mm/1 h versus 8 (6,14) mm/1 h, p = 0.000; 6 months: 2 (2,7) mm/1 h versus 9 (6.25,16) mm/1 h, p = 0.000] (Figure 1(a)).

Comparison of the disease alleviation between the TCZ and CTX groups after the treatments. (a) After treatment, the decrease in ESR in the TCZ group was larger than that in the CTX group (p = 0.000). (b) After treatment, the decrease in CRP level in the TCZ group was larger than that in the CTX group (p = 0.000). (c) After 3 months of treatment, the NIH score in the TCZ group was lower than that in the CTX group (p = 0.005). (d) After 3 months of treatment, the ITAS 2010 in the TCZ group was lower than that in the CTX group (p = 0.043). (e) After 3 months of treatment, the ITAS in the TCZ group was lower than that in the CTX group (p = 0.036). (f) The prednisone dose in the TCZ group was substantially smaller than that in the CTX group (p = 0.001). (g) The administration of GCs in both groups decreased after 6 months of treatment. The cumulative glucocorticoid dose was smaller in the TCZ group compared with the CTX group (1825 ± 1586 mg versus 4531 ± 1558 mg, p < 0.001).
After 6 months of treatment, the CRP level in the two groups was lower than that before treatment [TCZ group: 0.13 (0.05,1.01) mg/l versus 4.7 (2.0,16.8) mg/l, respectively; p = 0.000; CTX group: 1.25 (0.61,3.27) mg/l versus 7.22 (0.87,21.82) mg/l, respectively; p = 0.006]. In addition, the decrease in CRP level in the TCZ group was significantly larger than that in the CTX group after 1, 3, and 6 months of treatment [1 month: 2.6 (0.81,8.02) mg/l versus 0.51 (0.17,1.68) mg/l, p = 0.006; 3 months: 0.94 (0.40,3.92) mg/l versus 0.07 (0.45,0.84) mg/l, p = 0.000; 6 months: 1.25 (0.61,3.27) mg/l versus 0.13 (0.05,1.01) mg/l, p = 0.000] (Figure 1(b)).
After 6 months of treatment, there were reductions in the following scores in both TCZ and CTX groups: the NIH score [TCZ group: 0.00 (0.00,1.00) versus 2.00 (2.00,3.00), p = 0.000; CTX group: 0.00 (0.00,1.25) versus 3.00 (2.00,3.00), p = 0.000], ITAS 2010 [TCZ group: 1.00 (0.00,2.25) versus 7.00 (5.00,9.00), p = 0.000; CTX group: 0.00 (0.00,1.00) versus 7.00 (5.75,9.25), p = 0.000], and ITAS.A [TCZ group: 0.00 (0.00,2.00) versus 8.00 (4.00,10.00), p = 0.000; CTX group: 1.00 (0.00,3.50) versus 8.00 (6.00,12.25), p = 0.000]. After 1 month of treatment, there were no significant differences in the above-mentioned three scores between the two groups. After 3 months of treatment, the three scores were lower in the TCZ group compared with the CTX group [NIH score: 0.00 (0.00,1.00) versus 2.00 (1.00,2.00), respectively; p = 0.005; ITAS 2010: 0.00 (0.00,2.00) versus 3.00 (1.00,6.00), respectively; p = 0.043; ITAS.A: 0.00 (0.00,2.00) versus 3.00 (1.00,6.00), respectively; p = 0.036]. In the TCZ group, the proportion of patients with NIH scores ⩽1 reached 50% after 1 month, 90% after 3 months, and 96% after 6 months of treatment. In the CTX group, this proportion reached 36% after 1 month, 30% after 3 months, and 78% after 6 months of treatment. Thus, TCZ treatment of TAK is accompanied with a faster remission rate than CTX treatment (Figure 1(c)–(e)).
Regarding the comparison of the dose of GCs converted to prednisone before and after treatment, we should point out that the dose after 6 months treatment was smaller compared with the start of the treatment (4.8 ± 3.6 mg/d versus 22.8 ± 18.6 mg/d, p < 0.001). In the CTX group, the overall dose after 6 months of treatment was smaller compared with the start of the treatment (12.6 ± 4.6 mg/d versus 40.4 ± 17.2 mg/d, p < 0.001). The administration of GCs in both groups decreased after 6 months of treatment. The cumulative GC dose was smaller in the TCZ group compared with the CTX group (1825 ± 1586 mg versus 4531 ± 1558 mg, p < 0.001) (Figure 1(f) and (g)).
After treatment, the mural thickness of the SCA was decreased more significantly in the TCZ group
The images of thoracic aortic MRA, abdominal aortic MRA, or aortic CTA, together with peripheral arterial ultrasound, were compared before and after 6 months of treatment. No new vascular lesions and no progression of primary vessels were identified in both groups. We measured the mural thickness of the SCA and CCA for assessing the reactive vascular changes.
After 6 months of TCZ treatment, the mural thickness of the SCA was smaller than that before treatment (0.16 ± 0.04 cm versus 0.26 ± 0.13 cm; p = 0.000). The mural thickness of the SCA in the CTX group did not change significantly after treatment (0.22 ± 0.09 cm versus 0.23 ± 0.09 cm; p = 0.289). After 6 months of treatment, the mural thickness of the SCA in the TCZ group was smaller than that in the CTX group (0.14 ± 0.04 cm versus 0.22 ± 0.09 cm; p = 0.034) (Figure 2(a)).

Comparison of the changes in the intima-media thickness of the SCA and CCA before and after treatment in the TCZ and CTX groups. (a) The SCA thickness was smaller after treatment compared with before treatment (p = 0.000). (b) The CCA thickness in the TCZ group was smaller after treatment compared with before treatment (p = 0.005).
We should point out that while in the TCZ group, CCA mural thickness decreased after treatment (0.19 ± 0.06 cm versus 0.24 ± 0.09 cm; p = 0.005); no significant change in CCA mural thickness was identified in the CTX group (0.22 ± 0.06 cm versus 0.22 ± 0.07 cm; p = 0.368) (Figure 2(b)).
Remission rate in the TCZ group was higher
Remission was achieved in 29 patients of the TCZ group (remission rate = 93.5%) and 19 patients of the CTX group (remission rate = 59.3%). After 6 months of treatment, the remission rate in the TCZ group was significantly higher than that in the CTX group (p = 0.001).
Cumulative incidence of AEs was lower in the TCZ group
Side effects included neutrophil count <1.0 × 109/l, WBC count <4 × 109/l, ALT >2 times the upper normal limit or aspartate aminotransferase >2 times the upper normal limit, and infection. In the TCZ group, five patients (16.12%) had drug-related side effects, including one case of tuberculosis (TB), one case of varicella-zoster virus (VZV) infection, two cases of decreased WBC or neutrophil count, and one case of abnormal liver function. In the CTX group, twelve patients (37.5%) had drug-related side effects, including one case of lung infection. The WBC or neutrophil count decreased in six cases, and liver function was abnormal in five cases. There was a case of VZV infection and TB in the TCZ group. There was a case of lung infection in the CTX group. There were no deaths in two groups. No difference was noted when a comparison was made between the rate of serious AEs between the two groups (p = 0.056). The total cumulative incidence of AEs was lower in the TCZ group than in the CTX group (p = 0.035; Figure 3).

Cumulative incidence of side effects. The follow-up included at least one report once a month. The total cumulative incidence of AEs was lower in the TCZ group compared with that in the CTX group (p = 0.035).
Discussion
To the best of our knowledge, this is the first relatively large sample-size study to retrospectively compare the efficacy and safety of TCZ to CTX in the treatment of patients with TAK. In this study, TCZ treatment for 6 months showed a faster and safer effect by inducing a larger decrease in disease activity scores and better remission rates compared with the CTX treatment. Also, compared with CTX, TCZ was found to have a better effect on improving the intima-media thickness of the SCA. The reduced dose of prednisone achieved after TCZ therapy was smaller, which may also contribute to the less side effects observed in the TCZ group.
It has been found that the production of IL-6 increased in TAK,9,29 and IL-6 has been recognized as an important molecule in the pathophysiology of continuous inflammation in TAK. 30 The first successful treatment of a TAK patient by TCZ was reported by Nishimoto et al. 31 Since then, one randomized clinical trial12,13 and several observational studies16,18,20,24,25 and case reports32,33 have demonstrated that TCZ is effective in treating patients with refractory TAK. As a retrospective real-world study that included nonrefractory TAK patients, this study demonstrated that disease activity was improved in both TCZ and CTX groups after treatment. It is of note that after 3 months of treatment, the ITAS 2010 score, ITAS.A score, and NIH score were lower in the TCZ group compared with the CTX group. A higher reduction in ITAS 2010 score (which includes symptoms and arterial lesions, but not ESR and CRP) in TCZ group after 3 months of treatment further revealed that the extent of the alleviation of disease activity was higher in TCZ group compared with the CTX group. Also, in the TCZ group, the proportion of patients in remission reached 90% after 3 months of treatment and 96% after 6 months of treatment, which are higher than the corresponding proportions in the CTX group (30% at 3 months and 78% at 6 months). Thus, a higher disease remission rate after 3 months of treatment in the TCZ group suggested that TCZ rapidly reduced the status of the disease activity, and the extent of disease alleviation was better in TCZ group compared with the CTX group. In this context, Ferfar et al. 34 reviewed previous studies and found that TCZ treatment caused a reduction in disease activity in patients with TAK, of whom 87.5% experienced disease remission. Mekinian et al. 20 studied 13 patients with TAK and found a decrease in the median NIH score, ITAS 2010, and ITAS.A after 6 months of TCZ therapy.
After 6 months of treatment, the dose of GCs in both TCZ and CTX groups was substantially lower than that at the beginning of the treatment. In the TCZ group, the number of refractory and relapsed patients was higher. Furthermore, the number of patients of cardiovascular, cerebrovascular, and renal vascular involvements was higher. This study, however, showed that the patients in the TCZ group had a better treatment with less GCs compared with those in the CTX group. It is of note that the dose of GCs in the TCZ group was lower, suggesting that TCZ combined with a small dose of GCs was more effective than CTX combined with a larger dose of GCs.
In line with our results, Ferfar et al. 34 found that 79.1% of patients with TAK experienced a dose reduction or even discontinuation of prednisone after taking TCZ. Gon et al. 35 enrolled 12 TAK patients who were treated with infliximab or TCZ. The prednisolone dose decreased 12 months after the initiation of the biologics. In the study of Nakaoka et al., 36 the TAK patients resistant to GCs were treated with TCZ every 4 weeks for a total of 24 times. All of the patients showed good clinical responses during TCZ treatment. In one prospective study 21 that evaluated the efficacy and safety of TCZ monotherapy in three TAK and eight giant cell arteritis patients, no relapse was observed after TCZ discontinuation. In another study, 13 which was the extension of the randomized double-blind placebo-controlled clinical trial of Kong et al., 12 the results of a long-term (>2 years) efficacy and safety study of TCZ therapy in patients with TAK were reported, in which a clear steroid-sparing effect was observed with TCZ treatment in comparison with the GC dose used before the study entry.
TCZ not only inhibits disease activity but also improves mural thickness of SCA. After 6 months of treatment, the mural thickness of the SCA and the mural thickness of the CCA in the TCZ group were lower than those before treatment and the mural thickness of the SCA was lower than that in the CTX group. In the CTX group, there was no remarkable change in the mural thickness of the SCA or CCA after treatment. Although the improvement of the mural thickness of the SCA did not reflect a change in all of the arteries, at least TCZ was found to have a better effect on improving SCA lesions than CTX. Regenerative or anti-inflammatory activities of IL-6 are mediated by classic signaling, whereas pro-inflammatory activities of IL-6 are mediated by trans-signaling. 37 IL-6 inhibition with TCZ, which is a neutralizing antibody against IL-6 and IL-6R, blocks both classic and trans-signaling pathways. 38 As a result, IL-6 inhibition reduces the inflammatory response, and alleviates inflammation of the arterial wall. Consistent with this conclusion, Zhou et al. 22 reported that the mural thickness of both the CCA and SCA decreased after TCZ treatment. A study 39 comparing the involvement of coronary arteries in two groups of TAK patients, one treated with TCZ and another with conventional traditional disease-modifying anti-rheumatic drugs (DMARDs) including CTX, demonstrated that after 6 months of TCZ treatment, the total number of coronary artery lesions was reduced and vascular wall thickening was improved.
TCZ treatment yielded less adverse reactions overall. The cumulative incidence of AEs related to drugs was 16.12% in the TCZ group, which was lower than that in the CTX group (37.5%). TCZ was superior to CTX in terms of the safety, also. The WBC and lymphocyte counts decreased after CTX treatment, but there was no apparent change in the lymphocyte counts in the TCZ group, suggesting that TCZ had less effect on lymphocytes compared with CTX. There was no difference in incidence of serious AEs between the two groups, which may be related to the small number of samples. There was a case of VZV infection and TB in the TCZ group. In this context, it should be pointed out that in the results of the randomized clinical trial 12 only one patient was reported to experience serious AEs from TCZ. Ferfar et al. 34 reviewed the cases of 38 patients treated with TCZ. They found that there were two cases of liver damage, two cases of infection, and four cases of leukopenia, with an incidence of 21%, which is consistent with the incidence in this study. In summary, TCZ has shown good efficacy in treating TAK, but further confirmation by multicenter and large sample-size studies is needed.
This study has several limitations, such as its retrospective design and single-center of included patients. Despite these limitations, this trial is the first to show that a high remission rate can be achieved by TCZ treatment after only 3 months. Prospective studies with larger sample sizes and prospective designs to assess the efficacy and safety of TCZ in the treatment of TAK are still required.
Conclusion
In conclusion, this study demonstrated that TCZ might be a more effective alternative than CTX in the treatment of TAK by reducing the need for higher doses of GCs, improving the mural thickness of the SCA over time, and offering a superior safety profile with less side effects.
