Abstract
Background & Objective:
Although data on the effects of liraglutide and semaglutide in patients with diabetes have been reviewed, their therapeutic outcomes in obese/overweight individuals without diabetes have not been summarized. We conducted a systematic review to evaluate their effects on the latter population.
Methods:
We searched the PubMed, EMBASE, Cochrane, CNKI, and Wanfang databases. Studies regarding obese/overweight adults without diabetes treated with liraglutide/semaglutide compared with other active agents or placebos were accessed. The primary outcomes were the proportions of adults with at least 5% and 10% weight reduction. The secondary outcomes included metabolic indicators and adverse events.
Results:
Eighteen studies with 10,938 obese/overweight adults without diabetes were included. When stratified by the categories of at least 5% and 10% weight loss, the pooled data showed medians 27.7% and 10.3% of control groups versus 65.3% and 30.7% of liraglutide 3 mg once daily, respectively; whereas medians 47.6% and 20.4% of control groups vs 86.6% and 75.3% of semaglutide 2.4 mg once weekly were found in the two categories, respectively. Both agents either improved or had no impact on lipid or glycemia. Liraglutide or semaglutide therapy had discontinuation rates of 2.4%–11.4% which overlapped with 0.7%–8.6% in control groups. The frequency of adverse events was comparable between the treatment groups and the control groups (66.5%–95.8% vs 46.9%–96.1%), which were mild to moderate graded by studies.
Conclusion:
Liraglutide and semaglutide therapy led to a clinically relevant (⩾5%) weight loss of 48.2%–88.7% among obese/overweight adults without diabetes. Both liraglutide and semaglutide are associated with weight loss and are well-tolerated.
Introduction
Obesity is the most common chronic, progressive disease: approximately at least 30% of men and 35% of women are obese worldwide. 1 Obese/overweight adults are usually accompanied by various metabolic diseases. 2 Being obese/overweight is associated with higher all-cause mortality, as adults with a BMI of 27.5–30 kg/m2 had all-cause mortality that was 1.20 times higher than adults with a BMI of 20–25 kg/m2. 3 Being obese/overweight is now the sixth-largest risk factor contributing to the global burden of disease, placing enormous pressure on healthcare systems and societies. 4 Obesity deserves attention given its high prevalence, multiple complications, and unique reversibility.
Effective long-term weight loss and maintenance require continuous lifestyle interventions, including adjusted diet and increased physical activity, which perhaps are tough to keep for obese/overweight adults. 4 Pharmacotherapy, 5 surgery therapy, 6 and ancillary therapy are necessary and effective for some adults who have gone through countless weight loss failures. It is hard to make bariatric surgery widely applicable and tolerant due to its invasive and postoperative complications. 7 Several anti-obesity drugs approved for clinical use have been discontinued due to disappointing side effects. However, the development of glucagon-like peptide-1 receptor agonists (GLP-1RAs) drugs holds promise for obesity management. The glucagon-like peptide-1 (GLP-1) is a 36 amino acid peptide hormone with multifaceted actions such as glucose-dependent stimulation of insulin release, inhibition of glucagon secretion, and a decrease in gastric emptying. Several structurally refined GLP-1RAs with enhanced bioavailability have attracted extensive attention, including exenatide, liraglutide, dulaglutide, albiglutide, and semaglutide. 8 Among them, liraglutide and semaglutide are particularly noticeable.
Liraglutide is highly similar to the native GLP-1 sequence with only a few chemical modifications to improve bioavailability and extend the half-life, including replacing lysine at position 34 with arginine and adding a C16 fatty acid at the ɛ-amino group of lysine at position 26. 9 The once-daily 3.0 mg liraglutide has been approved by the Food and Drug Administration (FDA) and European Medicines Agency (EMA) for the treatment of obesity. 10 However, the once-daily injection model can cause physical and financial stress for some adults, so the once-weekly injection model for the GLP-1RAs was implemented. Semaglutide is an analogue of liraglutide with a substitution of alanine with an aminoisobutyric acid (Aib) at the 2nd position in the N-terminal. The C16 fatty acid is also exchanged for C18 fatty acid and linked by a synthetic spacer. The half-life of semaglutide extends to 160 h, supporting once-weekly administration. 11 Semaglutide once-weekly 2.4 mg was approved by the FDA in June 2021 for the treatment of overweight/obese individuals. 12
Liraglutide and semaglutide have shown beneficial effects in weight loss for patients with diabetes. While few reviews focused on obese/overweight individuals without diabetes, we aimed to perform a systematic review of semaglutide and liraglutide in randomized clinical trials (RCTs) and non-RCTs to provide information about their weight-loss effects and safety in obese/overweight individuals without diabetes.
Methods
Overview of the data search protocol
This systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement for Systematic Reviews and Meta-Analysis. The PROSPERO has been registered (ID: CRD42021272102). We included RCTs and observational studies if they studied liraglutide or semaglutide compared to either a placebo or other drugs in obese (BMI ⩾ 30 kg/m2) / overweight (BMI ⩾ 27 kg/m2) adults (age ⩾ 18 years) without diabetes, and reported either a proportion of adults achieving at least 5% weight loss or a change of weight outcome. Both English and Chinese language studies were included. We searched literature published before August 1, 2021, from PubMed, EMBASE, Cochrane, and two Chinese databases (CNKI and Wanfang). Details of the search strategies are shown in Sup. 2, and study selection procedures are shown in Figure 1.

Data search and collection process. The flowchart presented the process of identifying articles in the systematic review. Among 4,902 articles found, 621 were removed as duplicates, and 4,106 did not meet the inclusion criteria. After screening the rest of the 175 articles, 157 were removed due to the exclusion criteria. The remaining 18 articles were included in the systematic review (12 RCTs and 6 non-RCTs).
Eligibility criteria
We defined studies as eligible for inclusion in this analysis if they met the following criteria: (1) Obese/overweight adults who use liraglutide or semaglutide; (2) RCTs, observational studies; (3) reporting clinical events data of efficacy and safety; and (4) published before 2021/8/1, English or Chinese language. We excluded these publications: (1) age less than 18 years old; (2) with diabetes; (3) animal studies; (4) case reports, case series, or the sample size was too small (N < 10); and (5) unpublished studies.
Outcomes of interest
The primary outcomes were the proportions of adults with at least 5%, 10%, and 15% (if available) weight loss after liraglutide and semaglutide treatment. The secondary outcomes included the changes in metabolic-related indicators including glycosylated hemoglobin (HbA1c), glucose, insulin, total cholesterol (TC), triglyceride (TG), and low-density lipoprotein cholesterol (LDL-C), and high-density lipoprotein cholesterol (HDL-C). We were also concerned about adverse events, including the proportions leading to the discontinuation of drugs, any adverse events, and serious adverse events, such as the prevalence of dose-related gastrointestinal symptoms (nausea, diarrhea, and vomiting), gallbladder disorders, and acute pancreatitis.
Data search process
The literature search was performed by three authors (Y.D., A.P., and L.Z.). A list of eligible publications was generated by screening the titles, abstracts, and keywords of each record. Any record that met the inclusion criteria was obtained in full text. If any of the three authors had doubts based on the title and abstract, the full article was retrieved. Disagreements between reviewers were resolved by the corresponding author (C.Q.P.). Selected studies were further reviewed by all authors for the final confirmation based on the criteria.
Data extraction and synthesis
Three review authors (Y.D., A.P., and L.Z.) independently extracted data concerning details of the study designs, population characteristics, co-interventions, and outcomes of interest using a standard data extraction form designed for this review. If necessary, we sought information from the authors of the primary studies. For dichotomous outcomes, the number of events and the total number in each group was extracted. For continuous outcomes, the mean, standard deviation, and sample size of each group were extracted.
Risk of bias assessment
Three reviewers (Y.D., A.P., and L.Z.) independently assessed the risk of bias using the Cochrane Risk of Bias assessment tool (Table S1) for RCTs and the Newcastle-Ottawa Scale (NOS) (Table S2) for observational studies. The Cochrane Risk of Bias assessment tool and the Newcastle-Ottawa Scale (NOS) have become the standard approach to assessing the risk of bias in RCTs and observational studies, respectively. The quality of evidence was evaluated using the Grading of Recommendations Assessment, Development, and Evaluation approach. Criteria used to evaluate the quality of evidence were risk of bias, indirectness, imprecision, inconsistency, and publication bias. Each study was assessed by the three reviewers independently (Y.D., A.P., and L.Z.). Disagreements were resolved, when necessary, by recourse to a fourth reviewer (C.Q.P.).
Statistic consideration
No meta-analysis was performed due to the methodologic and statistic heterogeneity of the included studies. The descriptive analyses were performed when we summarized the data. Outcome assessments were presented by using the frequency, a proportion of the study group, or mean ± SD for variables when appropriate.
Overall assessment of data selected
Data inclusion
Through the keywords search, we identified 4,902 studies in PubMed, EMBASE, Cochrane Library databases, and two Chinese databases (CNKI and Wanfang) for eligibility for review, of which 621 duplicates were removed. After this, 4,106 studies were excluded due to the title and abstract. Of 175 full-text records, 157 were excluded, leaving 18 eligible studies for inclusion in the review, of which 12 studies were RCTs and 6 studies were non-RCTs. The details of screening and selecting are shown in Figure 1.
Quality of included studies
Results of the risk-of-bias analysis are shown in the Sup 3. Among 12 RCTs, 9 had a low risk of bias,13–21 and 3 had an unclear risk of bias22–24 according to the criteria. No studies were classified as having a high risk of bias. Among 6 observational cohort studies, 2 had a high risk of bias with a score of 6,25,26 while 4 had a low risk of bias with a score ranging from 7 to 8.27–30
Characteristics of included studies
There were 10,938 obese/overweight adults without diabetes enrolled in these 18 studies, where a total of 8797 patients completed these studies. There were 8 studies conducted in multiple countries around the world. Most studies were performed in Europe and America (16 articles) and published in English (17 articles). The enrolled population of different studies with different metabolic comorbidities except diabetes includes dyslipidemia, hypertension, prediabetes, moderate, or severe obstructive sleep apnea, or cardiovascular disease. The duration of these studies ranges from 12 weeks to 104 weeks. Among 10,938 obese/overweight adults, 4,146 were treated with liraglutide, 2,996 were treated with semaglutide, and 3,796 were treated with a placebo or other drugs as the control groups.
While the majority of studies (n = 9/18) compared the liraglutide-treated group with placebo or other drugs,13,16,19–23,29,30 four studies observed the efficacy and safety of liraglutide,25–28 four studies compared semaglutide to placebo or other drugs,14,17,18,24 and one study directly compared liraglutide with semaglutide. 15 The characteristics of these studies are summarized in Table 1.
The characteristics of studies for investigating liraglutide and semaglutide.

NR, not report; OSA, obstructive sleep apnea; RCT, Randomized Controlled Trial; UK, The United Kingdom; USA, United States of America.
Baseline characteristics of included studies population
The included studies’ populations had a baseline mean age ranging from 38.5 to 58.0 years, with the proportion of males ranging from 11.6% to 71.9%, and the baseline BMI (mean) ranging from 27.3 to 45.8 kg/m2. Among 18 studies, 16 provided lifestyle-based therapies including restricted caloric intake, forced exercise, and intervened behavior. The behavioral intervention was defined as offering participants an individualized behavioral consultation by a registered dietitian. The consultation provided patients with instructions on diet, physical activity, behavioral strategies, and other lifestyle modifications. Although lifestyle interventions were delivered together with GLP-1RAs therapy in most studies, the duration of the intervention was shorter than the duration of the treatment of GLP-1Ras in some studies. There were inconsistencies in providing details of the lifestyle intervention among studies. In addition, two studies did not mention the lifestyle cointervention and only presented GLP-1RAs therapy.25,26 The baseline characteristics of included studies’ populations are shown in Table 2.
Baseline characteristics of study population.

BMI, body mass index; NR, not report; WC, waist circumference.
Values are presented as mean ± standard deviation or medians (interquartile ranges).
Behavior intervention: participants were provided with individual intensive behavioral therapy visits with a registered dietitian, who instructed them in diet, physical activity, and behavioral strategies and so on. Different study have different behavior intervention.
Astrup A’s two studies of 2009 and 2012 used the same population and had the same baseline characteristics.
The study of O’Neil P.M compared semaglutide and liraglutide.
The primary and secondary outcomes of liraglutide and semaglutide
The primary outcomes of liraglutide and semaglutide
All populations treated with liraglutide or semaglutide were associated with effective, sustained, clinically relevant weight loss regardless of the study design and duration. The doses of liraglutide used in these studies include 0.6 mg, 1.2 mg, 1.8 mg, 2.4 mg, and 3.0 mg, where 3.0 mg once-daily was the most commonly treated dose. In the liraglutide 3.0 mg daily treated group, the ranges of patients who had more than 5% weight loss were 48.2% to 76.1% and 1.8% to 44.0% in control groups. The ranges of patients who lost ⩾ 10% and ⩾ 15% body weight were 20% to 46% and 8.4% to 28%, while the ranges were 1.8% to 26% and 1.8% to 12% in control groups, respectively. Semaglutide 2.4 mg once weekly was the most commonly used dose in clinical applications. In semaglutide treated groups, the ranges of patients who had more than 5%, 10%, and 15% weight loss were 86.4% to 88.7%, 69.1% to 79.0, and 50.5% to 63.7%, while it was 31.5% to 47.6%, 12.0% to 27.0%, and 4.9% to 13.2% in control groups, respectively. The data are shown in Table 3. The studies of Astrup et al.16,20 evaluated the efficacy and safety of different doses of liraglutide (1.2 mg, 1.8 mg, 2.4 mg, 3.0 mg, and the matching placebo group), and concluded that treatment with liraglutide led to a dose-dependent weight loss. They also stated that liraglutide 3.0 mg once daily demonstrated superior weight loss compared to other lower doses and control groups regardless of a 20-week or 104-week duration. One study directly compared liraglutide and semaglutide. O’Neil 15 randomly assigned participants to seven groups (semaglutide [0.05 mg, 0.1 mg, 0.2 mg, 0.3 mg, or 0.4 mg], liraglutide [3·0 mg], and a matching placebo group) and demonstrated that semaglutide 0.05–0.4 mg once-daily led to a statistically significant, clinically relevant, dose-dependent reduction of weight over 52 weeks compared to the placebo group and that semaglutide 0.2 mg once-daily showed superior weight loss than liraglutide 3.0 mg once-daily.
The primary endpoints of liraglutide and semaglutide.
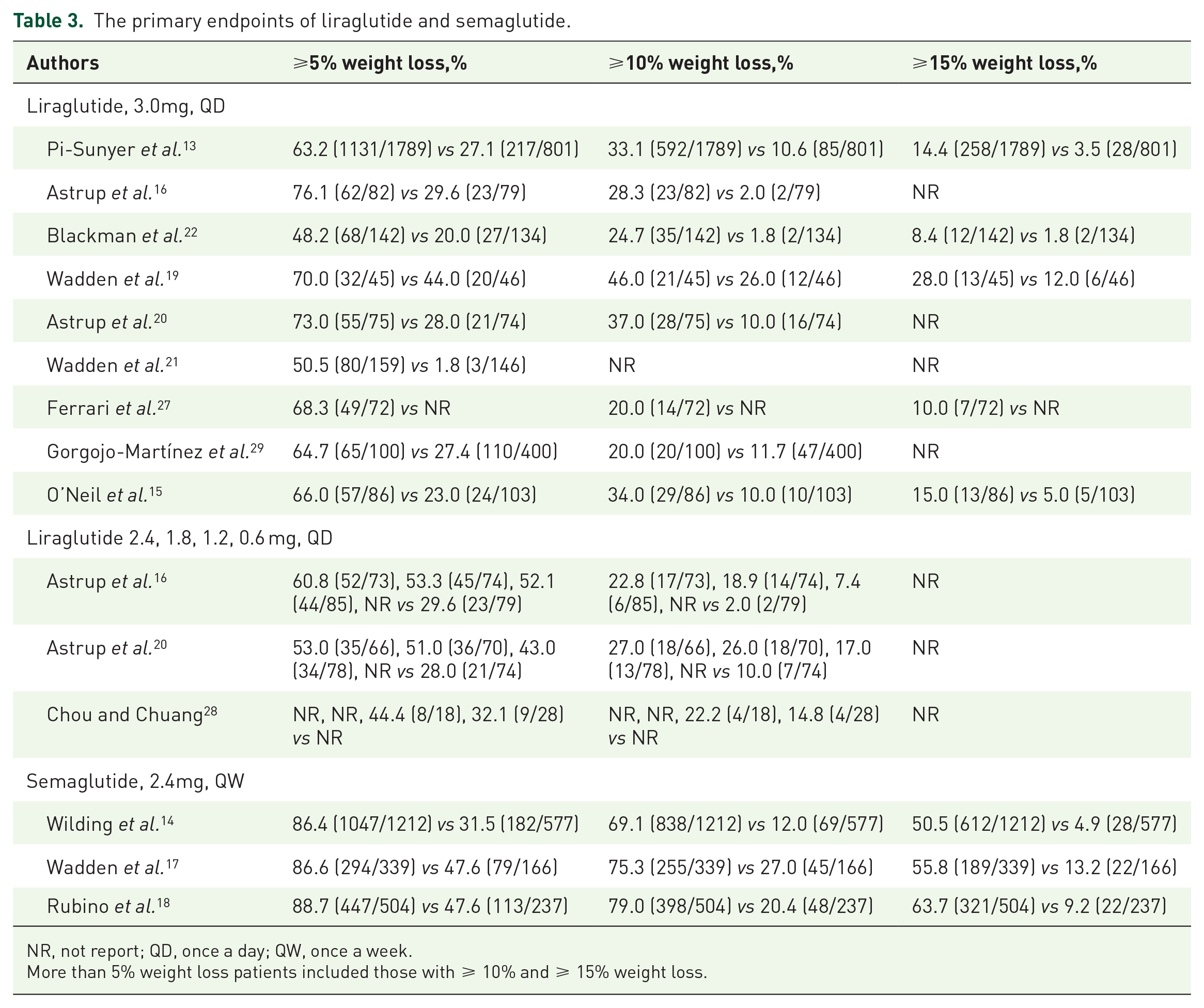
NR, not report; QD, once a day; QW, once a week.
More than 5% weight loss patients included those with ⩾ 10% and ⩾ 15% weight loss.
The secondary outcomes of liraglutide and semaglutide
The metabolic-related outcomes
Glucose and lipid metabolic risk factors were less studied in the included literature. Eleven studies evaluated the improvement of metabolic-related factors after the treatment of liraglutide 3.0 mg once-daily (n = 8)13,15,19–23,29 and semaglutide 2.4 mg once-weekly (n = 3).14,17,18 Nine studies found that liraglutide or semaglutide could statistically significantly improve the metabolic condition compared to control groups,13,15,17,18,20–23,29 including the decrease of HbA1c, glucose, insulin, TC, TG, LDL-C, and HDL-C. Two studies found numerically improved or unchanged metabolic indicators.14,19 An overview of the trend is shown in Table 4. In addition, consistent dose-dependent improvement in metabolic risk factors was seen for semaglutide 0.05–0.4 mg once daily compared to the placebo in the study O’Neil et al. 15
The secondary endpoints of liraglutide and semaglutide.

HDL-C, high density lipoprotein cholesterol; LDL-C, Low density lipoprotein cholesterol; NR, not report; QD, once a day; QW, once a week; TC, Total cholesterol; TG, Triglyceride.
Liraglutide compared to control or semaglutide compared to control, P < 0.05.
Adverse events outcomes
Adverse events were reported in 10 studies of liraglutide (n = 7) and semaglutide (n = 3). The proportion range leading to discontinuation was 5.4%–11.4% in liraglutide 3.0 mg once-daily groups and 0.7%–8.6% in control groups. The incidences of any adverse events and serious adverse events were 66.5%–96.7% and 0%–7.5% in the liraglutide 3.0 mg once-daily groups, while they were 46.9%–88.6% and 1.0%–8.0% in control groups, respectively. In semaglutide 2.4 mg per week groups, the proportions leading to discontinuation, any adverse events, and serious adverse events were 2.4%–7.0%, 81.3%–95.8%, and 7.7%–9.8% compared to 2.2%–3.1%, 75.0%–96.1%, and 2.2%–9.8% in control groups, respectively. The detailed data are shown in Table 5. The most common adverse events were gastrointestinal-related events, such as nausea and diarrhea. The incidences of common adverse events are summarized in Table S3. Serious adverse events were rarely reported and usually appeared to be mild to moderate in severity. It seems that liraglutide and semaglutide are well tolerated in real-world practice.
The tolerability and frequency of adverse events.

NR, not report; QD, once a day; QW, once a week.
Serious adverse events defined grade 2/3/4 adverse events. A serious adverse event (SAE) is defined as any untoward medical occurrence that at any dose: Results in death; is life-threatening; requires inpatient hospitalization or causes prolongation of existing hospitalization; results in persistent or significant disability/incapacity; may have caused a congenital anomaly/birth defect; requires intervention to prevent permanent impairment or damage.
Discussion
The systematic review summarized the efficacy and safety of liraglutide and semaglutide from 12 RCTs and 6 non-RCTs in 10,938 obese/overweight adults without diabetes. Liraglutide and semaglutide were both significantly associated with weight loss in obese/overweight adults without diabetes regardless of different study designs, population characteristics, cointerventions, background therapies, and durations across studies. They were often associated with improvements in metabolic factors and were well-tolerated in clinical practice.
Currently, four effective pharmacologic agents including orlistat, naltrexone-bupropion, liraglutide, and semaglutide have been approved by both the FDA and the EMA for obesity management. In addition, the FDA also approved lorcaserin and phentermine-topiramate for the same indication.12,31 Several systemic reviews and meta-analyses have compared weight loss among these five drugs except semaglutide, and have demonstrated that liraglutide was associated with the highest odds of achieving at least 5% weight loss compared to the others.32,33 Some studies found that semaglutide was more effective in reducing body weight than liraglutide; however, the vast majority of these studies are studies based on patients with diabetes, with weight loss not being the main primary outcome.34–36 Compared to patients with diabetes, obese/overweight individuals without diabetes should be assessed for the therapeutic outcomes as a unique group because of the different metabolic risks for the disease progression.
Liraglutide and semaglutide produced continuous weight loss in a dose-dependent manner.15,16,20 The current study found that GLP-1RAs administered linearly decreased glucose and HbA1c across a broad dose range in patients with type 2 diabetes.37,38 Pharmacokinetics revealed that high circulating levels of GLP-1RA were highly associated with the degree of weight loss in most GLP1-RAs. 39 There is a possibility that it is due to a unique molecular-specific interaction on GLP-1R. However, the incidence of gastrointestinal AEs also appeared to be dose-related. Therefore, concerning the tolerability of AEs may help in optimizing drug development.
Although current pieces of evidence are insufficient to conclude, few studies with preliminary data implied the superiority of semaglutide over liraglutide in obese/overweight adults without diabetes. In a head-to-head study that compares liraglutide with semaglutide by O’Neil et al., 15 semaglutide 0.2 mg per day produced a better effect on weight loss than liraglutide 3.0 mg per day in patients without diabetes. Several studies also explored the mechanistic aspects of the preclinical setting of animal studies and provided a hypothesis.39–41 The investigators indicated that the brain function associated with the localization of liraglutide and semaglutide in the brain might be explained how they worked. They observed that semaglutide affected larger brain areas that controlled hunger than those involved with liraglutide. In the lateral septal nucleus, semaglutide, but not liraglutide, was detected after peripheral administration. Transcriptome analysis showed ribosomes (eukaryotic initiation factor 2 and 4 signal transduction) and mitochondria (oxidative phosphorylation) were regulated differently between animals treated with liraglutide and semaglutide. 40 In addition, weight loss seemed to continue during the whole 52-week duration at higher doses of semaglutide. 15 This does not support the view that most anti-obesity drugs induce weight loss in the first 3–4 months. 41 Semaglutide enhances metabolic activity and prevents compensated downregulation of energy expenditure, which aids in prolonged weight loss or maintenance. 40 Importantly, weekly injections are considered better than daily injections, but there are no comparisons of once-daily liraglutide and once-weekly semaglutide in obese/overweight individuals without diabetes in the current works of literature, which makes it worthy of further investigation.
The treatment of liraglutide and semaglutide was accompanied by a positive change in metabolic condition, which seems to be an inevitable conclusion as it can promote insulin secretion, reduce glucagon production, and delay gastric emptying of GLP-1RAs. 42 A Danish study confirmed that treatment with GLP-1RAs has beneficial effects on cardiovascular, mortality, and kidney outcomes in patients with type 2 diabetes. 43 However, it is uncertain whether GLP-1RAs improve metabolism to the same extent in obese/overweight patients with and without diabetes.
An oral formulation of semaglutide is currently available and under investigation, which may increase the acceptability of this agent compared to the injectable form and has shown a significant ability to reduce HbA1c and body weight. 38 Given the results of semaglutide, researchers have shown great interest in whether binding one or more peptide epitopes together with GLP-1 can lead to greater weight loss. Several co-agonists including Glucagon-GLP-1R co-agonists, Amylin-GLP-1R co-agonists, and GIPR-GLP1R co-agonists have shown promising results in the management of obesity. 39 In preclinical studies, oxyntomodulin was shown to achieve significant weight loss by activating both the glucagon and GLP-1 receptors. Large clinical trials have been implemented to investigate the efficacy and safety of the AM833 (cagrilintide), a long-acting, non-selective amylin receptor agonist, together with semaglutide for the treatment of obesity. 44 In a recent study, tirzepatide, an effective GIPR-GLP1R co-agonist, was associated with greater weight loss than semaglutide 1 mg per week in T2DM patients. 45
Standard obesity management should be intensive, have multi-component interventions, and be step-by-step. The importance of combining lifestyle modifications such as dietary and physical activity should not be ignored in obesity treatment. Dietary modification is fundamental for the long-term management of obesity and exists in a variety of dietary interventions, including time adjustment (e.g. intermittent fasting), calorie restriction with different macronutrient compositions (e.g. low-fat diets, low-carbohydrates), avoidance of special foods, and following a certain dietary pattern (e.g. Mediterranean diet). Identifying dieters’ behavioral and metabolic phenotypes can optimize adherence and subsequent weight loss. 46 A new concept ‘instrumentalization of eating’ was recommended to be an effective predictor for sustained weight loss, which represents the extent to which participants have made their eating behavior an instrument for maintaining weight loss. The interplay between behavioral, emotional, and environmental changes is also noteworthy. 47 Physical activity is also crucial for successful weight loss, as it can induce energy deficit and preserve lean muscle mass. The American College of Sports Medicine (ACSM) recommended that individuals need to exercise 150–250 min/week, > 150–250 min/week, and 200–300 min/week to prevent weight gain, achieve weight loss, and maintain weight loss, respectively. 48 Nevertheless, the individual’s response to exercise is heterogeneous. A randomized clinical trial demonstrated that the exercise combined with liraglutide was approximately twice as much as either treatment alone in reducing the body weight and body-fat percentage. It was also associated with the improvements in insulin sensitivity, cardiorespiratory fitness, and maintaining a good mood. 49 In the area of surgical intervention, bariatric surgery has been applied in the clinical setting to help patients achieve significant weight loss. Currently, common procedure options include laparoscopic sleeve gastrectomy (LSG), the Roux-en-Y gastric bypass (RYGB), and the laparoscopic adjustable gastric band (LAGB). Bariatric surgery should be considered for patients who have failed to lose weight after undergoing various interventions. Bariatric devices are a growing field of intervention that can further support patients in achieving their weight loss goals based on their minimally invasive, reversible, and less expensive features. Recently, the FDA approved five devices, including 2 intragastric balloons, an aspiration device, superabsorbent hydrogel capsules, and the TransPyloric Shuttle. 50 These new options will add to the variety of options patients who fail pharmacological intervention and lifestyle modification can take to combat obesity.
Successful obesity management extends well beyond weight loss, as it is important to highlight the assessment of the life quality and the prevention of obesity-related comorbidities in outcome studies.51,52 The ideal treatment for obesity should be a highly individualized, personalized medicine. Treatment decisions will consider age, coexisting diseases, drug tolerance, and economic and local medical conditions. 53 For example, naltrexone/bupropion does not apply to uncontrolled hypertension patients and phentermine/topiramate treated patients should be monitored closely for depression. 41 However, there is no consensus or guidelines to help clinicians make decisions on which drugs to choose. Thus, more real-world studies, especially direct comparisons of different drugs that capture a broader range of outcomes, are needed.
There are some limitations to this review. First, several included studies were observational and did not design their control groups, which was unavoidable due to a limited number of large RCTs. Second, the methods, populations, study designs, and data collections vary considerably from one literature to the next, making it difficult to interpret data across studies. Third, the vast majority of studies for drug treatment are accompanied by lifestyle co-interventions which makes it difficult to evaluate the efficacy of drugs separately.
Conclusion
Liraglutide and semaglutide were both significantly associated with weight loss in obese/overweight adults without diabetes regardless of different trial designs, population characteristics, and durations. Few studies have directly compared the efficacy and safety profiles of liraglutide and semaglutide in obese/overweight individuals without diabetes, suggesting a need to develop trials directly comparing drugs for different obese/overweight subgroup populations.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221108064 – Supplemental material for Effect of semaglutide and liraglutide in individuals with obesity or overweight without diabetes: a systematic review
Supplemental material, sj-docx-1-taj-10.1177_20406223221108064 for Effect of semaglutide and liraglutide in individuals with obesity or overweight without diabetes: a systematic review by You Deng, Andrew Park, Lin Zhu, Wen Xie and Calvin Q. Pan in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We thank all participants in the study.
Ethics approval and consent to participate
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation Youth Fund Project (81600454), the National Natural Science Foundation of China (81770577), Clinical Medicine Development Project of Beijing Municipal Administration of Hospitals (ZYLX201828).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
All authors approve the manuscript and give the consent for submission and publication. All data generated or analysed during this study are included in this published article [and its supplementary information files].
PROSPERO had been registered
ID CRD42021272102.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
