Abstract
Background:
The impact of thyroid hormones within their normal ranges on skeletal muscle and bone in patients with type 2 diabetes mellitus (T2DM) remains unknown. The purpose of this study was to investigate the relationships of thyroid hormones with muscle and bone in euthyroid patients with T2DM.
Methods:
This cross-sectional study included 344 euthyroid T2DM patients. Muscle mass and bone mineral density were measured by dual-energy X-ray absorptiometry. The levels of thyroid-stimulating hormone (TSH), free triiodothyronine (FT3), and free thyroxin (FT4) were measured by electrochemiluminescence immunoassay.
Results:
The results revealed that FT3 was positively correlated with body mass index (BMI) in male patients after age correction. In men, FT4 was negatively correlated with body weight, BMI, total muscle mass, appendicular skeletal muscle mass (ASM), and ASM index (ASMI), while FT3/FT4 was positively correlated with body weight, BMI, total muscle mass, ASM, and ASMI after age correction. In women, FT4 was negatively correlated with ASM and ASMI, while FT3/FT4 was positively correlated with ASM and ASMI after age correction. FT3/FT4 was significantly lower in men with low muscle mass than in those with normal muscle mass. The age-adjusted odds for incident low muscle mass comparing the lowest and highest FT3/FT4 increased in men.
Conclusions:
FT3/FT4 was positively correlated with ASM and ASMI in both men and women. Therefore, FT3/FT4 may be a parameter indicative of low muscle mass in euthyroid men with T2DM.
Introduction
Thyroid hormones participate in important biological functions in the body and have key roles in determining growth, development, and energy metabolism. Muscle is the main target of thyroid hormone signal transduction. Triiodothyronine (T3) binds to thyroid hormone nuclear receptor and participates in muscle development, homeostasis, and regeneration. 1 Obvious thyroid dysfunction is an inducing factor for muscle wasting, 2 and even subclinical thyroid dysfunction has an adverse impact on muscle based on observed decreases in the muscle cross-sectional area in patients with subclinical hyperthyroidism or hypothyroidism.2,3 Thyroid hormones also coordinate endochondral ossification, regulate the number and activity of osteoblasts and osteoclasts, and participate in bone development and maintenance. 4 Excessive thyroid hormones lead to enhanced osteoclast activity and osteoporosis. 5 Patients with hypothyroidism have a two- to threefold increased risk of fractures. 6 In euthyroid postmenopausal women, thyroid function at the upper limit of the normal range is associated with reduced bone mineral density (BMD). 7
In patients with type 2 diabetes mellitus (T2DM), low muscle mass and osteoporosis are common complications. A cross-sectional survey showed that the prevalence of sarcopenia was 14.4% in Chinese T2DM patients. 8 In addition, a meta-analysis demonstrated that the prevalence of osteoporosis in T2DM patients was significantly increased in China, at 44.8% in women and 37.0% in men. 9 The state of muscle and bone in T2DM patients is influenced by many factors, including hormones, drugs, and complications. There is little research on the relationships of thyroid hormones with muscle and bone in euthyroid T2DM patients.
The aim of this study was to investigate the relationships of thyroid hormones with muscle and bone in euthyroid T2DM patients.
Materials and methods
Study design and participants
This retrospective cross-sectional study enrolled T2DM patients who were hospitalized in the Department of Endocrinology and Metabolism of Kunshan First People’s Hospital from 1 December 2018 to 14 August 2021. Based on the results of a power analysis using G*Power 3.1 and the preliminary results of this study, the correlation coefficient between skeletal mass and thyroid hormones was our primary outcome, and we expected an effect size|ρ| = 0.20. Based on an α = 0.05 (one-tailed) and β = 0.05 (power = 0.95), a minimum of 315 patients was required considering a 20% rate of loss to follow-up.
Patients
The inclusion criteria were an age of >50 years, presence of T2DM, and normal thyroid function. The diagnostic criteria for T2DM, according to the American Diabetes Association criteria, 10 were as follows: (1) fasting blood glucose (FBG) level of ⩾7.0 mmol/l, (2) random blood glucose level of ⩾11.1 mmol/l or stimulated blood glucose level of >11.1 mmol/l after standard oral glucose tolerance test, and (3) hemoglobin A1c (HbA1c) level of >6.5%. The exclusion criteria were type 1 diabetes mellitus (positive autoantibody and/or continuous need for insulin at diagnosis), thyroid disease, current treatment with thyroid hormone drugs or antithyroid drugs, current treatment with drugs that affect thyroid function (such as contraceptives, estrogen, amiodarone and other iodine-containing drugs, or lithium), malignant tumors, and renal insufficiency. The study followed the principles of the Helsinki Declaration and was approved by the Ethics Committee of Kunshan First People’s Hospital (ethics approval no. 2019-04-010-K01). All enrolled patients provided written informed consent.
Parameters
The patients’ age and disease course were recorded. Height and weight were measured, and the body mass index (BMI) was calculated as weight (kg) divided by height squared (m2).
Laboratory examination
Fasting blood samples were collected for FBG measurement using an automatic biochemical analyzer (AU5831; Beckman Coulter Life Sciences, Indianapolis, IN, USA). HbA1c was measured by high-performance liquid chromatography using a D-10 System (Bio-Rad Laboratories, Hercules, CA, USA) and expressed in appropriate units (%). Free triiodothyronine (FT3), free thyroxin (FT4), and thyroid-stimulating hormone (TSH) were measured by electrochemiluminescence immunoassay. The normal ranges of FT3, FT4, and TSH were 2.43–6.02 pmol/l, 9.01–19.04 pmol/l, and 0.350–4.940 mIU/l, respectively.
Dual-energy X-ray absorptiometry
Dual-energy X-ray absorptiometry (DXA) (Discovery DXA System; Hologic, Marlborough, MA, USA) was used to measure total muscle mass, trunk muscle mass, bone mineral content (BMC), BMD, appendicular skeletal muscle mass (ASM), and ASM index (ASMI). The ASMI was calculated as ASM (kg) divided by height squared (m2). According to the diagnostic criteria of the Asian Working Group for Sarcopenia, an ASMI of <7.0 kg/m2 in men and <5.4 kg/m2 in women was defined as low muscle mass. 11
Statistical analyses
IBM SPSS statistics version 22 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses. Continuous variables are expressed as mean ± standard deviation. Variables with a non-normal distribution are expressed as median (interquartile range). An independent t test was used for comparisons between groups of normally distributed variables and the Mann–Whitney U test was used for comparisons between groups of non-normally distributed variables. The classified variables are expressed as percentages and were compared by the chi-square test. Pearson’s correlation and partial correlation analyses were used to analyze the relationships of thyroid hormones with BMD and muscle mass. A binary logistic regression model was used to estimate the odds of low muscle mass according to tertiles of FT3/FT4 and the model was adjusted for age. Values of p < 0.05 were considered to indicate statistical significance.
Results
In total, 344 patients with T2DM aged >50 years were enrolled in the study. The patients’ characteristics are shown in Table 1. The cohort comprised 170 men with a mean age of 64.17 ± 8.62 years and 174 women with a mean age of 65.54 ± 8.55 years. Compared with the women, the men had significantly greater height, weight, FT3 level, total muscle mass, trunk muscle mass, ASM, ASMI, lumbar BMC, lumbar BMD, femoral BMC, and femoral BMD but a significantly lower TSH level (p < 0.05). The prevalence of low muscle mass was significantly higher in men than women (p < 0.05) (Table 1).
Characteristics of T2DM patients stratified by sex.

ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BMC, bone mineral content; BMD, bone mineral density; BMI, body mass index; FBG, fasting blood glucose; FT3, free triiodothyronine; FT4, free thyroxin; HbA1c, glycosylated hemoglobin A1c; T2DM, type 2 diabetes mellitus; TSH, thyroid-stimulating hormone.
The correlation analyses between FT3 and clinical indicators are shown in Table 2. In men, FT3 was positively correlated with BMI and this correlation was statistically significant after age correction (p < 0.05). FT3 was also positively correlated with ASMI in men, but the correlation was not significant after age correction. In women, FT3 was negatively correlated with BMI, but the correlation was not significant after age correction (Table 2).
Association between FT3 and clinical characteristics before and after adjusted by age in T2DM patients.

ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BMC, bone mineral content; BMD, bone mineral density; BMI, body mass index; FT3, free triiodothyronine; T2DM, type 2 diabetes mellitus.
The correlation analyses between FT4 and clinical indicators are shown in Table 3. In men, FT4 was negatively correlated with body weight, BMI, total muscle mass, and ASM and these correlations were statistically significant after age correction (p < 0.05). FT4 was negatively correlated with ASMI after age correction (p < 0.05). In women, FT4 was negatively correlated with ASMI and this correlation was statistically significant after age correction (p < 0.05). FT4 and ASM were negatively correlated after age correction (p < 0.05) (Table 3).
Association between FT4 and clinical characteristics before and after adjusted by sex in T2DM patients.
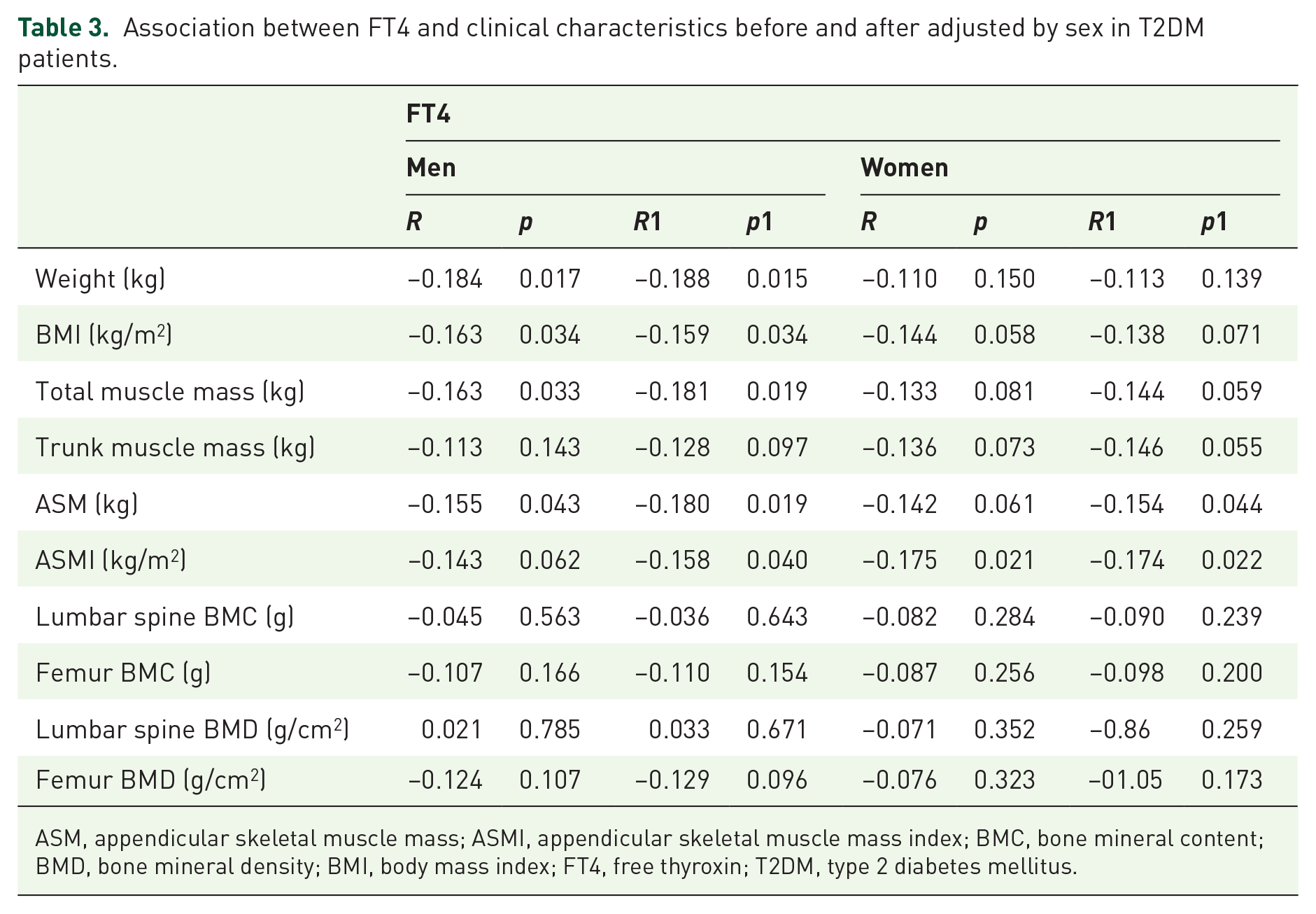
ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BMC, bone mineral content; BMD, bone mineral density; BMI, body mass index; FT4, free thyroxin; T2DM, type 2 diabetes mellitus.
The correlation analyses between FT3/FT4 and clinical indicators are shown in Table 4. In men, FT3/FT4 was positively correlated with body weight, BMI, total muscle mass, ASM, and ASMI and these correlations were statistically significant after age correction (p < 0.05). In women, FT3/FT4 was positively correlated with ASM and ASMI and these correlations were statistically significant after age correction (p < 0.05) (Table 4).
Association between FT3/FT4 and clinical characteristics before and after adjusted by sex in T2DM patients.

ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BMC, bone mineral content; BMD, bone mineral density; BMI, body mass index; FT3, free triiodothyronine; FT4, free thyroxin; T2DM, type 2 diabetes mellitus.
There were no correlations between TSH and the clinical indexes.
The comparisons based on muscle mass are shown in Table 5. FT3/FT4, total muscle mass, trunk muscle mass, ASM, and BMC and BMD of the femur were significantly lower in men with low muscle mass than in those with normal muscle mass (p < 0.05). FBG, total muscle mass, trunk muscle mass, ASM, and femoral BMC and BMD were significantly lower in women with low muscle mass than in those with normal muscle mass (p < 0.05) (Table 5).
The comparisons based on muscle mass.

ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BMC, bone mineral content; BMD, bone mineral density; BMI, body mass index; FBG, fasting blood glucose; FT3, free triiodothyronine; FT4, free thyroxin; HbA1c, glycosylated hemoglobin A1c; TSH, Thyroid-stimulating hormone.
According to the diagnostic criteria of the Asian working group for sarcopenia, ASMI < 7.0 kg/m2 in male patients and ASMI < 5.4 kg/m2 in female patients were defined as low muscle mass.
A binary logistic regression model was built to investigate the association of FT3/FT4 and incident low muscle mass. The age-adjusted odds for incident low muscle mass comparing the lowest and highest FT3/FT4 was 3.061 [95% confidence interval (CI) = 1.399–6.697] in euthyroid men with T2DM (Table 6).
Binary logistic regression, relationships of FT3/FT4 tertiles with low muscle mass among euthyroid T2DM patients, subdivided by sex, and adjusted for age.

FT3, free triiodothyronine; FT4, free thyroxin; SE, standard error; T2DM, type 2 diabetes mellitus.
Discussion
This study showed that FT3 was positively correlated with BMI in male euthyroid T2DM patients after age correction. Furthermore, FT4 was negatively correlated with body weight, BMI, total muscle mass, ASM, and ASMI, whereas FT3/FT4 was positively correlated with body weight, BMI, total muscle mass, ASM, and ASMI after age correction in male euthyroid T2DM patients. FT4 was negatively correlated with ASM and ASMI, whereas FT3/FT4 was positively correlated with ASM and ASMI in female euthyroid T2DM after age correction. The prevalence of low muscle mass in male T2DM patients was significantly higher than that in female T2DM patients. Among male T2DM patients, FT3/FT4 was significantly lower in those with low than normal muscle mass and the age-adjusted odds for incident low muscle mass comparing the lowest and highest FT3/FT4 increased.
In euthyroid patients, FT3 was found to be positively correlated with BMI and waist circumference. 12 In this study, FT3 was positively correlated with BMI in men with T2DM after age correction. Previous studies examined the relationship between FT3 and muscle mass, but the relationship showed opposite results under different weight states. FT3 was negatively correlated with lean weight, muscle cross-section, and SMI in individuals of normal weight13,14 but was positively correlated with skeletal muscle content and SMI in overweight or obese patients or patients with T2DM.15,16 In this study, FT3 was positively correlated with ASMI in men with T2DM without age correction. Furthermore, the mean BMI of the men with T2DM was >25 kg/m2, consistent with the results of Chen et al. 16 In individuals of normal weight, monodeiodination of T4 produced the same amount of T3 and reverse T3 (rT3), whereas in obese subjects, rT3 production was decreased and T3 production was increased. 17 These phenomena were associated with an increased leptin level in patients with obesity or T2DM, enhanced activity of type 1 deiodinase enzyme, 18 and insulin resistance–stimulated thymidine 5’-deiodinase enzyme activity, 19 resulting in increased T3 production.
Kong et al. 20 showed that FT4 was negatively correlated with ASMI in Korean euthyroid patients aged >60 years. Roef et al. 13 found a negative correlation between the muscle cross-sectional area and FT4 in young and middle-aged euthyroid patients in Italy. In this study, ASM and ASMI were negatively correlated with FT4 in euthyroid patients with T2DM. Furthermore, by analyzing the correlations between FT3/FT4 and muscle mass, ASM and ASMI were found to be positively correlated with FT3/FT4 in euthyroid patients with T2DM. A recent study on overweight or obese euthyroid subjects showed that skeletal muscle mass and limb skeletal muscle mass were positively correlated with FT3/FT4. 15 In studies on elderly euthyroid subjects in the community, those with higher FT3/FT4 had higher ASM and physical performance, and a reduced FT3/FT4 could be used as a reliable index to evaluate the decline in skeletal muscle in the elderly population.20,21 Skeletal muscle was confirmed to be a target tissue for thyroid hormones; specifically, contractile function, myogenesis, metabolism, and regeneration of skeletal muscle are affected by thyroid hormones. 22 Thyroid hormones act in the skeletal muscle through the interaction of intracellular T3 with thyroid hormone nuclear receptors and by regulating target gene expression. The intracellular concentration of thyroid hormones is tightly controlled by deiodinases. The three types of deiodinases are D1, D2, and D3. D1 and D2 activate T4 to T3 by removal of an outer-ring iodine, whereas D3 can convert T4 to rT3 or convert T3 to T2. D2 is present in key thyroid-responsive tissues such as brain tissue, brown adipose, and skeletal muscle. Expression of D2 in tissues can provide local control of intracellular T3. 23 Control of the intracellular T3 level is the basis for skeletal muscle generation and regeneration and is very important for the terminal differentiation of muscle cells. 24 D2 and D3 can control the uptake and activation or inactivation of T3 in skeletal muscle. 22 In D2-knockout mice, the T3 levels of skeletal muscle were reduced and muscle strength was decreased. 25 Deficiency of D2 resulted in changes in the muscle fiber type of the soleus, with an increase in major histocompatibility complex (MHC) I fibers and a decrease in MHC IIa fibers. 26 D2 converts T4 to T3 locally in skeletal muscle and FT3/FT4 may be an indirect index of the peripheral D2 activity. Actually, studies have shown that FT3/FT4 is positively associated with higher insulin levels and insulin resistance in euthyroid adults 27 and that FT3/FT4 is considered to be an index of D1 and D2 activity. 28 Therefore, high FT3/FT4 indicates an increased conversion rate of T4 to T3 in skeletal muscle, leading to promotion of muscle growth and regeneration.
In this study, an ASMI of <7.0 kg/m2 for men and <5.4 kg/m2 for women was defined as low muscle mass. Thus, the patients were divided into two groups of patients: those with normal muscle mass and those with low muscle mass. The prevalence of low muscle mass in men and women was 48.2% and 23.6%, respectively. The prevalence of low muscle mass was significantly higher in men than in women, similar to the findings reported by Park et al. 29 Further analyses demonstrated that FT3/FT4 was significantly lower in men with low muscle mass than in those with normal muscle mass and an increased odds of low muscle mass was correlated with a declined FT3/FT4 in men. The difference in the prevalence of low muscle mass between men and women is related to sex steroids. Testosterone has a strong role in promoting muscle anabolism and age-related alterations in testosterone are major contributors to muscle atrophy. The circulating level of testosterone in men decreases 1% per year after the age of 40 years, whereas the change in testosterone in women is minimal after the age of 50 years. 30 The greater decrease in testosterone in men than in women leads to more rapid aging-induced muscle loss in men than in women. 30 The high prevalence of low muscle mass in patients with T2DM is associated with impairment of insulin action, accumulation of advanced glycosylation end products, increased inflammation, and diabetic complications. 31 An appropriate marker is important for identifying and monitoring the intervention efficacy, and biomarkers that participate in different pathophysiological pathways may be associated with low muscle mass. A meta-analysis showed that some inflammatory biomarkers such as interleukin 6 and tumor necrosis factor alpha are associated with sarcopenia; however, the results were inconsistent in different studies, which may be explained by the differences in study populations, sources of biomarkers, and sample sizes. 32 Recent studies showed that microRNAs play an important role in the progression of muscle atrophy. The microRNAs expressed in muscle are associated with the differentiation of satellite cells, the switching of muscle fiber types, and the development of muscle. 33 MicroRNAs may be biomarkers for low muscle mass, but more clinical studies are needed to confirm the relationship between microRNAs and low muscle mass. According to the relationship between FT3/FT4 and skeletal muscle, FT3/FT4 may help to identify muscle wasting in patients with T2DM, especially men.
Thyroid hormones play important roles in bone development and growth as well as maintenance of bone mass and strength in adults. Hypothyroidism results in delayed skeletal development and bone aging with short stature, whereas hyperthyroidism can lead to bone loss and increased bone fragility. Research on the effects of thyroid hormones within their normal ranges on BMD has not produced consistent results. One study showed that TSH was positively correlated with BMD in euthyroid postmenopausal women, 34 whereas another study showed that femoral neck BMD was positively correlated with TSH and negatively correlated with FT4 in elderly euthyroid individuals. 35 However, other studies revealed no correlations between thyroid hormones and BMD in young euthyroid women. 36 Vendrami et al. 37 found that TSH and FT4 had no correlations with BMD at any site in euthyroid postmenopausal women. This study showed no correlations of FT3, FT4, TSH, or FT3/FT4 with BMC and BMD of the lumbar spine and femur. The relationships between thyroid hormones within their normal ranges and BMD in clinical research are contradictory and the discrepancies are likely related to differences in the age, sex, and size of the research cohort as well as whether any comorbidities exist. Therefore, further research is needed to explore the relationships between thyroid hormones within their normal ranges and BMD.
This study has some limitations. First, this retrospective cross-sectional study lacked data on the patients’ use of long-term hypoglycemic drugs. Second, the study did not evaluate grip strength and other indicators that reflect muscle strength. Finally, the study cannot explain the causality between thyroid hormones and skeletal muscle mass. Further prospective studies are needed to determine the effects of thyroid hormones within their normal ranges on the muscles of T2DM patients.
Conclusion
This study analyzed the relationships between thyroid hormones and muscle and bone in euthyroid patients with T2DM. The results showed that FT3/FT4 was positively correlated with ASM and ASMI in both men and women. Therefore, FT3/FT4 may be a parameter indicative of low muscle mass in male euthyroid T2DM patients.
Footnotes
Acknowledgements
Availability of data and materials
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical approval and consent to participate
The study followed the principles of the Helsinki Declaration and was approved by the Ethics Committee of Kunshan First People’s Hospital. All enrolled patients signed written informed consent.
Consent for publication
All authors in this study agreed to publication.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China, No. 82172441. Medical scientific research project of Jiangsu Provincial Health Commission, No. 2021201.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
