Abstract
Human apolipoprotein E (ApoE) is a 299-amino acid secreted glycoprotein that binds cholesterol and phospholipids. ApoE exists as three common isoforms (ApoE2, ApoE3, and ApoE4) and heterozygous carriers of the
Keywords
Introduction
The human apolipoprotein E (ApoE) is a 35 kDa glycoprotein encoded in three more frequent isoforms (ApoE2, ApoE3, and ApoE4) and one very rare (ApoE3r) isoform. The four variants (ε2, ε3, ε4, and ε3r) of the human
ApoE is involved in modulating synaptic function,5,6 blood–brain barrier (BBB) integrity,7–9 neuronal receptor recycling, 10 and physiological processes (including cytoskeletal assembly and stability, mitochondrial integrity and function, and dendritic morphology and function) and numerous metabolic pathways such as lipid transport, 11 glucose metabolism,12,13 and insulin signaling.14,15
ApoE was first identified in 1973 as a lipoprotein constituent of very-low-density lipoproteins (VLDL),
16
high-density lipoproteins (HDL), and chylomicrons, as it was established that dietary cholesterol altered APOE distribution in plasma. Thus, ApoE keeps the same structure in both VLDL and HDL and plays a pivotal role for cholesterol and other lipid transport, participating in their redistribution to cells and facilitating their internalization in cells. However, while the role of ApoE in lipid pathophysiology may be associated with normal aging, its function in central nervous system (CNS) pathophysiology needs clarification. Nevertheless,
HDL and low-density lipoprotein (LDL) are present in individuals with exceptional longevity and their offspring, but their particle sizes are larger compared with controls.
18
ApoE protein is required for healthy cholesterol metabolism and CNS cholesterol transport. In very old individuals, total ApoE levels in plasma seem to correlate with lower total cholesterol and LDL cholesterol levels, which in turn are correlated with allele ε2.
19
The presence of an abnormal cerebrospinal fluid (CSF) lipid profile and lower capacity to deliver neuronal cholesterol has been related to the ε4 allele.
20
Detrimental
About 25% of total body cholesterol resides in the CNS, playing an important role in synaptic plasticity. 24 Cholesterol metabolism can change with advancing age, and its alteration in the brain may be associated with AD development. 24 In addition, in the hippocampus and cortical areas of AD patients, there is decreased cholesterol compared with age-matched controls. 25 Moreover, modifiable lifestyle factors such as education, alcohol consumption, physical activity, and smoking may attenuate genetic risk for accelerated age-related cognitive decline. The complex interactions between genetics, lifestyle, and cognitive aging may encourage behaviors preserving cognitive health into later life. 26
ApoE is important not only for the pathophysiology of lipid metabolism
27
and CNS, but also for healthy aging and longevity.28–31 Longevity studies focus on lifespan, while healthy aging studies consider healthspan; they are correlated because persons who live long have a tendency to be healthy for much of their lives.
32
Healthy aging can be defined as reaching older age without multimorbidity (i.e. the coexistence of two or more chronic conditions in the same individual) or disabilities, and with intact cognition and/or mobility.
32
The probability of an extreme human lifespan is reduced by the detrimental effects on longevity related to the
Since the early 90s, several studies have pointed out that ApoE could have a central role in AD neurodegeneration. Allele ε4, linked to the ApoE4 isoform, represents a key genetic risk factor for noninherited forms of AD (NIAD),36–39 with a semidominant inheritance.
40
Nevertheless, in NIAD, the ApoE2 isoform might exert a protective effect.36,41–43 Although in
In AD pathogenesis, ApoE binds and transports amyloid β (Aβ) peptides,47–50 with differential affinity for Aβ according to ApoE isoform. Affinity is highest for lipid-associated ApoE4, intermediate for lipid-associated ApoE3, and lowest for lipid-associated ApoE2.51–54 Accordingly, distinct ApoE isoforms may exert different effects on Aβ aggregation and clearance,51,55–59 and also on Aβ production. 60 ApoE also modulates microglial responses to amyloid plaque pathology61,62 and can affect tauopathy and tau-mediated neurodegeneration.63–65
Thus, distinct ApoE isoforms may either increase the risk for AD48,52 or have a protective role, 66 depending on the different effects of fragments of distinct ApoE isoforms on deposition of Aβ.58,67,68 Brain ApoE is indeed present in smaller fragments (ApoE peptides), which are biologically active. 69 It has been shown that the concentration of specific proteolytic fragments of ApoE is increased in AD brain 70 and that some synthetic peptides of ApoE may be neurotoxic.71,72 The impact of ApoE fragments in AD pathogenesis remains unclear. Differences in the proteolytic processing of distinct ApoE isoforms are also under investigation. In the present narrative review, we have considered biochemical studies about APOE proteolysis, focusing on the enzymes involved in such processes and on the fragments produced by each enzyme. Furthermore, we have focused on recent studies about APOE fragments and their role in AD pathogenesis. In addition, we will review the current development status of different anti-ApoE drugs, and will discuss the feasibility of modulating ApoE processing as a new AD therapeutic approach.
Methods
The present was a narrative review article. We performed separate searches in the US National Library of Medicine (PubMed), Medical Literature Analysis and Retrieval System Online (MEDLINE), EMBASE, Scopus, Ovid, and Google Scholar databases to find original articles of interest. The search strategy used in PubMed and MEDLINE and adapted to the other four electronic sources was based upon searches using the following terms to identify risk exposure (apolipoprotein E OR APOE AND fragments AND enzymes) combined with terms to determine the outcomes of interest [Alzheimer’s disease OR dementia AND (pathogenesis OR development OR treatment OR therapeutics OR drugs OR compounds)]. Identified studies were analyzed for additional references of interest. The last search was performed on October 15, 2021. No language restriction was adopted. Two investigators (F.L.V., P.B.) independently and in duplicate searched for articles, screened titles and abstracts of the retrieved articles, reviewed the full texts, and selected articles for their inclusion. The following inclusion criteria were applied: original studies in cell and animal models and humans. Technical reports, letters to the editor, and systematic and narrative review articles were excluded. Data were cross-checked, any discrepancy was discussed, and disagreements were resolved by a third researcher (D.S.).
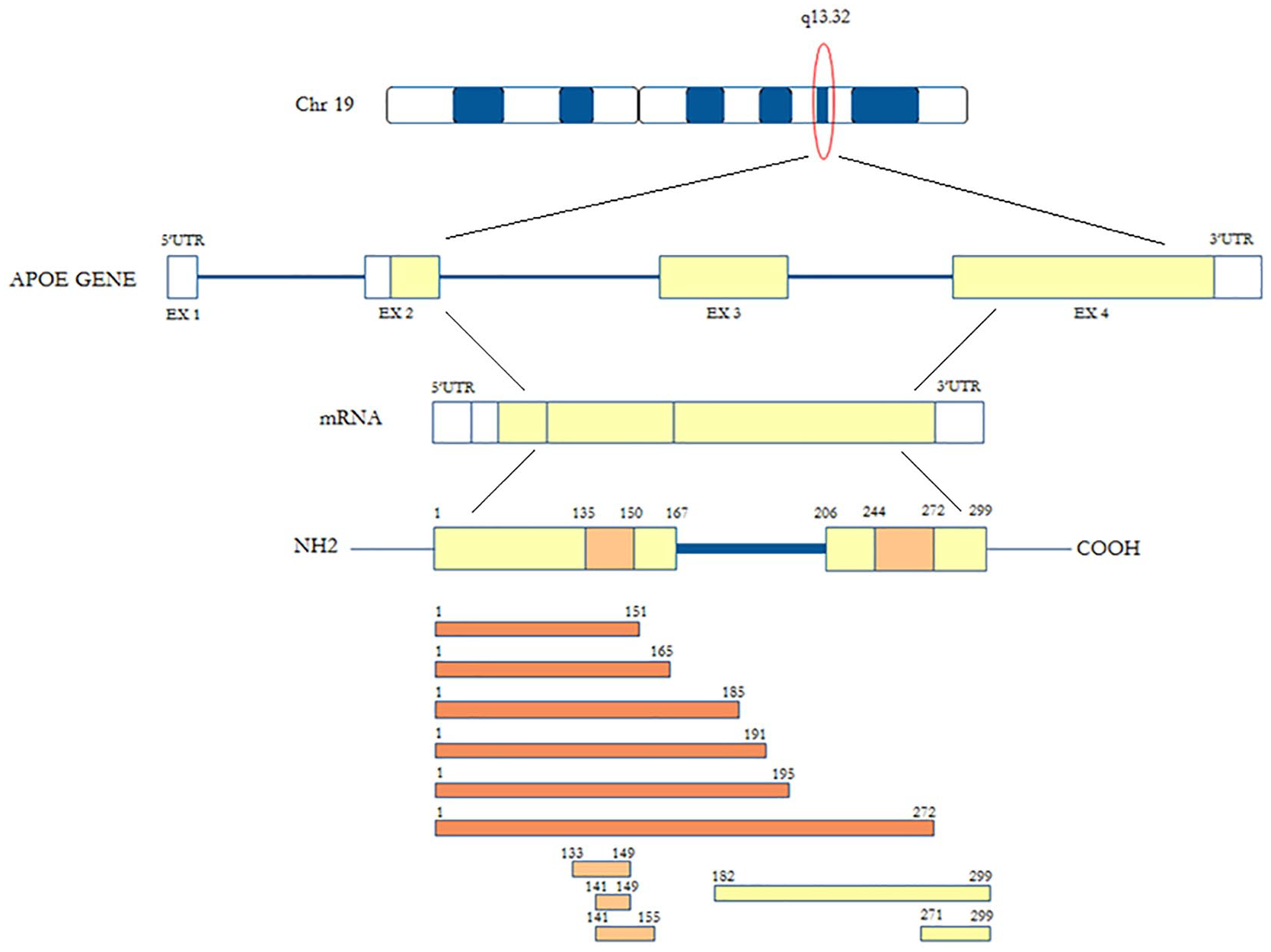
The ApoE structure
Encoded by the
ApoE is synthetized as a preprotein of 317 aa, with a signal peptide of 18 aa removed in the post-translational phase. ApoE is composed of two structural domains that are linked by an unstructured short hinge region. The
The carboxyl-(C)-terminal domain (aa 206-299) is composed of amphipathic α-helices [characteristic of the exchangeable apolipoprotein and contains the lipid-binding region (aa 244-272)]. As previously described, ApoE was identified as a main apolipoprotein/component in HDL particles, VLDL, and chylomicron remnants, being a fundamental ligand in the uptake of these lipoproteins by members of the LDL-R family, except for HDL particles. The structural organization of ApoE in HDL profoundly impacts its ability to regulate cholesterol homeostasis in AD and cardiovascular diseases.
ApoE also serves as the primary cholesterol chaperone in the neuropil and maintains brain cholesterol homeostasis. At residues 141-150, the dual-domain ApoE mimetic peptide Ac-hE18A-NH2 (ligand active site of the protein) co-localizes with a major heparin-binding site. 82 This peptide, the putative receptor-binding region of human ApoE, is covalently linked to a well-characterized class A amphipathic helix, 18A, which has no sequence homology to any other exchangeable apolipoprotein sequences. It demonstrates dramatic ability to reduce plasma cholesterol levels in dyslipidemic mouse and rabbit models. 82
The ApoE3 and ApoE3r structures
The ApoE3 isoform, characterized by the aa combination
Interestingly, the isoform ApoE3r, characterized by the aa combination
The ApoE4 structure
The ApoE4 isoform, the second most common allele, differs from ApoE3 in the change of
The ApoE2 structure
The ApoE2 isoform, the third most common allele, differs from ApoE3 by a single aa substitution
The role of ApoE in AD pathogenesis
The ApoE-cutting enzymes
As seen above, under various physiological and pathological conditions, CNS neurons can express ApoE, even if at lower levels than astrocytes.63,79,80 Indeed, various
Two classes of proteases have so far been suggested as mediators of ApoE fragmentation, targeting
Apolipoprotein E (ApoE) fragments generated by cutting of enzymes.
Thrombin
Thrombin, a serine protease belonging to chymotrypsin family,
115
has a 36-residue polypeptide A chain and a 259-residue B chain, linked by a disulfide bridge at the corresponding
In AD pathophysiology, a role for this enzyme has been suggested,
119
given that AD patients have amyloid deposits, senile plaques, NTFs, and micro-vessels, all containing thrombin. In addition, neuronal cells express prothrombin mRNA,120,121 and it has been observed that PAR-1, PAR-3 and PAR-4 are up-regulated in rat hippocampus.
122
Thrombin cannot process phosphorylated tau protein, resulting in intracellular aggregates of tau protein in hippocampal neurons, but it may induce proteolysis of microtubule-associated tau protein
Cathepsin D
The lysosomal enzyme cathepsin D is implicated in unfolded, unused, and damaged protein degradation and is highly expressed in the brain. Through autophagy and endocytosis processes, these damaged proteins are delivered into lysosomes and degraded. 130 Dysregulation of cathepsin D enzymatic activity may lead to accumulation and aggregation of various proteins, and is involved in several proteinopathies. Cathepsins are proteases including several members differentiating for the aa number in the active site: cathepsins B, C, F, H, K, L, O, S, V, W, and X comprise the cysteine cathepsin family; cathepsins A and G, the serine cathepsin family; while D and E are the aspartyl cathepsin family. All human tissues express cathepsins B, L, H, C and D, while A, G, K, S, V, X and W CatD are tissue-specific and based on cell type.131–137 Preprocathepsin D, the synthetized inactive zymogen, is proteolytically cleaved and cathepsin D is activated; preprocathepsin D contains an N-terminal signal peptide, a propeptide, and a catalytic domain.135–137 Cathepsin D is mainly involved in degradation of proteins in lysosomes, but also has key roles in signal transduction pathways, such as activation of enzymatic precursors, prohormones and growth factors, brain-specific antigen processing, neuronal cell homeostasis, and apoptotic processes.138,139 Cathepsin D is a pathology-specific biomarker involved in neurodegenerative disorders such as AD, but is also implicated in cancer, atherosclerosis, and inflammation.140–143 As cathepsin D might have neuroprotective effects by blocking abnormal tau accumulation, cathepsin D deficiency may induce elevation in C-terminally truncated tau variants, thus promoting neurotoxicity. 144
In human ApoE, 13 cathepsin D cutting-sites are found, which preferentially cleave aromatic
α-Chymotrypsin-like serine protease
α-Chymotrypsin-like serine protease cleaves aromatic and specific hydrophobic aa. ApoE has 67 sites for cleavage by α-chymotrypsin-like serine protease, and in the hinge, region
High-temperature requirement serine protease A1
The HtrA1 belongs to a four-enzyme subfamily of serine proteases in humans.
150
At variance with HtrA2 (which is a mitochondrial transmembrane protein), HtrA1, HtrA3 and HtrA4 have identical regional composition: an insulin-like growth factor–binding protein domain (IGF-BD), a Kazal motif followed by a trypsin-like catalytic domain (KM), and a PDZ region.
137
Although regulation of HtrA1 activity is still under debate, the IGF-BD and PDZ domains are dispensable for its activity.151,152 Indeed, a feature of an elastase-like serine protease activity is the cleavage of ApoE after valine (
The role of ApoE isoforms in fragment generation
The three ApoE isoforms differ in their physical properties, including lipid-binding capacity, type of lipids bound, domain-domain interactions, and stability. The isoform-dependent secretion of ApoE and its immunomodulatory effects could be attributable to post-translational modifications.
156
ApoE3 and ApoE2 are less sensitive to proteolytic cleavage than ApoE4.
157
The greater instability of ApoE4 is due to its presentation of numerous protease-sensitive sites in the hinge region, including to thrombin,
158
cathepsin D,
114
and HtrA1,155,159 and its vulnerable N- and C-terminal domains.
112
In addition, ApoE 14-20 kDa N-terminal fragments
111
and ApoE 10-15 kDa C-terminal fragments
109
have been also shown to be more frequent in AD patients than in age-matched controls. The 4 α-helix bundle of ApoE4 is partially opened and extended, so exposing the hydrophobic surface of the protein. Therefore, proteolytic enzymes may have greater access to the short hinge region
160
and the hydrophobic region, as compared to other isoforms. These differences may account for the lower AD risk in
Recently, it has been shown that the 25 kDa ApoE fragment may be neuroprotective, and is more abundantly produced in brains of
ApoE2 contains a
The role of ApoE peptides in neurodegeneration and AD
The interaction between ApoE and Aβ is influenced by various factors, including the ApoE isoform and its lipidation, and Aβ levels.
168
In healthy brain, Aβ can associate with ApoE, which is involved in its metabolism and clearance. The brains of both cognitively intact controls and AD patients contain full-length ApoE, high molecular weight (HMW-22–30 kDa) and low molecular weight (LMW-10–20 kDa) fragments in varying quantities, although fragments are more frequent in AD brains than in those of controls. Nonetheless, the proportions of the various fragments are similar in both groups, as well as among the various
Low lipidation of ApoE4 favors its binding to Aβ, which may induce generation of ApoE-fragment/Aβ heteromers, increasing Aβ accumulation and AD risk (Figure 2). 173 It is this ApoE4/Aβ interaction that plays a main role in AD development, while each component in isolation seems less critical. 174 ApoE fragments lacking N-terminus and C-terminus, together with Aβ accumulation, may also favor AD pathology. Such ApoE fragments are without the Aβ transporter-binding domain, and formation of ApoE-fragment/Aβ heteromers decelerates Aβ clearance and favors accumulation. ApoE4 is more efficient than other isoforms in favoring ApoE-fragment/Aβ heteromer formation. 170 Furthermore, ApoE can increase formation of Aβ oligomers.169,175 In AD brain, 3 ApoE fragments have been found – 18 kDa (ApoE18), 16 kDa (ApoE16) and 12 kDa (ApoE12) – with the 18 and 16 kDa forms being hybrid heteromers composed by Aβ1-42 peptides and ApoE middle fragments. One study showed that the formation of Aβ/ApoE16 and Aβ/ApoE12 heteromers (but not those involving ApoE18) seems to correlate with memory deficits in AD. 112 Only the ApoE18 fragment was significantly increased, while ApoE16 and ApoE12 fragments were less elevated. 109 Furthermore, the observation of increased hippocampal Aβ/ApoE18 heteromer suggested that it might serve as a biomarker for AD pathology. 112

The role of apolipoprotein E (ApoE) peptides in neurodegeneration and Alzheimer’s disease (AD). In the brain, astrocytes and activated microglia synthesize ApoE. In neurons, ApoE is cleaved in the C-terminal domain that binds to amyloid β (Aβ) and localizes to plaques; and in the N-terminal domain that localizes with NFTs. Low ApoE lipidation influences the binding of ApoE4 to Aβ, promoting ApoE-fragment/Aβ heteromer generation. ApoE fragments without N- and C-terminal domains (ApoE 133-149, ApoE 141-149, ApoE 141-155, 12 kDa fragment) do not have the Aβ transporter-binding domain – these fragments (together with Aβ accumulation) favor the formation of ApoE-fragment/Aβ heteromers, decelerating Aβ clearance and favoring Aβ accumulation in AD. The lipidated form of ApoE interacts with NFTs through its amino acid residues 245-260. Imbalance between the activities of tau protein kinases and phosphatases promotes accumulation of tau protein in neurons. C-terminal truncated ApoE fragments induce oxidative stress and inflammation, releasing pro-inflammatory cytokines. Increased MMP-9 leads to β-site amyloid precursor protein–cleaving enzyme-1 (BACE-1) activation and/or apoptosis, and interleukin (IL)-1β interacting with neurons by LDL-related protein receptor induces NFT formation.
ApoE mediates clearance of Aβ-protein from the neuropil by acting as a bridging protein between Aβ, via its C-terminal domain, and LRP, via its N-terminal domain.
176
A study in senile plaques showed that the N-terminal/C-terminal domain interaction is stronger in ApoE4 than in ApoE3. ApoE4, therefore, has a shorter inter-terminal domain distance, but a relatively longer and more exposed hinge, which is more susceptible to proteolysis. Increased hinge proteolysis in ApoE4 leads to more disassociated Aβ-bound C-terminal fragments. These events contribute to the loss of Aβ clearance function in brains with ApoE4, enhancing amyloid deposition,
169
given also that intracellular clearance of Aβ peptides is correlated with the
ApoE fragmentation presents as a potential AD-related pathological process (Figure 2). C-terminal truncated forms of ApoE induce intracellular NFT-like inclusions in neurons.70,157,177 ApoE fragments associate with or induce the formation of hyperphosphorylated tau and NFTs in an isoform-dependent manner. The lipidated form can also induce NFT formation, both
The ApoE receptor-binding N-terminal domain peptides
The potential neurodegenerative effects of ApoE N-terminal fragments are summarized in Table 1. In the BV2 mouse microglial cell line, the 17 kDa fragment has been reported to promote cell death.
178
It has been reported that intracellular Aβ1-42 accumulation is stimulated by the 19 kDa fragment, which produces reactive oxygen species (ROS) in the SK-N-SH human neuroblastoma cell line.74,179 In this cell line and SW-1783 human astrocytoma cells, the 21 kDa fragment has been also reported to promote MMP9/TIMP1 imbalance, by stimulating IL-1β and reducing IL-10 levels.
180
The 22 kDa thrombin cleavage ApoE4 fragment is also neurotoxic
Effect of apolipoprotein E (ApoE) N-terminal peptides in cell and animal models related to Alzheimer’s disease (AD) and differences in ApoE isoforms.

1Gene ID: 11816.
2Gene ID: 25728.
3Gene ID: 348.
Aβ, amyloid β; MMP9, matrix metalloproteinase 9; NFT, neurofibrillary tangle; ROS, reactive oxygen species; TIMP1, tissue inhibitor of metalloproteinase 1.
The 22 kDa fragments originated by thrombin cleavage lack the aa 244-272 lipid-binding site and are different from ApoE fragments generated in AD brains.
70
In contrast, this site seems to be essential for fragment-induced neurotoxic effects
The ApoE central domain peptides
The potential effects of the ApoE central fragments in neurodegeneration are summarized in Table 2. In BV2-microglia cells, release of tumor necrosis factor-α (TNF-α) and nitric oxide (iNOS) is induced by the 133-149 aa peptide by suppression of microglial activation.
184
This peptide also reduces inflammation response after lipopolysaccharide (LPS) injection in C57BL/6 J blastocysts,
185
suppressing the apoptosis and intracellular calcium increase caused by
Effect of apolipoprotein E (ApoE) peptides from central protein domain in cell and animal models related to Alzheimer’s disease (AD) and differences in ApoE isoforms.

Gene ID: -
Gene ID: 348.
Gene ID: 25728.
Gene ID: 394678.
LPS, lipopolysaccharide; nAChR, nicotinic acetylcholine receptor; NMDA,
The ApoE lipid-binding C-terminal domain peptides
The potential neurodegenerative effects of ApoE peptides originating from the C-terminal protein domain are summarized in Table 3. The C-terminal truncated ApoE fragment neither affects APP expression nor APP processing, but induces formation of NFTs. The C-terminal-derived fragment can stabilize Aβ to form pathogenic Aβ oligomers, but not Aβ fibrils. Furthermore, C-terminal fragments induce oxidative stress and inflammation, stimulating neuronal secretion of the pro-inflammatory cytokines IL-1β and MMP-9. Increased MMP-9 leads to BACE-1 activation and/or apoptosis, and IL-1β promotes NFT formation by interacting with neurons via the LRP receptor. In addition to aberrant activation or repression of tau kinases and phosphatases, the third α-helix of the C-terminal fragment is involved in abnormal tau phosphorylation and formation of NFT-like inclusions. Inclusions similar to NFTs containing the 272-299 aa fragment (also designated as 1-271 aa fragment) 70 are found in neuronal cells, and may cause AD-like tau pathology and behavioral disturbances in Neuro 2-a mouse neuroblastoma cells of transgenic mice C57BL/6 J overexpressing this peptide. 157 In Neuro-2a cells, removal of the first 20 aa, up to the first α-helix, of ApoE4 (272-299) does not modify its ability to induce intracellular NFT-like inclusions. Only a further truncation of its third α-helix decreases the induction of NFT-like inclusions by ∼50%. 70
Effect of apolipoprotein E (ApoE) C-terminal peptides in cell and animal models related to Alzheimer’s disease (AD) and differences in ApoE isoforms.

Gene ID: 11816.
Gene ID: 348.
Aβ, amyloid β.
In transgenic mice hAPPFA, overexpressing 272-299 aa peptide, there was lower Aβ clearance. 108 In Neuro-2a mouse neuroblastoma cells, production and stabilization of hexameric Aβ species may be induced by an ApoE C-terminal 13 kDa fragment that may also inhibit Aβ fibril formation. 190 ApoE fragments escape the secretory and endocytic–lysosome internalization pathways to induce the formation of NFTs and/or to associate with them. In Neuro-2a cells, the neurotoxic ApoE4 fragment is not found in either the Golgi apparatus or endoplasmic reticulum. In contrast, the fragment either interacts with the mitochondria or cytoskeletal elements to form filamentous inclusions containing phosphorylated tau and phosphorylated neurofilament proteins. 182 The mechanisms underlying this ability to evade the recycling pathway remain unknown.
It has been proposed that the LDL receptor–binding domain directs the C-terminal domain in initiating rapid lipid binding, followed by a slower N-terminal domain helix bundle opening, to yield discoidal reconstituted HDL. It appears that the bulky N-terminal domain determines the spatial organization of its C-terminal domain in reconstituted HDL, a finding that has significance for ApoE4, which is more susceptible to proteolytic cleavage in AD brains. 191
The ApoE mimetic peptides
In nature, potentially therapeutic peptides are present as 100 aa short-chain monomers. Such peptides may bind certain membrane receptors and activate specific signaling pathways. 192 Historically, the use of peptides as therapeutic agents has been ignored due to several limitations including dimension, degradation susceptibility, absence of effective delivery methods, fast excretion, poor distribution, low oral bioavailability, reduced cell permeability, and target specificity. 193 In 1953, oxytocin was the first therapeutic peptide synthesized. In 1982, recombinant human insulin was the first such peptide produced through recombinant fermentation. 194 Transformation of peptides into mimetic peptides is one intriguing strategy for using them as therapeutic agents. Mimetic peptides exert effects similar to those of the original molecule, but promise several advantages: increased structural consistency, good cell membrane permeability, and increased stability to proteolytic digestion and target specificity. Peptides have been chemically modified to induce non-natural aa changes, amide bond variations, scaffold rigidity, or hydrophobic residue inclusion.192,193,195
Mimetic peptide studies have been inspired by lipid-binding domains shared from apolipoproteins with a common structure to these mimetic peptides.196,197 Lipoprotein binding activity and clearance have been tested for peptides spanning the ApoE 130-169 region.71,198 Amphipathic α-helices within the C-terminal region are essential to its efflux capacity.
199
The ApoE mimetic peptide (ATI-5361) has been developed on the basis of a 26 aa peptide including residues 238-266.200,201 A peptide composed of a dimer of residues 141-155 with an N-terminal tyrosine residue, designated Y (141-155)2, binds to the LDL receptor. This latter peptide, upon acetylation of its N
The ATP-binding cassette transporter A1 (ABCA1) belongs to a large family of such transporters. It is localized on the plasma membrane of several cells where it mediates reverse cholesterol transport and (in particular) cholesterol and phospholipids efflux to extracellular apolipoprotein acceptors, such as ApoA-I, ApoE and small, lipid-poor HDL particles.199,203 This cholesterol efflux function is vital to the prevention of degenerative diseases, since intracellular cholesterol accumulation may have cytotoxic and inflammatory effects, and may induce structural and functional alteration in the plasma membrane.204,205 ABCA1 lipid efflux activity optimization may also be important in protecting against AD.206,207 ApoE lipidation and Aβ clearance could be facilitated by ABCA1 in brain cells208,209 and may protect against cognitive decline, particularly in the presence of ApoE4 phenotype.
209
This observation suggests that therapeutic strategies aimed at targeting ABCA1 with a small peptide might be beneficial to AD patients carrying the
Anti-ApoE therapeutics in AD
The complete absence of ApoE caused by a rare ablative
ApoE2 overexpression
Gene therapy methods may be a strategy to overexpress ApoE2 in
Downregulation of expression of ApoE4
In APP/PS1 transgenic mice homozygous for the
Anti-ApoE immunotherapy
The use of passive immunization targeting certain ApoE isoforms may be an alternative therapeutic strategy. The anti-ApoE4 monoclonal antibody 9D11 bound specifically to brain ApoE4 and not ApoE3. 221 For this monoclonal antibody, a direct intracerebroventricular application prevented ApoE4-driven accumulation of Aβ in hippocampal neurons, while repeated intraperitoneal injections of 9D11 in ApoE4 mice resulted in the formation of ApoE/IgG complexes, associated with reversal of cognitive impairments and ApoE4-driven pathologies, including hyperphosphorylated tau. 221 In APP/PS1 mice, the monoclonal antibody HJ6.3 delivered peripherally to either before or after plaque deposition reduced Aβ levels and fibrillary amyloid pathology, increased microglial activation, and improved spatial-memory performance. 222 Moreover, intraperitoneal HJ6.3 (10 mg/kg/week for 21 weeks) affected Aβ plaques, neuronal network function, and behavior in 7-month-old APP/PS1 mice after plaque onset. 223 In mice producing human ApoE3 and ApoE, central or peripheral administration of 4HAE-4, a monoclonal antibody that targets aggregated ApoE3 and ApoE4, reduced fibrillary amyloid plaque load without reducing cerebral or plasma ApoE levels. 224 Furthermore, this monoclonal antibody increased peri-plaque Aβ phagocytosis, 224 reduced cerebral amyloid angiopathy and amyloid plaque load, and improved cerebrovascular function. 225
ApoE mimetics
As previously discussed, in mouse models of amyloid pathology, treatment with peptides that mimic the structural and biological properties of native ApoE reduces Aβ deposition,226,227 tau hyperphosphorylation, 228 and glial activation.226–228 In male transgenic APP/PS1/APOETR mice, treatment with the ApoE mimetic CN-105 for 40 days beginning at 14–18 weeks old reduced Aβ pathology and rescued memory deficits. 229 Notably, delaying treatment onset to 25–28 weeks produced a less robust effect. At present, in 201 subjects with perioperative neurocognitive disorders, CN-105 is being tested in a double-blind, placebo-controlled study (NCT03802396). 230
ApoE4 structure correctors
The pathological conformation of ApoE4 may result from an interaction between its amino-terminal and carboxy-terminal domains,231,232 and several small organic molecules designed to block this interaction have been tested
Conversion of the ApoE4 amino acid sequence into that of ApoE3 or ApoE2
As the three ApoE isoforms only differ structurally by two aa residues, conversion of the ApoE4 sequence into that of ApoE3 or ApoE2 has been considered a logical approach to attenuating its neurotoxic effects. Indeed, conversion of
Inhibition of the ApoE-Aβ interaction
The synthetic peptide Aβ12-28P, which is homologous to the ApoE-binding site on the full-length Aβ molecule, inhibiting the ApoE-Aβ interaction reduced Aβ deposition25,238,239 and insoluble tau accumulation
25
in AD mouse models, and intraneuronal Aβ accumulation
240
in
Challenges of ApoE targeting therapeutic approaches
Given the poor clinical results obtained so far in AD with both anti-Aβ and anti-tau drugs, 242 anti-ApoE therapeutics represent a promising alternative approach. The most promising ApoE modulation approaches appear to be increasing ApoE2 expression or increasing ApoE4 clearance by immunotherapy. However, it is not clear whether chronic treatment with anti-ApoE drugs will be limited by the physiological role of this important glycoprotein in cytoskeletal assembly and stability, mitochondrial integrity and function, and dendritic morphology and function. 213 Indeed, clinical failures of unselective anti-Aβ and anti-tau therapies could be explained by their clearing effects of physiological forms of Aβ and tau protein. 243 Ideally, anti-ApoE therapeutics should attack the pathological forms of ApoE without interfering with its physiological functions.
Conclusion
Repeated studies on ApoE and
Recently, a catabolic pathway has been discovered, in which ApoE undergoes a proteolytic process to produce active peptides with neurotoxic or neuroprotective effects. This finding sheds new light on the possible role of ApoE and its peptides, particularly in AD. The influence that ApoE peptides may have in AD pathogenesis is currently under investigation, while their roles in the transport and metabolism of cholesterol and phospholipids are largely unknown. This may be due to ApoE catabolism being restricted to the CNS, as only astrocytes express the enzymes producing ApoE fragmentation. 159
Given the different roles of ApoE isoforms in AD, the relationship between these and ApoE cutting enzymes is also of interest. Production of ApoE peptides depends on the isoform, as distinct effects of ApoE fragments on CNS cells have been suggested. Several studies have investigated the impact of ApoE4-derived peptides, which may promote neurotoxicity, mitochondrial dysfunction, phosphorylation of tau protein, NFT-like inclusions, neurodegeneration, neuronal apoptosis or suppress microglial activation.
245
Mitochondrial dysfunction in AD also varies with
Understanding the differences between ApoE isoforms in different cell types will undoubtedly involve use of induced pluripotent stem cells that express physiological levels of endogenous genes, rather than relying on overexpression models.
159
Recent studies have shown the role of ApoE4 in AD neuropathology
Footnotes
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was fully supported by ‘Ministero della Salute’, I.R.C.C.S. Research Program, Ricerca Corrente 2018–2020, Linea n. 2 ‘Meccanismi genetici, predizione e terapie innovative delle malattie complesse’ and by the ‘5 x 1000’ voluntary contribution to the Fondazione I.R.C.C.S. Ospedale ‘Casa Sollievo della Sofferenza’.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
