Abstract
Objectives:
To evaluate the effectiveness of any form of physiotherapy intervention for the management of central neuropathic pain (cNeP) due to any underlying cause.
Methods:
Multiple databases were searched from inception until August 2021. Randomised controlled trials evaluating physiotherapy interventions compared to a control condition on pain among people with cNeP were included. Methodological quality and the quality of evidence were assessed using the Physiotherapy Evidence Database Scale and the Grading of Recommendations, Assessment, Development, and Evaluation tool, respectively.
Results:
The searches yielded 2661 studies, of which 23 randomised controlled trials met the inclusion criteria and were included in the meta-analyses. Meta-analyses of trials examining non-invasive neurostimulation revealed significant reductions in pain severity due to spinal cord injury (SCI; standardised mean difference (SMD): −0.59 (95% confidence interval [CI]: −1.07, −0.11),
Conclusion:
Evidence supports the use of non-invasive neurostimulation for the treatment of pain secondary to SCI and phantom limb pain. Beneficial pain management outcomes were also identified for acupuncture in stroke, TENS in multiple sclerosis, and mirror therapy in phantom limb pain.
Introduction
The International Association for the Study of Pain (IASP) defines neuropathic pain as pain caused by lesions or diseases of the somatosensory system. 1 Neuropathic pain describes the continuous shooting pains caused by endogenous chemicals in the body. 2 Pain initiated by lesions of the central nervous system, including the brain, brainstem, and spinal cord, has been categorised as central neuropathic pain (cNeP), 3 which is characterised by a throbbing clinical presentation and sensory impairments, manifested as the absolute or partial decline in sensory responses, resulting in pain, paraesthesia, and dysesthesia. 4 Stroke, traumatic brain injury, spinal cord injury (SCI), 5 and multiple sclerosis are common causes of cNeP. 6 cNeP is not uncommon, with 8% of stroke patients, 7 65% to 80% of individuals with SCI, 3 and 50% of multiple sclerosis patients 8 reporting pain. Due to the chronic nature of cNeP, those individuals diagnosed with neuropathic pain are among the most frequent consumers of healthcare services. 9 The annual per-patient indirect medical costs associated with neuropathic pain have been estimated at USD 19,000. 10 In addition, individuals with neuropathic pain experienced a decline in quality of life due to the necessity of increased drug prescriptions and regular visits to healthcare providers. 10
cNeP is commonly managed by pharmacotherapy, surgery, and non-surgical interventions. 3 Among existing non-surgical interventions, motor cortex stimulation, deep brain stimulation, and repetitive transcranial magnetic stimulation (rTMS) have gained interest.11,12 These neuromodulation techniques are thought to increase blood flow to the cingulate gyrus, reducing emotional affective pain, 13 increase the release of endogenous opioids, 13 and activation of pain inhibitory pathways. 14 Physiotherapy modalities used to manage cNeP include the application of heat and cold, massage, high-frequency currents (short-wave diathermy), low-frequency currents (such as transcutaneous electrical nerve stimulation (TENS)), high-voltage galvanic currents, and laser therapy. 12 These interventions have been tested across a spectrum of conditions associated with cNeP; however, the results remain inconclusive. 15 Rehabilitative interventions, such as psychotherapy, 16 relaxation therapy, 17 mirror therapy, 18 and graded motor imagery19,20 are considered to be useful adjuncts to pharmacotherapy, aiming to address the emotional, behavioural, and mental domains associated with pain. Unlike pharmacotherapeutic interventions, physiotherapy and rehabilitation interventions for neuropathic pain are less toxic and are often easily accessible.
Previously published reviews examining this field of research have restricted their focus to either one specific disease condition,21,22 or one specific treatment modality for the management of neuropathic pain.23,24 To the best of our knowledge, there are no previous systematic or Cochrane reviews on the efficacy of physiotherapy interventions for the management of cNeP due to any underlying cause. In 2020, a systematic review evaluated the effectiveness of pharmacological and non-pharmacological interventions, including non-invasive brain stimulation, TENS, invasive neurostimulation psychotherapy, and hypnosis, for the treatment of central and peripheral neuropathic pain. However, this review did perform any meta-analysis. 25 Therefore, the objective of the current systematic review and meta-analysis was to evaluate the effectiveness of any form of physiotherapy intervention for the management of cNeP due to any underlying cause.
Materials and methods
Search strategy
This systematic review was developed and is reported in accordance with the Preferred Reporting Items for Systematic review and Meta-Analyses (PRISMA) guidelines. 26 The review was registered with the International Prospective Register of Systematic Reviews (PROSPERO) before the search was initiated (CRD42020175111). The following databases were searched, from database inception to August 2021: Web of Science, Embase, EBSCO, MEDLINE, and CINAHL. Search terms were constructed as four themes, which included (1) diseases associated with neuropathic pain, (2) physiotherapy interventions, (3) outcome measures evaluating pain, and (4) randomised, controlled trial (RCT). The disease conditions that were included in the searches were based on the current grading system for neuropathic pain. 27 The search strategy for Medline is presented in Supplementary Appendix 1. The reference lists of all included studies and relevant systematic reviews were also manually searched. RCTs, pilot, cluster RCTs, cross-over trials (providing data prior to cross-over), and unpublished theses that compared any form of physiotherapy interventions against a control condition (no treatment, placebo, sham, or active control) for the management of cNeP associated with any underlying cause were included in the review. Trials that utilised the visual analogue scale (VAS) or numerical rating scale (NRS) to measure pain were included in the review. Trials published in languages other than English were also included in the review. Conference abstracts without full-text and quasi-experimental designs were excluded. Trials in which physiotherapy interventions were not delivered or supervised by physiotherapists were excluded. In this review, the usual care control was considered an active control.
Screening process
All identified trials were subjected to a four-step screening process. Duplicates were removed and titles were screened by one reviewer (UB). The abstract and full-text screening was conducted by two reviewers (SW and UB). Discrepancies were resolved by discussion until consensus was reached. If consensus was not reached between the two reviewers (SW and UB), a third reviewer (PK) was consulted. At the full-text screening level, we used the International Classification of Diseases (ICD)-11 classification model for cNeP to categorise the studies into central and peripheral neuropathic pain. 6 In this review, we considered cNeP secondary to SCI, post-stroke pain, and multiple sclerosis. We have included interventions for phantom limb pain, which is categorised as a type of neuropathic pain,27 –29 with a strong inclination towards central pain components. 30 The authors of the included trials were approached to obtain additional information if not reported in the publication.
Methodological quality and the quality of evidence
The methodological quality of all included trials was assessed using the Physiotherapy Evidence Database scale, and the quality of evidence using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) tool. PEDro scores for the included trials were obtained from the PEDro website (https://www.pedro.org.au/). If the score was not available, two independent reviewers (PK and UB) scored the methodological quality across the 10 items of the PEDro scale. Studies scoring 6 or above were considered high-quality studies, whereas studies scoring less than or equal to 5 were considered low-quality. 31
The GRADE quality of evidence was rated using the GRADE profiler software 3.6.1 (http://tech.cochrane.org/revman/other-resources/gradepro/download), for each intervention specific to the outcome measure, and was completed by one reviewer (PK). The quality of evidence was classified according to four levels: ‘very low’, ‘low’, ‘moderate’, or ‘high’. 32 The overall quality of evidence was based on the lowest quality of evidence for the outcome. 33 According to the GRADE system, evidence derived from RCTs is considered high-quality; however, the quality was downgraded for several reasons, including study limitations (risk of bias); the inconsistency of findings; the indirectness of evidence; imprecision; and reporting or publication bias. 32
Data extraction and analysis
Two independent reviewers (SW and PK) were involved in data extraction. Discrepancies were resolved by discussion. The following data were extracted from all included studies: (1) author and year of publication; (2) population, recruitment setting, country, language, and sample size; 34 (3) intervention(s) and the intervention dosage; (4) assessment time points; and (5) pre- and post-treatment means.
Meta-analyses were conducted using the Comprehensive Meta-Analysis (CMA) software version 3.0 (Biostat Inc., Englewood, New Jersey, USA). Trials of similar underlying causes for cNeP, interventions, outcome measures, and time points were grouped for pooling. A meta-analysis was planned if at least two trials evaluated an intervention for cNeP of a similar underlying cause. Pre- and post-intervention data were used to obtain the pooled estimates of differences between groups. Study authors were contacted in an attempt to obtain any missing data for the included studies. Data presented in graphical formats were extracted using the GetData Graph Digitizer 2.26 (http://getdata-graph-digitizer.com/).
35
Data reported as the median and range were converted to the mean and standard deviation, as described elsewhere.
36
To obtain pooled estimates of the differences between groups and associated 95% confidence intervals (CIs), the bias-adjusted, standardised, mean difference (SMD; Hedges’s g) or differences in means (weighted mean difference, WMD) were analysed. WMD was used to pool the effect of homogeneous studies that adopted similar outcome measures. The chi-square test (
Results
Figure 1 provides an overview of the search and selection process. The searches yielded 2661 studies. Twenty-three randomised controlled trials met the inclusion criteria and were included in the meta-analyses.
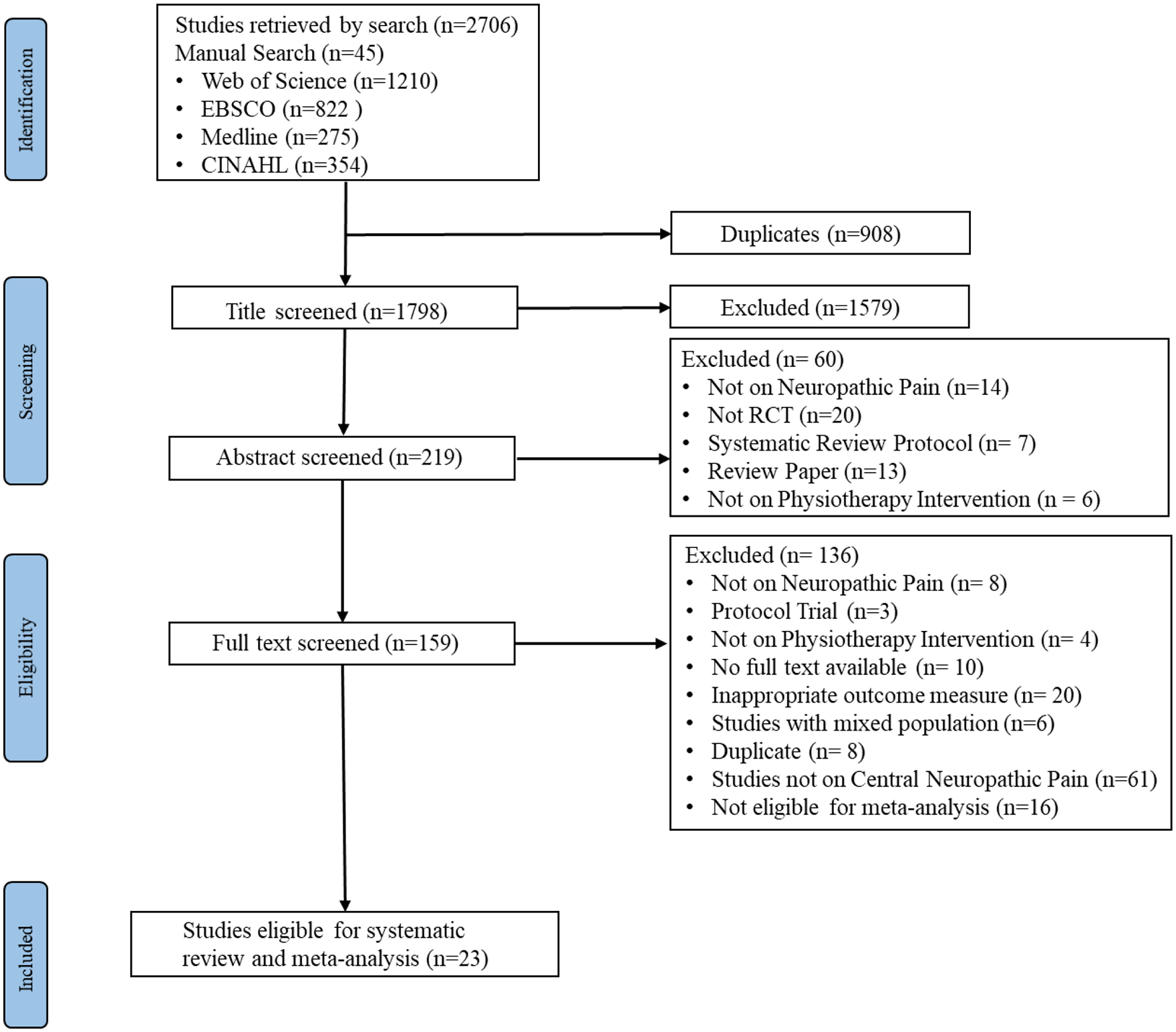
Flow of study process.
Characteristics of the included studies
The characteristics of each included study are presented in Table 1. The trials included in the review were published between 2003 and 2021. Among the included trials, cNeP was secondary to SCI (
Summary of the included studies (

Con, control; Exp, experimental; F, female; M, male; n/s, not stated; NRS, numerical rating scale; SD, standard deviation; SE, standard error; SEM, standard error of the mean; VAS, visual analogue scale.
Quality
The GRADE qualities of the 23 trials included in the meta-analyses ranged from ‘very low’ to ‘moderate’ (Table 2). The mean PEDro score of the trials included in the meta-analysis was 7, with scores ranging from 4 to 9. The PEDro quality scores of all included trials are reported in Table 3.
Summary of the findings for the effectiveness of interventions compared to control.

CI, confidence interval; cNeP, central neuropathic pain; GRADE, Grading of Recommendations, Assessment, Development, and Evaluation; NRS, numerical rating scale; TENS, transcutaneous electrical nerve stimulation; VAS, visual analogue scale.
Pain measured with 0–10 VAS or NRS, with lower scores indicating a better outcome.
The corresponding risk (and 95% confidence interval (CI)) is based on the assumed risk in the comparison group and the relative effect of the intervention (and 95% CI).
Lack of allocation concealment in six38 –42,45 out of seven studies pooled in the meta-analysis; lack of therapist blinding;43,45 and lack of assessor blinding. 39
Wide CI/ no overlap in CI
Lack of allocation concealment, lack of intention-to-treat (ITT)-based analysis, and loss to follow-up >15%; 53 lack of therapist blinding;53,54 lack of assessor blinding. 54
Evidence of clinical/methodological heterogeneity (I 2 > 50%)
Lack of random allocation, 55 insufficient information about the allocation concealment and lack of therapist and assessor blinding. 56
Strength of association
Lack of allocation concealment in all four studies pooled in the meta-analysis;46 –49 lack of therapist blinding47 –49 and assessor blinding; 49 and analysis not based on ITT.46,47,49
Lack of allocation concealment in two50,52 out of three studies pooled in the meta-analysis; lack of subject blinding,50 –52 therapist blinding50,51 and accessor blinding. 50
Lack of allocation concealment and assessor blinding in both studies pooled in the meta-analysis.59,60
Lack of allocation concealment; 57 lack of subject blinding, 58 therapist blinding and accessor blinding. 57
Summary of methodological quality of the included studies according to the PEDro scale (

Effectiveness of interventions
Non-invasive neurostimulation for cNeP, secondary to SCI
Eight trials evaluated the effectiveness of non-invasive neurostimulation on cNeP secondary to SCI. Neurostimulation was provided through 5–20 sessions of rTMS at a frequency of 5–10 Hz,38,41,43 tDCS at an intensity of 2 mA,
39
with a ramp on for 30 s and ramp off for 8 s;
40
and cranial electrotherapy, with a current intensity of 100 mA.
42
The methodological quality of the eight trials ranged from low to high and the GRADE quality of the evidence was very low. Pooled analysis of the eight trials (n = 152) showed a significant benefit of non-invasive neurostimulation for reducing pain severity compared to control (SMD – 0.59 (95% CI: –1.07 to –0.11);
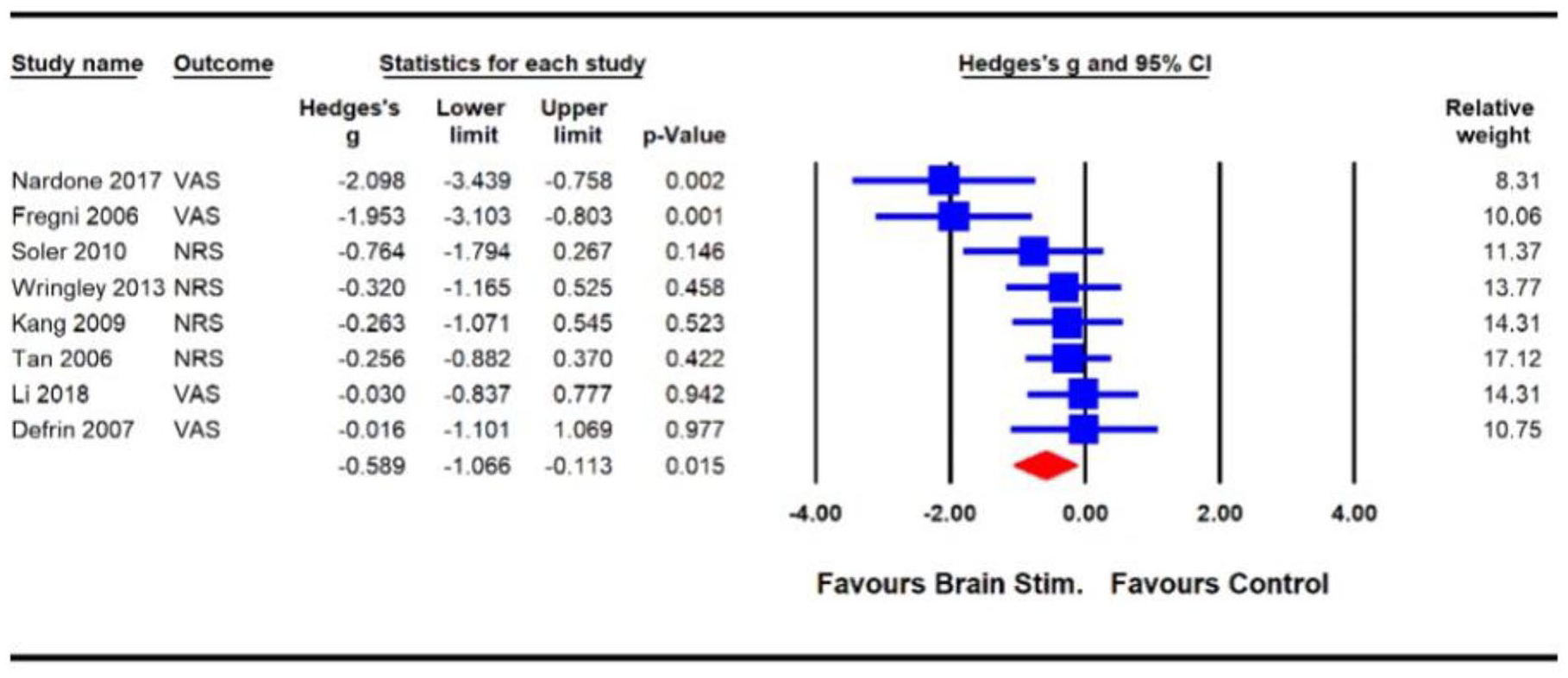
Effects of non-invasive neurostimulation on pain severity in individuals with spinal cord injury.
Non-invasive neurostimulation for cNeP secondary to stroke
Two trials53,54 evaluated the effectiveness of non-invasive neurostimulation on cNeP secondary to stroke. Both trials used rTMS to treat pain following stroke. rTMS was provided for 10 treatment sessions at a frequency of 10 Hz.53,54 Intensity of rTMS was set at 90%
53
% and 120%
54
of the resting motor threshold. Each train of stimulation lasted for 5 s, for a total of 20
53
and 25
54
trains of stimulation. The methodological quality of the two trials ranged from low to high and GRADE quality of evidence was very low. Pooled analysis of the two trials (n = 47) showed a non-significant effect of non-invasive neurostimulation (rTMS) for reducing pain compared to control (SMD –0.7 (95% CI: –2.09 to 0.69);

Effects of non-invasive neurostimulation on pain severity in individuals with stroke.
Acupuncture for cNeP secondary to stroke
Two trials55,56 evaluated the effectiveness of acupuncture for cNeP secondary to stroke. Both trials used acupuncture to treat shoulder pain following stroke. Acupuncture was provided in 9
55
and 18
56
treatment sessions using ten stainless steel needles (40 mm length and 0.25 mm diameter) to various acupoints (LI15, LI14, LI16, LI4, TE14, TE3, SI10, SI13, GB20, and ST36). Needles were inserted to a depth of 15–35 mm, retained for over 15 minutes during each treatment session, and manipulated until De Qi was elicited. The methodological quality of the two trials was high and the GRADE quality of the evidence was moderate. Pooled analysis (
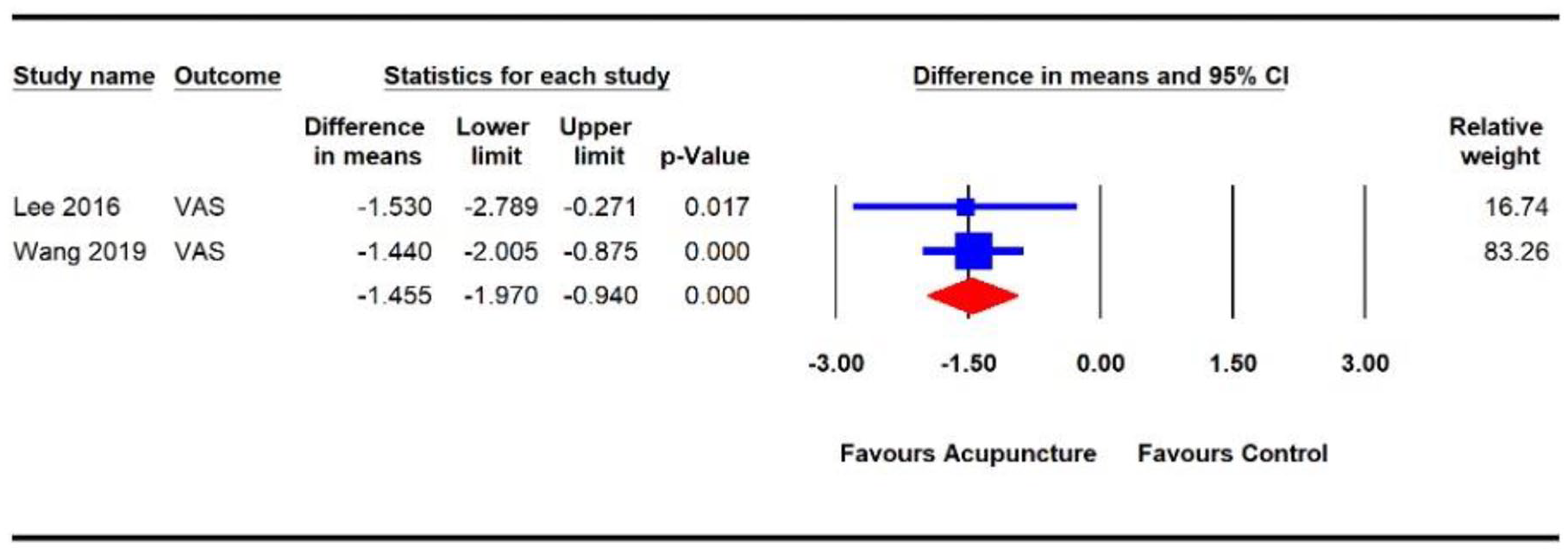
Effects of acupuncture on pain severity in people with stroke.
TENS for cNeP, secondary to multiple sclerosis
The effectiveness of TENS for cNeP secondary to multiple sclerosis was evaluated in four trials46
–49 (including six comparisons). In the four trials, TENS was delivered at frequencies of 4–110 Hz, with intensity ranging between 40 µs and 0.125 ms, for 20–60 mins over 12–24 treatment sessions.46
–49 The methodological quality of the four trials ranged from low to high and the GRADE quality of the evidence was very low. Pooled analysis (

Effects of TENs on pain severity in individuals with multiple sclerosis.
Exercise for cNeP, secondary to multiple sclerosis
The effects of different forms of exercise on cNeP secondary to multiple sclerosis was evaluated in three trials.50
–52 Among these three trials, exercises were delivered for 3 months (90 sessions of progressive muscle relaxation),
50
20 weeks (40 sessions of Ai-chi: Hydrotherapy)
51
or 5 weeks (15 sessions of strengthening, stretching, endurance, and balance exercises for 30 minutes each session).
52
The methodological quality of the three trials was high, and the GRADE quality of the evidence was moderate. The pooled analysis (
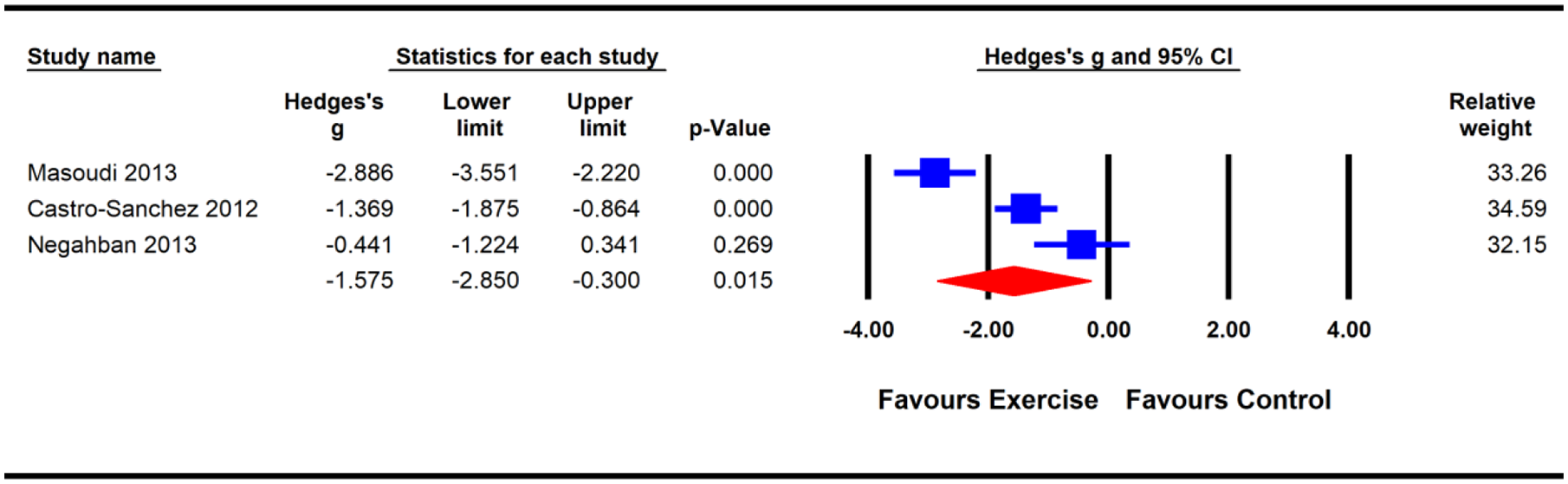
Effects of exercise on pain severity in individuals with multiple sclerosis.
Non-invasive neurostimulation for phantom limb pain
Two trials59,60 evaluated the effectiveness of non-invasive neurostimulation for phantom limb pain. The trials delivered rTMS at a frequency of 10 Hz,
59
and tDCS, at an intensity of 2 mA.
60
The methodological quality of the two trials was high and the GRADE quality of the evidence was low. The pooled analysis showed a significant benefit of non-invasive neurostimulation for reducing pain compared with controls (WMD –1.57 (95% CI: –2.85 to –0.29);
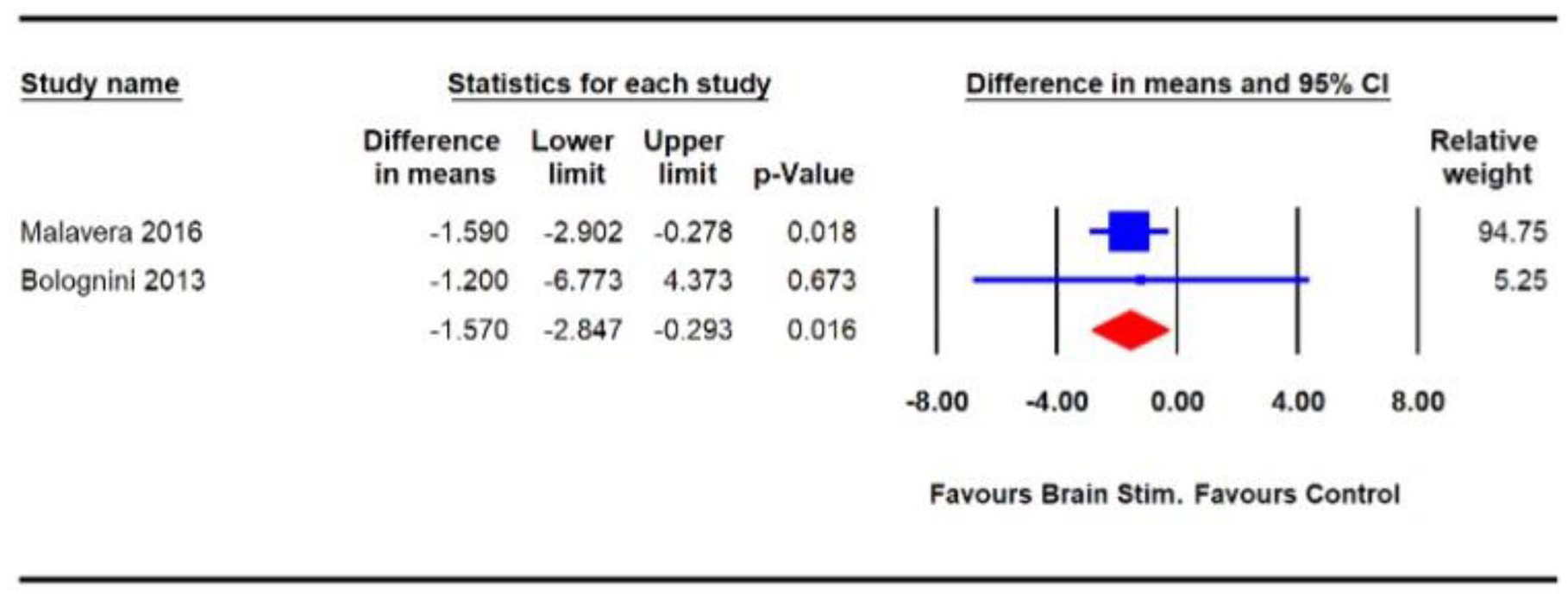
Effects of non-invasive neurostimulation on pain severity in individuals with phantom limb pain.
Mirror therapy for phantom limb pain
The effect of mirror therapy on cNeP among people with phantom limb pain was evaluated in two trials.57,58 One
57
of the two trials delivered mirror therapy in 15-minute sessions for a total of 20 sessions over the course of 4 weeks. The other trial
58
provided mirror therapy in 20-minute sessions for 4 consecutive days. The methodological quality of the two trials was high, and the GRADE quality of evidence was low. The pooled analysis (
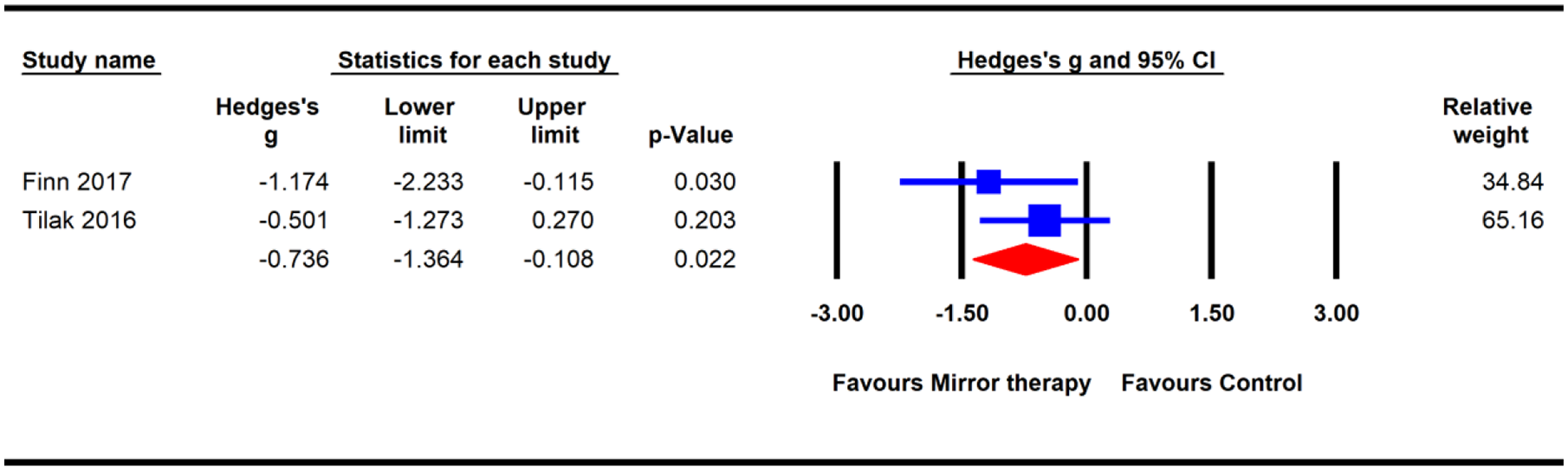
Effects of mirror therapy on pain severity in individuals with phantom limb pain.
Discussion
The current review identified significant reductions in pain severity in response to several physiotherapy interventions among individuals with cNeP resulting from various neurological conditions. Whether the meta-analysis-derived estimates for the average effects on pain and their confidence intervals represent clinically insignificant or clinically important effects must be carefully interpreted for each specific intervention, depending on the precise conditions to which they were applied, due to variations in the sizes and quality of trials and the parameters of the various interventions.
Different forms of non-invasive neurostimulation, including rTMS, tDCS, and cranial electrotherapy stimulation, were found to be beneficial for the treatment of pain in individuals with SCI and phantom limb pain. The meta-analysis of eight trials examining non-invasive neurostimulation identified significant effects for the relief of pain secondary to SCI. Although the mean estimate of the effect of the intervention (0.59) was small and the CI surrounding these estimates (1.07, 0.11) did not exclude the possibility that the effect was clinically trivial, the extent and quality of the obtained evidence and the minimal potential for a placebo effect suggested that non-invasive neurostimulation may be considered effective for the treatment of SCI-associated pain. Non-invasive neurostimulation has been proposed to reduce pain perception,
The pooled analysis of two trials of high methodological quality and very-low-GRADE evidence, examining non-invasive neurostimulation for the treatment of phantom limb pain identified a significant benefit of the intervention for reducing pain compared with the control, with a mean estimate of 1.57 (95% CI: 2.85, 0.29). Both phantom limb pain trials lacked allocation concealment and assessor blinding. Because these results were derived from two small, very-low-grade trials, other high-quality trials examining non-invasive neurostimulation remain necessary to confirm the effects of non-invasive neurostimulation and to narrow the CI obtained in this review.
Among the studies included in the meta-analysis that evaluated individuals with stroke, one study included participants with shoulder pain alone 53 whereas the other included cNeP that spread across the involved side. 54 Both studies used rTMS, and the technique was found to be effective for reducing shoulder pain alone. Because the cumulative effect of non-invasive neurostimulation was insignificant, we are unable to make a recommendation. Further studies that include a large sample are necessary to better evaluate the benefits of non-invasive neurostimulation for cNeP following stroke.
The pooled analysis of data from two55,56 methodologically high-quality, moderate-grade trials evaluating the effects of acupuncture revealed a significant reduction in pain in individuals with stroke. The 1.5 point reduction in pain approaches the MCID of 2 for the 0–10-point VAS. 63 Future research in this area would be useful to narrow the 95% CI, which currently extends up to 1.97 and down to 0.94 on the 0–10-point VAS. Both studies included in the meta-analysis used acupuncture to treat shoulder pain following stroke. Among the many theories postulated to explain the benefits of acupuncture, the included studies reported pain relief due to the closure of the pain gate, the release of endorphins, 55 and the adjustment of the primordial spirit that allows the flow of Qi energy. 56 The central phenomenon underlying pain relief mediated by acupuncture remains inconclusive and requires further exploration. Evoking de qi is considered to be the key to achieving desired therapeutic effects 64 and might represent a critical factor in the success of acupuncture-based analgesia. 65 De qi is a sensory response that occurs during acupuncture and is described as a sensation of warmth and tightening or deep soreness. Acupuncture needle manipulations that evoke de qi have been found to induce anti-nociceptive effects in both humans and animal models.66 –68 In both of the included acupuncture trials, de qi was reported to be evoked in the experimental groups but not in the control groups. Therefore, the control group participants may have been aware that they were receiving sham acupuncture, resulting in the biased reporting of outcomes. Because de qi is closely related to treatment efficacy, 65 future acupuncture trials should also include quantitative measurements of de qi using validated and standardised tools, such as the Massachusetts General Hospital Acupuncture Sensation Scale (MASS) or the Southampton Needle Sensation Questionnaire.
Mirror therapy, or mirror-induced visual illusion, entails the use of a mid-sagitally placed mirror to create the illusion of moving a hidden limb through the complete mirror inversion of the opposite limb.30,69 Although the neural mechanisms underpinning the analgesic effects of mirror therapy for central pain remain unclear, the restoration of mismatch between motor and sensory mechanisms mediated by the mirror illusion has been postulated as a potential explanation for this phenomenon.
70
The results obtained for mirror therapy in the treatment of phantom limb pain in the current review were obtained from two trials of high methodological quality and low-GRADE quality of evidence. We recommend the performance of further studies to substantiate these findings due to the limited number of studies that were included in the pooled analysis (
Meta-analysis of data from four TENS trials46 –49 for multiple sclerosis, of low to high methodological quality, very-low-GRADE evidence revealed a significant effect on cNeP and agree with the findings of a previous systematic review. 22 However, the mean estimate of the effect (0.32) was small and the 95% CI (0.57 to 0.06) does not exclude a clinically trivial effect. Due to the small effect size and quality of the included studies of TENS, we are unable to make any recommendations regarding the efficacy of TENS for the treatment of cNeP secondary to multiple sclerosis. Replication of this result in other methodologically high-quality studies of TENS should be sought. TENS parameters in the included trials of the current review varied greatly, which minimised the applicability of the review findings to clinical settings. Future studies and reviews are warranted to determine the optimal parameters for pain relief using TENS in individuals with cNeP.
Analgesic effects evoked by high-frequency, low-intensity TENS delivered via surface electrodes placed over the painful area or the innervating nerve have been proposed to be associated with a pain gating mechanism mediated by the intense activation of Aβ afferent fibres. 71 All four TENS trials46 –49 that were included in the review delivered TENS using surface electrodes placed over the painful area and delivered TENS at a high frequency and low intensity. However, one 46 of the four trials also provided low-frequency TENS (4 Hz) to one of three study groups and found that a greater number of participants in the high-frequency TENS group reported clinically significant effects compared with the low-frequency TENS group. The TENS intervention parameters in the trials included in the review varied greatly, which minimises the applicability of these findings to clinical settings. Future studies remain necessary to determine the optimal TENS parameters for improving pain severity in people with cNeP secondary to multiple sclerosis.
The pooled analysis of data from three exercise trials of high methodological quality and moderate quality of evidence showed the significant effects of exercise interventions on pain severity compared with control conditions among people with cNeP due to multiple sclerosis. The mean estimate of 1.6 reported in the meta-analysis approaches the clinically significant difference of 2 on the 0–10 VAS scale; however, the confidence interval extends below this threshold, indicating a clinically trivial effect. The proposed mechanisms underlying the beneficial effects of exercise interventions on central pain include altered serotonin transporter expression, increases in serotonin levels, increases in the opioid levels in central inhibitory pathways, and the facilitation of inherent inhibitory systems to modulate pain’. 72 Specific reductions in multiple sclerotic pain have been attributed to the physical and psychological health benefits of exercise and associated influences on strength and endurance.51,52 Although the analgesic benefits of exercise were significant, the evidence associated with this intervention modality must be interpreted with caution. The mean estimate was provided by a small number of studies, and the confidence interval did not exclude the possibility that the effect was clinically insignificant. The exercise interventions and parameters in the included trials varied greatly, minimising the applicability of these findings to clinical settings. Future studies remain necessary to substantiate the analgesic effects obtained in response to exercise interventions in the current review and to determine the optimal exercise types and parameters for the treatment of cNeP due to multiple sclerosis.
Significant benefits have been identified for several physiotherapy interventions, including non-invasive neurostimulation (for SCI and phantom limb pain), mirror therapy (for phantom limb pain), acupuncture (for stroke), and TENS and exercises (for multiple sclerosis). These results may potentially indicate useful approaches for the treatment of pain in neurological disorders. The other strengths of the review are as follows: (1) the comprehensive search strategy used to identify physiotherapy intervention trials for the treatment of cNeP; (2) this review is the most comprehensive review, to date, including all neurological disorders resulting in cNeP; and (3) language barriers were eliminated by the inclusion of studies published in all languages. A possible limitation of this systematic review was that heterogeneity and the limited number of studies for certain health conditions prevented the performance of meta-analysis for at least half of the identified interventions. Finally, we restricted our review to RCTs. Although systematic reviews of RCTs are considered to represent the highest level of evidence for investigating the efficacy of interventions, we excluded studies on pain management for cNeP beyond the five included health conditions due to the study design.
Conclusion
We identified five physiotherapy interventions for the management of cNeP secondary to SCI, multiple sclerosis, stroke, and phantom limb pain. Our meta-analysis provided evidence for the use of non-invasive neurostimulation to manage cNeP secondary to SCI and phantom limb pain, the use of mirror therapy to manage phantom limb pain, the use of acupuncture to manage cNeP secondary to stroke, and the use of TENS and exercises to manage cNeP secondary to multiple sclerosis. Further studies should include larger sample sizes to validate the benefits of non-invasive neurostimulation for the treatment of patients who require pain management following stroke and various stages of SCI. Because the treatment efficacy of acupuncture therapy depends on eliciting de qi, future acupuncture trials must utilise valid and standardised tools capable of performing quantitative measurement of de qi. Further studies also remain necessary to determine the optimal parameters for exercise and TENS interventions, including the most beneficial exercise types for improving pain severity in cNeP secondary to multiple sclerosis.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221078672 – Supplemental material for Physiotherapy interventions may relieve pain in individuals with central neuropathic pain: a systematic review and meta-analysis of randomised controlled trials
Supplemental material, sj-docx-1-taj-10.1177_20406223221078672 for Physiotherapy interventions may relieve pain in individuals with central neuropathic pain: a systematic review and meta-analysis of randomised controlled trials by Priya Kannan, Umar Muhammad Bello and Stanley John Winser in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was supported by the Dean’s reserve fund obtained by Dr Priya Kannan (Reference: ZVSV) Hong Kong Polytechnic University, Dean’s reserve fund.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Registration
Our systematic review was registered with the PROSPERO registry (CRD42020175111).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
