Abstract
Objective
To evaluate the effectiveness of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy (DPN).
Methods
This single-blind, randomized clinical trial enrolled patients with type 2 diabetes mellitus and moderate-to-severe DPN symptoms below the ankle. Patients were randomly assigned to receive INF or sham treatment. In the INF group, trained INF physical therapists provided therapy for 50–60 min, three times a week for 3 weeks. Sham treatment consisted of patients believing they received anodyne therapy for 3 weeks. Pre- and post-treatment data were compared between the two groups for quality of life, balance, gait, protective sensory function and pain outcome measures.
Results
A total of 28 patients (17 males) were enrolled in the study (INF group n = 17; sham group n = 11). There was a significant decrease in the overall pain score in both the INF and sham groups over time, but the decrease was greater in the INF group (1.11 versus 0.82). Between-group comparisons demonstrated significant differences in unpleasant pain and protective sensory function. The INF group showed post-treatment improvements in protective sensory function and composite static balance score.
Conclusions
INF treatment improved pain perception, the composite static balance score and protective sensations in patients with DPN.
Research Registry number: CNCT04025320
Introduction
In 2017, the International Diabetes Federation estimated that 651.1 million adults between the ages of 65–99 years to had diabetes mellitus (DM), with 90–95% of those having type 2 DM (T2DM). 1 Over 50% of patients with T2DM suffer from diabetic peripheral neuropathy (DPN).2,3 The annual cost of DM patients with DPN is $30 000, with this expense quadrupling if the DPN symptoms are severe and painful. 4 Additionally, DPN complications can negatively affect the patient's quality of life (QOL),5,6 with impaired balance,3,6,7 altered gait3,6,8 and increased pain.3,9,10
The current treatment for DPN consists of the following: (i) glycemic control; (ii) foot care; and (iii) pain management. 11 The American Diabetes Association recommends medication utilization for the relief of painful DPN.12,13 Despite ongoing progressive research, only modest benefits from pharmacology have been shown to slow disease progression and reduce pain associated with DPN. 14 Non-pharmacologic modalities such as infrared therapy, shoe magnets, Reiki therapy, exercise, acupuncture, transcutaneous electrical nerve stimulation, spinal cord stimulation and biofeedback behavioral therapy have attempted to reverse the debilitating effects on nerve axons and improve neural circulation in patients with T2DM. 15 Unfortunately, the American Academy of Neurology does not assign ‘A’ level evidence to any of the aforementioned interventions.
While non-pharmacologic and pharmacologic interventions may provide inconsistent symptomatic relief and improvement in function, a lack of a curative treatment remains. The authors posit that reversing DPN starts with the restoration of neurovascular circulation. Over time, hyperglycemia resulting from DM induces capillary endothelial basement membrane thickening, endothelial swelling and immune cell extravasation, resulting in capillary closure and neural ischemia.16,17 If this inflammatory cascade persists, axonal thinning and nerve fiber loss occur.16,17 It seems probable that the lack of a cure for DPN may be found in the practitioner’s inability to restore and maintain blood flow to the damaged neural capillary. Indeed, endoneurial capillary circulatory restoration for the damaged nerve results in nerve growth factor release that precedes axonal regrowth.16,17
It is with this background in mind that the authors assessed a manual therapy treatment that has been suggested to improve neurovascular circulation in the ischemic neurovascular capillary beds of patients with T2DM. Intraneural facilitation (INF) is a manual therapy approach that ‘aims to bias blood flow into the neural fascicle, improve the endoneurial capillary circulation, and reverse intrafascicular ischemia’. 7 This current study hypothesized that INF would decrease perceived pain, and improve balance, ambulation, QOL and protective sensory function in patients with moderate-to-severe DPN with a posited mechanism of action of neurovascular revascularization.
Patients and methods
Recruitment and patient criteria
This prospective, randomized, patient- and tester-blinded study enrolled eligible patients with T2DM in the Department of Allied Health Studies, School of Allied Health Professions, Loma Linda University, Loma Linda, CA, USA between 1 October 2019 and 8 October 2020. Recruitment activities included the following: distributing flyers in the Loma Linda Community; referrals from medical doctors at the Loma Linda University Medical Center and the Loma Linda Diabetes Treatment Center; emails to school staff; and word of mouth. The reporting of this study conforms to the CONSORT statements. 18 This study obtained approval from the institutional review board (IRB) of the University of Loma Linda, Loma Linda, CA, USA (reference no: IRB #5190128). The study was registered at ClinicalTrials.gov (no. CNCT04025320). Written informed consent was obtained from the participating patients.
The inclusion criteria were as follows: (i) patients with T2DM; (ii) below ankle DPN symptoms (e.g. numbness, tingling, burning, sharp pain, increased sensitivity, etc.); (iii) moderate-to-severe DPN as identified by scoring ≥10 on the Quality of Life – Diabetic Neuropathy (QOL-DN) scale; (iv) aged 50–75 years. The exclusion criteria were as follows: (i) any medical condition that suggested a possible decline in function over the next 6 months, such as a current regimen of chemotherapy, radiation therapy or dialysis; (ii) any lower extremity amputations or wounds; (iii) any documented active alcohol or drug misuse; (iv) any known health conditions: (v) patients with end-stage renal failure, uncontrolled hypertension, severe dyslipidemia, chronic liver disease, autoimmune disease, advanced chronic obstructive pulmonary disease and active inflammations; (vi) other inflammatory neuropathies including chronic inflammatory demyelinating polyneuropathy, proximal diabetes neuropathy, autonomic neuropathies or other neuropathies not associated with DM such as B12 deficiency, hypothyroidism and uremia.19–21 Patients with other severe chronic medical conditions requiring active treatment and those that were morbidly obese or pregnant (self-reported) were also excluded. Participants were screened using an IRB-approved telephone recruitment form and questioned to determine if they met the medical portion of the inclusion/exclusion criteria. Patients that met the medical portion of the inclusion criteria and had no exclusion criteria were then scheduled to visit the physical therapy laboratory at Loma Linda University to complete the last inclusion criteria, which was to score ≥10 on the QOL-DN scale. Eligible participants then signed the IRB consent form, completed baseline measurements and chose which leg to receive treatment on, so that they could compare the symptomology between the two lower extremities during the treatment weeks. Baseline measurements for the Pain Quality Assessment Scale (PQAS), Zeno™ Walkway Gait Analysis System, NeuroCom® SMART Balance Master computerized dynamic posturography (Limits of Stability and Sensory Organization Test) and Semmes–Weinstein Monofilament (SWM) test were completed as described below.
Randomization
Once baseline measurements were completed, participants visited the Neuropathic Therapy Center, Loma Linda University, Loma Linda, CA, USA for randomization and scheduling. Each participant was asked to choose between two sealed identical envelopes revealing if they were in group 1 or group 2. Group 1 was the INF group and participants were given INF treatment. Group 2 was the sham group and participants believed they were given anodyne light therapy. Patients were blinded to the intervention group. After completing treatments, participants returned to the physical therapy laboratory at Loma Linda University to repeat the baseline measurements, by the same tester, that was blinded to each participant’s treatment group.
Outcome measures
QOL-DN questionnaire
The QOL-DN questionnaire was used to measure the patient’s perception of the effects of diabetic neuropathy, assess neuropathy and differentiate between autonomic, large and small fiber neuropathy. 22 It was developed in the Department of Internal Medicine, Eastern Virginia Medical School, Norfolk, VA, USA. 22 The questionnaire was immediately scored and patients classified as having moderate-to-severe DPN (scoring ≥10) were invited to participate. A score range of 2–9 classified the neuropathy as mild, 10–19 as moderate and ≥20 as severe. 22
Pain quality assessment scale
The PQAS questionnaire was used to assess the numeric pain rating scale and distinct pain qualities for 20 pain (quality and spatial) descriptors. 23 After reading the introduction of the questionnaire, patients then measured their pain on a numeric scale 0 = ‘no pain’ or ‘no painful sensation’ to 10 = ‘worst imaginable pain sensation’. 23 There were also three pain quality domains; paroxysmal (this contained the average scores of shooting, sharp, electric, hot and radiating); superficial (this contained the average scores of itchy, cold, numb, sensitive and tingling); and deep (this contained the average scores of aching, heavy, dull, cramping and throbbing). 24
Zeno™ Walkway Gait Analysis System
The Zeno™ Walkway Gait Analysis System (ProtoKinetics, Havertown, PA, USA) was used to observe the spatiotemporal characteristics of gait in the patients, including velocity and stride length. 25 It contains a 16-level pressure-sensing pad that measures 6.10 × 1.22 m and captures data using ProtoKinetics Movement Analysis Software (PKMAS) version 5.08C1 (ProtoKinetics).26,27 The Zeno™ Walkway Gait Analysis System was directly connected to the PKMAS software and the data generated were analyzed on a computer that was able to record and store gait output. 28 Participants were instructed to wear comfortable walking shoes and to walk back and forth on the walkway four times.
NeuroCom® SMART Balance Master System computerized dynamic posturography
This apparatus is used to measure static and dynamic balance and was developed by Natus Medical Incorporated, Clackamas, OR, USA. 29 It consists of two force plates that can move up/down and in an anterior-posterior plane. 30 Two tests were administered: sensory organization test (SOT) and limits of stability (LOS) as described below.
Sensory organization test (SOT)
The SOT assesses the patient’s visual, somatosensory and vestibular systems for maintaining upright posture. 31 The standardized test instructions, per the NeuroCom® protocol, were either ‘stand quietly with your eyes open’ or ‘stand quietly with your eyes closed’, depending on the condition being tested. 31 This test was completed under six different sensory conditions lasting 20 s each and there were three trials of each condition. 32 The first three conditions examined static posturography and the last three conditions examined dynamic posturography. 32
In condition 1, the patient stands quietly with their eyes open as the force plate measures their sway. 33 In condition 2, the patient stands quietly with their eyes closed, which removes their visual aid. 33 In condition 3, the patient stands quietly with their eyes open as they sway forward or backward on the locked force plate, and the surrounding box moves with the patient. Thus, the patient’s visual information is telling the patient that they are not moving, but the proprioceptive information given to the patient is that he or she is moving. 33 In conditions 4, 5 and 6, the first three conditions are repeated consecutively, but with the force plate unlocked, which creates a situation in which the proprioceptive information is now incorrect because as the patient sways forward or backward, so does the force plate. 33
The composite equilibrium and balance equilibrium scores for each of the six trial conditions were assessed.7,34 The composite equilibrium score was defined as the patient’s anterior/posterior (AP) sway during each trial compared with the theoretical sway limit of 12.5.7,31 The composite equilibrium score was calculated by independently taking the mean for conditions 1 and 2, and adding this score to scores for all trials in conditions 3, 4, 5, and 6, then dividing that number by the total number of trials. 31 The total score is 100; therefore, those with little AP sway will acquire scores close to 100; scoring 0 means touching a support surface, shifting feet or falling.32,34
Limits of stability (LOS)
The LOS test assesses the dynamic balance by measuring the patient’s weight-shifting ability and voluntary limits to stability for eight directional targets set at 100% of theoretical limits of stability for an eight-second hold.30,35 During testing, patients were asked to lean away from midline toward the direction of one of the eight targets without stepping their feet or lifting from a standardized foot position. 36 The LOS test measures five parameters: reaction time (ReT), movement velocity (MVL), endpoint excursion (EPE), maximum excursion (MXE) and directional control (DC). 28 Researchers analyzed these five test parameters for eight different directions: forward (FW), forward right (FWRT), right (RT), backward right (BWRT), backward (BW), backward left (BWLT), left (LT) and forward left (FWLT).
Semmes–Weinstein monofilament (SWM) test
A Semmes–Weinstein 5.07/10 g (CH537 Retractable Accurate Monofilament 10 g; Bailey Instruments, Manchester, UK) monofilament (SWM) was used to test the protective sensory function in the third and fifth distal phalanx. This method was standardized according to generally accepted guidelines.37,38 The ‘yes/no’ method was used, meaning the patient responded with ‘yes’ each time they sensed the monofilament application. The inability to feel a 5.07 SWM (10 g of pressure) indicates that the individual has peripheral neuropathy. 39
INF versus sham treatment
Intraneural facilitation is a manual therapy proposed to reduce neuropathy symptoms through revascularizing ischemic peripheral nerves. 40 INF posits to improve the relationship between the macrocirculation and the microcirculation resulting in an increase of neurovascular pressure that sufficiently overcomes diabetes-induced neural capillary resistance. 40 INF postulates that this is achievable with a system involving three holds. 40 The first is the facilitation hold, in which the contralateral joint is placed in a maximal loose-pack position. 7 The purported physiologic reason is to stretch the connecting nutrient vessels creating a natural large to small vessel bias and enhanced neurovascular flow. This slight stretch in the contralateral joint is hypothesized to stretch the nerve further than the artery due to the increased elastin in the artery. 7 The increased neural excursion compared with the artery stretches the coiled nutrient vessel attached between the artery and the nerve. Thus, it is hypothesized that this enlarges the arterial junction's opening, allowing increased blood into the epineurium. 7 Now that pressure has increased, the secondary hold takes place to bias the increased epineurial blood into the transperineurial vessels that bridge the epiperineum and the endoneurial capillaries of the site being treated. 7 After the pressure has increased into the open endoneurial capillaries, the goal is to open up ischemic endoneurial capillaries, and this is hypothesized to open by performing the third hold. The third hold is known as the sub hold and encourages blood flow through ischemic endoneurial capillaries that have increased resistance/pressure through the application of Bernoulli's principle. The last two holds were repeated on the affected side for the duration of the allotted time. Trained INF physical therapists provided this therapeutic intervention for those in the INF group for 50–60 min, three times a week for 3 weeks.
The sham treatment lasted 50–60 min, three times a week for 3 weeks, and consisted of patients believing they were receiving infrared light therapy. The anodyne light therapy unit was applied to the patient, but the unit was not plugged in (the plug was not visible to the patient). Pads were placed on the patient in the following locations on the affected lower limb: two on the plantar aspect of the foot in a T formation and one pad on the medial and lateral side of the calf. A double-folded towel was wrapped around the patient’s foot at the electrode sites to blind the patients from the light not emitting from the electrodes due to the anodyne light therapy unit not being plugged in.
Statistical analyses
The sample size was estimated using an effect size of 0.9 between the INF and the sham group, a power of 0.8 and the level of significance α = 0.05. The total sample size needed was estimated to be 30. The baseline assessment consisted of baseline demographic data, QOL-DN, PQAS, Zeno™ walkway, NeuroCom® SOT and LOS, and SWM. Post-treatment data assessment consisted of QOL-DN, PQAS, Zeno™ walkway, NeuroCom® SOT and LOS, and SWM, all of which were collected after 3 weeks of treatment.
All statistical analyses were performed using IBM SPSS Statistics for Windows, Version 28.0 (IBM Corp., Armonk, NY, USA). Continuous data are presented as mean ± SD and categorical data are presented as frequency (%). Mann–Whitney U-test was used to compare the INF and sham groups for all variables at baseline. Wilcoxon’s signed-rank test was used to compare pre- and post-treatment data. A P-value <0.05 was considered statistically significant.
Results
Out of the 45 screened patients, 30 eligible patients were randomly assigned to the INF (n = 17) and sham (n = 13) groups. However, two patients dropped out from the sham group due to health-related issues, so only 28 patients completed the study (INF group n = 17; sham group n = 11) (Figure 1). There were 17 males (mean ± SD age, 66.55 ± 6.68 years) and eleven females (mean ± SD age, 67.29 years ± 4.73 years). There were no significant differences in the baseline characteristics of age, height, weight and sex between the two groups (see supplementary materials, Table 1).

Flow chart showing progress through enrolment, randomization and analysis of patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy. Each patient had a chance to randomly draw group 1 or group 2 therapy (INF or sham therapy).
Results of the Pain Quality Assessment Scale (PQAS) and Quality of Life – Diabetic Neuropathy (QOL-DN) questionnaires pre- and post-treatment in patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy.
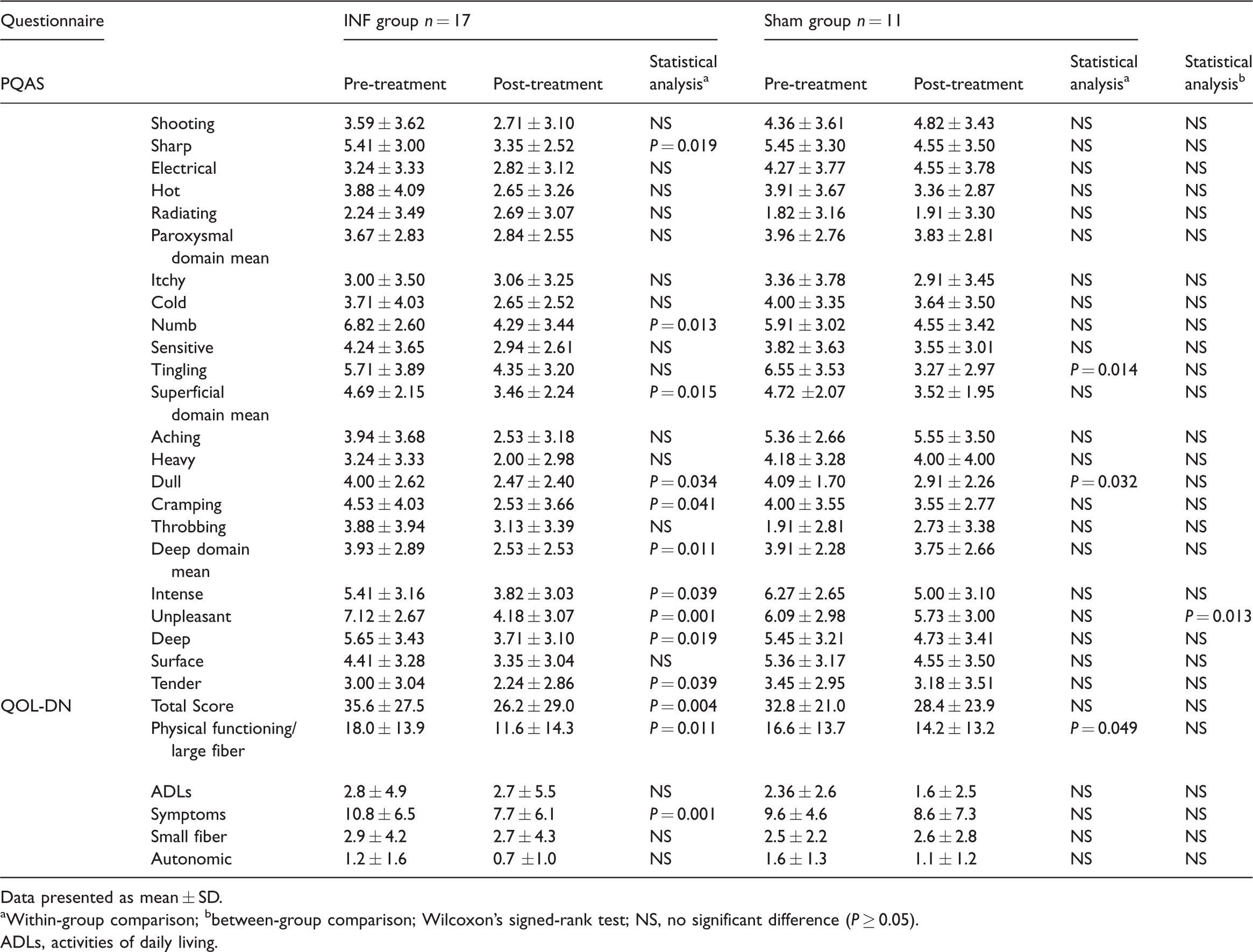
Data presented as mean ± SD.
aWithin-group comparison; bbetween-group comparison; Wilcoxon’s signed-rank test; NS, no significant difference (P ≥ 0.05).
ADLs, activities of daily living.
The results of pre- and post-treatment measurements for the Semmes–Weinstein monofilament tests are shown in Figure 2. Significant findings were observed within the INF group over time for both the third (P < 0.001) and fifth (P < 0.001) distal phalanx, as well as between the two groups post-treatment in the fifth (P = 0.003) distal phalanx.

Results of the Semmes–Weinstein monofilament test showing the mean percentage ‘yes’ responses pre- and post-treatment for the third and fifth distal phalanx in patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy. Data presented as mean; Wilcoxon’s signed-rank test.
The results of the PQAS and QOL-DN questionnaires pre- and post-treatment are shown in Table 1. There was a significant decrease in pain within the INF group over time for sharp (P = 0.019), numb (P = 0.013), dull (P = 0.034), cramping (P = 0.041), intense (P = 0.039), unpleasant (P = 0.001), deep (P = 0.019 and tender (P = 0.039) pain. There were significant decreases observed in the superficial (P = 0.015) and deep (P = 0.011) subcategories over time for the INF group. There were significant decreases in tingling (P = 0.014) and dull (P = 0.032) pain over time for the sham group and no significant findings for any of the domains. There was a significant decrease in unpleasant pain (P = 0.013) between the two groups. For the QOL-DN questionnaire, there was a significant decrease within the INF group over time in the total score (P = 0.004), physical functioning/large fiber (P = 0.011) and symptoms (P = 0.001). There was also a significant decrease in physical functioning/large fiber (P = 0.049) within the sham group over time. There were no significant differences between the two groups.
The numeric pain rating scale at baseline in the INF group had a mean of 3.76 with (median of 4) and the sham group had a mean of 4.27 (median of 4). There was a significant decrease in the overall pain score in both the INF and sham groups over time (P = 0.024 and P = 0.024, respectively), but the decrease was greater in the INF group (1.11 versus 0.82), but this was not significant. The frequency and percentage of each pain score (0–10) showed a change for severe pain (>7) in the INF group over time, but not in the sham group. The total percentage with severe pain levels in the INF group changed from two of 17 patients (11.8%) to 0 of 17 patients (0.0%). However, the total percentage with severe pain (>7) in the sham group remained the same at two of 11 patients (18.2%) pre- and post-treatment.
The results of the pre- and post-treatment measurements for the Zeno™ Walkway gait analysis are presented in Table 2. There were significant increases in mean gait velocity (P = 0.005) and stride length (P = 0.007) over time for the INF group. There were also significant increases in mean gait velocity (P = 0.014) and stride length (P = 0.007) over time for the sham group. No significant differences were observed between the two groups.
Changes in the spatiotemporal parameters of gait pre- and post-treatment in patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy.

Data presented as mean ± SD.
aWithin-group comparison; bbetween-group comparison; Wilcoxon’s signed-rank test; NS, no significant difference (P ≥ 0.05).
The results of the pre- and post-treatment measurements for the SOT are shown in Table 3. There was a significant increase in the mean composite score (P = 0.040) over time for the INF group. No significant differences were observed between the two groups.
Changes in the sensory organization test (SOT) pre- and post-treatment in patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy.

Data presented as mean ± SD.
aWithin-group comparison; bbetween-group comparison; Wilcoxon’s signed-rank test; NS, no significant difference (P ≥ 0.05).
1Composite score was the summation of all conditions; 2somatosensory system score was the condition comparison of condition 2 to condition 1; 3visual system score was the condition comparison of condition 4 to condition 1; 4vestibular system score is the condition comparison of condition 5 to condition 1; 5preference score was the condition comparison of conditions (3 + 6) to conditions (2 + 6).
The results of the pre- and post-treatment measurements for the LOS are shown in Table 4. Significant increases within the INF group over time were observed in the FWRT direction for MVL (P ≤ 0.001), the BW direction for MVL (P = 0.026), the BWRT direction for EPE (P = 0.018) and MXE (P = 0.046) and the FWLT direction for MXE (P = 0.047). For the sham group, there was a significant decrease in the RT direction for ReT (P = 0.005) and significant increases in the FWLT direction for MVL (P = 0.011) and MXE (P = 0.029). Significant differences between the two groups were seen in the FW direction for MVL (P = 0.034) and in the RT direction for ReT (P = 0.006).
Changes in the limits of stability (LOS) pre- and post-treatment in patients with type 2 diabetes mellitus and diabetic peripheral neuropathy that were enrolled in a prospective, randomized, single-blind study of intraneural facilitation (INF) for the treatment of diabetic peripheral neuropathy.

Data presented as mean ± SD.
aWithin-group comparison; Wilcoxon’s signed-rank test; NS, no significant difference (P ≥ 0.05).
FW, forward; FWRT, forward right; RT, right; BWRT, backward right; BW, backward; BWLT, backward left; LT, left; FWLT, forward left.
Discussion
The current study showed no significant differences in the baseline demographic characteristics of the two groups, confirming the random assignment of study participants into the INF and sham groups. The findings of the current study describe the effects of a manual therapy intervention on the pain symptoms associated with DPN. The baseline pain symptoms of both groups were almost comparable before the treatment intervention. There was a significant decrease in the overall pain score in both the INF and sham groups over time, but the decrease was greater in the INF group. There was a significant improvement over time in the INF group for pain quality factors found in the small nerve fiber symptoms as recorded by the superficial and deep pain domains of the PQAS questionnaire. DPN affecting the small nerve fibers involves multiple symptoms, including burning, shooting, prickling, cramping, numbness, heat sensation loss, allodynia and itching pain.41,42 This may suggest that INF also affected small nerve fibers at a neural capillary level potentially alleviating endoneurial microangiopathy and the neural ischemic conditions that have been shown to increase neural excitability and paresthesia. 43
In an earlier study, one of the highest mean severity scores on the neuropathic pain scale was unpleasant pain. 9 This previous study showed those with painful DPN also experienced a negative QOL. 9 In this current study, there was a significant improvement in the unpleasant pain quality in the INF group and the difference in the unpleasant pain quality between the two groups was also significant as recorded by the PQAS questionnaire. The significant reduction in some of the pain symptoms observed in both the INF and sham groups in the current study was consistent with the concept that the formation of a ‘sustained partnership’ between the healthcare provider and the patient can have direct therapeutic benefits. 44
There is little information on treatments that can improve gait parameters and decrease the risk of falls in patients with DM. 45 Improving gait velocity decreases the risk of falls in those with DPN. 46 In the current study, both the INF and sham groups showed significant improvements in gait velocity and stride length, but there were no significant differences between the two groups. An improvement in gait velocity and stride length post-intervention for DPN was observed in a previous study that investigated exercise as an intervention for those with T2DM with and without DPN, although no sham group was included in the study. 8 When comparing the baseline gait velocity and stride lengths in the previous study with those of the current study, 8 the higher baseline mean values in the current study suggest that the patients were higher functioning and that this could have created a ceiling effect.
There is a reasonable amount of research that shows decreased balance in individuals with DPN versus those without DPN.3,6,47–50 However, few published studies have used the NeuroCom® SMART Balance Master computerized dynamic posturography to measure SOT. In a previous study, SOT static balance was significantly lower in neuropathic patients than non-neuropathic patients, with no significant difference seen in dynamic balance. 32 This was expected because a decline in somatosensory function, along with damage to the peripheral nervous system, is defined as peripheral neuropathy for DM individuals.51,52 A previous study investigated the relationship between patients with DPN pre- and post-INF treatments and found a significant improvement in static balance with SOT. 7 However, their study lacked a control or comparative sham group. 7 This was similar to the findings in the current study in which the static balance composite score was significantly increased within the INF group. However, the composite score did not make the minimal detectable change, which required a change of greater than 8 points. 53 No significant differences were observed in the SOT between the INF or sham groups in the current study. This could be because the mean ± SD composite score pre-treatment in the INF group in this current study (71.9 ± 9.7) was considerably higher than the pre-intervention mean in the previous study (53.77 ± 21.81), suggesting that the participants in the current study had fewer balance deficiencies pre-treatment. 7
There is also limited research utilizing NeuroCom® LOS in individuals with DPN with only one comparable article being available. 7 In the current study, there was a significant difference for MVL in the FW direction between the two groups in favor of INF and for ReT in the RT direction in favor of the sham group. The MVL result was similar to that of the previous study in which MVL was also significant. 7 However, in the current study with a comparative sham group, the dynamic balance was improved in the INF group over the sham group. MVL had three directional improvements over time in the INF group suggesting the INF manual therapy approach affects the speed of the patient to change their center of gravity and reach their target without losing balance. The significant improvement in the ReT in the sham group was also found between the two groups in favor of the sham group. This result was not anticipated, but only one direction was significantly improved over time in the sham group. Further research into LOS with an increased sample size will help to increase the understanding of the relationship between INF and LOS. One possible explanation for the minimal significant findings for the LOS test in the current study is that this test focuses on the motor response and INF does not address motor system response. Future studies would benefit from investigating the use of INF in conjunction with specific exercises improving motor control.
In the current study, there were significant decreases within the INF group over time in the total score, physical functioning/large fiber and symptoms as reported by the QOL-DN questionnaire. The symptom category of the QOL-DN questionnaire included numbness, tingling, electric shocks, unusual sensations, superficial pain, deep pain, weakness and allodynia. These symptoms are also indicative of both small and large fiber nerve involvement. The large fiber pathophysiology involves more axonal neuropathy, suggesting possible changes are happening at the axon. The sham group had a significant decrease in the physical functioning/large fiber category over time. There were no significant differences between the two groups, which was possibly due to the unequal sample size in each group.
Diminished sensations in individuals with DPN can lead to diabetic ulcers and non-traumatic amputations. 54 The use of the 5.07 SWM test with 10 g of pressure is an essential tool used to diagnose the loss of protective sensory function and assess the risk of amputation. 12 This is important due to the rising healthcare costs associated with T2DM.4,20,55 There are typically 65 000 amputations per year in the US alone, which is one every 10 minutes; and neuropathy is the contributing factor in 87% of these cases. 20 The 10 g of pressure is important in the 5.07 SWM test because it ‘assesses the integrity of the Merkel touch domes and Meissner’s corpuscles and their associated large diameter fibers’. 56 In the current study, the INF group showed significant improvements in protective sensory function after INF treatment in the two tested areas. The INF results were significantly greater than those of the sham group, suggesting that INF could be a contributing factor in reducing amputations and ulcers related to DPN. The SWM test findings were also similar to those of the QOL-DN questionnaire findings, where the large fiber subcategory significantly improved following INF.
The results of the present study showed a strong placebo effect. This was anticipated as the protocol had mimicked the anodyne sham comparative group used in a previous study in which a large placebo effect was also observed. 57
This current study had several limitations. First, the small and uneven sample size might have prevented the observation of significant findings between the two groups or failed to make allowances for a possible placebo effect. Secondly, using the QOL-DN questionnaire to classify those with moderate-to-severe DPN reported symptoms could be significantly biased and may not be proportionate to the severity of the nerve damage, possibly contributing to the placebo effect. Thirdly, improving the pre-screening process to only include patients with diagnosed balance and gait issues would have provided a reduced ceiling effect and a greater chance of observing any possible post-treatment improvements. Finally, this current study did not include the duration of T2DM and DPN diagnosis or assess the long-term implications of the 3-week intervention. Future research should include a larger and equal sample size with gait and balance deficits in conjunction with DPN. It is strongly recommended that future research also include glycosylated hemoglobin changes in response to INF. Follow-up research might include tools that investigate the purported physiological mechanisms of INF such as laser Doppler flowmetry and near-infrared spectroscopy to assess for post-intervention cutaneous blood flow changes. Future studies should include follow-up assessments at 3 and 6 months to assess the long-term impact of INF.
In conclusion, this current study demonstrated that INF treatment for 3 weeks improved pain perception, the composite static balance score and protective sensory function in patients with DPN. These findings should be considered preliminary due to the small sample size and future studies should address the limitations of the current study.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221109390 - Supplemental material for Treating peripheral neuropathy in individuals with type 2 diabetes mellitus with intraneural facilitation: a single blind randomized control trial
Supplemental material, sj-pdf-1-imr-10.1177_03000605221109390 for Treating peripheral neuropathy in individuals with type 2 diabetes mellitus with intraneural facilitation: a single blind randomized control trial by Kyan Sahba, Lee Berk, Mark Bussell, Everett Lohman, Francis Zamora and Lida Gharibvand in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605221109390 - Supplemental material for Treating peripheral neuropathy in individuals with type 2 diabetes mellitus with intraneural facilitation: a single blind randomized control trial
Supplemental material, sj-pdf-2-imr-10.1177_03000605221109390 for Treating peripheral neuropathy in individuals with type 2 diabetes mellitus with intraneural facilitation: a single blind randomized control trial by Kyan Sahba, Lee Berk, Mark Bussell, Everett Lohman, Francis Zamora and Lida Gharibvand in Journal of International Medical Research
Footnotes
Acknowledgments
We thank everyone at the Neuropathic Therapy Center for their efforts and constant assistance with recruitment, organization, communication, treatment and data collection of this study: Dr Mark Bussell, Karla Pieters, Kevin Carrington, Dr Ron Coleman, Dr Trisha Gorny, Jay Alido, Paul Samosky, Ariana Martinez, and Jordan Lozano. Special thanks to Dr Candace Buckley from the Loma Linda Foot & Ankle Center and the Diabetes Treatment Center of Loma Linda University for her referrals. A big thank you to Kohei Suga for his assistance with data entry and all the participants in this study that volunteered their time. This project was brought to fruition by the McCarthy Foundation, specifically Edie McCarthy. Thank you for believing in the need for our study and for providing funding to us.
Declaration of conflicting interest
Dr Mark Bussell is the inventor on several patent applications filed by the Loma Linda University Medical Center that covers the subject matter of intraneural facilitation presented in this paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the McCarthy Foundation, which had no involvement in the study design, collection, analysis and interpretation of the data, the writing of the report or the decision to submit the paper for publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
