Abstract
Objective:
To investigate the optimal percutaneous coronary intervention (PCI) strategy in patients with ST-segment elevation myocardial infarction (STEMI) and multivessel coronary artery disease.
Methods:
Trials that randomized patients with STEMI and multivessel coronary artery disease to immediate multivessel PCI, staged multivessel PCI, or culprit-only PCI and prospective observational studies that investigated all-cause death were included. Random effect risk ratio (RR) and 95% confidence interval (CI) were calculated.
Results:
A total of 13 randomized trials with 7627 patients and 21 prospective observational studies with 60311 patients were included. In the pairwise and network meta-analysis based on randomized trials, immediate or staged multivessel PCI was associated with a lower risk of long-term major adverse cardiac events (MACE; RR: 0.58; 95% CI: 0.45 to 0.74) than culprit-only PCI, which was mainly due to lower risks of myocardial infarction (RR: 0.67; 95% CI: 0.51 to 0.88) and revascularization (RR: 0.38; 95% CI: 0.28 to 0.51), without any significant difference in all-cause death (RR: 0.85; 95% CI: 0.69 to 1.04; I2 = 0.0%). However, short-term outcomes were deficient in randomized trials. The results from real-world prospective observational studies suggested that staged multivessel PCI reduced long-term all-cause death (RR: 0.53; 95% CI: 0.39 to 0.71; I2 = 15.6%), whereas immediate multivessel PCI increased short-term all-cause death (RR: 1.58; 95% CI: 1.22 to 2.05; I2 = 43.8%) relative to culprit-only PCI.
Conclusion:
For patients in randomized trials, multivessel PCI in an immediate or staged procedure was preferred due to improvements in long-term outcomes. As a supplement, the results in real-world patients derived from prospective observational studies suggested that staged multivessel PCI was superior to immediate multivessel PCI. Therefore, staged multivessel PCI may be the optimal PCI strategy for patients with STEMI and multivessel coronary artery disease.
Keywords
Introduction
Primary percutaneous coronary intervention (PCI) remains the cornerstone for the treatment of patients with ST-elevation myocardial infarction (STEMI) when performed in a timely manner. 1 In patients diagnosed with STEMI, it is estimated that approximately 40–65% exhibit multivessel coronary artery disease and are associated with worse short- and long-term mortality and morbidity than subjects with single-vessel disease. 2 Three different revascularization strategies are available for the treatment of multivessel coronary artery disease at the time of primary PCI: (1) immediate multivessel PCI (MV-PCI), in which the infarct-related artery (IRA) and non-IRA are treated during the index procedure; (2) staged MV-PCI strategy, in which the IRA is treated at the index procedure followed by a planned PCI of the non-IRA at a later time within 1 month; and (3) culprit-only PCI (CO-PCI) strategy, in which the only treated vessel is the IRA. The results based on earlier observational studies demonstrated that an immediate MV-PCI strategy was associated with worse short-term outcomes than a CO-PCI strategy.3,4 However, recent randomized trials including the PRAMI (Preventive Angioplasty in Myocardial Infarction), 5 CvLPRIT (Complete Versus Lesion-Only Primary PCI Trial), 6 DANAMI-3-PRIMULTI (Third Danish Study of Optimal Acute Treatment of Patients with ST-segment Elevation Myocardial Infarction: Primary PCI in Multivessel Disease), 7 COMPARE-ACUTE (Comparison Between FFR Guided Revascularization Versus Conventional Strategy in Acute STEMI Patients With Multivessel Coronary Artery Disease), 8 and COMPLETE (Complete vs Culprit-only Revascularization to Treat Multi-vessel Disease After Early PCI for STEMI) trials 9 as well as meta-analyses10,11 all demonstrated that immediate or staged MV-PCI was superior to CO-PCI in reducing the risks of long-term revascularization, cardiac death and myocardial infarction in the absence of short-term outcomes. Therefore, the utility and strategy of MV-PCI in patients with STEMI and multivessel coronary artery disease remain difficult to perform in real-world practice in China. 12 Meanwhile, differences exist with regard to the guidance [angiography or fractional flow reserve (FFR)] of non-IRA revascularization. Thus, we sought to conduct a comprehensive pairwise and network meta-analysis of randomized trials to assess the relative merits of different PCI strategies in patients with STEMI and multivessel coronary artery disease, and subgroups were designed based on the guidance of revascularization (angiography or FFR) and MV-PCI strategy (Immediate, staged or mixed). Moreover, considering that many early deaths occur within several days after STEMI 13 and that short-term outcomes were not reported in randomized trials, we resorted to prospective observational studies to investigate short-term (in-hospitalization or within 30 days) and long-term all-cause death (⩾6 months) among different PCI revascularization strategies.
Methods
Data sources
This study was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement for pairwise and network meta-analysis.14,15 An electronic search of PubMed, Web of Science, the Cochrane Library, ClinicalTrials.gov, and Google Scholar along with major conference proceedings was conducted using the Medical Subject Heading and the keyword search terms ‘percutaneous coronary intervention(MESH)’, ‘myocardial infarction(MESH)’, ‘PCI’, ‘angiography’, ‘STEMI’, ‘multivessel’, ‘non-IRA’, ‘culprit-only’, ‘staged’, ‘immediate’, ‘simultaneous’, ‘incomplete’ and ‘complete revascularization’ from inception through November 2020 with no language restriction. In addition, we searched the presentations at major cardiovascular scientific sessions and the bibliographies of original trials, meta-analyses, and review articles to find other eligible studies. This meta-analysis was registered at the PROSPERO international prospective register of systematic reviews (CRD42020218552). We obtained summary data from published studies, which has been approved by the institutional review committee in their respective studies. Therefore, no further sanction was required for our meta-analysis.
Selection criteria
We only included randomized trials and prospective observational studies (observational studies must investigate all-cause death) that compared any combination of CO-PCI, immediate MV-PCI or staged MV-PCI in patients with STEMI and multivessel coronary artery disease. Studies focused on patients diagnosed with cardiogenic shock or chronic total occlusion (CTO) were excluded to ensure similar baseline characteristics. The quality of the included randomized trials was evaluated using Review Manager 5.3.
Data extraction
Two independent authors (M.-J.H. and J.-S.T.) extracted information regarding the study design, interventions performed, number and characteristics of patients enrolled, definition of multivessel coronary artery disease, inclusion and exclusion criteria, clinical outcomes, follow-up duration and baseline characteristics of the included patients. Any discrepancies were resolved by consensus with third-party adjudication (X.-J.G.).
Outcomes
In analyses based on randomized trials, the primary outcomes were major adverse cardiac events (MACE), all-cause death, myocardial infarction and revascularization. We preferentially utilized data from the longest available follow-up as long-term outcomes. Secondary outcomes defined as cardiac death, angina, heart failure and rehospitalization together with safety outcomes defined as major bleeding, renal failure and stroke were also investigated. In prospective observational studies, short- and long-term all-cause death were investigated.
Statistical analysis
Raw, unadjusted data from the included randomized trials and prospective observational studies were extracted. Random-effects models of DerSimonian and Laird were used to construct the summary estimated risk ratio (RR) and the corresponding 95% confidence interval (CI). Statistical heterogeneity was examined using the I2 statistic, with I2 being considered substantial when it was >50%. 16 Begg’s method and funnel plot were used to estimate publication bias. 17 Sensitivity analysis was performed using a leave-one-out analysis to assess whether the pooled results were influenced by a single trial. All analyses for the pairwise meta-analysis were performed using STATA software version 14 (STATA Corporation, College Station, Texas). Meanwhile, network meta-analysis was carried out using the ‘network’ command in STATA software. 18 We performed trial sequential analysis (TSA) to assess the reliability and conclusiveness of the present evidence, anticipating a 25% RR reduction for efficacy outcomes, α = 5%, 1 − β = 80%. 19 TSA was conducted using TSA software, version 0.9 beta (Copenhagen Trial Unit, Copenhagen, Denmark).
Results
Search process, study characteristics and quality assessment
Our initial search yielded 5286 articles. Ultimately, 13 randomized trials enrolling 7627 patients and 21 prospective observational studies enrolling 60311 patients met our inclusion criteria. Figure 1 reports how the eligible studies were identified. Table 1 reports the characteristics of the included randomized trials. Overall, two trials5,20 compared immediate MV-PCI with CO-PCI, four trials6,8,21,22 compared mixed MV-PCI (either immediate or staged) with CO-PCI, five trials7,9,23–25 compared staged MV-PCI with CO-PCI and two trials26,27 compared staged MV-PCI with immediate MV-PCI. Meanwhile, in three trials,7,8,23 non-IRA was revascularized with the guidance of FFR. Table 2 summarizes the baseline characteristics of the included patients. The patients were more likely to be old males with a history of hypertension and diabetes. Over time, more drug-eluting stents (DES) were adopted. Figure 2(a)–(d) and Figure 2(e) and (f) report the evidence of the included randomized trials and prospective observational studies, respectively. Figure 2(g) summarizes the measures of study quality. The characteristics of the included prospective observational studies are shown in Supplementary Table 1.
Main characteristics of included randomized trials.
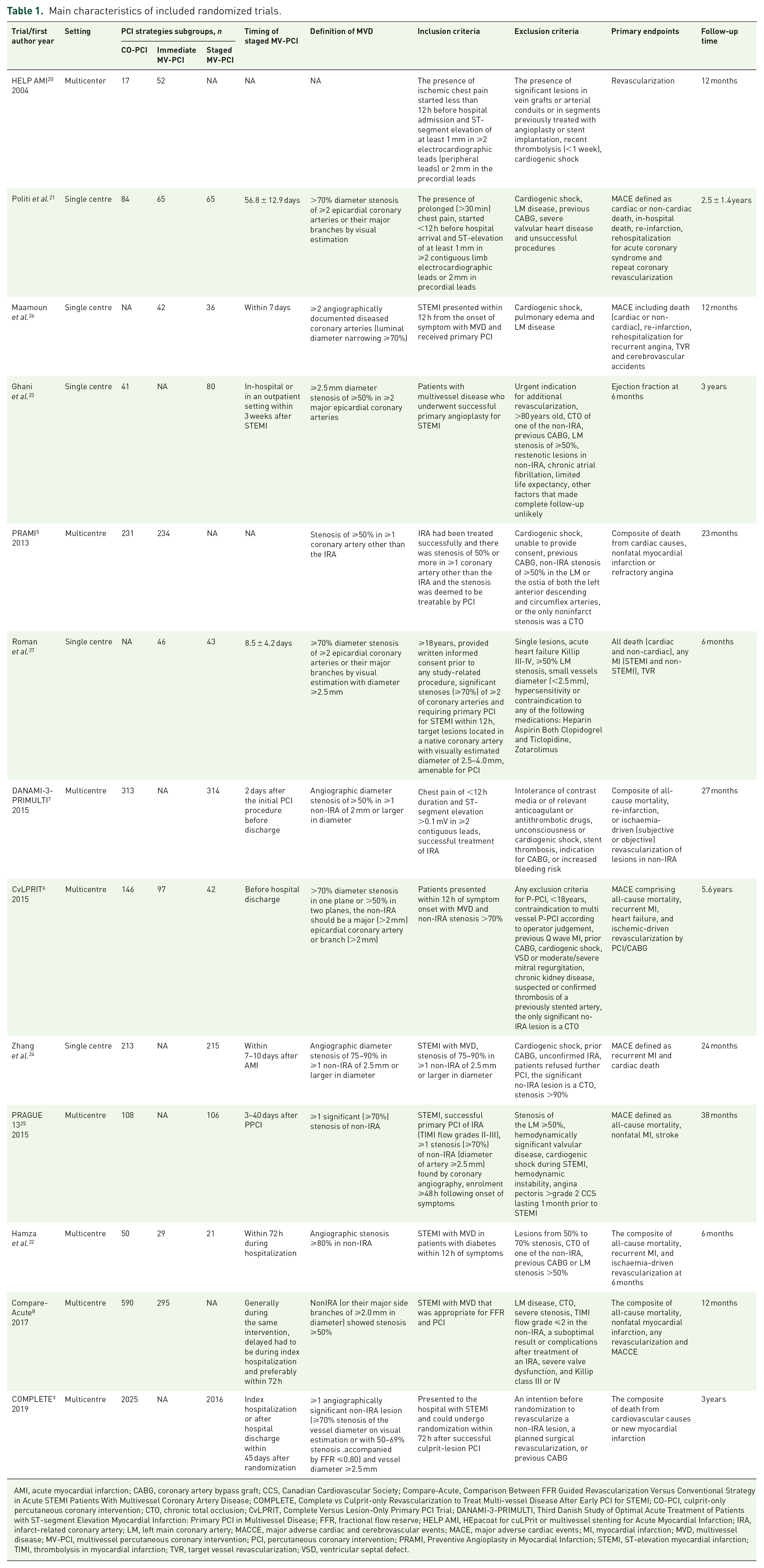
AMI, acute myocardial infarction; CABG, coronary artery bypass graft; CCS, Canadian Cardiovascular Society; Compare-Acute, Comparison Between FFR Guided Revascularization Versus Conventional Strategy in Acute STEMI Patients With Multivessel Coronary Artery Disease; COMPLETE, Complete vs Culprit-only Revascularization to Treat Multi-vessel Disease After Early PCI for STEMI; CO-PCI, culprit-only percutaneous coronary intervention; CTO, chronic total occlusion; CvLPRIT, Complete Versus Lesion-Only Primary PCI Trial; DANAMI-3-PRIMULTI, Third Danish Study of Optimal Acute Treatment of Patients with ST-segment Elevation Myocardial Infarction: Primary PCI in Multivessel Disease; FFR, fractional flow reserve; HELP AMI, HEpacoat for cuLPrit or multivessel stenting for Acute Myocardial Infarction; IRA, infarct-related coronary artery; LM, left main coronary artery; MACCE, major adverse cardiac and cerebrovascular events; MACE, major adverse cardiac events; MI, myocardial infarction; MVD, multivessel disease; MV-PCI, multivessel percutaneous coronary intervention; PCI, percutaneous coronary intervention; PRAMI, Preventive Angioplasty in Myocardial Infarction; STEMI, ST-elevation myocardial infarction; TIMI, thrombolysis in myocardial infarction; TVR, target vessel revascularization; VSD, ventricular septal defect.
Baseline characteristics of included patients in randomized trials.
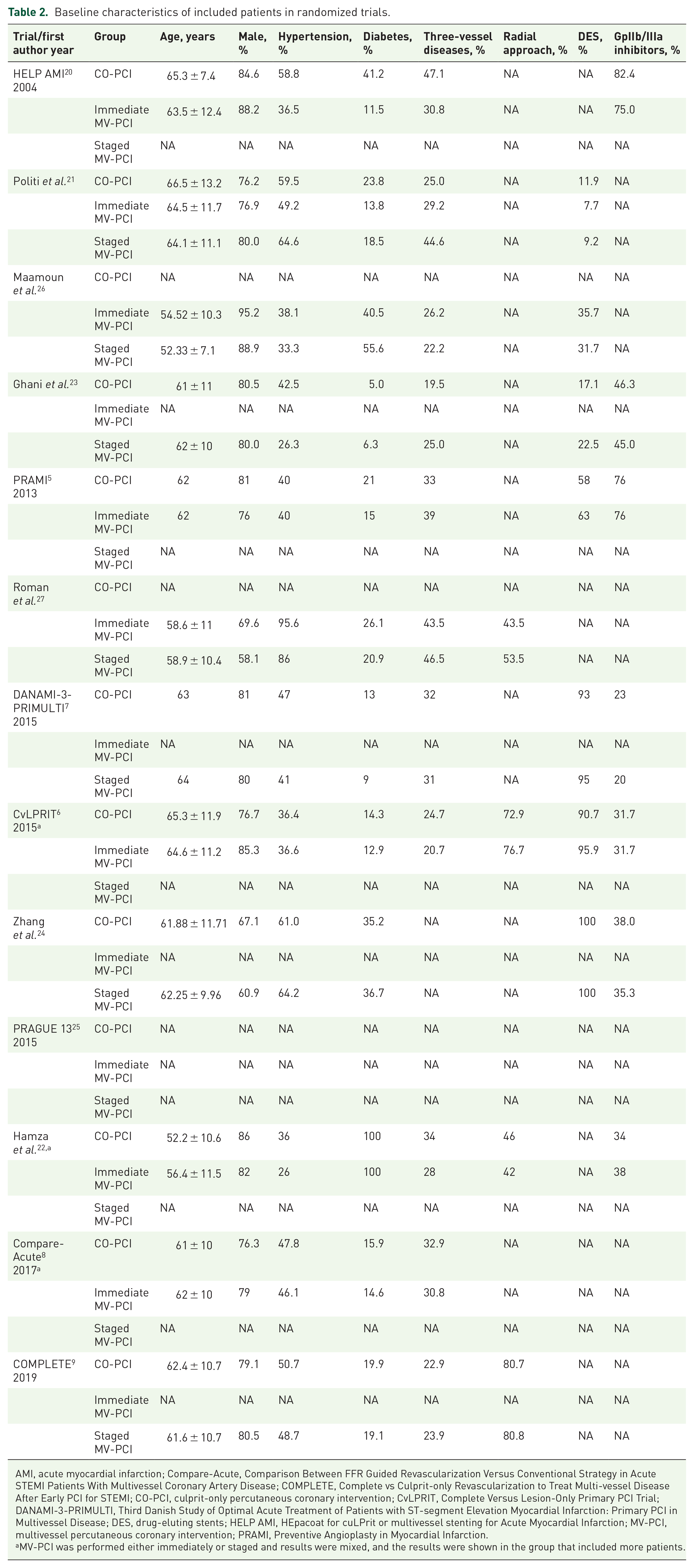
AMI, acute myocardial infarction; Compare-Acute, Comparison Between FFR Guided Revascularization Versus Conventional Strategy in Acute STEMI Patients With Multivessel Coronary Artery Disease; COMPLETE, Complete vs Culprit-only Revascularization to Treat Multi-vessel Disease After Early PCI for STEMI; CO-PCI, culprit-only percutaneous coronary intervention; CvLPRIT, Complete Versus Lesion-Only Primary PCI Trial; DANAMI-3-PRIMULTI, Third Danish Study of Optimal Acute Treatment of Patients with ST-segment Elevation Myocardial Infarction: Primary PCI in Multivessel Disease; DES, drug-eluting stents; HELP AMI, HEpacoat for cuLPrit or multivessel stenting for Acute Myocardial Infarction; MV-PCI, multivessel percutaneous coronary intervention; PRAMI, Preventive Angioplasty in Myocardial Infarction.
MV-PCI was performed either immediately or staged and results were mixed, and the results were shown in the group that included more patients.

PRISMA flow of the study search.

Network evidence and risk of bias of included studies. Network evidence plot for primary outcome of randomized trials (a–d), all-cause death of prospective observational studies (e and f), and risk of bias of included randomized trials (g).
Pairwise meta-analysis of MACE, myocardial infarction and revascularization based on randomized trials
Compared with CO-PCI, MV-PCI was associated with lower risks of long-term MACE (RR: 0.58; 95% CI: 0.45 to 0.74; I2 = 57.0%), myocardial infarction (RR: 0.67; 95% CI: 0.51 to 0.88; I2 = 18.8%) and revascularization (RR: 0.38; 95% CI: 0.28 to 0.51; I2 = 63.5%) (Supplementary Figures 1–3), which were not influenced by the strategy (immediate, staged or mixed) or the guidance of MV-PCI.
Pairwise meta-analysis of all-cause death based on randomized trials and prospective observational studies, respectively
Analyses based on randomized trials revealed that the risk of all-cause death was similar between MV-PCI and CO-PCI (RR: 0.85; 95% CI: 0.69 to 1.04; I2 = 0.0%) (Figure 3). However, analyses based on prospective observational studies suggested that compared with CO-PCI, immediate MV-PCI increased the risk of short-term all-cause death (RR: 1.58; 95% CI: 1.22 to 2.05; I2 = 43.8%), whereas the risk of long-term all-cause death (RR: 1.15; 95% CI: 0.83 to 1.58; I2 = 91.6%) was similar (Figure 4(a)). The risk of short-term all-cause death (RR: 0.29; 95% CI: 0.03 to 2.58; I2 = 62.0%) was similar between staged MV-PCI and CO-PCI, yet staged MV-PCI decreased the risk of long-term all-cause death (RR: 0.53; 95% CI: 0.39 to 0.71; I2 = 15.6%) (Figure 4(b)). Immediate MV-PCI increased the risks of both short-term (RR: 3.11; 95% CI: 2.11 to 4.57; I2 = 0%) and long-term all-cause death (RR: 2.24; 95% CI: 1.37 to 3.66; I2 = 80.1%) compared with staged MV-PCI (Figure 4(c)).

Pairwise meta-analysis of long-term all-cause death based on randomized trials.

Pairwise meta-analysis of all-cause death based on prospective observational studies. (a) Immediate MV-PCI vs Culprit-only PCI, (b) Staged MV-PCI vs Culprit-only PCI, and (c) Immediate MV-PCI vs Staged MV-PCI.
Meta-regression based on randomized trials
The publication year of the study, age, sex, hypertension, diabetes, radial access, DES and administration of glycoprotein IIb/IIIa inhibitor were not associated with long-term MACE, all-cause death, myocardial infarction or revascularization results. However, there was a trend that the prevalence of three-vessel disease was positively associated with MACE (p = 0.086) (Supplementary Table 2).
Pairwise meta-analysis of secondary and safety outcomes based on randomized trials
Compared with CO-PCI, MV-PCI (immediate or staged) was associated with lower risks of long-term cardiac death (RR: 0.73; 95% CI: 0.54 to 0.98; I2 = 6.6%), angina (RR: 0.49; 95% CI: 0.39 to 0.61; I2 = 0.0%) and rehospitalization (RR: 0.44; 95% CI: 0.29 to 0.66; I2 = 4.9%). The risk of long-term heart failure was similar between MV-PCI and CO-PCI (Supplementary Figure 4). Safety outcomes including major bleeding, renal failure and stroke were also similar between MV-PCI and CO-PCI (Supplementary Figure 5). The results of the sensitivity analyses were consistent with the main analyses (Supplementary Figure 6). There was no evidence of publication bias (Supplementary Figures 7).
TSA results of randomized trials
Regarding MACE, myocardial infarction and revascularization, the cumulative z-curve crossed both the conventional boundary (p = 0.05) and the trial sequential boundary, indicating that compared with CO-PCI, MV-PCI reduced the risks of long-term MACE, myocardial infarction and revascularization by 25% with firm evidence (Figure 5). However, regarding all-cause death, the cumulative z-curve crossed the futility boundary, indicating that MV-PCI failed to reduce the risk of long-term all-cause death by 25% compared with CO-PCI. The TSA results for secondary and safety outcomes are shown in Supplementary Figures 8 and 9, respectively.

Results of the TSA for the risks of long-term primary outcomes based on randomized trials. (a) MACE, (b) All-cause death, (c) Myocardial infarction, and (d) Revascularization.
Network meta-analysis of randomized trials
The mixed treatment model showed that MV-PCI in an immediate or staged procedure was associated with lower risks of long-term MACE, myocardial infarction and revascularization than CO-PCI. However, there was no significant difference between immediate and staged MV-PCI regarding MACE, myocardial infarction and revascularization. The risk of long-term all-cause death was similar between any combination of the three different revascularization strategies (Figure 6(a)). CO-PCI showed the highest cumulative probability for increasing the risks of long-term MACE, all-cause death, myocardial infarction and revascularization, followed by staged and immediate MV-PCI strategies (Figure 6(b)–(e)). The contribution, loop consistency, comparison-adjusted and predictive interval plots for network meta-analysis are shown in Supplementary Figures 10–13, respectively.

Forest plot and cumulative probability rankings for the network meta-analysis. Forest plot for the network meta-analysis (a) and cumulative probability rankings (b–e) for long-term primary outcomes based on randomized trials.
Discussion
In the meta-analysis based on randomized trials, we demonstrated that MV-PCI (immediate or staged) was associated with a 42% lower risk for long-term MACE, which was mainly due to a 33% lower risk for myocardial infarction and a 62% lower risk for revascularization. The results above were consistent with the network meta-analysis. However, real-world prospective observational studies suggested that staged MV-PCI decreased both short- and long-term all-cause death, whereas immediate MV-PCI increased the risk of short-term all-cause death relative to CO-PCI.
Our results are consistent with previous meta-analyses of randomized trials suggesting that MV-PCI was associated with reduced MACE.28–30 However, these analyses focused on comparing pooled MV-PCI with CO-PCI rather than exploring the relative benefit from immediate MV-PCI versus staged MV-PCI. This comparison is significant, as immediate MV-PCI is different from staged MV-PCI from both technical and pathophysiological perspectives.31,32 Moreover, immediate and staged MV-PCI strategies have some individual advantages and disadvantages. 33 In our pairwise and network meta-analysis of randomized trials, we did not find that the timing of MV-PCI (immediate or staged) had an impact on long-term clinical outcomes, which means there was a consistent treatment effect for MV-PCI versus CO-PCI, regardless of the timing when MV-PCI was achieved. Meanwhile, the largest randomized trial in the field at present, the COMPLETE trial, 34 showed that the benefit of MV-PCI over CO-PCI was consistent irrespective of the timing of non-IRA intervention (index hospitalization or after hospital discharge). Therefore, achieving MV-PCI, rather than its timing, is the most important determinant of long-term clinical outcomes according to the results of randomized trials.
It is noteworthy that in our included randomized trials, patients were strictly selected, and those with high-risk conditions such as cardiogenic shock, left main coronary artery disease and CTO were excluded from 12 of the included 13 studies. Therefore, the included patients in randomized trials represented strictly selected patients with relatively low-risk profiles compared with patients from real-world scenarios, and caution is advised when extrapolating our findings to real-world populations. Meanwhile, short-term all-cause death within hospitalization or 30 days was not reported, and we are unable to exclude the possibility that the higher rates of long-term MACE and revascularization in the CO-PCI group could be a consequence of competing risks. After all, the pathological inflammatory process in STEMI involves not only the IRA but also the entire coronary tree and can lead to the destabilization and rupture of multiple atherosclerotic plaques, resulting in a sharply increased risk of death. 35 Worse still, the dynamics of this specific inflammatory process are greatest in the first month after STEMI.36,37 If patients receive immediate MV-PCI, unforeseen periprocedural complications in the non-IRA region may be poorly tolerated due to the ‘double jeopardy’ of the IRA and non-IRA regions. 31 Increased radiation exposure caused by prolonged procedure time 38 and a higher risk of contrast-induced nephropathy triggered by increased contrast load may further deteriorate patients’ condition. 6 If patients in the immediate MV-PCI group were unable to tolerate the extremely prothrombotic and inflammatory milieu 39 and died early, they therefore did not survive long enough to develop MACE and revascularization in the long-term course. Correspondingly, the long-term risks of MACE and revascularization were lower in patients undergoing immediate MV-PCI than in those receiving CO-PCI. Based on our abovementioned discussion, investigating the short-term outcomes may provide more information about which one is better when considering immediate and staged MV-PCI. However, the short-term outcomes were unavailable in randomized trials. Therefore, we turned to real-world prospective observational studies for answers. Both short- and long-term follow-ups in prospective observational studies showed that immediate MV-PCI increased the risk of all-cause death when compared with staged MV-PCI. In addition, immediate MV-PCI paradoxically increased the risk of short-term all-cause death compared with CO-PCI, which seems contrary to the results from randomized trials. The randomized CULPRIT-SHOCK trial 40 was dedicated to comparing immediate MV-PCI versus CO-PCI in high-risk patients with STEMI and multivessel coronary artery disease complicated by cardiogenic shock. The results showed that at 30 days, the composite primary endpoint of all-cause death or renal-replacement therapy was 45.9% in the CO-PCI arm versus 55.4% in the immediate MV-PCI arm (p = 0.01). All-cause death was higher in immediate MV-PCI than in CO-PCI (51.6% versus 43.3%, p = 0.03). Therefore, in high-risk patients with STEMI and multivessel coronary artery disease complicated by cardiogenic shock, CO-PCI instead of immediate MV-PCI was advocated due to the reduced risk of short-term all-cause death. However, the rates of long-term (1-year) all-cause death (50.0% versus 56.9%; RR: 0.88; 95% CI: 0.76 to 1.01) and renal-replacement therapy (11.6% versus 16.4%; RR: 0.71; 95% CI: 0.49 to 1.03) were similar between CO-PCI versus immediate MV-PCI, yet rehospitalization for heart failure (5.2% versus 1.2%; RR: 4.46; 95% CI: 1.53 to 13.04) and revascularization (32.3% versus 9.4%; RR: 3.44; 95% CI, 2.39 to 4.95) occurred more frequently with CO-PCI. 38 Based on long-term results from the CULPRIT-SHOCK trial, we may wrongly conclude that immediate MV-PCI was superior to CO-PCI because of the reduced risks of rehospitalization for heart failure and revascularization. It is thought-provoking that during short-term follow-up, immediate MV-PCI increased the risk of all-cause death. Therefore, based on results from real-world prospective observational studies and the CULPRIT-SHOCK trial, a staged MV-PCI strategy may be the best option. Meanwhile, staged MV-PCI enables operators to have more time to appropriately evaluate the risks and benefits of additional revascularization, perhaps resulting in better patient selection 41 and avoiding the overestimation of stenosis severity in the acute phase of STEMI. 42 However, because of the deficiency of randomized trials comparing immediate and staged MV-PCI directly, the benefits of staged MV-PCI should be evaluated in future randomized trials. Two ongoing trials, MULTISTARS AMI (NCT03135275) and BioVasc (NCT03621501), which test the outcomes between immediate MV-PCI and staged MV-PCI, will help to further clarify the options of different MV-PCI strategies.
Another finding of our meta-analysis is the consistent benefit of MV-PCI guided by angiography or FFR. Meanwhile, two ongoing trials, FLOWER-MI (NCT02943954) and FRAME-AMI (NCT02715518), are comparing clinical outcomes following FFR-guided versus angiography-guided PCI in the treatment of non-IRA stenosis.
Our meta-analysis included studies from 2004 to 2019, and it is obvious that the use of DES increased with time in the included studies. Moreover, a study suggested that for STEMI patients receiving DES, a trend towards lower long-term mortality at 1 year was observed in comparison to the bare metal stent (BMS). 43 Therefore, meta-regression was performed to investigate whether the publication year and the percent of DES used may influence outcomes. However, the results indicated that publication year and DES did not exert effects on the association between the PCI strategy and clinical outcomes. In addition, during the past years, the recommendation for the PCI strategy for STEMI patients with multivessel coronary artery disease has changed oppositely. The 2013 American College of Cardiology Foundation/American Heart Association (ACCF/AHA) guidelines did not recommend the MV-PCI strategy for STEMI patients with multivessel coronary artery disease (Class III, Level B), mainly based on observational studies. 44 In 2015, they upgraded the MV-PCI recommendation to Class IIb (Level B), 45 which was similar to the European Society of Cardiology/European Association of Cardiothoracic Surgery (ESC/EACTS) guideline in 2015. 46 With the publication of more well-designed randomized trials and meta-analyses, the latest 2017 ESC guideline has upgraded MV-PCI to Class IIa (Level A). 47 Therefore, recommendations are revised with the emergence of randomized trials and meta-analyses, and further randomized trials are needed to establish a solid conclusion on the optimal PCI strategy for STEMI patients with multivessel coronary artery disease.
Study limitations
First, the PRAGUE 13 trial has not yet been published, and we were unable to obtain the baseline characteristics. Meanwhile, the number of patients in the COMPLETE trial was large (4041, 53.0%); therefore, the results of our meta-analysis could have been skewed towards biases within the COMPLETE trial. However, sensitivity analyses performed by excluding these studies yielded similar results to the main analysis. Second, data are from different health care systems, different populations and different endpoint definitions, which might potentially increase the heterogeneity and impact the outcomes. Moderate degrees of heterogeneity were observed in MACE, revascularization and heart failure in the pairwise meta-analysis. We attempted to mitigate this heterogeneity with several strategies, including using a random effects model and further subgroups of the MV-PCI strategy or the guidance of revascularization in our analysis. Third, although the included patients were all diagnosed with STEMI, yet the exclusion criteria were a little different, such as CTO, with some trials excluding CTO, yet others did not mention it. Finally, short-term outcomes were not reported in randomized trials and we were unable to evaluate the relative merits during short-term follow-up based on randomized trials.
Conclusion
Based on randomized trials, our findings demonstrated that MV-PCI in an immediate or staged procedure should be preferred for patients with STEMI and multivessel coronary artery disease compared with CO-PCI, which improved the long-term prognosis, but no data were reported on the short-term prognosis. As a supplement, the results in real-world patients derived from prospective observational studies suggested that staged MV-PCI was superior to immediate MV-PCI in the consideration of both short- and long-term all-cause death. Therefore, staged MV-PCI may be the optimal PCI strategy for patients with STEMI and multivessel coronary artery disease.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-docx-2-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-1-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-1-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-10-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-10-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-11-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-11-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-12-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-12-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-13-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-13-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-2-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-2-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-3-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-3-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-4-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-4-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-5-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-5-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-6-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-6-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-7-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-7-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-8-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-8-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-9-taj-10.1177_20406223221078088 – Supplemental material for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis
Supplemental material, sj-tif-9-taj-10.1177_20406223221078088 for The optimal percutaneous coronary intervention strategy for patients with ST-segment elevation myocardial infarction and multivessel disease: a pairwise and network meta-analysis by Meng-Jin Hu, Jiang-Shan Tan, Wen-Yang Jiang, Xiao-Jin Gao and Yue-Jin Yang in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors thank Xin-Hui Wang and Song Hu for their guidance in the software manipulation.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the CAMS Innovation Fund for Medical Sciences (CIFMS) (2016-I2M-1-009) and the Twelfth Five-Year Planning Project of the Scientific and Technological Department of China (2011BAI11B02).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
