Abstract
Aim:
The purpose of this study was to investigate whether there is an association between smoking, the extent of the degeneration process in the biceps tendinopathy, including cells and extracellular matrix (ECM) alterations, and long-term surgical results.
Methods:
This study comprised 40 consecutive patients admitted for shoulder arthroscopy due to symptomatic biceps tendinopathy and classified into three groups based on smoking status: active smokers, former smokers, and non-smokers. According to the classical Bonar score criteria, the histopathologic evaluation of the harvested intra-articular portion of the tendon was done. The follow-up examination was based on the American Shoulder and Elbow Surgeons Score (ASES).
Results:
A cohort of 32 patients was enrolled in the final follow-up examination; mean 37.56 months. Histopathological evaluation according to the classical Bonar score revealed degeneration of the tendinous tissue in each group but there was no correlation between the extent of degeneration, smoking indexes and the ASES. After revision of Bonar scale within the vascularity criterion, we found a correlation between the extent of degeneration of tendinous tissue, smoking data, ASES score, and the severity of rotator cuff injury.
Conclusion:
In this paper, we indicate the ambiguous role of the neovascularization in the biceps tendinopathy, and it was used for modification of the classical Bonar score. Consequently, recalculated, modified Bonar score was correlated positively with smoking indexes and functional outcomes. Furthermore, the morphological alterations of rotator cuff tendons also correlated positively with the extent of biceps tendon degeneration, measured according to the modified scoring system.
Introduction
Tobacco smoking is a serious threat, especially to respiratory, circulatory, and immune systems. An aerosol of hazardous substances contained in tobacco smoke also negatively influences the musculoskeletal system.1–3 Tobacco smoking causes osteoporosis and alveolar bone loss and also delays fracture healing. In soft tissue, it causes muscle mass and strength loss, causing muscle pain, and increases the risk of pathologies such as intervertebral disc degeneration and tendinitis, and reduces tendon healing. 3 Thus, tobacco smoke adverse effects are concentrated on different tissues, leading to a specific alteration in cells, vascular bed, and extracellular matrix (ECM).4,5
Tendinopathy is a chronic tendon disorder caused by impaired regeneration, which is characterized by disruption of the collagen architecture, increased proteoglycans, altered tenocyte morphology, and vascular bed expansion. 6 Rotator cuff (RC) tendinopathy and long head of biceps (LHB) tendinopathy, which are usually seen together, constitute an important part of shoulder disorders.7–9 At the microscopic level, Lundgreen et al. observed pronounced degenerative changes with reduced local cell density and increased apoptotic cell death in smokers’ tendons obtained from RC. The authors linked this finding clinically with reduced tendon healing capacity. 9 Furthermore, Mallon et al. found that smokers who underwent RC repair have worse postoperative outcomes than non-smokers. 10
Moreover, Baumgarten et al. revealed a dose-dependent and time-dependent relationship between smoking and rotator cuff tears (RCTs). 4 Overall, smoking is considered as an important risk factor for the development and progression of RC pathology. Despite a few studies evaluating the relationship between smoking and rotator cuff pathology, no study to date connects the extent of histopathologic alterations in the biceps tendinopathy with functional results after shoulder surgery in a smoker population.11–14
The most popular and established scale with which to assess the microscopic alterations of tendons is the Bonar score. This semiquantitative scoring system evaluates four main variables: tenocyte morphology alterations, the ground substance accumulation, the extent of neovascularization, and collagen bundles architecture. However, a few authors tried to modify the Bonar score, adding some new variable or changing the present one.15–17 Okazaki et al. and Docking et al. expanded the classical Bonar score with a fifth variable, counting the number of fibroblasts. Fearon et al. in their study revealed that a complete lack of vascularity in tendinopathy should be rather treated as a pathology, similarly to accumulation of newly formed vessels. The neovascularization variable seems to be ambiguous and should be reconsidered in the future scales to assess the tendon degeneration.
This study aimed to investigate whether there is an association between smoking, the extent of the degeneration process in the biceps tendinopathy, including cells and ECM alterations, and long-term surgical outcomes.
Based on our previous study, we hypothesized that smokers have worse clinical outcomes pre- and post-operatively with more advanced histopathologic alterations in tendinous tissue, increased rate of failures, and a strong association between microscopic alterations and functional results. 18
Methods
Patients and smoking status
This study comprised 40 consecutive patients admitted for shoulder arthroscopy to the Department of Orthopedic Surgery (2016–2018) due to symptomatic LHB tendinopathy. Inter-ventions on the biceps tendon were generally performed as part of a complex surgical procedure. Patients with additional pathologies, such as RCTs and subacromial impingement, were not excluded from the study. All included subjects were diagnosed with LHB tendinopathy based on the clinical examination – tenderness over the bicipital groove test, Speed’s test, Yergason’s test, Abbott–Saunders test, sonographic examination, and non-contrast magnetic resonance imaging of the shoulder. Inclusion criteria were: no response to conservative treatment with a minimum of 3 months of physiotherapy and age >18 years. Exclusion criteria were: the history of systemic inflammatory diseases, previous surgical treatment of the concerned shoulder or corticosteroid injections in the year preceding surgery. Pre-operatively, demographic data and the American Shoulder and Elbow Surgeons Score (ASES) were recorded.
The population was divided into three subgroups according to smoking status: active smokers (did not give up smoking during surgery and after surgery, former smokers (gave up smoking minimum 1 year before surgery), and non-smokers (never used any nicotine supplement, e.g. nicotine gum or patch, oral snuff/moist snuff, cigars and cigarettes). We also recorded dose-dependent and time-dependent data: the period of cigarette smoking – smoking years, the mean number of cigarettes smoked per day and pack-years index (one pack contains 20 cigarettes in our country). Surgeons were blinded to smoking status.
Surgery
Shoulder arthroscopy of all patients was performed with a standard 30° arthroscope (Smith&Nephew) in the beach chair position, under general anesthesia or interscalene brachial plexus block. Additional working portals were used in addition to the standard posterior portal. During the surgery the intracapsular part of the LHB tendon was tested by a probe and investigated for the presence of tears, fraying or any findings for tendinopathy, from its insertion in the labrum, till the bicipital groove. The biceps pulley condition, the LHB tendon stability in the groove, and the extracapsular part of the tendon were also inspected. The arthroscopic classification of LHB tear was made according to the Laffose classification: grade 0 (normal tendon), grade I (minor lesion), grade II (major lesion). 19 All patients underwent biceps tenodesis or tenotomy, depend on the surgeon’s decision, followed by the excision of the intracapsular part of LHB tendon – a sample for further histological study. Concomitant lesions were treated arthroscopically, and RCTs were classified according to Snyder Classification. 14 The rehabilitation protocol after shoulder arthroscopy varied according to surgical procedures and consisted of a six-week physiotherapy program. The rehabilitation protocol after tenotomy included an arm sling, worn for 3 weeks, with early passive range of motion exercises and active shoulder range of motion exercises. Active flexion of the elbow was allowed as of the third week post-surgery. On the other hand, the rehabilitation protocol after the tenodesis procedure included an arm orthosis, worn for 5 weeks, and passive shoulder range of motion exercises, subsequently followed by an active exercise started from the fourth to fifth week. However, if there was an RCT repair an arm abduction orthosis was recommended.
Histopathological evaluation
The resected biceps tendon fragments were fixed in fresh and sterile 10% buffered formalin. After 24 h they were dehydrated and paraffin embedded, prepared for further histologic evaluation, and stained using the hematoxylin and eosin, Alcian blue and Trichrome protocols. Alcian blue and Trichrome staining were performed to inspect the ground substance elements (glycosaminoglycans and mucopolysaccharides). The prepared slides were then examined using light microscopy (Olympus BX46, Tokyo, Japan). The evaluation was conducted by three observers experienced in tendinous tissue, blinded to the sample identity. The extent of histopathologic alterations was evaluated according to the classical Bonar score assumptions.17,20 This scoring system evaluates four main variables: tenocyte morphology, ground substances elements accumulation, neovascularity, and collagen architecture. For each variable 0–3 points were attributed (0 – normal tissue, 3 – extreme pathology), a completely normal tendon would score 0 and a maximally degenerated tendon would score 12.
In our previous study, we observed less new vessel formation in the tendinopathy process in the smoker group than in the non-smoker group. 18 Thus, we decided to reverse the attributes of neovascularization variable in the Bonar scale; three points are given for normal tendon with an average occurrence of the blood vessels – absent neovascularization; two points for incidental cluster of capillaries less than one per 10 high power fields (HPFs) – mild neovascularization; one point for 1–2 clusters per 10 HPFs – moderate neovascularization; zero points for more than two clusters per 10 HPFs – abundant neovascularization (Table 1).
Comparison between classical Bonar score and modified Bonar score.
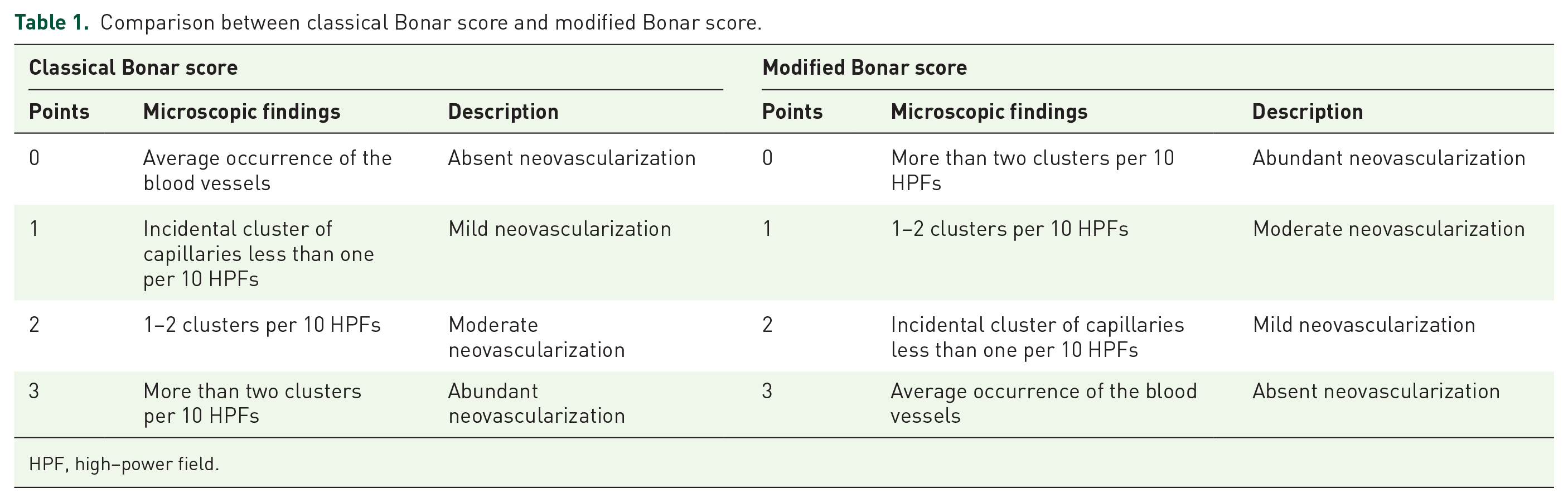
HPF, high–power field.
This decision was linked with our previous findings, where we proved that the neovascularization process was remarkably reduced in patients with smoking history due to ECM alterations. Smoking impairs vascularization of the biceps tendon in chronic tendinopathy cases, by deposition of highly compacted, non-collagenous ECM. 18
Outcome evaluation
The follow-up examination 2–4 years after surgery included functional outcomes assessment by the physical examination, the ASES scale, reoperations rate and smoking habits re-evaluation. Of those 40 patients, eight patients were lost in the follow-up because they were unavailable for final follow-up or did not respond to contact.
Statistical analysis
All the comparisons between groups and statistical analyses were performed by two independent investigators using Prism software (GraphPad). p-value < 0.05 was considered to be statistically significant. Circular plots were generated by Circos software. Variables were tested for normality by the Shapiro–Wilk test. Relations between the studied parameters were assessed using the Spearman Rho correlation coefficient. The data were compared with the non-parametric Mann–Whitney U and analysis of variance tests.
Bioethics
The study was performed following the Declaration of Helsinki for experiments involving humans, after receiving permission from the local Bioethics Committee (approval number KB 598/2016). All patients provided written informed consent before participating in the study.
Results
Patient characteristics
The 32 patients included in the study were divided into three groups according to their smoking history: active smokers (15 patients with 17.8 mean pack-years history, 46.87%), former smokers (six patients with 12.5 mean pack-years history, 18.75%), and non-smokers (11 patients who never smoked, 34.37%). The mean age at diagnosis was 51.75 years (range, 28–75 years); 53.73 years (range, 33–62) for active smokers, 54.17 years (range, 33–74 years) for former smokers, and 47.72 years (range, 28–75 years) for non-smokers. The mean number of cigarettes smoked per day was 17.5 in former smokers (range, 10–40), whereas it was 16 in active smokers (range, 10–40). In addition, the mean period of smoking was 12.5 years in former smokers (range, 5–20 years) and 20.13 years in active smokers (range, 5–30 years). Patient demographic data and characteristics are summarized in Table 2.
Summary of demographic and clinical characteristics.

ANOVA, analysis of variance; min., minimum; max., maximum; pts, points.
Surgery
In the examined cohort the LHB tenodesis (26 patients) or tenotomy (six patients) was performed on each patient. Moreover, each patient underwent additional procedures: eight patients had RCT repair (C-2 according to Snyder classification) and 13 patients had massive RCT repair (C-4 according to Snyder classification), 11 patients had subacromial impingement with RC tendinopathy (B-1 and B-2 changes in RC according to Snyder classification), with acromion-clavicular joint (ACJ) and acromion resections, joint debridement and coracohumeral ligament resection. There were no patients with isolated LHB tendinopathy. Among the 32 cases all of the LHB tendon tears were grade II, according to Lafosse classification.
Histopathological assessment
The histological evaluation of the microscopic slides revealed degeneration of the tendinous tissue according to classical Bonar score in each group; active smokers 8.13/12 points (range 4–11), former smokers 8.17/12 points (range 7–10), and non-smokers 7.72/12 points (range 5–11). There were no statistical differences between the Bonar scores in groups (Figure 1A–D). There was no correlation between the mean extent of degeneration measured in classical Bonar score and smoking variables (Figure 1E–G). There was also no correlation between the mean extent of degeneration measured in classical Bonar score and age (Figure 1H).

Summarized statistical analysis depending on the smoking status, classical Bonar score, age, Snyder classification, and pre- and post-operative ASES. A, B: comparison and dependence of classical Bonar score according to smoking status. C, D: comparison and dependence of classical Bonar score according to smoking history. E–H: correlation between the classical Bonar score and the smoking years, number of cigarettes smoked per day, pack-years, and age. I–K: correlation between the classical Bonar score and pre-operative ASES, post-operative ASES, and Snyder classification.
We analyzed the neovascularization variable of the Bonar score in each group: active smokers (mean 1.46 points), former smokers (mean 1.5 points), and non-smokers (mean 2 points). Moreover, the neovascularization was absent only in subjects from the active and former smokers groups. Furthermore, the active smokers had the most severe ECM alterations (mean 2.66 points) compared with former smokers (mean 2.5 points) and non-smokers (mean 2.09 points).
According to the modified Bonar scoring, the non-smoker group had 6.73/12 points (range 5–8). In contrast, the former smoker group had 8.17/12 points (range 7–11), and the active smoker group had 8.2 points (range 5–11) (Supplemental material Table 3 online). There were no statistical differences between the modified Bonar scores in groups except the non-smoker and active smoker groups (p = 0.0212).
Outcome evaluation
The average follow-up period after surgery was 37.56 months (range 24–44 months). There were no wound infections or reoperations reported during the follow-up period. Patients admitted that they did not change their smoking habits during the follow-up period. The clinical tests for LHB tendinopathy were positive in one patient from the non-smoking group, three patients from the former smokers group and four patients from the active smokers group.
In the non-smoking group, the mean preoperative ASES score was 49.36/100 (range, 38–60), in the former smokers group it was 43.66/100 (range, 38–52), whereas in the active smokers group it was 42.46/100 (range, 34–50). The mean post-operative ASES score was 77.45/100 (range, 60–100) in the non-smoking group, in the former smokers group it was 71.5/100 (range, 50–86), whereas in the active smokers group it was 70/100 (range, 58–85). The improvement from pre-operative to post-operative ASES was statistically significant in all groups (Supplemental Table 4).
There was no correlation between the extent of degeneration in tendons in the classical Bonar score and the ASES, measured pre- and post-operatively, and RCT morphology according to Snyder classification (Figure 1I–K). Also, after changing the neovascularization variable in the classical Bonar scale, we found statistically significant differences only between non-smokers and active smokers (p = 0.0212) (Figure 2A–D). However, there was a positive correlation between the extent of degeneration measured in the modified Bonar score and all smoking variables, which were not statistically significant using the classical Bonar score (Figure 2E–G). Similarly, the modified Bonar score positively correlated with the age (p = 0.0317, Spearman r = 0.3320) (Figure 2H). Also, we found a statistically significant negative correlation between the modified Bonar score and the ASES score, measured pre- and post-operatively (p = 0.0462; Spearman r = 0.3025 and p = 0.0023; Spearman r = 0.4889, respectively) (Figure 2I and J). Furthermore the modified Bonar score positively correlated with degree of RCT morphology according to Snyder classification (p = 0.0327; Spearman r = 0.3297) (Figure 2K).

Summarized statistical analysis depending on the smoking status, modified Bonar score, age, Snyder classification, and pre- and post-operative ASES. A, B: comparison and dependence of modified Bonar score according to smoking status. C, D: comparison and dependence of modified Bonar score according to smoking history. E–H: correlation between the modified Bonar score and the smoking years, number of cigarettes smoked per day, pack-years, and age. I–K: correlation between the modified Bonar score and preoperative ASES, post-operative ASES, and Snyder classification.
Discussion
To the best of our knowledge, this is the first study which assesses the association of smoking, the biceps tendon degeneration, and long-term clinical outcomes after surgery. This paper presents the ambiguous role of the neovascularization variable in the Bonar scoring system and the possible implications in the smoking population.
The associations between smoking and persistent shoulder pain, the size of lesions in RC tendons, and impaired healing process with poor outcomes are well described. Moreover, tobacco smoking is known and accepted as a detrimental agent in RC tendon disorders.10,12,13,21,22 The relationship between RC and LHB tendon pathology is firm and these pathologies may often appear simultaneously. Smoking severely impairs regeneration in RC and leads to poor functional outcomes.2,4,9,14,18 In our study there were no patients with isolated biceps pathology and the most often accompanying pathology was massive RCTs. Furthermore, when we assessed the degeneration of biceps tendon according to the modified Bonar score, it positively correlates with the morphology of RC tendon tears. This observation enhances the relationship between biceps and RC tendon pathologies.
It is known that a broad spectrum of mechanical pathologies can affect the LHB tendon, including instability, subluxation, luxation, and tears.23,24 The transverse humeral ligament is not a separate structure and is formed by the subscapularis tendon, supraspinatus tendon, and posterior lamina of pectoralis major muscle. 24 Moreover, it is often damaged due to RCTs with subsequent biceps tendon instability. It may be the cause of complex shoulder lesions and their influence on the clinical outcomes. The ASES score results from various shoulder lesions (prevalent in all of the patients in this study), simultaneously with RCTs, subacromial impingement, and superior labrum antero-posterior (SLAP) tears. However, all of these pathologies are also under the influence of hazardous substances from tobacco smoke. Park et al. revealed controversies about the impact of smoking on the clinical outcomes after shoulder mini-invasive surgery. 21 On the other hand, Baumgarten et al. have found a relationship between smoking data and a higher rate of RCTs. 12 However, in the present study, the association between the smoking indexes, tendinous tissue degeneration, and post-operative ASES score was found in cases with chronic biceps tendinopathy.
As previously stated, the aerosol of tobacco smoke contains quite a large group of hazardous substances. Therefore, it is not possible to evaluate the effects of nicotine and other substances in tobacco smoke separately. Nicotine is a potent vasoconstrictor and anti-proliferative substance – it decreases collagen synthesis.2–4 Carbon monoxide decreases microperfusion and tissue oxygenation. However, the exact toxic dose of smoke and its dose-dependent effects on tendinous tissue remain unknown. During the 2–4 year follow-up, the active and former smokers had the worst clinical outcomes. Probably all of the hazardous substances of tobacco smoke had a negative influence on the regenerative process in tendinous tissue after shoulder surgery.
Often the shoulder surgeons insist on patients giving up smoking before surgery or even will not operate on severe smokers. Patients who undergo shoulder surgery or conservative treatment should be aware of the effect of smoking on tendon recovery and functional outcomes. Smoking cessation should be advised before treatment. Our study showed decreased functional outcomes in patients with smoking history compared with non-smokers. Moreover, both active and former smokers groups had an advanced degeneration in tendinous samples, in either classical or modified Bonar score. Park et al. and Lundgreen et al. revealed that heavy smokers had a greater incidence of high-grade tendinosis in RCTs.10,21 In our study, the scores gained in the modified Bonar system positively correlated with smoking indexes. According to the findings of our previous study on the neovascularization process in the biceps tendon pathology, we suggest to modify the classical Bonar score and reverse the attributes of the neovascularization variable. 14 If there was more abundant neovascularization, the modified Bonar score was lower, and the degeneration of the tissue was classified lower. On the contrary, if there was no vascularization, the modified Bonar score was higher, and the degeneration was usually classified as severe. Consequently, the modified Bonar score was correlated with smoking indexes and outcome scores, and revealed a statistically significant relationship. The presence of neovascularization, impaired in smokers, is necessary in the repair of the tendinous tissue, specifically, in the initial phase.
Furthermore, Lundgreen et al. noted that pronounced degenerative changes in tendinous tissue might be associated with reduced tendon healing capacity. 10 Functional scores negatively correlated with smoking status in our study may indicate the healing capacity of the tendon. Moreover, the age of the population positively correlated with the degree of tendon degeneration measured in the modified Bonar score, which may indirectly indicate the better regeneration capabilities in a younger population. Sethi et al. showed in their study that the degree of the Bonar score of the degenerated RC and gross tendon appearance did not correlate with multivariate analysis of the ASES score, which is similar to our outcomes. 25 However, when the attributes of neovascularization were reversed in our study, according to the introduced modified Bonar score, we found a correlation between the extent of degeneration of tendinous tissue, post-operative ASES score, and all smoking variables. Results of our previous study suggest that smoking impairs vascularization of the biceps tendon in chronic tendinopathy by deposition of highly compacted, non-collagenous ECM, inhibiting the neoangiogenesis. 18 The balanced neoangiogenesis is critical to tissue repair and wound healing, which, as we suggest, could be affected by smoking.
The main limitation of our study was a relatively modest cohort of patients; however, studies that deal with tendinous tissue pathology have similar patient numbers. The findings should be interpreted with caution because of the simultaneous presentation of the LHB tendon and RC disorders. Still, as we emphasized, this association is unavoidable, for example, due to the biceps tendon microinstability phenomenon. During the arthroscopic procedure we were unable to obtain a complete proximal tendon of the biceps; however, we tried to obtain a part which was the most exposed to friction and compression forces in the shoulder joint. The medical discussion and data about smoking could be biased because they are reported subjectively by patients, but this limitation concerns all authors. Moreover, we could not obtain reliable information about the nicotine concentration that the subjects consumed – the dose-dependency rate cannot be estimated from this study and is, rather, related to animal studies.
Conclusion
In this paper, we presented an ambiguous role of the neovascularization in the biceps tendinopathy. The neovascularization process is crucial in biceps tendinopathy and was significantly reduced in patients with smoking history. It is one of the main variables in the Bonar scoring system, assessing the tendon degeneration. Assuming that neovascularization is an important smoking-dependent variable we suggested to modify the Bonar score and reverse the attributes of neovascularization. Consequently, recalculated, modified Bonar score was correlated positively with smoking indexes and functional outcomes. Furthermore, the morphological alterations of RC tendons correlated with the extent of biceps tendon degeneration measured according to the modified scoring system.
Supplemental Material
sj-pdf-1-taj-10.1177_2040622321990262 – Supplemental material for Relationship between long head of the biceps tendon histopathology and long-term functional results in smokers. A time to reevaluate the Bonar score?
Supplemental material, sj-pdf-1-taj-10.1177_2040622321990262 for Relationship between long head of the biceps tendon histopathology and long-term functional results in smokers. A time to reevaluate the Bonar score? by Jan Zabrzyński, Maciej Gagat, Łukasz Łapaj, Łukasz Paczesny, Alper Yataganbaba, Dawid Szwedowski and Gazi Huri in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-pdf-2-taj-10.1177_2040622321990262 – Supplemental material for Relationship between long head of the biceps tendon histopathology and long-term functional results in smokers. A time to reevaluate the Bonar score?
Supplemental material, sj-pdf-2-taj-10.1177_2040622321990262 for Relationship between long head of the biceps tendon histopathology and long-term functional results in smokers. A time to reevaluate the Bonar score? by Jan Zabrzyński, Maciej Gagat, Łukasz Łapaj, Łukasz Paczesny, Alper Yataganbaba, Dawid Szwedowski and Gazi Huri in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We would like to thank J. Szukalski MD PhD and Prof. Dariusz Grzanka MD PhD for assistance with histopathologic examination and helpful suggestions during manuscript preparation.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
