Abstract
Aims:
We aimed to verify the efficacy and safety of tacrolimus as long-term immunotherapy for the treatment of neuronal surface antibody-mediated autoimmune encephalitis (AE) during the first attack.
Methods:
In this retrospective observational cohort study, patients with neuronal surface antibody-mediated AE who experienced the first attack were enrolled. We compared the outcomes of 17 patients who received tacrolimus with those of 47 patients treated without tacrolimus. Patients were assessed at onset and 3, 6, and 12 months, as well as at the last follow-up, by using the modified Rankin scale (mRS) and the Clinical Assessment Scale in Autoimmune Encephalitis (CASE). The efficacy of tacrolimus was also compared in a subgroup of patients with anti-NMDA receptor encephalitis.
Results:
Among all patients with neuronal surface antibody-mediated AE, those receiving tacrolimus had lower median mRS scores [1 (IQR = 0–1) versus 2 (IQR = 1–3) in controls, p = 0.001)], CASE scores [2 (IQR = 1–3) versus 3 (IQR = 2–7), p = 0.006], and more favorable mRS scores (94.1% versus 68.1%, p = 0.03) at the 3-month follow-up. No difference was found at the last follow-up. There was no significant difference in the occurrence of relapse and adverse events between the two groups (11.8% versus 14.9%, p = 0.75). In the subgroup of patients with anti-NMDA receptor encephalitis, patients treated with tacrolimus had a lower median mRS score at the 3-month follow-up [1 (IQR = 0–2) versus 2 (IQR = 1–3), p = 0.03]; however, no difference in the outcome was detected at the last follow-up.
Conclusion:
Tacrolimus can be used as long-term immunotherapy in patients with neuronal surface antibody-mediated AE during the first attack. Treatment with tacrolimus appears to accelerate the clinical improvement of neuronal surface antibody-mediated AE.
Keywords
Introduction
Autoimmune encephalitis (AE) is acute or subacute onset encephalitis that occurs via autoimmune processes. 1 Since anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis was first reported in 2007, 2 an increasing number of antibodies against neuronal cell-surface antigens have been identified. 3 AE presents with a wide spectrum of symptoms with limbic and extra-limbic dysfunctions. The neurological symptoms may be reversible and respond relatively well to immunosuppressive therapy. 1 Treatment approaches for AE broadly include immunosuppressive drugs and those targeting antibody-mediated immune processes. 4 Corticosteroids, intravenous immunoglobulin (IVIG), and plasma exchange are the commonly used first-line treatments in the acute phase of AE. 5 When the first-line immunotherapy is insufficient, second-line immunotherapy, including rituximab and cyclophosphamide, may be initiated.5,6 Long-term immunosuppression is recommended after relapse following treatment. Azathioprine and mycophenolate mofetil (MMF), as well as rituximab, have been used to reduce subsequent relapse in patients who experienced a clinical relapse.4,7 However, the optimal timing to initiate long-term immunotherapy has not yet been determined. Furthermore, several well-recognized immunotherapeutic agents are still not used in long-term immunotherapy for AE.
Tacrolimus is a common immunosuppressant drug that suppresses the immune system by inhibiting the activation of calcineurin in T cells. 8 It has been widely used to treat most organ transplants and autoimmune neurological diseases, including myasthenia gravis, inflammatory myopathy, and neuromyelitis optica spectrum disorder.9–11 To date, only a few case reports have described tacrolimus immunotherapy for AE.5,12,13 In this cohort study, we evaluated the efficacy and safety profile of tacrolimus as long-term immunotherapy for the treatment of neuronal surface antibody-mediated AE during the initial attack.
Methods
Ethics statement
This study was approved by the ethics committee of Tongji Medical College, Huazhong University of Science and Technology (TJ-IRB20190502). Written informed consent was obtained from all patients. All data analyzed in this study were strictly anonymous.
Study design and participants
In this retrospective observational cohort study, patients with AE were evaluated at the Tongji Hospital of Tongji Medical College, Huazhong University of Science and Technology between January 2017 and January 2020. The inclusion criteria were (1) a diagnosis of definitive AE according to accepted diagnostic criteria, 14 (2) serum and/or cerebrospinal fluid (CSF) tested positive for neuronal cell-surface autoantibodies, and (3) a minimum 1-year follow-up from the onset. The exclusion criteria were (1) diagnosis of possible or antibody-negative AE; (2) tumors, such as ovarian teratomas, extra-ovarian teratomas, small cell lung carcinoma, thymoma, and breast or testicular tumor; (3) lack of first-line immunotherapy; and (4) insufficient clinical data.
All patients were tested for neuronal surface autoantibodies by transfected cell-based assay with serum and CSF, including anti-NMDAR, contactin-associated protein 2 (CASPR2), leucine-rich glioma-inactivated protein 1 (LGl1), γ-aminobutyric acid receptor-B (GABABR), α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR), dipeptidyl aminopeptidase-like protein 6 (DPPX), glycine receptor (GlyR), metabotropic glutamate receptor 5 (mGluR5), dopamine-2 receptors (D2R), and IgLON5.
Clinical data collected from medical records included demographics, clinical features, and CSF, electroencephalogram (EEG), and brain magnetic resonance imaging (MRI) findings. All patients were screened for tumors at the onset. First-line immunotherapy included corticosteroids, IVIG, and plasma exchange and second-line immunotherapy consisted of rituximab and cyclophosphamide. Tacrolimus, azathioprine, and MMF were used for long-term immunotherapy. For analysis, patients with neuronal surface antibody-mediated AE were divided into two groups: a tacrolimus-treated group and a no tacrolimus-treated (control) group. In the tacrolimus group, tacrolimus was initially administered at a dose of 2–3 mg/day after first-line immunotherapy was completed [in most cases with concomitant oral prednisolone (1 mg/kg/day) for 2 weeks]. Prednisolone was then gradually decreased 5 mg every 2 weeks and weaned to 5–10 mg or stopped. Tacrolimus was used for a minimum of 1 year. In the control group, patients underwent first-line immunotherapy and received oral prednisolone. If first-line immunotherapy was insufficient, second-line or long-term immunotherapy was administered. The patients in the control group were not treated with tacrolimus. Follow-up information was obtained at regular intervals after onset and at 3, 6, and 12 months. Neurological status and outcomes were assessed using both the modified Rankin scale (mRS) and Clinical Assessment Scale in Autoimmune Encephalitis (CASE) (Supplemental Table 1).15,16 A favorable outcome was defined as an mRS score ⩽2 at the last follow-up. Relapse was defined as new onset or worsening of symptoms, after at least 2 months of stabilization or improvement.
Statistical analysis
All data were analyzed using SPSS, version 20.0 (IBM Corp., Armonk, NY, USA) and figures were generated using GraphPad Prism 6.0. Data were presented as median ± interquartile range (IQR) or the number of subjects (%). Pearson’s chi-square test was used for comparisons of categorical variable and the Mann–Whitney U test was used for continuous variables. The mRS and CASE scores in the different groups were compared using the Wilcoxon test. The relapse frequency was analyzed with Kaplan–Meier curves using log-rank statistics. A p value of <0.05 indicated a statistically significant association.
Results
Patient characteristics
A total of 64 patients with neuronal surface antibody-mediated AE were included in the study (Figure 1). Seventeen patients (nine men, eight women) with a median age of 29 years were treated with tacrolimus (tacrolimus group). The remaining 47 patients (27 men, 20 women) with a median age of 30 years were included in the control group. There was no significant difference in age (p = 0.38) or sex (p = 0.75) between the two groups. The results of the autoantibody tests are shown in Figure 2. In the 64 patients, the most common neuronal surface antibody was anti-NMDAR (38/64, 59.4%), followed by anti-LGI1 (8/64, 12.5%) and anti-GABABR (6/64, 9.4%). In the tacrolimus group, anti-NMDAR antibodies were detected in 12 patients (70.6%), while anti-DPPX, anti-LGI1, and anti-CASPR2 antibodies were detected in three, one, and one patients, respectively.

Flow chart of the present study.

Distribution by autoantibodies.
Comparative analysis of the groups with or without tacrolimus
Clinical characteristics of the treatment groups are summarized in Table 1. The most common clinical features of the neuronal surface antibody-mediated AE were seizures and psychosis in both groups. There was no difference in the median mRS score [3 (IQR = 3–5) versus 3 (IQR = 3–4), p = 0.70] and CASE score [12 (IQR = 6–18) versus 10 (IQR = 4–18), p = 0.50] at onset between the two groups. The median time from onset to treatment was 18 (IQR = 10–30) days for the tacrolimus group and 15 (IQR = 8–30) days for the control group (p = 0.72). One patient in the tacrolimus group received rituximab as second-line immunotherapy. One patient underwent early rituximab therapy and three underwent additional long-term immunotherapy in the control group. Other clinical, laboratory, and treatment profiles were comparable between the two groups.
Demographic and clinical data of patients with neuronal surface antibody-mediated AE.

AE, autoimmune encephalitis; CASE, Clinical Assessment Scale in Autoimmune Encephalitis; CSF, cerebrospinal fluid; EEG, electroencephalogram; ICU, intensive care unit; MRI, magnetic resonance imaging; mRS, modified Rankin scale; IQR, interquartile range.
Table 2 provides the follow-up data. In the tacrolimus group, the median follow-up duration was 20 (IQR = 13–30) months. The median mRS scores were 1 (IQR = 0–1), 0 (IQR = 0–1), and 0 (IQR = 0–1) at 3, 6, and 12 months of follow-up, respectively. At the last follow-up, the median mRS score was 0 (IQR = 0–0) and 16 patients (94.1%) displayed favorable mRS scores. The CASE score showed a similar tendency toward a gradually decreasing severity over 12 months (Table 2 and Figure 3). Two patients (11.8%) experienced relapse while undergoing tacrolimus treatment. Tacrolimus was well tolerated in all 17 patients and two patients adhered to treatment despite mild adverse events, including mild hepatic insufficiency and elevation of blood glucose. Compared with the control group, patients who received tacrolimus treatment had a lower median mRS score (p = 0.001), CASE score (p = 0.006), and more favorable mRS scores (p = 0.03) at the 3-month follow-up. No difference in the outcome was found at the last follow-up (p = 0.26) (Figure 3). There was no significant difference in the occurrence of relapse between the two groups (p = 0.75). The use of tacrolimus was not statistically associated with the relapse frequency (p = 0.66) (Figure 4).
Follow-up data of patients with neuronal surface antibody-mediated AE.
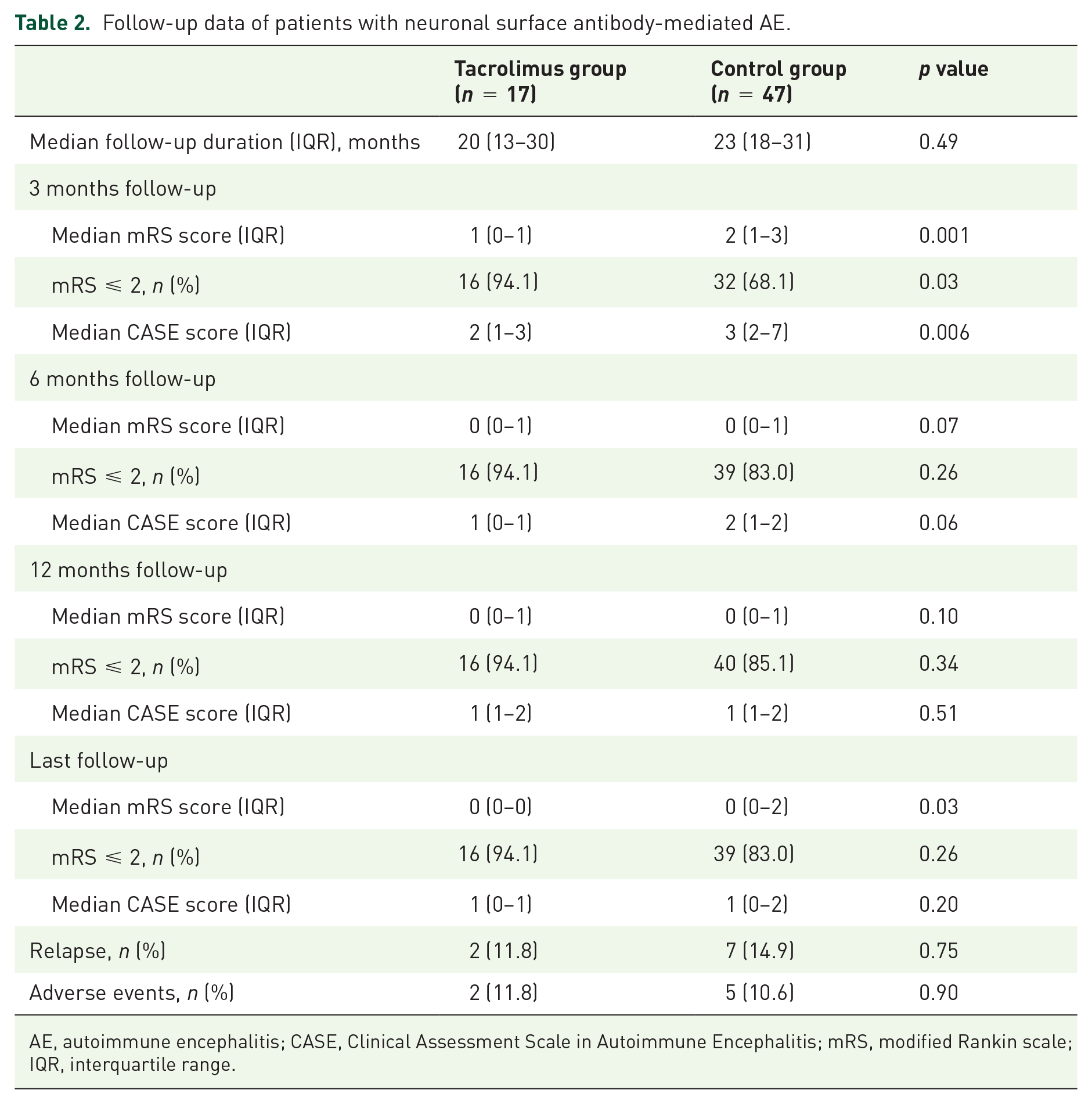
AE, autoimmune encephalitis; CASE, Clinical Assessment Scale in Autoimmune Encephalitis; mRS, modified Rankin scale; IQR, interquartile range.

Distribution of mRS and CASE scores at onset and follow-up in patients with neuronal surface antibody-mediated AE: (a) mRS scores in the control group; (b) mRS scores in the tacrolimus group; (c) CASE scores in the control group; and (d) CASE scores in the tacrolimus group.
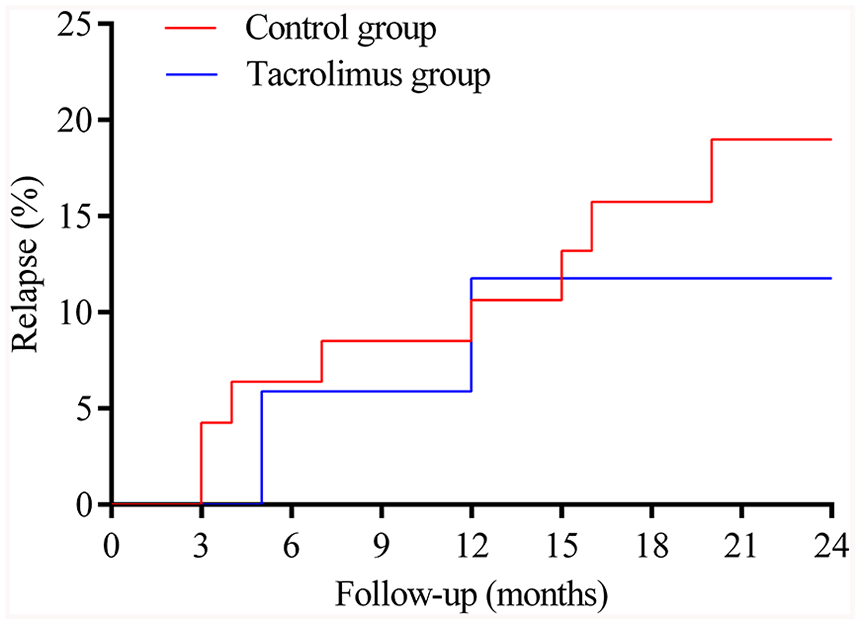
Frequency of relapses in patients with neuronal surface antibody-mediated AE.
Comparative analysis of anti-NMDA receptor encephalitis subgroup
Table 3 shows the demographic, treatment, and outcome data of anti-NMDAR encephalitis patients. A total of 12 patients who received tacrolimus and 26 patients who were treated without tacrolimus were included. We observed no differences in the demographic and clinical profiles between the two groups (Supplemental Table 2). Compared with the control group, patients with tacrolimus treatment had a lower median mRS score [1 (IQR = 0–2) versus 2 (IQR = 1–3), p = 0.03] at the 3 months follow-up, but no difference in the median CASE scores or favorable mRS scores at 3, 6, and 12 months and the last follow-up (Table 3, Supplemental Figure 1). There was no significant difference in the occurrence of relapse between the two groups (16.7% versus 19.2%, p = 0.85). Moreover, tacrolimus treatment was not statistically associated with relapse frequency (p = 0.88) (Table 3, Supplemental Figure 2).
Demographic, treatment, and outcome data of patients with anti-NMDAR encephalitis.

AE, autoimmune encephalitis; CASE, Clinical Assessment Scale in Autoimmune Encephalitis; mRS, modified Rankin scale; NMDAR, N-methyl-D-aspartate receptor; IQR, interquartile range.
Discussion
This study is the first observational cohort study to evaluate the efficacy and safety profile of tacrolimus in the treatment of AE. Our data revealed that tacrolimus is an effective long-term immunotherapy option for neuronal surface antibody-mediated AE during the first attack. Tacrolimus treatment mediated clinical improvement and favorable outcomes at 3 months after disease onset. Furthermore, tacrolimus exhibited a safety profile, with infrequent adverse events.
AE includes a range of neurological disorders that can result in severe disability or death if not recognized and treated early. 17 To date, there is still a lack of randomized trials available to guide immunotherapy of AE. Current treatment regimens are based on expert opinions or experiences from other autoimmune neurological diseases, such as myasthenia gravis, neuromyelitis optica spectrum disorder, and Guillain–Barré syndrome. The clinical effectiveness of immunotherapy in the acute phase has been confirmed by previous studies; however, the clinical experience of long-term management of AE is currently insufficient.1,4,7 The conventional long-term immunotherapeutic agents include azathioprine and MMF. A few reported studies include the use of immunosuppressants such as tacrolimus, cyclosporine, and methotrexate.5,18 In addition, rituximab is recognized as both a second-line treatment for acute immunotherapy and a long-term agent for relapse cases. 1 In this study, we focused on tacrolimus, which is used to treat several autoimmune disorders involving humoral immunity such as rheumatoid arthritis, systemic lupus erythematosus, myasthenia gravis, polymyositis, and neuromyelitis optica spectrum disorder.9–11,19,20 Tacrolimus, an orally available macrolide calcineurin inhibitor, inhibits the production and release of cytokine [e.g. tumor necrosis factor α (TNF-α), interferon γ (IFN-γ), and interleukin-2] and the activation of T lymphocytes, as well as B-cell functions that are dependent on T-cell activation, and reduces the production of antibodies by inhibiting B cells. 20 In addition, this agent can also affect natural killer (NK) cell by reducing cell degranulation and IFN-γ production. 21 Different from paraneoplastic AE targeted by T-cell-mediated mechanisms, AE associated with antibodies against neuronal surface antigens is functionally disrupted by abnormal humoral immunity. 22 Autoantibodies to the neuronal cell-surface antigens can be directly pathogenic by altering the function of the target protein. 17 Antibody binding may also lead to complement deposition and the activation of NK cells, resulting in disrupted neuron function or cell death. 17 It follows that tacrolimus exerts its effects on humoral immunity to reduce the production of antibodies by regulating B cells via the inhibition of T lymphocytes derived cytokines production, as well as modulating complement and NK cell activation.
Recommendations on the timing of long-term immunotherapy initiation differ based on clinical specifics and there are no clear criteria to determine when to initiate long-term immunotherapy in patients with AE. In a recent survey, 70% of clinicians indicated that they would initiate long-term treatment for neuronal surface antibody-mediated AE after a second attack while 50% indicated that they would start treatment after the initial attack. 1 Several studies have found benefits of initiating long-term immunotherapy after failure of first-line treatments or a relapse.4,5,7,13 However, it remains to be discussed whether long-term immunotherapy used during the first attack of AE is effective. The beneficial effect of tacrolimus during the first episode was a remarkable finding of this study. Moreover, we found that tacrolimus led to a good recovery at early follow-up based on both the mRS and CASE scores. This may be explained by the mechanism influencing glucocorticoid receptor hormone-binding function and enhancing the effect of additional steroid treatment. 23 Therefore, tacrolimus may be effective in accelerating neuronal surface antibody-mediated AE remission. In this study, we did not observe substantial satisfactory clinical outcomes in the tacrolimus group when compared with that in the control group, possibly because most neuronal surface antibody-mediated AE patients responded relatively well to early immunotherapy.
In general, autoantibodies to neuronal cell-surface antigens are associated with a higher recurrence rate than antibodies that recognize intracellular antigens.1,24 The frequency of relapse in the neuronal surface antibody-mediated AE ranges from 10% to 35%.1,25 The recurrence rates in our study are consistent with previous observations. Tacrolimus did not show a lower relapse rate in our study possibly because of the relatively small sample size in our cohort. Therefore, larger prospective trials are required. Relapses often occur within 24 months when immunotherapy is reduced or discontinued.26,27 In the present study, we found that relapses commonly occurred at 6 and 12 months after the initiation of tacrolimus therapy. One of the two patients took tacrolimus for 1 year and relapsed soon after unauthorized discontinuation. First, we speculate that tacrolimus may take several months or more to show therapeutic effects in some AE patients, which was also observed in other autoimmune disorders treated with tacrolimus.28,29 Furthermore, the optimal duration of long-term immunotherapy is currently unclear. Empirical approaches typically involve continuation of long-term immunotherapy from 6 months to 3 years after stability is achieved.1,4,24 However, prolonged use of immunosuppressive agents is a double-edged sword because of a series of side effects that may occur. Tacrolimus may cause various severe adverse effects, such as infection, lymphoproliferative disorders, and progressive multifocal leukoencephalopathy.30,31 Clinical management of tacrolimus can be challenging due to its narrow treatment range and significant variability within and among individuals. In our study, the low incidence of adverse events associated with tacrolimus may be attributed to maintaining a blood tacrolimus concentration below 10 μg/ml. Overall, tacrolimus has relatively mild adverse effects and improves after symptomatic therapy, suggesting the good tolerability of this agent.
One limitation of this study is the observational cohort study design. Neuronal surface antibody-mediated AE is uncommon. To demonstrate the efficacy and safety of tacrolimus as long-term immunotherapy, large-sample prospective controlled studies are warranted. In addition, longer follow-up is needed because relapse up to 6 years after onset has also been reported in patients. 32 Despite these limitations, this study expands the current knowledge of the treatment of neuronal surface antibody-mediated AE and is a precursor for future prospective studies to validate the efficacy of tacrolimus.
Conclusions
Our data suggest that tacrolimus is an effective and tolerable long-term immunotherapy option for treating neuronal surface antibody-mediated AE during the first attack. The use of tacrolimus can maintain favorable outcomes and accelerate recovery. However, tacrolimus is not successful preventing further relapse in our study cohort.
Supplemental Material
sj-docx-1-taj-10.1177_20406223211063055 – Supplemental material for Efficacy of tacrolimus as long-term immunotherapy for neuronal surface antibody-mediated autoimmune encephalitis
Supplemental material, sj-docx-1-taj-10.1177_20406223211063055 for Efficacy of tacrolimus as long-term immunotherapy for neuronal surface antibody-mediated autoimmune encephalitis by Chenchen Liu, Suqiong Ji, Huajie Gao, Zhuajin Bi, Qin Zhang, Ke Shang, Jie Cao and Bitao Bu in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
We are grateful to the patients for their contributions to the study. And we also thank Wuhan Kindstar Diagnostics Co., Ltd., China, for technical support.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 81873758).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
