Abstract
Objective:
The study purpose was to determine the outcomes and factors predictive of primary stent patency for the treatment of central venous occlusive disease (CVOD) in hemodialysis patients.
Methods:
Data of 71 patients with CVOD treated with stent placement from January 2012 to December 2017 were analyzed. Univariate and multivariate analysis was performed to determine factors associated with stent patency. Adverse events related to stent placement were also examined.
Results:
The median primary patency duration of the 71 patients was 16 ± 2.2 months. The cumulative 3-, 6-, 9-, and 12- month primary patency rates were 93%, 72%, 55%, and 51%, respectively. Independent predictors of longer primary patency were vessel diameter > 12 mm, the use of a covered stent, and absence of vessel calcifications. Median primary patency of covered stents was 21 months as compared with only 10 months for bare stents (
Conclusions:
A vessel diameter > 12 mm, the use of a covered stent, and no vessel calcifications are independently associated with a higher primary patency rate after treatment of CVOD in hemodialysis patients.
Introduction
Hemodialysis is the most effective option of dialysis and relies on functional vascular access including an arteriovenous graft (AVG), arteriovenous fistula (AVF), and central venous catheter. 1 Central venous obstruction disease (CVOD) is common and disrupts the function of vascular access. 2 Superior vena cava syndrome caused by CVOD presents as progressive ipsilateral upper extremity swelling, venous hypertension, prolonged bleeding, and low vascular access flow is an important predictor of dialysis access failure. Treatment traditionally involves wire and catheter techniques, followed by angioplasty with or without stenting. 3 Stent placement is indicated in the case of lesions that do not respond to percutaneous angioplasty (PTA). 4 However, more than 75% of stents do not remain patent without any additional intervention within 1 year because of restenosis caused by endothelial hyperplasia in and around the stent. 2 Thus, it is important to identify factors associated with restenosis after stent placement. Primary patency is the key parameter that indicates the effect of the primary treatment, and secondary patency is affected by subsequent treatments. Few studies, however, have examined predict factors for primary patency after stenting to treat CVOD in hemodialysis patients.
In this study, we retrospectively reviewed the data of hemodialysis patients with CVOD treated at our center to identify independent predictors of higher primary patency.
Methods
This study was approved by the Ethics Committee of the First Affiliated Hospital, Sun Yat-sen University (approval number [2020]075). Because of the retrospective nature of the study, the requirement of informed patient consent was waived.
Patients and definitions
Data of 110 consecutive hemodialysis patients with CVOD who received endovascular treatment at our center between January 2012 and December 2017 were retrospectively reviewed. Patients who met the following inclusion and exclusion criteria were included in the analysis.
Inclusion criteria: (1) Hemodialysis performed
Exclusion criteria: (1) Treated without stenting or stenting combined with open surgery; (2) no multi-slice computed tomography angiography examination before procedure; (3) primary patency could not be evaluated for some reason, such as unexplained death, kidney transplantation, or conversion to peritoneal dialysis.
CVOD was defined as total vascular obstruction by a lesion located in central venous system, consisting of the subclavian vein (SCV), innominate/brachiocephalic vein (BCV), and the superior vena cava (SVC) (internal jugular vein excluded). Primary stent patency was defined as the interval between placement of the stent and the next stent-related intervention or ⩾ 50% restenosis confirmed by imaging studies. 5 Patency rates were defined according to the Committee on Reporting Standards for Arterio-Venous Accesses of the Society for Vascular Surgery and the American Association for Vascular Surgery. 6 Anatomical success was defined as <30% residual stenosis after stent placement and demonstrated continuity of the central vein to the right atrium on venography. Procedure complications were recorded and classified according to the Society of Interventional Radiology Standards of Practice Committee Classification of Complications by Outcome (ACR-SIR classification). 4
Stent placement procedure
After application of local anesthesia, percutaneous puncture access in the femoral vein was performed, and a catheter was inserted for venography. After identification of the occlusion lesion, a 4 F sheath (Terumo) was inserted for access. Venography was performed by injecting contrast media on both sides of the lesion to identify the length and position of the occluded segment, and to determine the diameter of normal vein close to the lesion. A conventional technique, sharp recanalization, or a flossing wire technique 7 was performed to cross the occlusive lesion. Indications for the placement of a stent were recoiled lesion after PTA or sharp recanalization in this study. The covered stent used was a Viabahn® (W. L. Gore & Associates Inc., CA, USA), and the bare stents used were a ProtégéTM GPSTM (ev3 Inc., MN, USA) and Absolute Pro (Abbott Vascular, CA, USA). The use of a particular stent was based on the lesion characteristics. Post-stent dilation with a corresponding larger balloon was considered if necessary. After stent placement, venography was performed to identify patency.
Statistical analysis
All statistical analyses were performed with SPSS version 21 software (IBM Corp., Armonk, NY, USA). Continuous data were reported as mean ± standard deviation. The Kaplan–Meier method was used to calculate the cumulative rates of post-intervention primary patency. Univariate and multivariate Cox regression analysis was used to determine independent predictors of primary patency after stent placement. Variables with a value of
Results
We reviewed data of 110 consecutive patients with CVOD treated with an endovascular procedure. A flow diagram of patient inclusion is shown in Figure 1. Of the 110 patients, 5 were not initially treated at our center, 9 could not be treated successfully, 15 received only balloon dilatation, and 7 did not have complete follow-up; therefore, these patients were excluded from the analysis. Thus, 71 patients were included in the study. The procedures of central venous stent placement are shown in Figure 2.

Flow diagram of patient inclusion.

Representative images of CVOD treatment. (a), (b), and (c) are one patient and (d), (e), and (f) are another patient. (a) Computed tomography (CT) reconstruction of occlusion in the initial segment of the brachiocephalic vein. (b) Venography images show the occlusion and collateral circulation. (c) Venography after covered stent placement show blood flow was restored. (d) Venography of another patient showing a long segment occlusion in the left brachiocephalic vein. (e) Venography after bare stent placement. (f) Venography showing bare stent restenosis 4 months later. Stent lumen is vascular endothelial hyperplasia is present.
All patients without restenosis had a follow-up period more than 1 year. The mean follow-up time was 19 months (range: 3–40 months). A Kaplan-Meier graph of post-intervention primary patency is shown in Figure 3. The mean primary patency time was 16 ± 2.2 month.
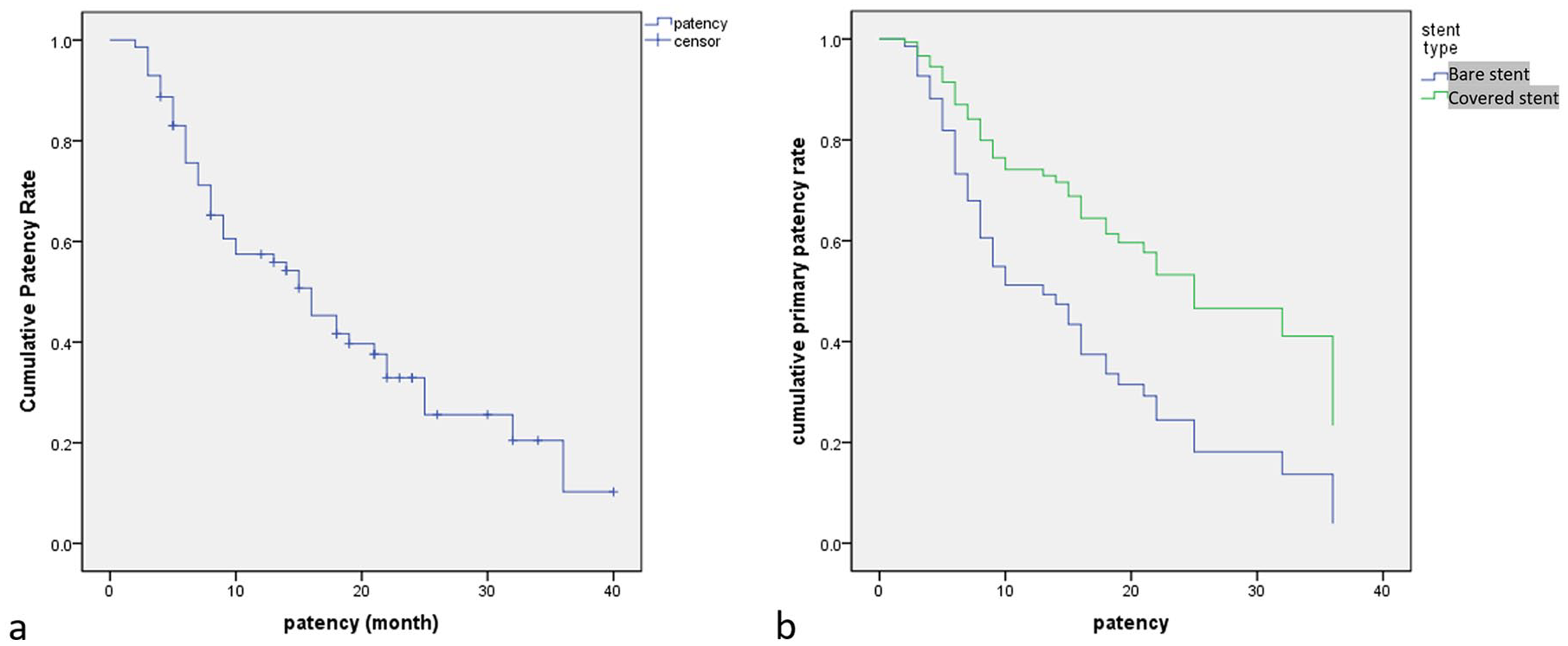
Cumulative primary patency rates of the 71 patients. (a) The median primary patency was 16 ± 2.2 months. (b) KM curve comparing the covered stent and bare stent.
Baseline variables that were considered clinically relevant, or that had a value of
Procedure-related complications and outcomes.

Data are numbers of patients. ACR, American College of Radiology; SIR, Society of Interventional Radiology.
There were 17 procedure-related adverse events (Table 2). Severe adverse events of pericardial tamponade and acute pleural effusion occurred in one patient, who recovered after drainage. Acute heart failure was observed in two patients who were recovered after hemodialysis and anti-hypertension treatment.
Univariate and Multivariate Analysis of Prognostic Factors for primary patency.

Data are numbers of patients. CVC, central venous catheter.
Discussion
The incidence of central venous stenosis or obstruction in hemodialysis patients ranges from 19% to 41%.8,9 The major causes of subclavian or brachiocephalic venous stenosis are prolonged central venous catheterization, extrinsic compression by the first rib and clavicle, and high-flow rate in an AVF or graft. Injury to the venous endothelium and the resultant inflammatory response within the vessel wall are considered possible mechanisms. 10 The diagnosis and indication for treatment of CVOD are based on clinical manifestation such as ipsilateral arm or neck swelling, elevation of venous pressure during hemodialysis, appearance of collateral branches on the neck and chest, and hemodialysis access failure. The guidelines of the Kidney Disease Outcomes Quality Initiative (KDOQI) for CVOD suggest that placement of a stent may be performed for elastic recoil that leads to remarkable residual stenosis after balloon dilation, or for restenosis that occurs within 3 months. 11 At the same time, study has indicated that the high rate of restenosis may be attributed to multiple lesion or disease factors. 12
In this study, we analyzed the data of 71 hemodialysis patients with CVOD treated at our center. To the best of our knowledge, this is the largest case series regarding CVOD in hemodialysis patients, and the overall treatment success rate was superior to that reported in most studies. The 3-, 6-, 9-, and 12-month primary patency rates were 93%, 72%, 55%, and 51%, respectively, and the median patency rate was 16 months.12,13 We believe there are some reasons for the superior results shown in this case series. At our center, patients were treated with appropriate size covered stents, whereas in previous reports most patients were managed with bare self-expanding stainless-steel stents with diameters of 8–14 mm (ref). Our analysis of patency rate showed there was a significant difference between covered and bare stents (P < 0.05). Stent size was also confirmed as an independent predictor of primary patency with multivariate analysis in this study. Based on our clinical experience and literature review, we selected 26 clinical factors and imaging characteristics for input into the univariate analysis, and screened out 8 factors to include in the multivariate analysis. Finally, we identified that vessel diameter >12 mm, stent type, and vessel calcifications were independent predictors of primary patency.
Most investigators considered the choice of intervention has an important influence on the long-term effect of CVOD treatment. However, the optimal endovascular intervention for CVOD is unknown. The treatment options include PTA, placement of bare stents, and placement of covered stents. Hemodialysis Reliable Outflow (HeRO) graft has become a well-known and highly effective treatment for CVOD over the past 10 years.14 –16 The KDOQI guidelines recommend PTA, with or without stent placement, as the preferred treatment approach to CVOD. 17 HeRO graft was applied as a hybrid alternative access once the occlusion is bypassed in KDOQI for management of CVOD. 18 Several investigators reported that angioplasty with bare stents does not provide a better primary patency rate than PTA, although the technical success rate is higher.13,17 Other studies indicate there are significant limitations of bare stents with respect to long-term outcomes. Furthermore, bare stent placement may preclude future endovascular procedures or surgical revision. It is also clear that bare stents induce intimal hyperplasia leading to restenosis and multiple repeat interventions to maintain patency.11,19 A retrospective study of treatment for CVOD showed the 3-, 6-, and 12-month primary patency rates with PTA were 58%, 25%, and 29%, respectively, and that of bare stents were 65%, 54%, and 45%, respectively, and analysis showed the rates were not significantly different. 12
A mechanical advantage of covered stents is that they provide a relatively inert and stable intravascular matrix for endothelialization.
10
However, covered stents are limited by restenosis at the junction between the covered stent and vessel wall. Compared with bare stents, covered stents have been found to provide superior results in many vascular stenosis conditions. A study of transjugular intrahepatic portosystemic shunt (TIPS) found that the cumulative restenosis rates of covered stents at 1, 2, 3, 4, and 5 years were 6.9%, 11.5%, 19.1%, 26.0%, and 35.9%, respectively, and that of bare stents were 27.6%, 37.0%, 49.6%, 59.8%, 74.8%, respectively, and the patency rates of covered stents were significantly better than those of bare stents (
Several recent studies found that covered stents provide several advantages in the treatment of CVOD in hemodialysis patients.20,21 In hemodialysis patients with autogenous arteriovenous access and CVOD, several recent studies have shown that covered stents are associated with significantly improved patency compared with the use of bare stents.20,22 In patients with central venous stenosis, a study reported the primary patency rates at 3, 6, 12, and 24 months when covered stents were used were 97%, 85%, 67%, and 45%, respectively, and the secondary patency rates at 12 and 24 months were 80% and 75%, respectively. 23 For bare-metal stent, primary patency at 3, 6, 12, and 24 months was 84%, 80%, 75%, and 46%, respectively. 20 Another study reported the primary patency rate of covered stents was 56%, the secondary patency rate was 100% at 12 months, and the technical success rate was 100%. 5
Our results showed that vessel diameter was an independent predictor of primary patency after treatment for CVOD. The larger diameter of the vein and larger sized stent, the better the patency rate. Vessel and stent diameter have been shown to be important factors for post-procedure blood flow volume through the stent and the rate of restenosis. In a randomized clinical trial, larger diameter and short-length drug-eluting stents were shown to have a significant advantage with respect to treatment effect and cost-effectiveness. 24 Most investigations of endovascular treatment of occlusion of lower veins show that large size stents provide better patency. 25
However, few studies have examined the association between vessel/stent diameter and patency in hemodialysis patients. A study of the influence of cephalic vein diameter and diabetes on primary maturation and patency of autogenous radiocephalic arteriovenous fistulas (RCAVF) indicated that a minimal cephalic vein diameter of >2 mm was an independent predictor of RCAVF maturation and was associated with a significantly higher primary patency rate than when the diameter was ⩽2 mm. 26
Severe vessel wall calcification is frequently observed in patients affected by metabolic disorders, especially in chronic renal disease. Patency rates are reported to be lower in the case of very calcified arteries. 27 A study that investigated vascular histology and collagen fiber configuration in patients with advanced chronic kidney disease found that the anisotropy index (anisotropy index is a measure of the randomness of the fiber network, ranging from ‘0’ for totally no preferential directionality to ‘1’ for totally aligned in one direction) of medial collagen fibers was lower in both arteries and veins in patients with chronic kidney disease as compared with patients with no kidney disease. 28 The patients with advanced chronic kidney disease also had greater arterial micro-calcifications. These results indicate that calcifications indicate abnormalities in arterial and venous histology and collagen fiber configuration. 28 The mechanism of angioplasty relies on plaque fracture and vessel wall stretching, and severe calcifications completely alters the morphology and compliance of the vessel wall, which reduces the effect of both angioplasty and stenting. Moreover, calcifications increase the risk of stent sub-expansion, malposition, and fractures.29 –31 Our results also showed that calcifications were associated with a lower primary patency rate after stent placement for treatment of CVOD. The reasons were inferred as the more severe thrombin generation, platelet activation, and inflammatory reaction. And vessel wall hyperplasia was secondary to hemodynamics changes and oxidative stress.2,8,28
Our univariate analysis revealed a number of factors that could possibly impact the primary patency rate after CVOD treatment including sex, heart failure history, smoking tobacco, duration of CVC placement, and failed procedure history, but these were found not to be significant in the multivariate analysis. Whether or not sex and heart failure history affect the primary patency rate requires further study, and we did not find relevant reports in our literature review. Smoking is associated with a lower success rate of target lesion revascularization in patients undergoing percutaneous coronary intervention (PCI), but the effect of outcomes of stent placement is not clear. 32 On the contrary, a report found that the patients who smoke 10 or more cigarettes daily had less risk of intermediate-term restenosis after endovascular interventions in the lower limbs. 33 Smoking was not a predictor of the primary patency in the study. Mechanism of vascular sclerosis and stenosis caused by smoking is sophisticated. Although it is clear that CVC placement duration is an important factor associated with central venous stenosis and occlusion, we did not find an association with primary patency after treatment. This may be because the CVC had already been removed before stent deployment in almost all patients. Failed procedure history did not demonstrated as an independent factor of the outcomes in our study as well, in spite of the fact that it had been proven as crucial barrier to recanalize the coronary artery in PCI procedure. 34
There are strengths and limitations of this study that should be considered. The sample size is larger than most similar reports, and our medical staff has a rich experience in interventional therapy of vascular access in hemodialysis patients.
However, this was a retrospective study performed at a single center. Another limitation was that the power analysis for determining the sample size was not performed, partially due to the retrospective design. Although we identified 26 possible factors related to patency after treatment, it is impossible to include all potential factors in an analysis, some of which might be considered more important at other hospitals.
Conclusion
Our results showed that a vessel diameter >12 mm, the use of a covered stent, and no vessel calcifications were independently associated with a higher primary patency rate after treatment of CVOD in hemodialysis patients.
Footnotes
Appendix 1
Univariate and multivariate analysisof prognostic factors for primary patency.

| Univariate analysis of prognostic factors for primary patency | Multivariate analysis of prognostic factors for primary patency | |||||||
|---|---|---|---|---|---|---|---|---|
| Factors | No. of patients | Mean | Median ± SD | Coefficient | 95% CI | |||
| Age | ⩾60 years | 31 | 18.12 | 10 ± 4.88 | .815/.852 | |||
| <60 years | 40 | 18.75 | 16 ± 1.06 | |||||
| Gender | Male | 38 | 21.80 | 18 ± 3.37 | .023/.046 | 0.426 | 0.815, 2.879 | .185 |
| Female | 33 | 13.84 | 9 ± 1.37 | |||||
| BMI | ⩾24 | 38 | 16.54 | 16 ± 2.37 | .642/.548 | |||
| <24 | 33 | 19.36 | 15 ± 4.89 | |||||
| NYHA classification | I | 45 | 18.78 | 16 ± 1.94 | .768/.575 | |||
| II | 26 | 17.21 | 9 ± 5.95 | |||||
| Heart failure history | Yes | 5 | 8.00 | 9 ± 2.12 | .056/.165 | –0.380 | 0.208, 2.255 | .533 |
| No | 66 | 18.84 | 16 ± 1.95 | |||||
| Vessel disease | Yes | 24 | 18.67 | 15 ± 5.94 | .783/.957 | |||
| No | 47 | 17.13 | 16 ± 1.41 | |||||
| Hypertension | Yes | 42 | 18.91 | 15 ± 3.98 | .726/.856 | |||
| No | 29 | 17.99 | 16 ± 5.59 | |||||
| Smoker | Yes | 28 | 21.61 | 22 ± 5.31 | .108/.182 | –0.013 | 0.407, 2.389 | .976 |
| No | 43 | 15.83 | 15 ± 4.83 | |||||
| Dyslipidemia | Yes | 26 | 20.19 | 21 ± 5.80 | .278/.305 | |||
| No | 45 | 16.23 | 15 ± 3.83 | |||||
| Diabetes | Yes | 12 | 17.82 | 10 ± 1.47 | ||||
| No | 59 | 18.49 | 16 ± 2.13 | |||||
| Number of CVC | 1 | 15 | 15.79 | 15 ± 6.90 | .942/.932 | |||
| 2 | 22 | 16.45 | 15 ± 5.41 | |||||
| ⩾3 | 34 | 19.07 | 16 ± 4.71 | |||||
| Duration of CVC | >7 month | 33 | 21.54 | 18 ± 4.90 | .089/.176 | –0.448 | 0.323, 1.263 | .198 |
| <7 month | 38 | 15.50 | 10 ± 3.46 | |||||
| Previous failed lesion | Yes | 5 | 9.40 | 8 ± 4.38 | .185/.097 | 0.736 | 0.729, 5.977 | .170 |
| No | 66 | 18.67 | 16 ± 1.92 | |||||
| Symptom | >1 month | 44 | 17.36 | 14 ± 2.65 | .469/.464 | |||
| ⩽1 month | 27 | 18.93 | 21 ± 5.27 | |||||
| Access Site | CVC | 11 | 17.44 | 16 ± 7.11 | .731/.925 | |||
| Fore arm | 59 | 18.88 | 16 ± 2.04 | |||||
| Upper arm | 1 | 9.00 | 9.00 | |||||
| Access type | CVC | 13 | 18.33 | 21 ± 9.99 | .241/.235 | |||
| AVG | 54 | 11.00 | 12 ± 6.06 | |||||
| AVF | 4 | 11.56 | 10 ± 8.90 | |||||
| Location of lesion | Left | 34 | 15.05 | 15 ± 2.92 | .280/.682 | |||
| Median | 14 | 19.70 | 22 ± 7.87 | |||||
| Right | 23 | 21.30 | 15 ± 6.88 | |||||
| Site of lesion | Subclavian | 20 | 18.65 | 18 ± 2.01 | .552/.284 | |||
| Brachiocephalic | 37 | 16.67 | 10 ± 3.86 | |||||
| SVC | 14 | 19.70 | 22 ± 7.87 | |||||
| Side branches | 0 | 36 | 17.66 | 13 ± 4.85 | .793/1.00 | |||
| 1 | 19 | 14.76 | 16 ± 0.82 | |||||
| ⩾2 | 16 | 19.62 | 16 ± 5.71 | |||||
| Blunt | Yes | 23 | 17.33 | 16 ± 4.96 | .761/.414 | |||
| No | 48 | 18.15 | 15 ± 2.58 | |||||
| Calcification | Yes | 18 | 15.48 | 9 ± 4.83 | .289/.161 | 0.663 | 0.999, 3.765 | .050 |
| No | 53 | 19.15 | 18 ± 2.42 | |||||
| Bending | Yes | 24 | 17.21 | 16 ± 4.63 | .617/.870 | |||
| No | 47 | 18.92 | 15 ± 3.01 | |||||
| Lesion length >20 mm | Yes | 34 | 17.08 | 14 ± 3.83 | .354/.228 | |||
| No | 37 | 19.16 | 18 ± 5.49 | |||||
| Multiple lesions | Yes | 13 | 20.85 | 8 ± 8.99 | .591/.948 | |||
| No | 58 | 17.43 | 16 ± 1.86 | |||||
| Vessel diameter | ⩽12 mm | 25 | 12.19 | 8 ± 1.59 | .002/.007 | 1.115 | 1.628, 5.716 | .001 |
| >12 mm | 46 | 21.67 | 18 ± 3.03 | |||||
| Stent type | Covered | 22 | 22.44 | 21 ± 3.70 | .108/.056 | 0.805 | 1.081, 4.625 | .030 |
| Bare | 49 | 16.54 | 10 ± 3.39 | |||||
| Stent diameter | ⩽12 mm | 26 | 18.02 | 18 ± 2.56 | .789/.430 | |||
| >12 mm | 45 | 17.98 | 15 ± 3.04 | |||||
| Stent angle | ⩽120° | 29 | 16.88 | 15 ± 3.54 | .530/.867 | |||
| >120° | 42 | 18.14 | 16 ± 4.45 | |||||
Data are numbers of patients.
AVF, arteriovenous fistula; AVG, arteriovenous graft; BMI, body mass index; CI, confidence interval; CVC, central venous catheter; SVC, superior vena cava; NYHA, New York Heart Association.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work received funding from National Natural Science Foundation of China (81671792).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
