Abstract
Lurbinectedin is responsible for DNA recognition and binding, producing double-strand DNA (dsDNA) breaks thus resulting in apoptosis. Sensitivity to lurbinectedin is linked to the nucleotide excision repair (NER) system. Furthermore, irinotecan, a topoisomerase I inhibitor, provokes dsDNA breaks that could be reinforced abrogating the NER system using lurbinectedin. BRCA-mutated patients, already treated with platinum-derived drugs, who suffered DNA damage, cannot repair the breaks due to lurbinectedin interaction, whereas irinotecan provokes a dsDNA break that promotes synthetic lethality. This article describes an exceptional response to lurbinectedin alone followed by the association with irinotecan in a BRCA-mutated platinum-resistant ovarian cancer patient. A 44-year-old BRCA1-mutated ovarian cancer patient was treated in sixth line with lurbinectedin and irinotecan with a time to further progression (TTFP) equal to 8 months. In our case, the association with irinotecan overcame the resistance to lurbinectedin alone. In conclusion, lurbinectedin and irinotecan demonstrated a promising response in platinum-resistant patients. However, further studies should be conducted to validate our findings and future trials will be important to further define the clinical utility of lurbinectedin.
Introduction
Lurbinectedin shares its structure with trabectedin, containing two tetrahydroisoquinoline domains, named subunits A and B, that recognize and bind DNA. Unlike trabectedin, a third module (named ring C) is represented by tetrahydro β-carboline, rather than tetrahydroisoquinoline, that improves drug pharmacokinetics and activity. 1 By binding to CG-rich sequences, the DNA transcription is blocked, causing double-strand DNA (dsDNA) breaks and therefore apoptosis. 2 Furthermore, the lurbinectedin inhibits inflammatory factors, reducing tumor-associated macrophages and the inflammatory microenvironment. 3
Lurbinectedin sensitivity depends on the nucleotide excision repair (NER) efficiency along with the homologous recombination deficiency (HRD), suggesting more drug activity in tumor cells with mutation in BRCA genes. Indeed, lurbinectedin activity has been shown in BRCA2-mutated cells, where R-loops occur as consequence of HRD. 4
Recently, lurbinectedin was compared to topotecan in a phase-II study of platinum-resistant ovarian cancer patients, showing an impressive activity in terms of overall response rate (ORR), progression-free survival (PFS), and overall survival (OS). 5
A phase-I study of lurbinectedin and olaparib, a PARP-inhibitor used in patients with BRCA1/2 mutations, has been performed in advanced solid tumors regardless BRCA mutation showing a good tolerability and a disease control rate of 60%. 6
Irinotecan is a DNA-damage agent provoking a double strand-break that can be significantly reinforced by lurbinectedin which abrogates NER system, necessary to repair bulky adducts to DNA caused by platinum-derived drugs.
BRCA-mutated patients with DNA damage previously treated with platinum-derived drugs are unable to repair the break due to lurbinectedin interaction, whereas irinotecan provokes a double strand-break that activates the non-homologues end-joining and finally the synthetic lethality.
A strong synergistic effect between lurbinectedin and SN-38, an active metabolite of irinotecan, has been shown in a preclinical study, in the RMG1 and RMG2 cells, two different kinds of human ovarian clear cell carcinoma, likely due to the induction of multiple DNA-damage mechanisms. 7
This article describes an exceptional early response to lurbinectedin alone and subsequently to the association with irinotecan in BRCA-mutated platinum-resistant ovarian cancer patient.
Case report
A 44-year-old woman experienced progressive abdominal increase associated with pelvic pain. Gynecological ultrasound showed the presence of diffuse ascites in lower abdominal quadrants with a large mass involving bilateral ovarian region, whereas the computed tomography (CT) scan revealed a right pleuric effusion with multiple pleural and peritoneal nodes. Diagnostic and exploratory laparoscopy and thoracoscopy, with multiple diffuse biopsies on peritoneal and pleural nodes were performed, resulting compatible with high-grade serous carcinoma, FIGO stage IV. Before starting neoadjuvant treatment, genetic counseling was carried out since significant family history for breast and gynecological cancers was referred by the patient. The pedigree showed multiple tumors on both genealogies as represented in Figure 1. Genetic testing for BRCA1 and BRCA2 genes revealed a likely pathogenic BRCA1 mutation on exon 23 named c.5509 T > C, p. (Trp1837Arg). The same mutation was also found on DNA of the patient’s father.
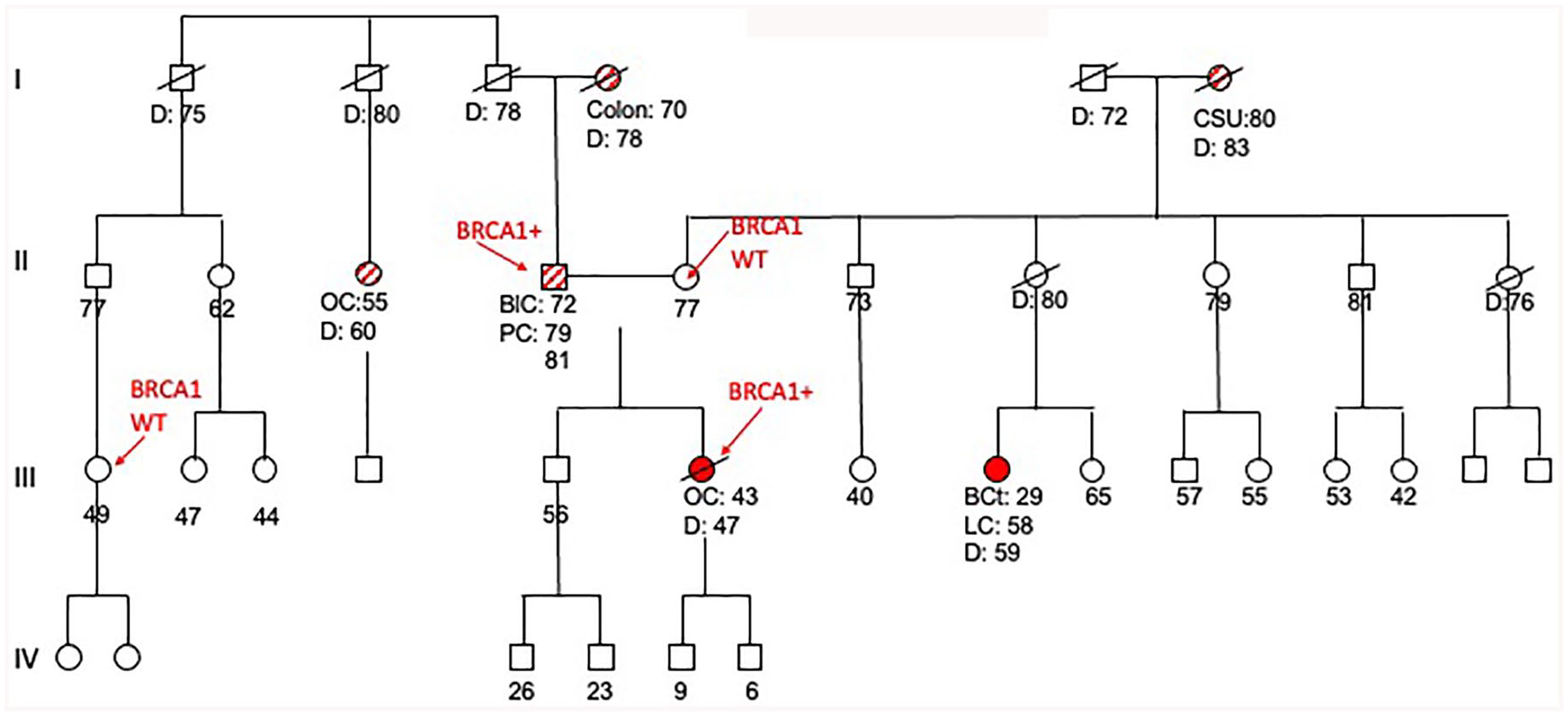
Pedigree of patient.
On June 2016, considering patient’s young age, neoadjuvant treatment was initiated with carboplatin in combination with paclitaxel and bevacizumab, with the aim to achieve optimal debulking surgery with initial unresectable tumor, as reported in the Anthalya phase-II study. 8 After four cycles, the re-staging revealed response of the disease with reduction of pleural and peritoneal nodules. However, the multidisciplinary team recommended two further courses of chemotherapy since complete cytoreduction was deemed not possible. Bevacizumab was administered only for one more cycle and a new radiological evaluation after six courses of carboplatin plus paclitaxel was planned. The CT scan, performed on November 2016, showed good partial response (PR) with disappearance of pleuric and peritoneal effusions, reduction of ovarian lesions, and persistent peritoneal thickening in supra-hepatic loggias and right peritoneal sub-hepatic region.
Interval debulking surgery (IDS) was performed at the end of November 2016 in a Regional Reference Center for Ovarian Surgery. The time between the last bevacizumab administration and the IDS was equal to 7 weeks. The patient underwent bilateral annessiectomy, hysterectomy, pelvic, pre-vesical, peri-cecal peritonectomy, appendectomy, sigma-rectal resection, omentectomy, and thoracoscopy with diaphragm-plasty. Unfortunately, the histological examination revealed the persistence of disease on one lymph node out of four examined and on hepatic round perilegmentum and meso-ileal peritoneum, whereas sigma-rectum wall, right pleura and distal intestinal resection were free of the disease. The Peritoneal Cancer Index (PCI) was 21 and the Completeness of Cytoreduction (CC) was equal to 2.
The CT scan performed after surgery, before starting bevacizumb maintenance therapy, showed progression of the disease (PD) at peritoneal level, and on February 2017, the patient started a second-line chemotherapy with trabectedin, at 1.3 mg/m2 administered every 3 weeks for a total of six cycles, according to the MITO 15 trial results. 9 Trabectedin was given at 75% of entire dosage after the third cycle for grade-II liver toxicity. The patient developed transient neutropenia (grade 3), anemia (grade 2), and fatigue (grade 2) that spontaneously recovered without dose adjustment or specific therapies. The treatment induced a rapid symptom improvement with partial remission (PR) at radiological evaluation. After 9 months, CT scan showed a further PD, also evidenced by positron emission tomography/CT (PET/CT) imaging performed with the aim to confirm pleural and pelvic disease involvement. Following an extended response period of treatment with trabectedin, a re-challenge with platinum-based therapy (carboplatin-gemcitabine) was attempted, with the aim to test the sensitivity restoration, as claimed by the theory that NER-proficient cells are platinum resistant and following trabectedin treatment, NER-proficient cells are reduced, rendering the tumor platinum sensitive again. 10 Due to anemia G3, thrombocytopenia G2 and Carboplatin toxicity, occurred after the first cycle of treatment, cisplatin alone was administered for five more courses, without gemcitabine. The PET/CT showed a PR, therefore, Olaparib was offered as maintenance therapy after platinum-derived response at the standard dosed of capsules 400 mg × 2/die. Since anemia G3 occurred, a dosage reduction to 200 mg × 2/die was performed, subsequently, due to further hematological toxicities (G3 neutropenia, G1 anemia and G1 piastrinopenia) 100 mg × 2/die was administered for a total of 6 months of therapy. Moreover, liver PD was evidenced, and the patient was offered Caelyx at 50 mg/mq q21 for nine courses. Time to further progression (TTFP) measured 9 months. As subsequent well-tolerated treatment, gemcitabine at 1000 mg/mq q14 was offered, excluding a topoisomerase inhibitor such as topotecan, which can cause substantial and highly clinically relevant hematological toxicity, in a patient so heavily affected in the bone marrow; meanwhile, lurbinectedin was requested. After 3 months of gemcitabine treatment, peritoneal PD with carcinosis was shown (Figure 2(a)).
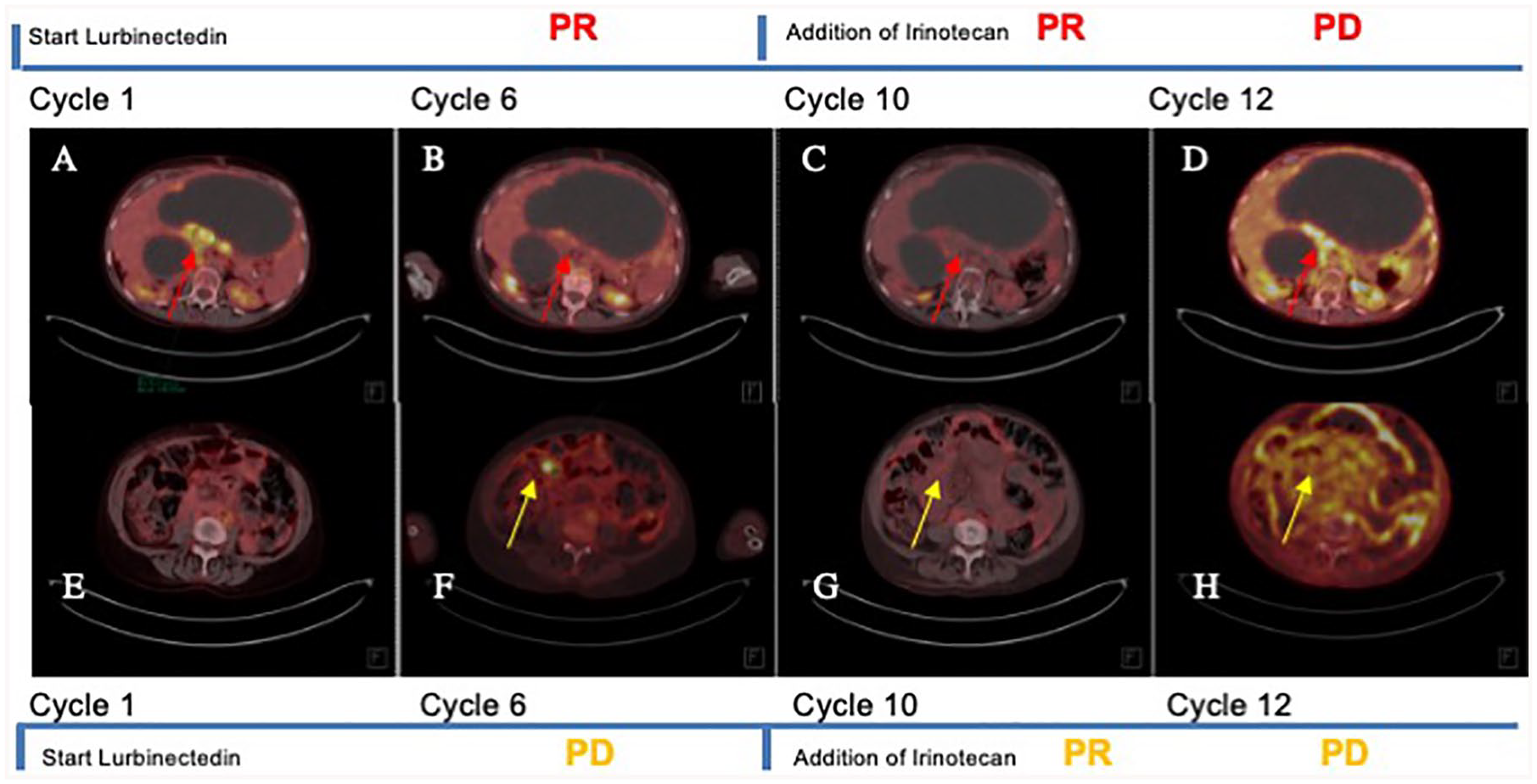
PET/CT showed widespread metastases throughout the entire body of patient: (a–d) describe the peritoneum in upper abdominal quadrant while (e–h) describe the peritoneum in central abdomen. (a and e) represent baseline situation in May 2020, before patient began treatment with lurbinectedin. PET/CT showed diffused peritoneal disease (red arrow in (a)) while no evidence of any uptake was observed in (e). (b and f) represent first revaluation in September 2020. Imaging showed PR at peritoneal level (red arrow in (b)) and PD in right flank (yellow arrow (f)). After PET/CT imaging, the patient started irinotecan and continued with lurbinectedin. (c and g) represent first revaluation, in December 2020, during lurbinectedin and irinotecan treatment. PET/CT imaging showed PR, in particular in right flank (yellow arrow in (g)) and continued to respond at peritoneal level (red arrow in (c)). (d and h) represent last revaluation in February 2021 with a PD in right flank (yellow arrow in (h)) and peritoneal level (red arrow in (d)). Following documented progression, the patient was offered best supportive care and died 2 months later.
The patient started lurbinectedin therapy at 3.2 mg/mq q21 on May 2020. Due to grade-3 gastrointestinal toxicity (nausea and vomiting), the dosage was reduced to 75% without any grade toxicity >2. Performance status (PS) conditions at the beginning of lurbinectedin treatment were poor (Eastern Cooperative Oncology Group (ECOG) 2). The PET/CT scan, performed after six courses, showed PR at peritoneal level, but PD in right flank (Figure 2(b) and (f)). Patient improved PS until 1, despite the pain in right flank. The TTFP of lurbinectedin alone was equal to 4 months.
In order to maintain PR at peritoneal level and improve response in right flank, on October 2020, irinotecan at 75 mg/day 1,8 q21 was added to lurbinectedin at 2 mg/mq day 1 q21, according to recent data presented at the American Society of Clinical Oncology (ASCO) 2020 meeting. 11 Due to previous hematological toxicity, both drugs were administered at 75%, supported by granulocyte colony-stimulating factor (G-CSF) for four courses. By reducing irinotecan dosage to 75%, we were able to maintain administration at day 8. Consequently, the pain disappeared, patient’s quality of life significantly improved in absence of any toxicity. The first evaluation, performed at the end of December 2020, showed PR at peritoneal level and right flank (Figure 2(c) and (g)). Unfortunately, after two more cycles of treatment, patient’s symptoms and PS deteriorated and subsequent PET/CT confirmed tumor progression (Figure 2(d) and (h)). Therefore, on February 2021, lurbinectedin and irinotecan were discontinued, and hospice care was suggested. The patient died 2 months after treatment discontinuation. The TTFP of association between lurbinectedin and irinotecan was equal to 4 months. Overall, the number of lurbinectedin administrations was equal to 12, throughout a period of 8 months, being irinotecan able to provide a synergistic effect after previous PD.
Discussion
To our knowledge, this is the first case report on ovarian cancer treatment with lurbinectedin plus irinotecan in BRCA-mutated patient with poor PS. Recently, lurbinectedin showed activity in small-cell lung cancer (SCLC) as second-line therapy. 12 In a phase-II study, Cruz et al. 13 reported that lurbinectedin showed a notable efficacy in BRCA1/2-mutated advanced breast cancer. Overall, 22 of 54 BRCA1/2-mutated patients responded (ORR, 41%); two complete responses (CRs) were registered, with 6.1 months of median response duration (range, 3–11 months). Median PFS and OS were 4.6 and 20.0 months, respectively. Of interest, BRCA2-mutated patients showed the most significant responses (ORR, 61%; PFS, 5.9 months; OS, 26.6 months), particularly in patients not previously treated by PARPi and platinum therapy, where ORR increased to 72% and 71%, respectively. Data from a phase-III trial with lurbinectedin versus topotecan or pegylated liposomal doxorubicin (PLD) in relapsed platinum-resistant ovarian cancer patients (CORAIL trial) were recently presented at the European Society of Medical Oncology (ESMO) 2020 meeting. No differences in median PFS were shown (3.5 months with lurbinectedin vs 3.6 months with standard treatment), whereas the primary endpoint (30% of reduction in PFS) was not met. 14
Our results with lurbinectedin alone compared those provided by the CORAIL study, with a PFS equal to 4 months in sixth line. Furthermore, they were in line with those presented by Aix 11 at the ASCO 2020, where the combination of lurbinectedin and irinotecan in the endometrial carcinoma had a median PFS of 7.1 months.
Lurbinectedin lead to an oligometastatic progress, which could be then stabilized for another 4 months by adding a topoisomerase inhibitor in a most heavily pre-treated patient.
Currently, another platinum-resistant BRCA-mutated ovarian cancer patient is under treatment as seventh line, after a PARPi re-challenge PD, showing a Ca.125 decrease at the fourth administration.
This case report provides a possible new approach for BRCA-mutated ovarian cancer patients, although it should be interpreted with caution as a single experience, in the context with other existing data.
Conclusion
The purpose of this study was to investigate and evaluate treatment with lurbinectedin and irinotecan as a promising therapy in ovarian cancer patients no longer sensitive to platinum-derived drugs.
Further evaluation of this combination, perhaps in earlier lines of treatment, could be of interest. Our findings need to be validated by further studies useful to define the clinical implications of lurbinectedin in ovarian cancer.
Footnotes
Acknowledgements
The authors thank and express our gratitude to Tamara Sassi and Johanna Chester for their editorial assistance.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Associazione Angela Serra per la Ricerca sul Cancro, Modena (Angela Serra Association for Cancer Research).
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LC: honoraria: AstraZeneca, MSD, Pfizer; Consulting or Advisory Role: Pfizer, Novartis, GSK, Clovis. No other potential conflicts of interest were reported.
Ethics approval statements
The local Ethical Committee ‘Area Vasta Emilia Nord (AVEN)’ does not require official approval for the publication of single case reports. The Ethical Committee approved the Lurbinectedin compassionate use with protocol N° 0007474/20. Written consent to publish information and images included in this report was obtained from the patient’s legal guardian.
