Abstract
Objective:
To compare the clinical outcomes of patients with active immunoglobulin G (IgG) 4 related disease (IgG4-RD) receiving tocilizumab versus those receiving cyclophosphamide (CYC).
Methods:
This IgG4-RD registry study was a prospective cohort study conducted among patients with active IgG4-RD hospitalized at Zhongshan Hospital, Fudan University. Patients who were treated with tocilizumab or CYC along with glucocorticoids (GCs) were enrolled. All participants were followed up at the hospital clinic at 3 and 6 months after discharge. Primary clinical outcomes were measured via the IgG4-RD responder index (RI), complete response (CR), and partial response (PR), as well as side effects.
Results:
From January 2015 to June 2020, 29 patients enrolled. Fourteen and 15 patients were treated with tocilizumab and CYC, respectively. At the 6-month follow-up, disease activity parameters including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), IgG4, and IgG4-RD RI, decreased significantly in both groups. At 6 months, tocilizumab demonstrated its superiority, with 50% of patients achieving CR in the Tocilizumab group versus 20% in the CYC group. However, no statistical significance was identified (p = 0.128). The GC dosage at 6 months was significantly lower in the tocilizumab group than in the CYC group [10 (9.4–15) mg/d versus 15 (15–15) mg/d, p = 0.025]. In the CYC group, two patients experienced lumbar vertebral compression fractures related to GCs. Other patients in both groups showed mild adverse effects.
Conclusions:
Tocilizumab could be a better steroid-sparing agent, with a comparable curative effect and tolerance, than CYC, in the treatment of IgG4-RD.
Introduction
Immunoglobulin G (IgG) 4 related disease (IgG4-RD) is a systemic and inflammatory autoimmune disease. 1 It is a new type of disease that was named 10 years ago. With advancements in understanding of the disease, the rate of IgG4-RD diagnosis has been steadily increasing. The disease commonly manifests with multiple organ involvement, accompanied by organ fibrosis and even dysfunction. Without prompt and effective treatment, the disease can greatly affect patients’ quality of life and life span.
At present, patients with IgG4-RD are usually treated with glucocorticoid (GC) based immunosuppressive therapy. In a study conducted in 1220 patients with IgG4-RD, 89% achieved complete remission or partial remission with GC treatment, and 11% did not respond to GCs. 2 In studies conducted among patients with IgG4-RD in whom GC treatment was reduced, 40–64% of patients showed disease recurrence, and only 27% could completely stop receiving GCs.2–5
However, GC therapy is associated with many adverse reactions. Approximately 67% of patients treated with GCs experienced adverse reactions, which mainly included diabetes, infection, and femoral head necrosis. 6 Therefore, in recent randomized controlled trials (RCTs) conducted in patients with IgG4-RD, attention has been paid to the effectiveness of steroid-sparing agents known as immunosuppressors.7–9 It has been suggested that leflunomide or mycophenolate mofetil (MMF), used in combination with GCs, was superior to GC therapy alone.7,8 A prospective study also indicated that the use of GCs plus cyclophosphamide (CYC) reduced the relapse rate better than did GCs plus MMF. 10 Regarding biological agents, the other type of steroid-sparing agents, in an open-label study in which 30 patients were enrolled, 47% achieved CR with rituximab monotherapy. 11 However, more RCTs assessing the efficacy of rituximab are needed.
Given that the conventional therapeutic approach for IgG4-RD management is associated with a high recurrence rate, side effects, and difficulty in reducing GC dose, it is important to further explore new therapies based on the pathogenesis of the disease. Our recent study showed that interleukin 6 (IL-6) plays an important role in the pathogenesis of IgG4-RD, 8 that is, IL-6/IL-6R can promote fibroblasts releasing follicular helper T (Tfh) and B cell differentiation factors. Tocilizumab, a humanized IL-6 receptor monoclonal antibody, has been widely used in the management of various autoimmune diseases.12,13 Furthermore, it has been shown to be effective in treating giant cell arteritis 14 and rheumatoid arthritis. 15 However, to date, the outcomes of tocilizumab treatment for IgG4-RD have not been reported. Therefore, we designed this prospective cohort study to investigate the clinical outcomes of tocilizumab treatment versus CYC in combination with GCs among Chinese patients with active IgG4-RD.
Materials and methods
Study design
This was a prospective cohort study based on an ongoing prospective observational cohort of IgG4-RD patients at our center (Department of Rheumatology, Zhongshan Hospital, Fudan University, Shanghai, China). All patients were diagnosed with IgG4-RD according to the 2011 Japanese comprehensive clinical diagnostic criteria: 16 (1) clinical examination shows characteristic diffuse/localized swelling or masses in single or multiple organs; (2) hematological examination shows elevated serum IgG4 concentrations (⩾135 mg/dl); (3) histopathologic examination shows marked lymphocyte and plasmacyte infiltration, and fibrosis and infiltration of IgG4-positive plasma cells (ratio of IgG4/IgG positive cells >40% and >10 IgG4-positive plasma cells/HPF). Patients who met (1), (2), and (3) were categorized as having definite IgG4-RD. Those who met (1) and (2) or (1) and (3) were classified as having possible or probable IgG4-RD, respectively. The inclusion criteria were as follows: (a) patients with active disease [IgG4-RD responder index (RI) ⩾ 3]; (b) patients who received induction therapy of GCs combined with tocilizumab or CYC; (c) patients without concurrent carcinoma, an infection, a hematological disease, or another rheumatic disease; and (d) patients with a history of tocilizumab or CYC treatment were excluded.
In a cohort of 151 IgG4-RD patients, 122 patients who did not satisfy the above criteria were excluded (Figure 1). Finally, 14 patients in the tocilizumab group and 15 patients in the CYC group were enrolled between January 2015 and June 2020. Among all the 29 patients, 28 met the 2011 Japanese criteria for definite IgG4-RD, while the remaining patient met the criteria for possible IgG4-RD. The patient with possible IgG4-RD had multiple lesions including bile duct, peri-aortic, and renal pelvic lesions. This patient was finally diagnosed with IgG4-RD clinically by two rheumatologists. In the tocilizumab group, three patients with disease refractory to GC plus at least one type of immunosuppressor (with a lack of an apparent response and decline of <2 in IgG4-RD RI 17 ) and three patients who showed relapse (recurrence of clinical symptoms or on imaging after remission 7 ) were enrolled. Of these three patients, two patients showed disease refractory to GC plus leflunomide, and one patient had disease refractory to GC plus CYC. The CYC group had no patient with treatment-refractory disease or patient showing relapse.

Follow-up chart.
This study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University [B2013–115(3)] and conformed to the tenets of the 1975 Declaration of Helsinki. Written informed consent was obtained from all patients.
Treatment and follow-up
The initial GC dose was 0.6–0.8 mg/kg/d prednisone for 4 weeks in 25 patients, and treatment was gradually tapered by 5 mg daily every 2 weeks in the first 3 months and 5 mg per month during the remaining follow-up period. The maintenance dose was 0.1–0.2 mg/kg/d. The remaining four patients who received lower doses of GC were all in the tocilizumab group. Two patients with refractory disease were treated with 0.5 mg/kg/d prednisone combined with leflunomide 20 mg/d as before. In these two patients, prednisone was tapered by 5 mg daily per month until a maintenance dose of 0.1–0.2 mg/kg/d and leflunomide was continued at a dose of 20 mg/d. Of the two patients showing relapse, one continued prednisone 5 mg/d as before, the other one was given 0.5 mg/kg/d of prednisone. Tocilizumab was administered at a dose of 8 mg/kg div once every 4 weeks for 6 months. CYC (0.5–0.6 g/m2, intravenously) was administered every 4 weeks for 6 months. All patients were followed up for at least 6 months. Data on patients’ clinical features, laboratory examinations, imaging findings, treatment response, and side effects were collected at each visit.
Treatment response and definitions
Disease activity was measured using the IgG4-RD RI at 3 and 6 months, based on the patients’ clinical symptoms and signs, laboratory data, and imaging findings. An expert team of two rheumatologists was responsible for the follow-up and treatment response assessment.
Clinical response was assessed at the end of 6 months and was classified into three types: complete response (CR), partial response (PR), and no change (NC). The responses were defined as follows: 1) CR was defined as follows: (a) IgG4-RD RI ⩽ 2 after a decline of ⩾2 in IgG4-RD RI;18,19 (b) GC tapered to the maintenance dose (prednisone ⩽ 0.2 mg/kg/d); and (c) no relapse during GC tapering (within 6 months).
20
2) PR was defined as follows: (a) IgG4-RD RI ⩾ 3 after a decline of ⩾2. If a patient’s IgG4-RD RI score was 3 points at the beginning, the patient was considered as PR if the score decreased to 2 points after therapy.
17
(b) no relapse during GC tapering (within 6 months).
20
3) NC was defined as a lack of apparent changes in mass size and/or clinical manifestations and a decline of <2 in the IgG4-RD RI.
17
Statistical analysis
Continuous normally distributed variables were summarized as means ± standard deviation. Non-normally distributed data were represented by median and quartiles, and categorical variables were represented as frequencies and percentages. The Mann-Whitney U test, independent two-sample Student’s t-test, Kruskal–Wallis test, or Chi-square test was used, as appropriate, for comparisons between groups. A two-tailed p-value of ⩽0.05 was considered statistically significant. Because data were collected from an observational registry designed to provide real-world clinical settings instead of an RCT, multiple logistic regression models were used to determine the relative risk (RR) and 95% confidence interval (CI) of CR with treatment status. Covariates included male sex, number of organs involved, and an elevated erythrocyte sedimentation rate (ESR); missing data were not imputed. Statistical analysis was performed using SPSS for Windows, Version 22.0 (IBM Corp., Armonk, NY, USA).
Results
Patient demographics and baseline characteristics
In all, 29 patients were enrolled in the present study. The ratio of male to female patients was 1.64:1, and the disease onset age was 60 (42–66) years. The disease duration was 7 (2–24) months. Among these patients, 26 (89.7%) showed the involvement of more than two organs. The common clinical manifestations (Supplemental Table S1) were eye manifestation (8/29, 27.6%); ear, nose, and throat (ENT) manifestation (8/29, 27.6%); digestive manifestation (7/29, 24.1%); submandibular gland mass (5/29, 17.2%); and low back pain (4/29, 13.8%). The involved organs (Supplemental Table S1) included the mediastinum/retroperitoneum (58.6%), lymph nodes (41.4%), lung/pleura (41.4%), ear/nose/throat (31%), salivary glands (27.6%), lacrimal gland/orbit (24.1%), and pancreas (24.1%).
A comparison of the patient demographics and baseline characteristics between the tocilizumab and CYC groups (n = 14 and 15, respectively) is shown in Table 1. There was no significant difference in age, sex, and the number of organs involved between the two groups. A longer disease course in the tocilizumab group might have contributed to the disease being refractory and relapse in six patients in this group. A higher serum IL-6 level was noted in the tocilizumab group because most patients who were treated with tocilizumab had a high serum IL-6 level at baseline. The commonly used parameters for disease activity in IgG4-RD, such as ESR, C-reactive protein (CRP), and IgG4-RD RI were comparable.
Patient demographics and baseline characteristics.

Data are presented as median (interquartile range).
Statistically significant p-values are in bold font.
C3, compliment 3; C4, compliment 4; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IgE, immunoglobulin E; IgG, immunoglobulin G; IgG4-RD RI, IgG4-related disease response index; IL-6, interleukin-6; IL-2R, interleukin-2 receptor; TNF-α, tumor necrosis factor-α.
Treatment response
Response rate
In both groups, significant improvements in clinical manifestations, imaging results, and IgG4-RD RI were seen in the 29 patients. All patients achieved CR or PR, and the overall rate of CR and PR at 6 months was 34.4% and 65.5%, respectively. In the tocilizumab group, the prevalence of both CR and PR was 50%. In the CYC group, the rate of CR and PR was 20% and 80%, respectively. No significant difference was observed in the prevalence of CR or PR between the tocilizumab and CYC groups [p = 0.128, Figure 2(a)]. Among the six refractory and relapsed patients in the tocilizumab group, the prevalence of CR and PR was 50% and 50%, respectively, which indicated that tocilizumab could also be used in patients with refractory and relapsed disease.

(a) CR and PR rates in different treatment groups. (b) CR rates in patients with GC doses of ⩽10 mg/d in the two groups.
Disease activity assessment
At the end of the 6-month treatment period, clinical symptoms improved in 92.9% (13/14) of the patients in the tocilizumab group, and 93.3% (14/15) of those in the CYC group. Significant improvements in disease activity in comparison with the baseline, including lower ESR, reduced levels of CRP and IgG4, and decreased IgG4-RD RI, were seen in both tocilizumab and CYC groups (Table 2, Figure 3). The changes in these disease activity markers from baseline to 3 and 6 months are shown in Supplemental Table S2.
Comparison of the clinical characteristics and laboratory examinations before and after therapy in the tocilizumab and CYC groups.

Data are presented as median (interquartile range).
Statistically significant p-values are in bold font.
C3, compliment 3; C4, compliment 4; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IgE; immunoglobulin E; IgG, immunoglobulin G; IgG4-RD RI, IgG4-related disease response index; IL-2R, interleukin-2 receptor; IL-6, interleukin-6; TNF-α, tumor necrosis factor alpha; E, eosinophil.

The disease activity parameters and GC doses in different treatment groups during the 6-month follow-up.
Laboratory examinations
The patients in both tocilizumab and CYC groups showed lower levels of IgG, globulin, and IL-2R after the 6-month treatment in comparison with the baseline values (Table 2). The C3 level also decreased in the tocilizumab group but not in the CYC group at 6 months (p = 0.043). A decline in the white blood count (WBC) (p = 0.033) and platelet (PLT) counts (p = 0.003) was seen in the CYC group at 6 months (Supplemental Table S3). Though a slight increase was observed in the alanine aminotransferase (ALT) level in the tocilizumab group (p = 0.007) after treatment, the values were still in the normal range. Total cholesterol was elevated at 6 months in the CYC group (p = 0.016), but not in the tocilizumab group, which might be associated with the combined use of GC. There was no significant difference in the levels of kidney function parameters, triglyceride (TG), low density lipoprotein (LDL), and tumor necrosis factor alpha (TNF-α) between 0 and 6 months.
Baseline demographics and clinical characteristics can predict the risk of 6-month CR
Multiple logistic regression models showed that male sex (p = 0.057), number of involved organs ⩾3 (p = 0.006), and elevated ESR (p = 0.054) were associated with CR. The RR (95% CI) for these three factors is shown in Table 3. This result indicated that the involvement of ⩾3 organs was negatively correlated with CR. All of the above covariates did not differ between the tocilizumab and CYC groups at baseline.
Adjusted RR (95% CI) of CR stratified by baseline demographics and clinical characteristics predict risks.
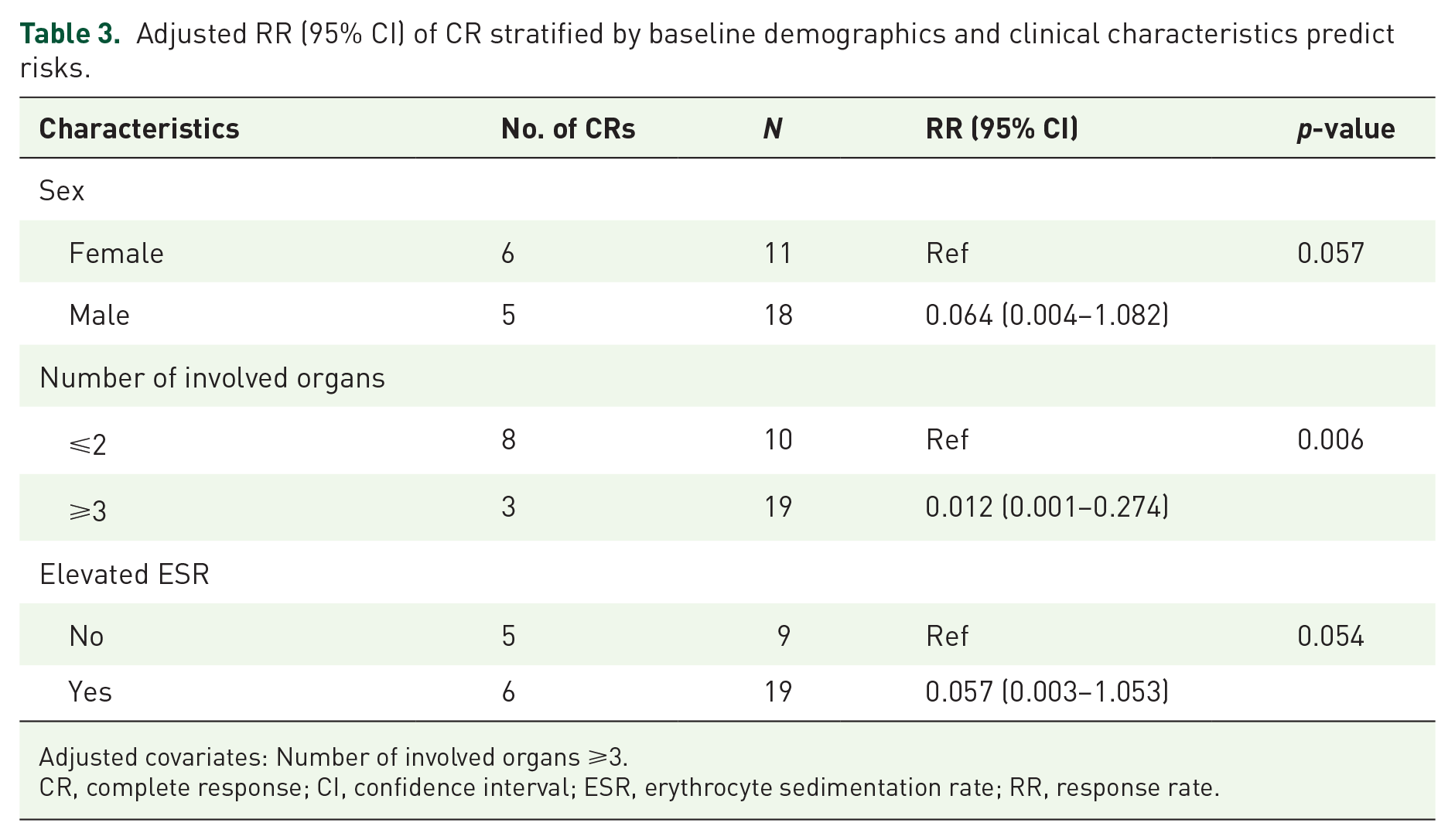
Adjusted covariates: Number of involved organs ⩾3.
CR, complete response; CI, confidence interval; ESR, erythrocyte sedimentation rate; RR, response rate.
GC reduction
The GC doses at baseline and 6 months are shown in Figure 3. The GC dosage at 6 months was significantly lower in the tocilizumab group than in the CYC group [10 (9.4–15) mg/d versus 15 (15–15) mg/d, p = 0.025]. A GC dose ⩽15 mg/d was seen in 13 patients (92.9%) in the tocilizumab group and 12 patients (80%) in the CYC group (p = 0.316). A GC dose ⩽10 mg/d was seen in more patients in the tocilizumab group than in the CYC group [8 (57.1%) versus 3 (20%), p = 0.039].
Among the patients whose GC dose declined to ⩽15 mg at 6 months, the CR rates in the tocilizumab group and CYC group were 53.8% and 33.3%, respectively (p = 0.302). Among the patients whose GC dose declined to ⩽10 mg, the CR rates in the tocilizumab and CYC groups were 50% and 33.3%, respectively (p = 0.621, Figure 2b).
Safety
Of the 15 patients in the CYC group, two experienced lumbar vertebral compression fractures (GC was subsequently discontinued for one of them at 2 months), two showed urinary tract infection, one showed hematuria, and one showed colonic adenoma during a 6-month follow-up period. Of the 14 patients in the tocilizumab group, two had mild infection, including pharyngitis and bacterial vaginitis, one showed mild abnormal liver function [aspartate aminotransferase (AST) 86 U/l], and one showed slight thrombopenia with the number of platelets at 92 × 109/l. Two patients in the tocilizumab group had concomitant diabetes and controlled blood glucose effectively, while 40% (6/15) and 28.6% (4/14) of the patients in the tocilizumab and CYC groups, respectively, gained ⩾5 kg of weight. However, the adverse effects, including lumbar vertebral compression fracture and weight gain, were mainly due to the use of glucocorticoids. Other patients in both groups showing mild adverse effects (mild infections, slight thrombopenia, and abnormal liver function) experienced relief soon after receiving appropriate treatment. The colonic adenoma in one patient in the CYC group was not necessarily related to the use of CYC or GC.
Discussion
This prospective cohort study investigated the clinical outcome of tocilizumab versus CYC for IgG4-RD in combination with glucocorticoids. We found that (1) all patients in both groups achieved various degrees of remission, (2) the GC dosage at 6 months was significantly lower in the tocilizumab group than in the CYC group, indicating that tocilizumab could be a better steroid-sparing agent than CYC, (3) good curative effect was also seen in the refractory and relapsed patients in the tocilizumab group, and (4) tolerability was acceptable in both tocilizumab and CYC groups.
In a recent study, we showed that IL-6 could promote the pathogenesis of IgG4-RD by inducing fibroblasts releasing Tfh and B cell differentiation factors. 8 However, the therapeutic effect of the IL-6R antibody tocilizumab on IgG4-RD has not been reported to date, and the effectiveness of tocilizumab has only been reported in several retroperitoneal fibrosis and chronic periaortitis cases.21,22 Retroperitoneal fibrosis and chronic periaortitis are thought to have overlap with IgG4-RD. 23 Here, we explore the efficacy and safety of tocilizumab in IgG4-RD for the first time.
In both groups, the disease activity parameters, including the levels of ESR, CRP, and IgG4 and the IgG4-RD RI all decreased significantly at 6 months. The levels of IL-2R, IgG, and globulin, which have been also reported to be disease activity markers,24,25 declined remarkably in both treatment groups. After 6 months of treatment, all the patients in both groups showed various degrees of relief. The CR rate of patients in the tocilizumab group was higher than that in the CYC group (50% versus 20%). However, no statistical significance was identified (p = 0.128), probably due to the small sample size, which compromised the power of the test. These findings indicated that the therapeutic effect of tocilizumab was comparable to that of CYC.
Treatment with GC is associated with various adverse reactions, 6 including severe osteoporosis, fragility fracture, and significant weight gain, all of which were seen in our patients. Considering these adverse effects of GC, reducing the dose of GC as soon as possible is extremely important. The combination therapy of GC with tocilizumab GC showed a better steroid-sparing effect than with CYC. Therefore, tocilizumab might be more suitable for patients who are intolerant or show contraindications to GC.
CYC is a widely used disease-modifying antirheumatic drug (DMARD) administered for a variety of autoimmune disease. Several studies have investigated the curative effect of CYC in IgG4-RD.10,17 In a retrospective study with 102 patients, combination therapy of GC and CYC was superior to GC monotherapy in protecting patients with IgG4-RD from disease relapse within 1 year. 17 In another prospective study, GC plus CYC reduced the relapse rate better than GC plus MMF. 10 Both studies indicated the good therapeutic effect of GC plus CYC. However, CYC also has some adverse effects, of which reproductive toxicity is an important issue, since it was seen in 30% of patients within 25 years of follow-up. 26 In addition, hemorrhagic cystitis, a less common adverse effect of CYC in the Chinese population, was still seen in one of our patients in the CYC group. In contrast, the adverse reactions of tocilizumab, such as transient mild liver function impairment and thrombocytopenia, were tolerable.
The present study was the first prospective cohort study to evaluate the efficacy of tocilizumab versus CYC for IgG4-RD treatment. Because of the inherent limitations of a registry and the lack of patient randomization to groups, there may have been a selection bias. However, we adopted several methods to control bias. First, we eliminated the potential confounding factors of previous treatment by only including patients who were new tocilizumab/CYC users. Second, because there may have been a selection bias if patients dropped out of the registry, we adopted a variety of follow-up incentives, and no patients were lost to follow-up in this study. Third, we used a multivariate regression model to adjust the potential confounders, and the results indicated that the correlation was reliable. However, the small sample size was a limitation of our study. IgG4-RD is a rare disease. Therefore, it was not easy to recruit drug-naïve patients with IgG4-related disease. Nevertheless, this study provides a novel comparison of the effectiveness of tocilizumab and CYC. To our knowledge, no similar study has been reported previously. In such a situation, our focus shifted from sample size to research quality. In our study, active IgG4-RD patients, including some patients with refractory and relapsed disease, were enrolled in the tocilizumab group. In a further study, more refractory and relapsed cases should be enrolled to further evaluate the therapeutic efficacy of tocilizumab in the treatment of IgG4-RD.
In conclusion, we found that tocilizumab had a comparable curative effect and fewer adverse effects than CYC in active IgG4-RD. Tocilizumab is expected to be a superior steroid-sparing agent to CYC in IgG4-RD, especially in patients with contraindications or intolerance to GC. A further randomized, double-blind, controlled study would be appropriate to confirm these conclusions.
Conclusion
Tocilizumab could be a better steroid-sparing agent, with a comparable curative effect and tolerance, than CYC in the treatment of IgG4-RD. Good curative effect was also seen in the refractory and relapsed patients who were treated with GC plus tocilizumab.
Supplemental Material
sj-pdf-1-taj-10.1177_20406223211028776 – Supplemental material for Improved clinical outcomes of tocilizumab versus cyclophosphamide for IgG4-related disease: insights from a prospective IgG4-related disease registry
Supplemental material, sj-pdf-1-taj-10.1177_20406223211028776 for Improved clinical outcomes of tocilizumab versus cyclophosphamide for IgG4-related disease: insights from a prospective IgG4-related disease registry by Ji Zongfei, Zhang Lijuan, Sun Ying, Liu Dongmei, Wu Sifan, Kong Xiufang, Ma Lingying, Liu Yun, Ma Lili, Chen Huiyong and Jiang Lindi in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC 81771730); Clinical Cultivation Project of Shanghai Shenkang Science and Technology Development Center (SHDC12019X05).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
