Abstract
The parathyroid glands, one of the last organs to be discovered, are responsible for maintaining calcium homeostasis, and they continue to present the clinician with diagnostic and management challenges that are reviewed herein. Primary hyperparathyroidism (PHPT) comprises the vast majority of pathology of the parathyroid glands. The classic variant, presenting with elevated calcium and parathyroid hormone levels, has been studied extensively, but the current body of literature has added to our understanding of normocalcemic and normohormonal variants of PHPT, as well as syndromic forms of PHPT. All variants can lead to bone loss, kidney stones, declining renal function, and a variety of neurocognitive, gastrointestinal, and musculoskeletal complaints, although the majority of PHPT today is asymptomatic. Surgery remains the definitive treatment for PHPT, and advances in screening, evolving indications for surgery, new imaging modalities, and improvements in intra-operative methods have greatly changed the landscape. Surgery continues to produce excellent results in the hands of an experienced parathyroid surgeon. For those patients who are not candidates for surgery, therapeutic advances in medical management allow for improved control of the hypercalcemic state. Parathyroid cancer is extremely rare; the diagnosis is often made intra-operatively or on final pathology, and recurrence is common. The mainstay of treatment is normalization of serum calcium via surgery and medical adjuncts.
Keywords
Introduction
The parathyroid glands, first reported by Sir Richard Owen during a rhinoceros necropsy in 1862 and studied in detail by Swedish anatomist Ivar Sandstrom in 1880, were one of the last organs to be discovered in the body, 1 and insufficient knowledge about their location and blood supply was the source of inadvertent postoperative tetany in the early 20th century. 2 Early parathyroidectomies for primary parathyroid disease ranged from successful adenoma excision by Felix Mandl in 1927, to multiple reoperations for persistent hyperparathyroidism in Charles Martell, until eventually his mediastinal parathyroid gland was discovered and removed. 3 The parathyroid glands continue to pose challenges as we learn more about their often nuanced pathophysiology and clinical manifestations.
The vast majority of pathology associated with the parathyroid glands, which control calcium homeostasis through the secretion of parathyroid hormone (PTH), is from primary hyperparathyroidism (PHPT), which will be the focus of this review. There have been enormous developments in our understanding of this disease and its more subtle variants. Traditional treatment of the hypercalcemic state with hydration and loop diuretics has given way to bisphosphonates. Moreover, changes in criteria for surgical management, along with advances in preoperative and intra-operative localization modalities and minimally invasive methods have changed the surgical landscape. The purpose of this review is to highlight these advances and provide clinicians with the most up-to-date information on treatment of the hypercalcemic state due to excess PTH secretion.
Part I: Epidemiology and clinical manifestations
Prevalence
Primary hyperparathyroidism (PHPT) is the third most common endocrine disorder and the most common cause of hypercalcemia in the outpatient setting. PHPT was initially described in the 1920s independently in both Europe and North America and was thought of primarily as a rare disease affecting the bones, in the form of osteodystrophy which is rarely seen today. 4 The clinical presentation of PHPT has evolved over the years and across regions. Into the 1960s, many patients presented with the classic ‘stones, bones, groans, psychiatric overtones’ – referring to the kidney stones, bone loss, abdominal pain, and neurocognitive symptoms associated with PHPT – but as biochemical screening has become more routine, patients are now diagnosed with the disease before the development of any overt symptoms. 5 In regions where biochemical screening is not routine, symptomatic PHPT still predominates.
Regardless of region, the prevalence of PHPT increases with age and affects postmenopausal women more commonly than men. In North America, where the prevalence of PHPT is close to 1% in the general population, the disease affects one in 500 postmenopausal women and one in 2000 men and premenopausal women. 6 The incidence of PHPT varies with ethnicity, affecting black individuals more than white individuals, and is less commonly seen in Asian-American and Hispanic populations. 7 A 2015 population-based study performed by the Mayo Clinic on all patients in the Rochester area diagnosed with PHPT from 1970 to 2010 parsed out causes of the increasing prevalence of PHPT over time. As expected, the incidence of asymptomatic PHPT increased with automated serum calcium levels in the 1970s. Interestingly, they noted another peak PHPT incidence after the late 1990s, coinciding with the introduction of national osteoporosis screening guidelines and the increased use of bone mineral density scans. 8
Manifestations
Even with the increasing prevalence of disease, the vast majority of PHPT today is asymptomatic, and the condition is often discovered incidentally on routine lab work. When patients do present with symptoms, the most common manifestation is kidney stones, seen in approximately 40–60% of PHPT patients in the 1970s and in about 15–20% today. 9 Recent estimates suggest that 7–18% of patients with PHPT have kidney stones, and 2–8% of patients with kidney stones have PHPT. 10 Routine abdominal imaging for patients diagnosed with PHPT also demonstrates a higher prevalence of silent kidney stones than previously thought. 11
Bone loss is another measurable symptom of hypercalcemia, manifesting with worsening osteopenia or osteoporosis, particularly in the distal radius, and contributing to increased fracture risk. Osteitis fibrosa cystica, characterized by bone pain, skeletal deformities, and pathological fractures, occurs in advanced disease and is now rare in regions where biochemical screening for hypercalcemia is routinely done. 6
Patients can also present with a variety of neurocognitive, musculoskeletal, and gastrointestinal complaints that are associated with hypercalcemia. 6 Often patients will complain of uncharacteristic fatigue, ‘brain fog’, loss of short-term memory, difficulty concentrating, and insomnia, symptoms that the patients often associate with normal aging. Abdominal pain, constipation, and reflux can also be signs of hypercalcemia, although peptic ulcer disease is rarely seen today. Patients with hypercalcemia also endorse bone and muscle pain. Whatever the symptomatology, symptoms do not correlate with calcium level. There are some data on the effects of hypercalcemia on cardiovascular health, particularly with regard to hypertension, premature atherosclerosis, and valve calcification; however, these studies were inconclusive. 6
As many of the symptoms associated with PHPT can be non-specific, PHPT should be suspected in patients with recurrent kidney stones, osteoporosis, on long-term lithium therapy, prior radioactive iodine ablation, or head and neck radiation in childhood.
Establishing the diagnosis
Definition of PHPT
The diagnosis of PHPT is a biochemical one, based on elevated calcium (after correcting for albumin) and elevated or inappropriately normal PTH level. PHPT is caused by loss of control of calcium homeostasis, either because of increased PTH secretion by individual parathyroid cells, or from increased parathyroid cell proliferation. PTH acts on the bone and kidney. In healthy individuals without parathyroid disease, PTH has balanced anabolic and catabolic effects on bone, and it stimulates calcium reabsorption, phosphate excretion, and 1 alpha-25-hydroxy vitamin D hydroxylase activity in the kidney. These coordinated activities help normalize serum calcium within hours of a hypercalemic event, with decreased PTH secretion leading to decreased bone resorption, increased renal calcium loss, and decreased renal hydroxylation of 25-hydroxy-vitamin D.6,12
Diseased states with unregulated PTH secretion lead to bone loss and fragility fractures, as well as hypercalciuria and kidney stones. Parathyroid adenomas, parathyroid hyperplasia, and parathyroid carcinoma can all cause PHPT.
Other lab parameters, such as creatinine, albumin, phosphorus, chlorine, and 25-hydroxy vitamin D, are helpful adjuncts. Hypoalbuminemia can mask overt hypercalcemia; corrected calcium (Ca) should be calculated using the following formula: corrected Ca = serum Ca level (mg/dL) + 0.8 × (4 − serum albumin [g/dL]). Ionized calcium may be used for confirmatory testing. Vitamin D deficiency can cause elevated PTH levels, and calcium and PTH should be rechecked once the deficiency has been corrected. Patients with PHPT have a chloride:phosphorus ratio greater than 33. Urine studies are also helpful to distinguish PHPT from familial hypocalciuric hypercalcemia (FHH). Patients with FHH generally have 24-hour urine calcium less than 100 mg. The calcium to creatinine clearance ratio (CCCR), calculated as (24-hour urine calcium/plasma total calcium)/(24-hour urine creatinine/plasma creatinine), can help differentiate PHPT from FHH, as FHH has a CCCR less than 0.01. If 24-hour urine calcium excretion is greater than 400 mg per day, a complete stone risk profile analysis should be measured according to most recent guidelines. 13
Advancements in diagnosis of variants of PHPT
With improved and more frequent biochemical testing, PHPT is recognized earlier, with improved recognition of subtle biochemical findings and variants such as normocalcemic and normohormonal PHPT. Although there has been much published on these two entities in recent years, they remain incompletely characterized, and further large-scale studies with longer-term follow-up are necessary to understand these variants better.
Normocalcemic PHPT has a reported a prevalence between 0.5% and 16%, 14 and secondary causes of hyperparathyroidism such as vitamin D deficiency, kidney disease, malabsorption, and idiopathic hypercalciuria must be ruled out to make this diagnosis. Corrected serum calcium must be measured twice in a 6-month period to distinguish this entity from classic PHPT. The pathophysiology of this disease has been hypothesized as: an early form of classic PHPT; relative hypercalcemia compared to a patient’s individual baseline; less PTH secretion; and partial tissue resistance to PTH. 15 The natural history of normocalcemic PHPT is similar to that of classic PHPT, with many presenting asymptomatically and greater than one-third progressing to symptomatic disease characterized by bone loss and kidney stones. 16 More recent data suggest that patients with normocalcemic PHPT develop kidney stones and bone loss at similar or higher rates than patients with classic PHPT despite normal calcium levels, 15 although other studies have found that the rate of bone turnover in normocalcemic PHPT is more similar to that of normal controls than to that of patients with classic PHPT. 17 Admittedly there is selection bias in patients who present with symptoms of kidney disease or bone loss and are evaluated for parathyroid disease, and reasons why normocalcemic patients develop nephrolithiasis or osteoporosis remain unclear. 18
The Fourth International Workup ‘Guidelines for the Management of Asymptomatic Primary Hyperparathyroidism’ addressed normocalcemic PHPT and proposed an algorithm for monitoring these patients with calcium and PTH annually and dual-energy X-ray absorptiometry (DEXA) every 1–2 years, with surgery recommended for those patients who have progression of disease as evidenced by worsening bone mineral density or fracture or the presence of kidney stones. 13 Medical management has not been well studied in this population, although in a small study of 30 postmenopausal women with normocalcemic PHPT, the 15 women who received alendronate appeared to show improvements in bone mineral density in the lumbar spine compared to the control group. 19 There have been more case series on surgical management and subsequent improvements in nephrolithiasis and bone mineral density. When surgery is performed, the most common finding is a single adenoma, and intra-operative parathyroid hormone (IOPTH) can be used in a similar fashion to classic PHPT to determine the extent of surgery. 20 However, multi-gland disease is more common in normocalcemic PHPT than in the classic variant (12% versus 4% in a retrospective review of 616 patients from 2004 to 2014 at Johns Hopkins), and surgeons should have a low threshold for conversion to bilateral exploration if the IOPTH does not decline appropriately. 21 The prevalence of multi-gland disease, as well as decreased sensitivity of established diagnostic modalities for localization, could also be the reason there is a lower surgical cure rate (normalization of serum PTH by 6 months after surgery) compared to the hypercalcemic form. 18 A query of the Collaborative Endocrine Surgery Quality Improvement Program (CESQIP) database reinforced these findings of higher prevalence of multi-gland disease, as well as higher rates of reoperation. 22
Normohormonal PHPT, in which calcium is elevated but the PTH level is normal, has been reported in 5.5–17% of patients with PHPT and has only recently been recognized as a distinct form of PHPT. 23 In a retrospective study examining 516 consecutive patients undergoing parathyroidectomy, 22.5% had PTH levels in the normal range (less than 75 pg/mL). These patients were less likely to have an enlarged parathyroid gland localized on ultrasound or sestamibi, and intra-operatively were almost twice as likely to have multi-gland hyperplasia than patients with classic PHPT. 23 As IOPTH is so commonly used these days to determine the extent of parathyroidectomy, Trinh et al. studied the utility of using IOPTH in patients who are normohormonal and have a normal baseline PTH at the beginning of surgery. 24 The authors found that patients with normohormonal PHPT had a slower rate of PTH decline; nonetheless, a PTH drop of greater than 50% was still a reliable marker for a successful operation. 24
Hollowoa et al. performed a literature review comparing classic, normocalcemic, and normohormonal PHPT. 25 Demographic profiles were similar across the groups. Rates of nephrolithiasis and decreased bone mineral density were higher in the normocalcemic and normohormonal PHPT groups, but this could be selection bias, as these are the symptoms that that caused them to be referred for evaluation; still, due to these end-organ effects, surgery is still recommended as treatment of these more subtle variants of PHPT. 25
Diagnostic challenges
In any hyperparathyroidism work-up, it is important to rule out benign FHH, an autosomal dominant disorder that is due to inactivating mutations in the calcium-sensing receptor gene (CASR) causing decreased receptor activity. CASR is expressed on parathyroid cells and cells of the thick ascending loop of the renal tubule. Loss or reduction of function of CASR impairs the inhibitory effect of serum calcium on PTH regulation and reduced calcium excretion. These patients have mild hyperparathyroidism, hypercalcemia, hypocalciuria, hypomagnesemia, and hypophosphatemia, and manifest few symptoms if any. A thorough family history and a low urine CCCR can differentiate this disease from PHPT. This distinction is important, because FHH does not require parathyroidectomy, and subtotal parathyroidectomy does not cure the disease. 26
Kidney stones and hypercalciuria (calcium excretion greater than 250 mg/day in women and 300 mg/day in men) are features of PHPT, but idiopathic hypercalciuria should also be ruled out in the evaluation of PHPT. Idiopathic hypercalciuria, which is a polygenic trait, causes increased calcium turnover. Features of this disease include increased gut calcium absorption, decrease renal calcium absorption, and increased bone turnover, the reasons for which are not clear. Unlike PHPT, patients have normal calcium and PTH levels, and the trait is affected by levels dietary calcium. 27 Long-term idiopathic hypercalciuria, however, can lead to parathyroid stimulation and in turn lead to tertiary hyperparathyroidism with concomitant parathyroid hyperplasia or adenoma; in these cases, even after definitive parathyroidectomy, hypercalciuria can persist, thus presenting both a diagnostic and management challenge. 28
Secondary hyperparathyroidism (SHPT) is not a hypercalcemic state; rather, it is a maladaptive process whereby low extracellular calcium causes increased PTH synthesis and parathyroid cell proliferation, which eventually becomes autonomous. This disease is characterized by four-gland hyperplasia. It has many different causes, including vitamin D deficiency, history of bariatric surgery, other causes of malabsorption, restricted calcium intake, renal leak, or decreased glomerular filtration rate (GFR). The most common cause of SHPT is end stage renal disease (ESRD). Decreased calcium absorption due to renal failure leads to PTH hypersecretion, and laboratory data demonstrate low-normal calcium, elevated phosphorus, and markedly elevated PTH. The initial treatment of SHPT is medical management with phosphate binders, vitamin D analogs, and calcimimetics with goal PTH 150–550 pg/mL. 29 Indications for surgery in SHPT include osteodystrophy, bone pain, soft tissue calcifications, calciphylaxsis, severe vascular calcifications, and pruritus, as well as medically refractory hypercalcemia, hyperphosphatemia, product of calcium and phosphorus greater than 70, and PTH greater than 800 pg/mL. Tertiary hyperparathyroidism (THPT) is persistent hyperparathyroidism following kidney transplant. Laboratory values are similar to that of PHPT, with elevated calcium and PTH. It is reported in 20–50% of patients. The most important predictors of THPT are severity of SHPT and length of time on dialysis. Approximately 1–5% of patients require surgery for THPT. Indications for surgery (generally subtotal parathyroidectomy) include hypercalcemia >11.5 mg/dL, persistent hypercalcemia greater than 10.2 mg/dL more than 12 months after transplantation, and the presence of symptoms. 30
Syndromic hyperparathyroidism
The vast majority of PHPT (>90%) is sporadic and can be caused by single gland disease characterized by enlargement of one parathyroid gland (~85%); multi-gland disease due to parathyroid hyperplasia of all four glands (~10–15%); and rarely by parathyroid carcinoma (<1%). Other causative agents implicated in the development of PHPT include a history of external beam radiation to the neck, exposure to nuclear fallout, and long-term lithium therapy. 6
PHPT can also be part of a genetic syndrome. The most common associations are multiple endocrine neoplasia type 1 (MEN1), multiple endocrine neoplasia type 2A (MEN2A), hereditary hyperparathyroidism-jaw tumor syndrome (HPT-JT), and familial isolated hyperparathyroidism (FIHP). These hereditary syndromes are summarized in Table 1. They should be suspected if the patient has a known family history of hypercalcemia, young age at presentation, or multi-gland hyperplasia.
Inherited forms of primary hyperparathyroidism.
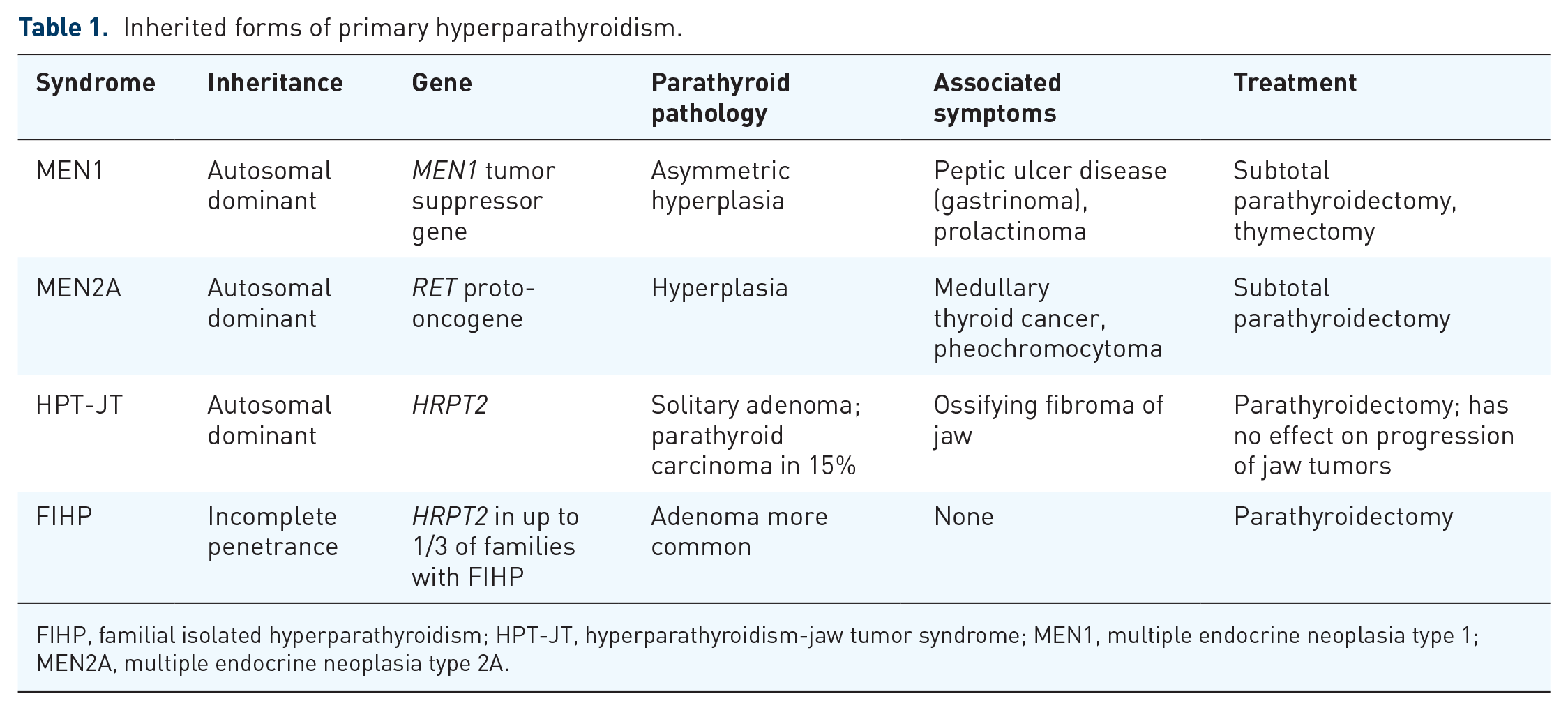
FIHP, familial isolated hyperparathyroidism; HPT-JT, hyperparathyroidism-jaw tumor syndrome; MEN1, multiple endocrine neoplasia type 1; MEN2A, multiple endocrine neoplasia type 2A.
MEN1 is an autosomal dominant disorder characterized by a mutation in the MEN1 tumor suppressor gene encoding menin. It primarily affects the parathyroid glands, pancreatic endocrine cells, and anterior pituitary. Most patients with the disease manifest symptoms between the ages of 20 and 40 years; mutations in MEN1 are highly penetrant, and almost half of affected patients are symptomatic by age 20 years and nearly 100% are symptomatic by age 60 years. PHPT is the initial clinical and biochemical manifestation of MEN1 in 60–90% of patients; conversely, MEN1 is the cause of PHPT in 2–4% of cases in the general population. 31 MEN1 patients develop hypercalcemia and PHPT at an earlier age than those with sporadic PHPT, with studies describing elevated ionized serum calcium levels in 66% of patients before 25 years of age. 32 Surgical indications for PHPT due to MEN1 are the same as for sporadic disease (see below). An additional indication for parathyroidectomy in patients with MEN1 is the presence of severe peptic ulcer disease or other symptoms caused by a gastrinoma (the Zollinger–Ellison syndrome) that are difficult to control with medications. Hypercalcemia typically worsens hypergastrinemia, and parathyroidectomy may markedly reduce gastrin secretion in patients with a gastrinoma. 32
Unlike patients with sporadic PHPT, in whom 85% have a solitary adenoma, MEN1 pathology is asymmetric hyperplasia involving all parathyroid glands. The procedure of choice, therefore, is subtotal parathyroidectomy, leaving an approximately 50 mg, well-vascularized remnant of the most normal-appearing tissue, with cervical thymectomy to rule out a supernumerary or ectopic gland, which is seen in as many as 20–30% of MEN1 patients. Alternatively, the surgeon can perform a total parathyroidectomy with parathyroid implantation into the forearm; while this approach has a lower rate of recurrence of PHPT, it also carries a higher risk of permanent hypoparathyroidism.
Both persistence and recurrence of hyperparathyroidism occur more frequently in MEN1 than in sporadic disease. Persistence, defined as a failure to normalize serum calcium and PTH levels following the initial surgery, occurs in 12% of cases of MEN1. Recurrent disease, defined as reappearance of hyperparathyroidism following at least 3 months of normocalcemia, is seen in as many as 44% of these cases 8–12 years following surgery, 31 likely owing to the high prevalence of supernumerary and ectopic glands in this disease process.
MEN2A
MEN2A is an autosomal dominant disorder characterized by a gain-of-function mutation in the RET proto-oncogene. The most common tumors associated with MEN2A are medullary thyroid cancer (MTC), pheochromocytoma, and PHPT. Unlike MEN1, PHPT presents many years after the development of MTC, with average age of presentation at 38 years old, and symptoms tend to be mild. These patients can have single gland disease, but the majority have parathyroid gland hyperplasia. Similar to MEN1, MEN2A patients are at increased risk for persistent or recurrent disease, and bilateral neck exploration is recommended. 33
Hereditary hyperparathyroidism-jaw tumor syndrome
Hereditary hyperparathyroidism-jaw tumor syndrome (HPT-JT) is a rare autosomal dominant disorder. Hyperparathyroidism is a result of parathyroid tumors (typically a solitary adenoma), and parathyroid carcinoma has a prevalence as high as 15%. 34 These patients also develop ossifying fibromas of the mandible and maxilla that are distinct from osteitis fibrosa cystica, and rarely develop kidney and uterine lesions. Excision of solitary parathyroid tumor leads to normocalcemia, but does not appear to have any effect on the development or progression of jaw tumors. 35
Familial isolated hyperparathyroidism
Familial isolated hyperparathyroidism (FIHP) is a familial form of PHPT without other associated features of syndromic hyperparathyroidism. This can be the result of incomplete penetrance. The HRPT2 germ-line mutation has been reported in up to one-third of families with FIHP, and these individuals present with more severe hypercalcemia and at a younger age than those without an HRPT2 mutation. 36
Special patient populations
The manifestations and management of PHPT in special patient populations deserve mention, as treatment has evolved over the years.
Pregnant women
The incidence of PHPT in women of childbearing age is eight per 100,000, 37 and although gestational PHPT is estimated to be less than 1%, the true incidence is likely to be higher given the overlap of PHPT and pregnancy symptoms and under-recognition of the disease. 38 In addition to the typical manifestations of PHPT, the consequences of untreated PHPT in pregnancy include hyperemesis gravidarum, preeclampsia, miscarriage, intrauterine growth restriction, and neonatal tetany secondary to fetal parathyroid gland suppression.37–39 Although there are limited data on the treatment of PHPT in pregnancy, a few studies have lent clarity to issues such as medications safe to use in pregnancy and appropriate work-up and surgical management during pregnancy.
Medical management has been used for mild to moderate hypercalcemia, and intravenous fluids are often first-line treatment. Bisphosphonates, which cross the placenta, can impair fetal skeletal development and are contraindicated in pregnancy. 39 Calcitonin, which blocks bone resorption and does not cross the placenta, has been described in case reports, but the calcium-lowering effects are short lived. 40 Similarly, the calcimimetic cinacalcet has been shown to lower serum calcium levels in pregnancy and has been used as a bridge to surgery in the postpartum period, but there are insufficient data on the fetal effects of cinacalcet to recommend its use in pregnancy. 41
Surgery may be safely performed in the second trimester, thus avoiding the teratogenic effects of anesthesia on the growing fetus in the first trimester, and the risk of preterm delivery in the third trimester. 37 Surgery during pregnancy is safe and effective, avoids potentially harmful side effects of continued medical management in pregnancy, and decreases the risk of neonatal hypocalcemia. 39 A recent systematic review found that surgery results in better fetal outcomes, and recommends surgery in the second or third trimester regardless of patient symptomatology. 38 Preoperative localization with ultrasound is safe in any trimester. Recently, the American College of Obstetricians and Gynecologists guidelines for diagnostic imaging in pregnancy have stated that sestamibi scan at doses of less than 5 mGy does not increase the risk of birth defects 42 and thus may be used for parathyroid localization.
While clear guidelines on treatment of PHPT in pregnancy are lacking, a multidisciplinary approach among members of the obstetrics, endocrinology, surgery, anesthesia, and neonatal teams is critical for the wellbeing of the mother and baby.
Pediatric patients
PHPT in pediatric patients is much rarer than in adults, affecting approximately two to five children in 100,000. Moreover, at diagnosis children often already have kidney stones or bone loss, in part due to delay in diagnosis. 43 In a retrospective study of 63 children, El Allali et al. found that 52% of cases were caused by a genetic mutation, 43 either in genes involved in parathyroid proliferation (MEN1, CDKN1B, CDKB2B, CDKN2C, CDC73, RET, GMC2), as was the case in the adolescent population, or those encoding the calcium-sensing receptor (CaSR) or downstream signaling components (GNA11, AP2S1), found in 94% of infants with PHPT-causing genetic mutations. 43
As with adults, parathyroid adenoma is the most common etiology for PHPT, although parathyroid hyperplasia is much more common in pediatric populations (up to 40% versus 10–15% in adults), even in sporadic PHPT. 44 Surgery is still the standard of care in the pediatric population, and Ramonell et al. recently demonstrated that surgery for PHPT can be safely performed in pediatric patients in the outpatient setting, with low complication rates in the hands of experienced endocrine surgeons. 44
The elderly
Although the prevalence of PHPT increases with age, and the elderly are the most likely to benefit from the improvement in end-organ damage after parathyroidectomy, they are less likely to have surgery. Two separate studies found the rate of parathyroidectomy in patients aged 65 years and over who meet criteria for surgery is only around 30%, regardless of whether they have Medicare 45 or private insurance. 46 Sieb et al. looked at 210,206 Medicare beneficiaries with PHPT and found that increasing patient age (particularly over 75 years), comorbidities, and frailty score were associated with lower use of parathyroidectomy, despite multi-disciplinary guidelines recommending its use. 45 Further studies are needed to address the underutilization of parathyroidectomy in the geriatric population and provide more specific guidelines for surgery in this patient group.
Hypercalcemic crisis
Hypercalcemic crisis, defined by a serum calcium level exceeding 14.5 mg/dL, can be caused by large parathyroid adenomas, parathyroid carcinoma, or hyperplasia. Clinical manifestations of hypercalcemic crisis include polyuria and extreme thirst, abdominal complaints such as nausea and constipation, and cognitive impairment. Diagnostic work-up should include albumin-corrected calcium levels, PTH, and electrolytes. Electrocardiogram (EKG) may demonstrate a shortened QT interval.
There have been numerous therapeutic advances in the medical management of hypercalcemia in recent years. While rapid volume re-expansion with 0.9% normal saline remains the mainstay of treatment, loop diuretics have fallen out of favor. Previously, loop diuretics after rehydration were thought to aid calciuresis; however, they have recently been shown to be ineffective in reducing serum calcium, and should only be used if the patient develops volume overload. 47 Instead, intravenous bisphosphonates have emerged as the recommended treatment for continued hypercalcemia after rehydration. Caution must be used in renally impaired patients, and the dose of medication can be reduced and/or infused more slowly. The serum calcium is expected to reach its nadir 2–4 days after bisphosphonate infusion. 47
Second-line treatments for hypercalcemic crisis include glucocorticoids, which inhibit the conversion of vitamin D to calcitriol and thus decrease calcium absorption, as well as calcimimetics, denosumab, and calcitonin. 47 Calcimimetics, which interact with the calcium-sensing receptor on parathyroid cells to downregulate PTH, often take longer to titrate appropriately and are not commonly used in the acute setting. Denosumab, an osteoclast inhibitor, has been used more recently for patients in hypercalcemic crisis, particularly those who have renal impairment that can be made worse by the nephrotoxic side effects of bisphosphonates. Eremkina et al. performed a retrospective study of 10 patients with hypercalcemic crisis who were treated with 60 mg of denosumab; normocalcemia was observed on average by day 7 post-treatment. 48 The study was limited by its small sample size, heterogeneous causes of hypercalcemic crisis in study patients, and lack of standardization of treatments, including some patients treated concurrently with cinacalcet; however, denosumab was able to aid calcium normalization without causing further renal impairment. 48 Calcitonin, which inhibits bone resorption, has rapid onset but short duration of effect, and may cause rebound hypercalcemia.
Ultimately, parathyroidectomy is the definitive treatment for hypercalcemic crisis secondary to PHPT and should be performed expeditiously once the patient has been medically optimized.
Part II: Management
Operative management
Surgery remains the first-line treatment for PHPT, and indications for surgery, preoperative work-up, and type of surgery have seen many advances in the past few decades. Surgery is recommended for all symptomatic patients. In 2009, the Third International Workshop on ‘Guidelines for the Management of Asymptomatic Primary Hyperparathyroidism’ recognized that up to one-third of patients with PHPT have disease progression 49 and outlined indications for surgery in asymptomatic patients. These criteria included age less than 50 years; serum calcium greater than 1 mg/dL above the upper limit of normal; bone density T-score less than −2.5 at the lumbar spine, femoral neck, total hip, or distal one-third radius in peri or postmenopausal women, and Z score less than −2.5 in premenopausal women and men younger than 50 years. 50 The most recent summary statement released in 2014 by the Fourth International Workshop differs in also taking into account subclinical kidney stones and increased risk of vertebral fractures. 13 The kidney is a primary target of PHPT. Although the 2009 guidelines did not recommend 24-hour urine calcium like the First and Second International Workshops did, the 2014 guidelines reintroduced urine calcium excretion greater than 400 mg per day as a guideline for surgery, and recommended stone risk analysis for these patients, as well as renal imaging with either kidney–ureter–bladder (KUB), ultrasound, or computed tomography (CT) to look for silent nephrolithiasis or nephrocalcinosis. 13 With regard to the skeletal system, the most recent guidelines stress the importance of measuring cortical bone density in the distal one-third radius, and also recommend other imaging modalities to evaluate trabecular bone involvement. Tests for cardiovascular involvement are still not recommended in the evaluation of PHPT as there are no prospective data on cardiovascular outcomes in mild PHPT. The newer guidelines also recommend genetic testing for PHPT patients who are younger than 30 years of age, have syndromic findings, and have a family history of syndromic PHPT. 13
Surgery is the only definitive treatment for PHPT. After successful parathyroidectomy, bone mineral density improves, fracture risk decreases, kidney stone formation decreases, and there is improvement in subjective symptoms associated with PHPT. Even patients with advanced osteitis fibrosa cystica can have bone mineral density improve into the normal range after parathyroidectomy. 51 Although parathyroidectomy decreased the risk of kidney stone formation, in a recent publication, we demonstrated that nearly one-quarter of our study population with concurrent PHPT and kidney stones present with recurrent stones despite normalization of serum calcium after definitive parathyroidectomy; these patients were younger and warrant closer monitoring after surgery. 52 Improvements in subjective neurocognitive complaints are harder to measure than improvements in bone and stone disease. In 2009, Walker et al. compared 39 postmenopausal women with mild disease before and after parathyroidectomy to 89 age-matched controls. 53 The patients with PHPT scored worse in terms of depression, anxiety, and memory tests than the control group at baseline; however, 6 months after parathyroidectomy, the scores between the patients who were now cured of their PHPT and that of the healthy controls showed no significant differences. 53 More recent quality of life studies have demonstrated improved neurocognitive symptoms after surgery in patients with even mild or normocalcemic PHPT. 54
In addition to symptom improvement, successful surgery has been shown to be more cost-effective than monitoring. Zanocco et al. published their findings on cost-effectiveness of parathyroidectomy after taking into account the fracture risk reduction post-parathyroidectomy. 55 The authors found that for patients with mild, asymptomatic PHPT, parathyroidectomy is the dominant strategy, resulting in cost savings compared to observation, and improving quality of life. 55
Although mild PHPT may be safely monitored without surgery, Rubin et al. demonstrated that bone mineral density starts to decrease at cortical sites before 10 years, and 37% of patients in this study had enough bone loss to meet surgical guidelines by 15 years. 49
Advances in localization modalities
Once the biochemical diagnosis has been established and the decision to proceed to surgery has been made, the next step is imaging to help the surgeon plan the operation. Historically, patients with PHPT underwent bilateral neck exploration. In the hands of an experienced surgeon, cure rates exceeded 95%. 56 Today, improvements in non-invasive imaging have dramatically changed the surgical approach to PHPT and allowed surgeons to perform directed, minimally invasive parathyroidectomy (MIP) with similar cure rates.
The most widely available and cost-effective localization study is a simple neck ultrasound. A normal-sized parathyroid gland is too small to be seen on any imaging study, but an enlarged parathyroid gland can often be seen as a hypoechoic structure posterior to the thyroid gland (Figure 1). Doppler imaging can identify the parathyroid feeding vessel and distinguish parathyroid from lymph node. 57 In experienced hands, ultrasound has a high sensitivity for localizing parathyroid adenomas, but sensitivity is reduced in the setting of large thyroid nodules and goiters or by patient body habitus, and it is more difficult to assess parathyroid glands in ectopic locations.3,57
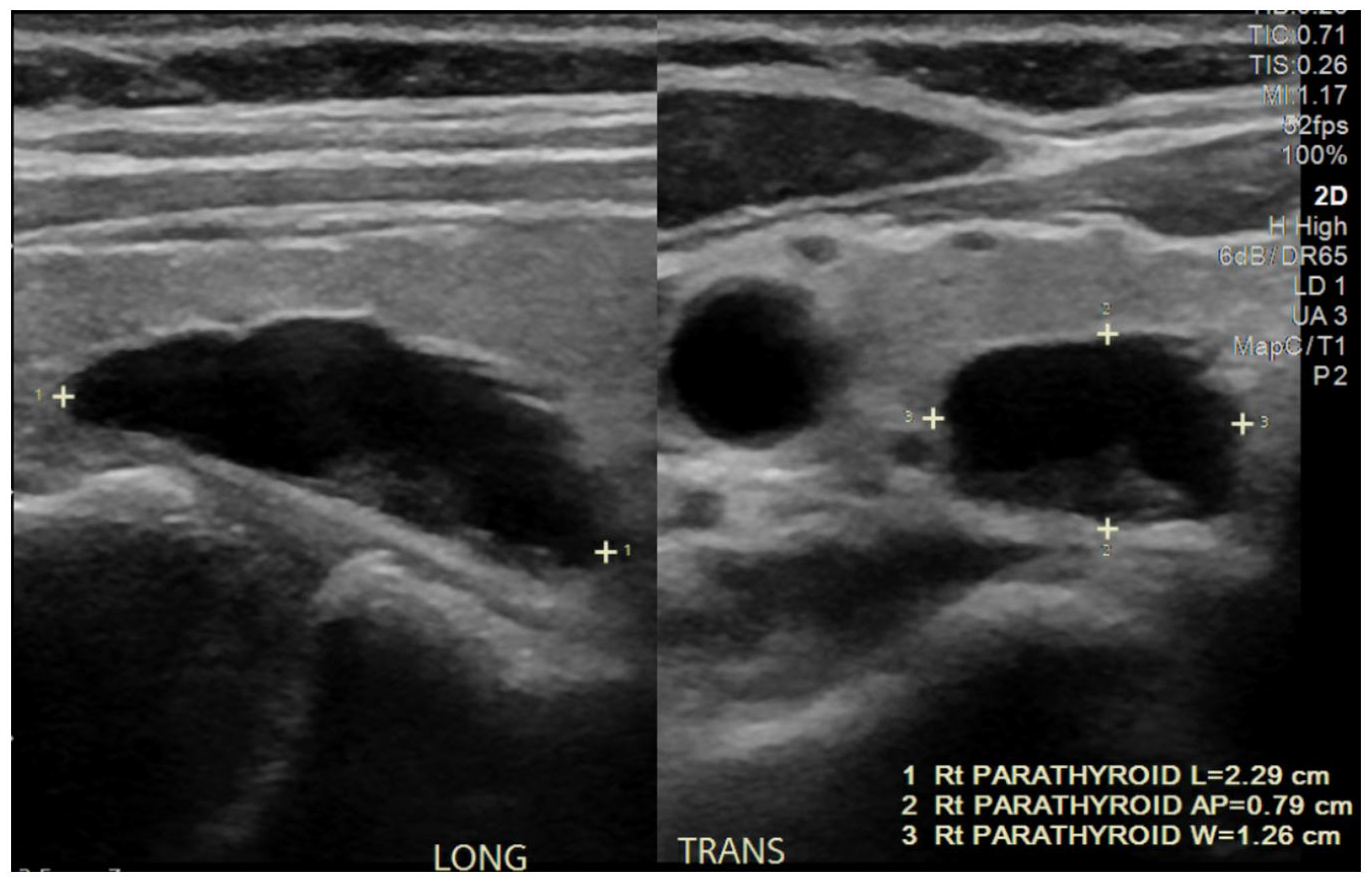
This is a 72-year-old woman with PHPT. Ultrasound demonstrated an enlarged parathyroid gland on the right. She underwent minimally invasive parathyroidectomy with excision of a right superior parathyroid adenoma. Baseline PTH was 285 pg/mL, final post-excision PTH normalized to 40 pg/mL.
If the ultrasound is non-localizing, radionuclide imaging with 99mTc-sestamibi scan can help detect an abnormal parathyroid gland. Sestamibi concentrates in the mitochondrial-rich oxyphilic cells of the parathyroid glands and helps distinguish it from surrounding thyroid tissue. 57 The most common type of sestamibi scan provides immediate and delayed planar images over a 2-hour period, with focal radiotracer activity retention in an abnormal parathyroid gland seen on the 90–180 min post-injection images. The addition of single-photon emission computed tomography (SPECT) allows for the detection of parathyroid glands that are located in the posterior retroesophageal or retropharyngeal spaces, which would be hidden on planar images. 57 SPECT may be further fused with CT scan for anatomical correlation, known as SPECT-CT, which allows more precise localization for the abnormal parathyroid gland and differentiation from surrounding tissue. Although SPECT and SPECT-CT have a higher sensitivity for abnormal parathyroid detection than planar sestamibi, they are not widely available. Moreover, sestamibi scans can produce false-positive results in the setting of thyroiditis, thyroid cancer, Hurthle cell adenomas, follicular adenomas, and lymph node metastases, which can be sestamibi-avid. Lastly, sestamibi scintigraphy, like ultrasound, has a limited ability to detect multi-gland hyperplasia.3,57
A newer localization modality is four-dimensional computed tomography (4D-CT), so called because of the addition of time as the fourth dimension. The multiphase contrast CT allows for the identification of abnormal parathyroid glands because of these glands’ rapid uptake and washout of contrast. In addition to functional information, 4D-CT can also give detailed anatomical information (Figure 2). Naqvi et al. found this modality to have exceptionally high sensitivity and specificity for locating ectopic glands, as well as a 93.3% negative predictive value, thereby limiting unnecessary bilateral exploration. 58 Limits of this 4D-CT imaging include the fact that it is not widely available, as well as higher thyroid-specific radiation exposure. There has been a lot written in the literature recently comparing 4D-CT to sestamibi. The largest study to date, evaluating 400 patients who underwent 4D-CT alone or in combination with sestamibi SPECT-CT at a single institution, found 4D-CT to have a higher sensitivity for both single-gland (92.5% versus 72.1%) and multi-gland disease (58.2% versus 30.8%). Moreover, the addition of SPECT-CT did not improve localization compared to 4D-CT alone. 59 Conversely, a meta-analysis of nine studies specifically comparing sestamibi SPECT-CT to 4D-CT (including the study by Yeh et al.) 59 found just borderline increased sensitivity of 4D-CT, which may not outweigh the risk of higher radiation exposure. 60 Still, there are certain situations in which 4D-CT outperforms other imaging modalities, including in the re-operative setting for persistent or recurrent hyperparathyroidism (4D-CT found to have a sensitivity of 77% versus 46% for SPECT-CT for correct lateralization), 61 mild or normocalcemic PHPT, 62 and inconclusive imaging. These different imaging modalities are summarized in Table 2.

This is a 53-year-old woman with PHPT. CT demonstrated an ectopic 1.3 × 0.9 cm parathyroid gland in the anterior mediastinum, just inferior to the innominate vein. The patient underwent robot-assisted thoracoscopic thymectomy. The PTH declined from 128 pg/mL at baseline to 42 pg/mL at the end of the case.
Imaging modalities.

4D-CT, four dimensional computed tomography; SPECT-CT, single-photon emission computed tomography fused with computed tomography scan.
Although not commonly used, magnetic resonance imaging (MRI) has been an available adjunct for localizing parathyroid glands after a failed previous parathyroidectomy or in the case of recurrent disease. Recently, Kluijfhout et al. examined the use of MRI for parathyroid localization in 84 patients with recurrent or persistent disease and 41 patients who had not yet undergone parathyroidectomy. 63 MRI was shown to have a sensitivity of 79.9% for the detection of parathyroid adenomas, with increased sensitivity of 91.5% when MRI was combined with ultrasound and sestamibi, in both patients who had and had not undergone previous operation. 63 MRI shows promise as a localizing modality, particularly as it does not subject the patient to ionizing radiation.
Whatever the imaging modality, it is important to keep in mind imaging pitfalls: multinodular thyroid goiter, abnormal parathyroid glands that are less than 200 mg, multi-gland hyperplasia, and suboptimal imaging and interpretation. If the patient has the biochemical diagnosis of PHPT, negative imaging does not rule out parathyroid disease, and the patient should undergo full neck exploration. 3
There are some intra-operative localization techniques that have been used with varying degrees of success. Gamma probes, used in radio-guided parathyroidectomy, work by localizing an abnormal parathyroid gland by its higher radionuclide counts compared to surrounding tissue. Disadvantages of this include the patient needing to be injected with sestamibi 1–2 hours prior to surgery, and gamma probe counts can be inaccurate or confusing. Moreover, the usefulness of radio-guided parathyroidectomy in the setting of a negative preoperative sestamibi scan was questionable, but Chen et al. demonstrated that gamma probes are effective in locating parathyroid glands even in cases of negative preoperative localization. 64 Methylene blue, which is selectively taken up by abnormal parathyroid glands, has also been used to guide surgery; however, side effects including neurotoxicity, nausea, spillage of methylene blue into the operative field, and false-positive staining of lymph nodes, thyroid, and nearby thymus and adipose tissue 65 limit its usefulness. More recently, intra-operative cameras that detect the parathyroid glands’ natural autofluorescence with near-infrared fluorescence imaging can illuminate parathyroid glands before direct visualization. This technology is limited in its ability to detect abnormal parathyroid glands and is more useful for preserving normal parathyroid glands during thyroid surgery; however, Kose et al. recently found that hyperfunctioning parathyroid glands emit a more heterogeneous pattern and lower intensity autofluorescence than normofunctioning parathyroid glands, aiding in their detection. 66
Operation
The normal human parathyroid glands, measuring 2–4 mm in size and weighing 20–40 mg on average, are located on the posterior surface of the thyroid gland and are supplied by the inferior thyroid artery. The superior and inferior parathyroid glands are typically found within a 1 cm radius cephalad and caudal, respectively, to the intersection of the inferior thyroid artery and recurrent laryngeal nerve. The inferior parathyroid glands, derived from the third branchial pouch, demonstrate greater variability in their location than the superior parathyroid glands, which are derived from the fourth branchial pouch. 67
The traditional surgical approach is a bilateral exploration, wherein all four parathyroid glands are identified and the abnormal parathyroid gland excised. However, with the advent of improved imaging modalities and reliable IOPTH management, surgical management has evolved to more limited dissections that shorten operating time, hospital length of stay, and postoperative recovery; 68 provide cosmetic benefit; and improve patient quality of life in the short and long term. 69 There was some concern about increased recurrence due to missed multi-gland disease in focused parathyroidectomy; however, systemic reviews comparing MIP to bilateral exploration demonstrate similarly high cure rates and low complication rates (hypocalcemia, recurrent laryngeal nerve injury, bleeding, infection, and mortality) for both approaches, although literature review is limited by variable endpoints to determine cure, treatment of hypocalcemia, and consistency in the evaluation of recurrent laryngeal nerve injury. 70 Ahmadieh et al. reviewed five randomized controlled trials comparing MIP and bilateral exploration, all based out of European university hospitals. 71 The authors found similar success rates between the two operative approaches at 6 months and 5 years post-parathyroidectomy when IOPTH monitoring is used. 71
Minimally invasive (directed) parathyroidectomy is typically performed through a 2–3 cm transverse collar incision, and the operation is targeted to the enlarged gland identified on preoperative imaging. The surgeon needs to have an intimate knowledge of parathyroid anatomy, and IOPTH guides the extent of surgery. Typically, an IOPTH level is sent prior to incision, and then 5, 10, and 15–20 min after excision of the suspected abnormal parathyroid gland. PTH has a half-life of less than 5 min, and the expectation is for the PTH to decrease by at least 50%, otherwise known as the Miami criteria,72,73 if the abnormally secreting parathyroid gland has been excised. Some institutions use an absolute rather than relative PTH value to determine the extent of surgery, concluding when PTH is normal. This is problematic in patients who may have mild or normohormonal PTH values. Most institutions will use criteria of a 50% drop in PTH and/or normalization of PTH to determine successful surgery. All of these criteria have similar accuracy and result in eucalcemia when discerned appropriately by the surgeon. 74 Importantly, PTH kinetics can be affected by other factors such as decreased creatinine clearance or long-term bisphosphate use, and it may take longer for PTH to reach its nadir. 75 Red flags to alert the surgeon to the presence of more than one abnormal parathyroid gland include insufficient IOPTH drop, slow rate of IOPTH decline, an IOPTH nadir instead of continued decline, and multiple grossly abnormal parathyroid glands visualized during the operation. 76 In these cases, a full four-gland exploration should be performed. Bilateral exploration is also recommended as the initial operation in the case of non-localizing or discordant preoperative imaging, unavailability of intra-operative PTH, and high suspicion for multi-gland disease, for example, in a patient with long-term lithium use or syndromic PHPT. 77 In the latter case, particularly in patients with MEN1, a subtotal parathyroidectomy is recommended.
Ectopic parathyroid glands can be a source of great frustration, and the surgeon must have an awareness of their most likely locations. Superior gland adenomas are rarely truly ectopic, and can be predictably located retropharyngeal, retroesophageal, paraesophageal, or in the tracheoesophageal groove. Other superior parathyroid gland adenomas may be compressed behind the superior pole of the thyroid or in the carotid sheath. Ectopic inferior parathyroid glands are not infrequently found in the cervical or upper mediastinal thymus, and in some cases may be intra-thyroidal. If the missing parathyroid gland is still not found, the carotid sheath should be opened from the thoracic inlet to the carotid bifurcation (Figure 3).78,79 The surgeon may also consider drawing PTH levels from the upper and lower internal jugular vein on either side, using the gradient to narrow the location of the missing gland.

Normal and ectopic locations of parathyroid glands.
If all of these maneuvers fail to identify the missing gland, the surgeon should terminate the procedure and consider SPECT-CT, 4D-CT, or selective venous sampling to locate the missing gland. In the case of an ectopic mediastinal or aortopulmonary window parathyroid gland that is not accessible via a cervical approach, video-assisted thoracoscopic surgery (VATS) presents a minimally invasive alternative to the traditional median sternotomy performed for these lesions. VATS parathyroidectomy is associated with shorter hospitalization, less postoperative pain, and fewer complications than median sternotomy and is a safe and effective procedure for mediastinal parathyroid glands. 80
In 2006, Perrier et al. developed a novel nomenclature for parathyroid localization to facilitate communication among multi-disciplinary team members in the care of PHPT by giving them a common language to describe the precise locations of these enlarged glands (Figure 4). 81 Use of this anatomical nomenclature can aid re-operative surgery by giving the surgeon a precise map of locations explored. One limitation of this nomenclature is that it was developed at an institution which routinely uses 4D-CT as the primary imaging modality, a modality that is not widely available. Still, Mazeh et al. demonstrated that this nomenclature is reproducible. 82

Perrier nomenclature for locations of parathyroid adenomas. Locations A, B, and C are associated with superior glands; D, E, and F with inferior glands. A. Posterior to the superior pole. B. Tracheoesophageal groove, including retroesophageal, retropharyngeal, and carotid sheath locations. C. Tracheoesophageal groove, caudal to the inferior pole. D. Midpole, directly over the recurrent laryngeal nerve. E. Anterior to the inferior pole. F. Thyrothymic ligament. G. Intrathyroidal.
Ultimately, the success of parathyroid surgery is correlated with the experience of the surgeon, and parathyroidectomy should be performed by surgeons with appropriate training. Iacobone et al. performed an evidence-based literature analysis of operative volumes and outcomes in parathyroid surgery. 83 Low volume centers had higher rates of vocal cord injury, hypoparathyroidism, and neck hematomas, as well as longer lengths of stay. The authors recommend that more challenging parathyroidectomies should be directed to high-volume surgeons, whom they define as someone performing greater than 40 parathyroid operations annually. 83 In experienced surgical centers, regardless of the imaging modality or operative approach, surgical cure rates are greater than 98% with a low risk of complications. 56
Re-operative surgery
Surgical cure after parathyroidectomy is defined as normalization of calcium and PTH 6 months after surgery. 77 In cases of persistent or recurrent PHPT, re-operative surgery may be necessary. It is imperative to re-establish the diagnosis with elevated calcium, elevated or unsuppressed PTH, and a 24-hour urine CCCR >0.01 to rule out FHH. Review of previous operative records and prior pathology is essential to the understanding of what was and was not accomplished at past exploration, including the number and location of parathyroid glands visualized.3,77 Because risk of complications is higher in a re-operative setting due to scarring, loss of normal tissue planes, and the increased likelihood of ectopic or supernumerary glands, two concordant localization studies should be performed to achieve a focused exploration. Selected venous sampling, in which a catheter is placed through the femoral vein and PTH samples are obtained from the vena cava, brachiocephalic, internal jugular, thymic, thyroid ima, vertebral, inferior, idle, and superior veins, requires a high degree of skill to perform and is typically reserved for re-operative parathyroid exploration when imaging is inconclusive. 3 The goal of re-operative surgery is not only to remove the diseased gland, but to avoid any potential devascularization or destruction of normal parathyroid glands and permanent hypoparathyroidism.
A direct fiberoptic laryngoscopy should be performed prior to re-operation to assess vocal cord mobility.3,77 In addition, an assessment of PHPT-associated morbidity should be ascertained (presence of kidney stones, kidney function, and bone mineral density) to evaluate the risk–benefit ratio for re-operation.
Cryopreservation, which permits parathyroid tissue to be stored for later re-implantation in cases of permanent hypoparathyroidism, should be considered in the re-operative setting. The cryopreserved tissue viability decreases over time, however, and should be used within 6 months if needed. 3
The success of re-operative surgery with an experienced surgeon ranges from 82% to 98%. 77 Patients who are not suitable operative candidates or have a high risk of morbidity with re-operation may be managed medically as detailed below.
Non-operative management
For patients who cannot tolerate or are not good candidates for surgery, monitoring entails annual measurements of serum calcium, creatinine, and GFR, as well as bone mineral density measurements every 1–2 years, and 24-hour biochemical stone profile and renal imaging if kidney stones are suspected. The most important consideration for hypercalcemia is adequate hydration and avoidance of dehydration. Calcium intake does not need to be restricted, and patients should follow national dietary guidelines. Vitamin D should be corrected to levels above 20 ng/mL. 84 The majority of patients with mild asymptomatic PHPT do not need medical therapeutic agents.
Cinacalcet, a calcimimetic that works by activating the extracellular calcium-sensing receptor (CaSR) on parathyroid cells, works by inhibiting PTH secretion, decreasing parathyroid cell proliferation, and increasing calcium excretion by the kidney. It has been approved to treat secondary hyperparathyroidism in ESRD patients on dialysis, hypercalcemia in patients with parathyroid carcinoma, and severe hypercalcemia in patients with PHPT who are not appropriate for surgery. 6 Although it effectively lowers serum calcium, it has no effect on bone mineral density. 13
Bisphosphonates can improve bone mineral density in patients with PHPT, but do not affect serum and urine concentrations of calcium and PTH. 85 Faggiano et al. studied the effects of combination therapy of cinacalcet and alendronate for up to 2 years in patients with sporadic PHPT. 86 There was no synergistic effect of the two drugs on serum or urine calcium or PTH, but the addition of alendronate did improve bone mineral density significantly compared to the cinacalcet-only group. 86 This combination therapy may be an option for patients with hypercalcemia and osteoporosis who are not suitable for operative intervention.
Part III: Parathyroid cancer
The vast majority of parathyroid disease is benign. Although parathyroid cancer also manifests as hyperparathyroidism, it is the cause of PHPT in only 0.1–0.4% of cases. Because it is so rare, our understanding of the natural history of parathyroid cancer is limited. It appears to affect men and women equally and most commonly manifests in the fifth decade of life.
The calcium and PTH elevations associated with parathyroid cancer are higher than those of benign disease, with calcium levels not uncommonly greater than 14 mg/dL and PTH levels at least five times the upper limit of normal. The majority of these patients are symptomatic, with bone and kidney-related symptoms, compared to their counterparts with benign parathyroid disease. In addition, parathyroid glands are not palpable in benign disease, but 40% of patients with parathyroid cancer can present with a palpable neck mass. 87
The diagnosis of parathyroid cancer is similar to that of PHPT, with suspicion for parathyroid cancer increased if the patient’s calcium and PTH levels are extremely high and the patient has a palpable neck mass. Ultrasound can identify an abnormally enlarged parathyroid gland. Fine needle aspiration (FNA) biopsy is not recommended due to the risk of seeding the tumor. Intra-operatively, the surgeon should consider parathyroid cancer in the differential if the gland is grey and firm, appears fibrotic, and demonstrates local invasion. In the latter case, an en bloc resection of the parathyroid gland and adjacent tissue into which it is invading should be performed. Lymph node metastases are seen in 15–30% of patients at initial presentation; any suspicious lymph nodes seen during surgery should be removed, but prophylactic neck dissections have not been shown to improve prognosis. 88 Histologically, parathyroid cancer demonstrates fibrous bands that form trabeculae, capsular or vascular invasion, and the presence of mitotic figures, and diagnosis can be aided by histological biomarkers.77,89
As with benign disease, surgery remains the mainstay of treatment of parathyroid cancer. The local recurrence rate for parathyroid cancer has been estimated at between 36% and 80%, 87 necessitating repeat surgery. The primary cause of mortality and poor quality of life in these patients is from hypercalcemia, and surgery is the best option for treatment. When surgery is not possible, medical management with calcimimetics can help control hypercalcemia, which is the main cause of morbidity and mortality in parathyroid cancer. 90 Chemotherapy and radiation have generally been ineffective in treating parathyroid cancer. Adjuvant external beam radiation is reserved as a palliative option. 77
Conclusion
Tumors of the parathyroid glands are benign in the vast majority of cases. Parathyroid enlargement can lead to excess PTH secretion, which in turn results in hypercalcemia. Over time, untreated PHPT can lead to bone loss and declining renal function. The most effective treatment for PHPT is surgery after confirmation of diagnosis and localization studies. Indications for surgical management, preoperative and intra-operative imaging, and type of operation have evolved considerably over the years, and surgery has excellent results when performed at a specialized center. Re-operative surgery is sometimes necessary, and in those cases, re-establishing the diagnosis, reviewing prior operative and pathology reports, obtaining imaging studies, and assessing the risk to benefit ratio is essential prior to embarking on surgery. In patients who are not operative candidates, numerous therapeutic advancements have been made in the treatment of hypercalcemic crisis. Calcimimetics and bisphosphonates can help treat hypercalcemia and bone loss long term in these patients. Parathyroid cancer is present in fewer than 1% of patients with hypercalcemia. Surgery is the standard of care; unfortunately, recurrence is common, and calcimimetics and external beam radiation can be used for palliation.
