Abstract
Aims:
The study investigates the effectiveness and safety of methotrexate (MTX)
Methods:
This was a cohort study. Patients diagnosed with TAK between 1 January 2013 and 1 January 2019 were enrolled from First Hospital of China Medical University and Zhongshan Hospital of Fudan University. Patients had active disease and were treated with glucocorticoid combined with LEF or MTX. Treatment response, imaging assessment and side-effects were evaluated during 12-month follow-up.
Results:
In total, 68 patients were enrolled (40 cases treated with LEF and 28 treated with MTX). At baseline, age, sex, disease duration and disease activity index showed no significant differences between groups. Prevalence of complete remission (CR) at 6 months was significantly higher in the LEF group than that in the MTX group (LEF
Conclusion:
LEF might provide a quicker treatment response with lower prevalence of disease relapse compared with that elicited in MTX during 12 months follow-up for TAK.
Introduction
Takayasu arteritis (TAK) is a granulomatous inflammatory large-vessel vasculitis that involves primarily the aorta and its main branches. 1 TAK is seen more commonly in young Asian women, with an incidence of 12.9–40 cases per million.2,3 Persistent inflammation and subsequent vascular remodeling leads to stenosis and/or occlusion of the involved vessels which, finally, results in ischemic manifestations and organ dysfunction. 4 Thus, timely and efficacious treatment against inflammation is essential.
The recommended treatments for TAK are usually a glucocorticoid (GC) combined with one or more disease-modifying anti-rheumatic drugs (DMARDs). The combination with DMARDs instead of monotherapy with a GC is to prolong disease remission and help tapering of the GC dose.5 –7 Furthermore, one present study indicated that concomitant conventional DMARDs would have a significant positive effect on biological DMARDs drug retention rate. 8 However, evidence pointing to which DMARDs should be the first-line choice is lacking.
Methotrexate (MTX) is a traditional DMARD used as first-line treatment for rheumatoid arthritis (RA). It is extremely efficacious and shows good tolerance.9 –12 In recent years, MTX has also been used against other autoimmune diseases, such as vasculitis and systemic lupus erythematosus.13 –17 However, for TAK treatment, no randomized clinical trial has been carried out until now. A few case reports or small-sample studies have suggested that low-dose MTX appeared to be an efficacious, safe, and GC-sparing agent in TAK treatment.18 –21 Additional large-cohort and long-term studies are required to confirm the effects of MTX as induction treatment and to assess the durability of disease remission for TAK.
Leflunomide (LEF) is another DMARD that could be considered as alternative treatment for TAK. However, data on the effectiveness and safety of LEF in TAK treatment are also scarce. A small study of 15 patients with TAK showed the favorable effectiveness of LEF in TAK, with a clinical response documented in 80% of cases. 22 A prolonged study with 4-year follow-up showed that 41.6% of patients who had LEF treatment continued to have sustained remission from disease. 23 Our previous studies have also indicated that LEF can lead to quick induction (clinical response in 83.9% of cases at 6 months) and sustained remission (clinical response in 69.6% of patients) from TAK until 12 months. 24 Thus, the underlying effectiveness and good tolerability of LEF against TAK show promise.
Until now, no study has reported on the effectiveness and safety of MTX compared with that of LEF as induction treatment for TAK. Thus, we designed this cohort study to investigate the effectiveness and safety of MTX
Materials and methods
Ethical approval of the study protocol
Procedures were in accordance with the tenets of the Helsinki Declaration 1975 and its later amendments. The study protocol was approved by the Ethics Committees of Zhongshan Hospital, Fudan University (Shanghai, China). Written informed consent was obtained from all patients to use their data.
Patients
This was a cohort research study. Patients diagnosed with TAK according to 1990 American College of Rheumatology classification criteria 25 between 1 January 2013 and 1 January 2019 were enrolled. Enrolled patients came from two centers: First Hospital of China Medical University, and Zhongshan Hospital of Fudan University. Patients enrolled in the present study had to satisfy all of the following criteria: (i) with active disease; (ii) with induction therapy of a GC combined with LEF or MTX; (iii) without other DMARDs or biologic agents used before or together with MTX or LEF; (iv) without an infection or tumor.
Treatment
Enrolled patients were treated with a regimen of a GC and MTX or LEF according to the experts’ experience. GC (prednisone) was initiated (0.8–1.0 mg/kg/day, p.o.) for 4 weeks and tapered gradually to a maintenance dose of 0.1–0.2 mg/kg/day within the next 5 months. MTX was administered at 10–15 mg/week (p.o.). LEF was administered at 20 mg/day (p.o.). The duration of induction treatment was 6 months. If disease remission was not achieved, the induction treatment was prolonged to 9 months. The follow-up duration was 12 months.
Disease assessment and follow-up
National Institutes of Health (NIH, Bethesda, MD, USA) criteria 26 were use as the “gold standard” to assess disease activity. A NIH score ⩾2 was identified as “active disease.” The follow-up frequency was once a month in the active phase and once every 3 months in the remission phase. Symptoms/physical signs, laboratory profiles, and imaging results were documented at each visit.
Treatment response
Treatment response was defined as “complete remission” (CR), “partial remission” (PR) or “treatment resistant” (TR). All the following criteria had to be satisfied for CR to be reported: (i) no new/worsened systemic symptoms; (ii) no new/worsened vascular symptoms or signs; (iii) erythrocyte sedimentation rate (ESR) was normal (⩽40 mm/h); (iv) GC dose ⩽15 mg/day. PR was denoted if item (ii) was satisfied combined with at least one of the other three items. TR was denoted if patients did not achieve CR when treated by GC combined with LEF or MTX for 6 months, even if induction treatment was prolonged to 9 months. “Disease relapse” was defined as: (i) achievement of CR at the end of induction treatment; (ii) recurrence of active disease during the remaining follow-up. The prevalence of side-effects was also calculated.
Imaging assessment
At baseline and every 6 months, whole-body enhanced magnetic resonance angiography (MRA) was done instead of conventional angiography. Angiography findings were classified according to the classification set by Numano and colleagues in 1996. 27 Disease progression according to imaging findings was denoted as “imaging progression.” The latter was defined as new lesions or vascular stenosis and/or progression of wall thickening ⩾20% as confirmed by MRA. Improvement in disease according to imaging was denoted as “imaging improvement,” and was defined as an increase ⩾20% of the lumen of the original lesion according to MRA. Angiograms were read in a blinded manner by two radiologists who were not aware of the treatment regimen. Dispute was resolved by discussion.
Statistical analyses
Data were analyzed using SPSS v22.0 (IBM, Armonk, NY, USA). Continuous variables are presented as the mean ± SD for a normal distribution or median and interquartile range (IQR) for a non-normal distribution. Between-group differences were analyzed using the Student’s
Results
Patient characteristics at baseline
In total 68 patients were enrolled in this study. The median age at the diagnosis was 35 (24–45) years, with median disease duration of 11 (4–56) months. Most of the study cohort were women (56/68, 86.15%). Headache/dizziness (21/68, 30.88%) as well as weakness (17/68, 25.00%) were the manifestations of TAK seen most commonly. Vascular murmur was observed mostly commonly in the left area of upper and lower clavicle (11/68, 16.18%), followed by right-neck area (9/68, 13.24%). Hypertension (17/68, 25.00%) was the most common complication. Type V (31/68, 45.59%) was the most common imaging type, followed by type I (13/68, 19.13%) and type IIb (8/68, 11.76%).
A total of 40 cases were treated with GC plus LEF, whereas the other 28 cases were treated with GC plus MTX. There were no significant differences in age, sex, disease duration, clinical manifestations, or imaging types between the two groups. The disease activity index, which comprised the NIH score, ESR level, and C-reactive protein (CRP) level, showed no significant difference between LEF and MTX groups at baseline. No significant difference in the GC dose at baseline was observed between the two groups. The characteristics of patients at baseline are shown in Table 1.
Characteristics of patients at baseline.

Imaging results: patients were grouped according to the angiography classification of the International TA Conference in Tokyo (1996) based on lesion distribution: type I, branches of the aortic arch; IIa, ascending aorta, aortic arch, and its branches; IIb, ascending aorta, aortic arch, its branches, and thoracic descending aorta; III, thoracic descending aorta, abdominal aorta, and/or renal arteries; IV, abdominal aorta and/or renal arteries; V, combined features of IIb and IV;
CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IQR, interquartile range; LEF, leflunomide; MTX, methotrexate.
Treatment response at 6 months
At the end of 6 months of treatment, a significant improvement in disease activity compared with that at baseline (lower ESR and CRP levels and lower NIH scores) was observed (

Changes of disease activity index including ESR and CRP levels and NIH score, as well as daily GC dose in different treatment groups during the 12 months follow-up.
The overall prevalence of CR and PR at 6 months was 64.71% and 16.18%, respectively. In the LEF group, the prevalence of CR and PR was 72.50% and 12.50%, whereas the prevalence of CR and PR was 53.57% and 21.43% in the MTX group, respectively. A higher prevalence of CR was demonstrated in the LEF group compared with that in the MTX group (
Relative risks (95% confidence interval) of CR, PR, TR and disease relapse during follow-up.
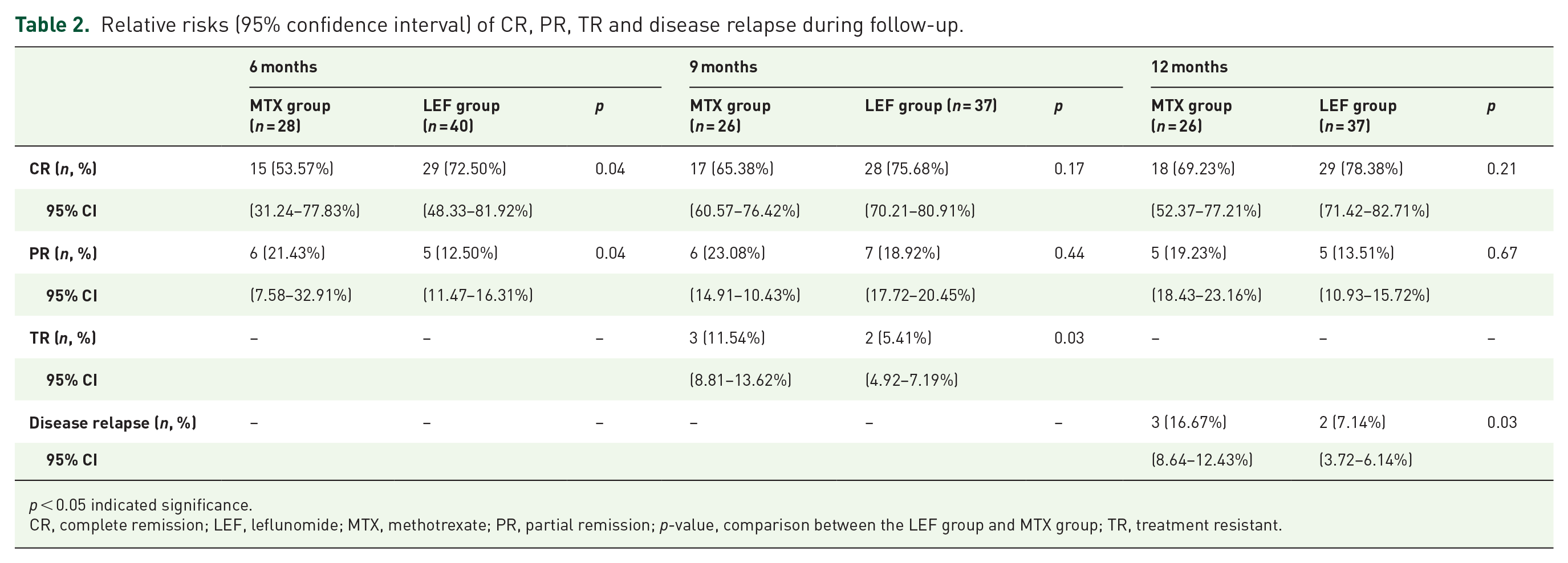
CR, complete remission; LEF, leflunomide; MTX, methotrexate; PR, partial remission;
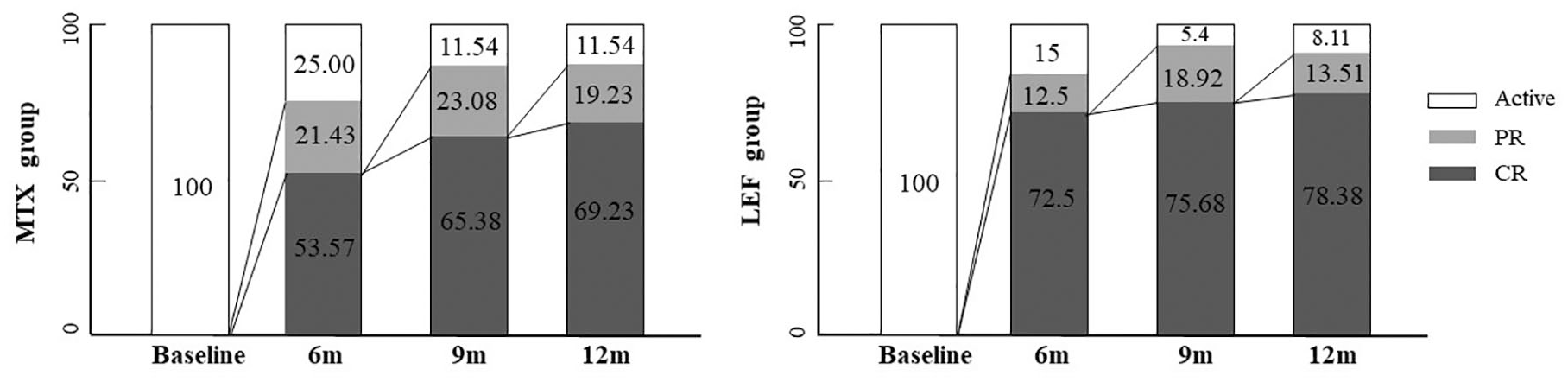
Treatment response in different treatment groups during the 12 months follow-up.
Treatment response at 9 months
Three patients discontinued LEF treatment after 6 months: one case switched to GC treatment alone due to pregnancy preparation; one patient turned to MTX treatment due to intolerance to alopecia caused by LEF; one case was lost to follow-up. Two patients discontinued MTX treatment after 6 months: one case switched to azathioprine after disease remission; one patient turned to cyclophosphamide due to persistent active disease.
For patients treated with LEF, at 9 months, the disease activity index was similar to that at 6 months. The daily dose of GC was 12 (10–15) mg, which was also similar to that at 6 months. In the MTX group, the daily dose of GC was 15 (10–15) mg, and the disease activity index was also similar to that at 6 months (Figure 1).
The overall prevalence for CR, PR and TR at 9 months was 71.42%, 20.63% and 7.94%, respectively. In the LEF group, the prevalence of CR and PR was 75.68% and 18.92%, respectively, whereas the TR prevalence was 5.41%. In the MTX group, the prevalence of CR, PR and TR rates was 65.38%, 23.08% and 11.54%, respectively. No significant difference in the prevalence of CR or PR was demonstrated between the LEF and MTX groups at 9 months. The TR prevalence was significantly higher in the MTX group compared with that in the LEF group (
Treatment response at 12 months
In the LEF group, the disease activity index observed at 12 months was similar to that seen at 9 months. The daily dose of GC was 10.00 (8.00–10.00) mg at 12 months, which was significantly lower than that at 9 months (
The overall prevalence of CR and PR at 12 months was 74.60% and 15.87%, respectively. In the LEF group, the prevalence of CR and PR was 78.38% and 13.51%, respectively. In the MTX group, the prevalence of CR and PR was 69.23% and 19.23%, respectively. No significant difference in the prevalence of CR or PR was demonstrated between the LEF and MTX groups at 12 months. (Table 2 and Figure 2).
Disease relapse was demonstrated in five patients during the follow-up, with medium relapse time 9 (9–11) months. Two (7.14%) in the LEF group, and three (16.67%) in the MTX group suffered from disease relapse. For cases with disease relapse in the LEF group, one switched to tofacitinib treatment and one turned to tocilizumab treatment. For cases in the MTX group, one switched to cyclophosphamide treatment, one turned to tofacitinib treatment, and one switched to tocilizumab treatment. The prevalence of disease relapse was significantly higher in the MTX group than that in the LEF group (
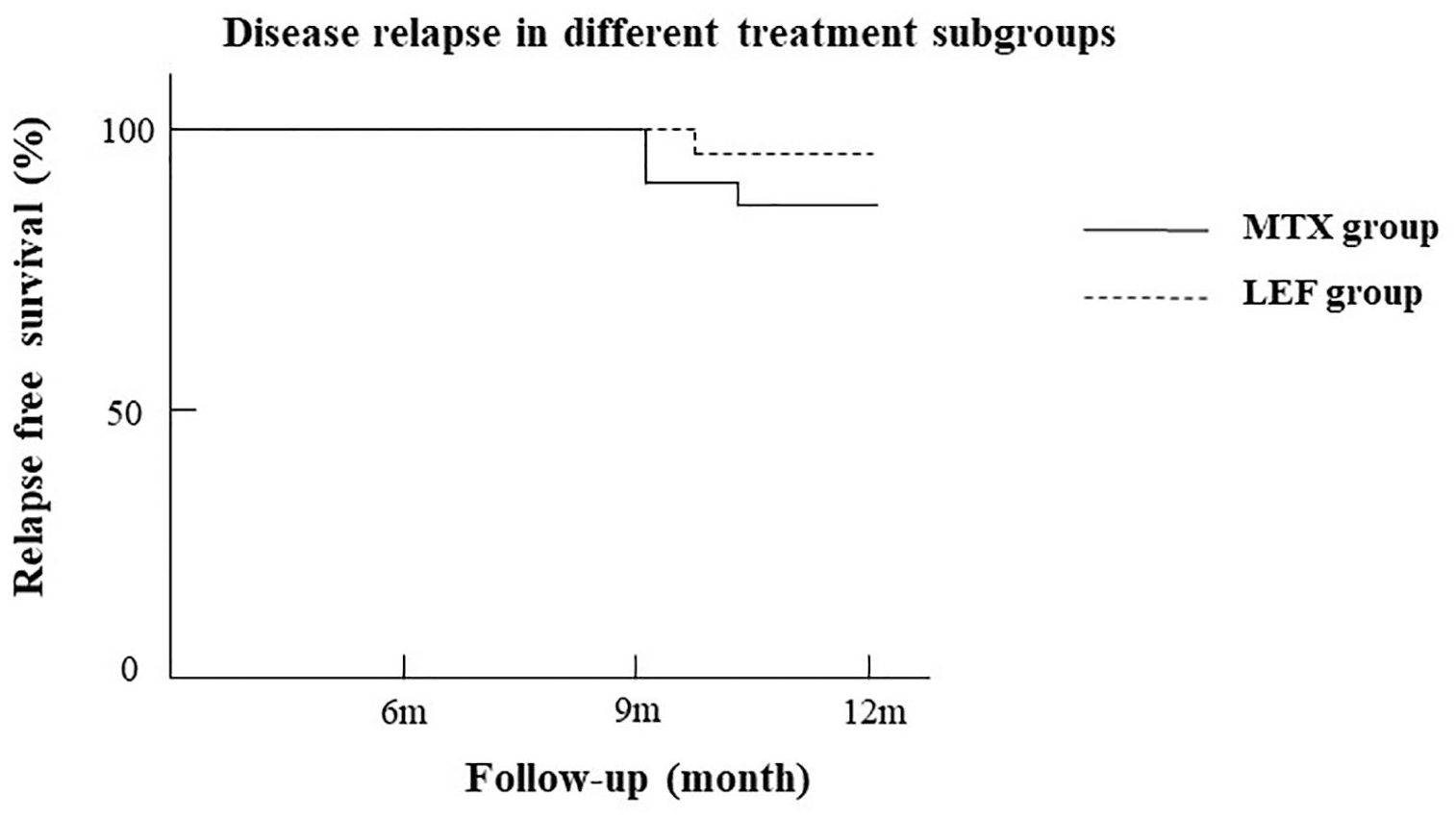
Disease relapse in different treatment groups during the 12 months follow-up.
Potential risk factors for treatment resistance and disease relapse
Multivariate logistic regression analysis indicated that a high baseline CRP levels (⩾15 mg/L) was associated significantly with TR [odds ratio (OR) = 1.36, 95% CI 1.07–13.41,
Furthermore, Cox proportional hazards regression analysis revealed the independent predictors of disease relapse to be a high baseline CRP level (HR = 2.51, 95% CI 1.36–12.98,
Imaging assessment
At 6 months, two cases (5.00%, both with worsening lesions) in the LEF group and two patients (7.14%, one with new lesion and one with worsening lesion) in the MTX group demonstrated imaging progression. Stable imaging results were observed in the other patients. At 12 months, imaging progression was observed in four (10.81%) cases in the LEF group and three (11.54%) patients in the MTX group, including five worsening lesions (three in LEF group and two in MTX group) and two new lesions (one in LEF group and one in MTX group). Imaging improvements were not observed at 6 months or 12 months. There was no significant difference in the prevalence of imaging progression between the two groups at 6 months or 12 months.
Safety
We found that nine (22.50%) patients in the LEF group suffered side-effects of treatment. The most common side-effect was infection: there were four cases of infection (three pulmonary infections and one urinary infection). Hair loss was found in two cases, and one patient quit LEF treatment after 6 months and switched to MTX treatment. Mild liver dysfunction (liver enzymes <3 times the normal level) was observed in two cases, which decreased to normal levels after treatment with hepatic protectors (polyunsaturated phosphatidylcholine in one case and glycyrrhizin in one case). A menstrual disorder was seen in one patient after 6 months of treatment.
In the MTX group, six (21.43%) patients suffered side-effects. The most commonly seen side-effect was also infection (two pulmonary infections and one urinary infection). Hair loss was observed in two cases, which could be tolerated. Intolerable weakness was demonstrated in one case after 6-month treatment of MTX, which resulted in switching to azathioprine. Two cases had mild liver dysfunction (liver enzymes <3 times the normal level) during follow-up, and achieved improvement after liver-protection treatment.
Discussion
TAK is a rare disease characterized by large-vessel vasculitis seen more commonly in Asian people. Guidelines for TAK treatment are lacking. Thus, the therapeutic strategy is decided mainly by professional experience. We undertook a cohort study to identify the effectiveness of LEF
We found that: (i) LEF provided a quicker treatment response compared with MTX (higher CR prevalence at 6 months); (ii) treatment resistance and disease relapse were more commonly observed in patients treated with MTX; (iii) tolerability was acceptable both in LEF and MTX groups; (iv) patients with high baseline CRP level carried a higher risk of treatment resistance and disease relapse.
MTX is an inhibitor of dihydrofolate reductase that was applied first to cancer treatment (e.g. leukemia, lymphoma).28 –30 With the development of pharmacology, MTX began to be used gradually for the treatment of autoimmune diseases, especially for RA.9 –17 The effects of MTX in the treatment for large-vessel vasculitis are controversial. For TAK treatment, randomized clinical trials have not been carried out until now. Only a few case reports or small-sample, single-center studies indicated that low-dose MTX appeared to be an efficacious, safe, and GC-sparing agent for TAK treatment.18 –21 In the present study, we found that MTX showed relatively good effectiveness for TAK treatment with a CR prevalence of 53.57% at 6 months, which could be maintained to 65.38% and 69.23% at 9 and 12 months, respectively. The doses of MTX used in our study (as well as in Asians) were relatively lower than those used in the Caucasian population (mainly 20–25 mg/week). It has been reported that the dose of 10–15 mg/week for MTX was effective and safe for TAK in previous studies.31,32 In the meanwhile, to avoid the adverse events, especially abnormal liver functions and myelosuppression, the doses of MTX we used were low. However, whether the lower dose of MTX could have significantly affected the efficacy of the drug itself still needs to be confirmed in further randomized controlled trials.
In recent years, LEF has become a promising immunosuppressive agent for the treatment of large-vessel vasculitis. 33 In one former study, we showed that LEF could improve the inflammatory status and reduce the GC dose after 6 months of treatment, and that this improvement could be sustained until 12 months. 24 In the present research, the efficacy of LEF treatment was also satisfactory. The prevalence of CR after using LEF was 72.50% and 75.58% at 6 months and 9 months, respectively, which could be sustained in 78.38% of cases at 12 months. These data are in accordance with a study from South America.22,23 What is more, though the CR prevalence showed no significant differences between LEF and MTX at 9 and 12 months, LEF achieved a higher CR prevalence at 6 months compared with MTX, which indicated that LEF could induce disease remission more quickly. In addition, patients treated with LEF showed a lower prevalence of TR and disease relapse compared with MTX, which may also support the more promising role of LEF in TAK treatment.
Treatment resistance and disease relapse was the biggest concern in clinical practice. Previously, Comarmond and colleagues reported that CRP ⩾7 mg/L was an independent risk factor for disease relapse in TAK. 34 Accordingly, in the current research, we found that patients with high baseline CRP level carried a higher risk of treatment resistance and disease relapse, which indicated that cases with high inflammatory condition might show inadequate treatment response to traditional DMARDs. Thus, for cases with refractory disease and/or high inflammatory condition, more aggressive treatment and more closely follow-up was needed to improve the treatment response.
In the present study, the side-effects of treatment with LEF or MTX (infection, mild liver dysfunction, menstrual disorder) were well tolerated except for one case of intolerable hair loss in the LEF group and one case of intolerable weakness in the MTX group. Reproductive toxicity is always a concern during TAK treatment because most patients are women of childbearing age. Even though our study revealed relatively good tolerability and few effects on reproduction, LEF and MTX should be discontinued before a planned pregnancy according to the recommendation for RA treatment.35,36
Our study had two main limitations. First, there may have been a bias in patient enrollment. Only initial-onset cases were enrolled in the current study, thus the effectiveness of LEF and MTX for refractory cases needs to be further investigated. Second, the follow-up time was relatively short: a longer-term study will be carried out in the future.
Conclusion
This was a multicenter cohort study to evaluate the efficacy of LEF
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (NSFC 81771730, 81901639, 81801598) and Clinical Cultivation Project of Shanghai Shenkang Science and Technology Development Center (SHDC12019X05).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
