Abstract
Anastrozole is a selective non-steroidal aromatase inhibitor that blocks the conversion of androgens to estrogens in peripheral tissues. It is used as adjuvant therapy for early-stage hormone-sensitive breast cancer in postmenopausal women. Significant side effects of anastrozole include osteoporosis and increased levels of cholesterol. To date, seven case reports on anastrozole hepatotoxicity have been published. We report the case of an 81-year-old woman with a history of breast cancer, arterial hypertension, type 2 diabetes mellitus, hyperlipidemia, and chronic renal insufficiency. Four days after switching hormone therapy from tamoxifen to anastrozole, icterus developed along with a significant increase in liver enzymes (measured in the blood). The patient was admitted to hospital, where a differential diagnosis of jaundice was made and anastrozole was withdrawn. Subsequently, hepatic functions quickly normalized. The observed liver injury was attributed to anastrozole since other possible causes of jaundice were excluded. However, concomitant pharmacotherapy could have contributed to the development of jaundice and hepatotoxicity, after switching from tamoxifen to anastrozole since several the patient’s medications were capable of inhibiting hepatobiliary transport of bilirubin, bile acids, and metabolized drugs through inhibition of ATP-binding cassette proteins. Telmisartan, tamoxifen, and metformin all block bile salt efflux pumps. The efflux function of multidrug resistance protein 2 is known to be reduced by telmisartan and tamoxifen and breast cancer resistance protein is known to be inhibited by telmisartan and amlodipine. Moreover, the activity of P-glycoprotein transporters are known to be decreased by telmisartan, amlodipine, gliquidone, as well as the previously administered tamoxifen. Finally, the role of genetic polymorphisms of cytochrome P450 enzymes and/or drug transporters cannot be ruled out since the patient was not tested for polymorphisms.
Introduction
Tamoxifen is a mixed estrogenic antagonist/partial agonist that is used in breast cancer to block the estrogenic stimulation of tumor cell growth. 1 Anastrozole is a selective third-generation aromatase inhibitor.2,3 Anastrozole competitively inhibits the enzyme cytochrome P450 (CYP) 19A1, which converts androgens, produced in the adrenal glands, to estrogens. 4 Therefore, aromatase inhibition leads to a decrease in the concentration of estrogens in serum and breast cancer tissue, which slows tumor growth. Anastrozole is indicated for adjuvant treatment of advanced breast cancer, in postmenopausal women, following tamoxifen therapy.4,5 Unlike tamoxifen, aromatase inhibitors do not increase the risk of thromboembolic complications.2,5 Both tamoxifen and anastrozole are extensively metabolized by several CYP enzymes. 6 After enterohepatic circulation, metabolites of tamoxifen appear in the stool. The major excretory pathway for anastrozole is the liver and biliary tract.3,6 Drug-induced liver injury (DILI) is a serious medical condition that can be induced by various medicinal products. It can be caused by anti-infective drugs such as amoxicillin/clavulanate, isoniazid, and certain macrolide antibiotics. It is also caused by anti-seizure drugs (carbamazepine, valproate), immune modulators (interferon-alpha, methotrexate), non-steroidal anti-inflammatory drugs (diclofenac and nimesulide), antidepressants (agomelatine), and certain tyrosine-kinase inhibitors.7–11 Three major types of DILI have been described: hepatocellular, cholestatic, and mixed liver injury due to a major underlying mechanism.12,13 The fourth type of hepatotoxicity, that is, indirect liver injury, is not a completely accepted category of DILI. 14 In the United States, pharmaceuticals were linked to about 20% of the jaundice cases seen in the elderly in the year 2000.15,16
Case report
We report the case of an 81-year-old woman who presented with a history of breast carcinoma pT1a pN0 M0, G1, ER 100%, PR 80%, HER2 negative, KI-67 10–15%. In November 2015, she underwent breast resection followed by radiotherapy and was treated with tamoxifen thereafter. The patient had regular follow-ups without signs of disease reoccurrence. In addition, she was treated for arterial hypertension, type 2 diabetes mellitus, chronic renal insufficiency, hyperlipidemia, and non-alcoholic fatty liver disease. The patient was admitted to the hospital in September 2016 for painless icterus that had started 2 days prior. She was afebrile, weak, tired, and dehydrated at the time of admission. She described nausea and abdominal discomfort in the epigastrium; palpation found no pain or tenderness. Serum analysis found hyperbilirubinemia and elevated levels of ALT, AST, ALP, and GGT (Table 1). C-reactive protein was mildly elevated but, without leukocytosis, there was a progression of her chronic renal insufficiency as well as asymptomatic bacteriuria.
The patient’s laboratory results (2016).

ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transferase.
Abnormal values are shown in bold.
The patient’s medical history showed that the patient’s oncologist had changed her hormone therapy 4 days prior to symptom onset. The oncologist switched tamoxifen, which the patient had taken for more than 1 year, to anastrozole. Other medications, which the patient had been taking for several years, remained unchanged. At the time of her visit to her oncologist, the patient was symptom-free, had no reported problems, and none of the examinations, including a computer tomography scan, indicated a relapse of her oncologic disease. During hospitalization, a differential diagnosis of the icterus was made, mainly because elevated conjugated bilirubin; the blood count was normal (without atypical blood cells or blast cells), a diagnosis of hemolytic anemia was excluded. Due to the rapid onset of the disease and well-documented normal liver function only a few weeks before neither autoimmune hepatitis nor primary biliary sclerosis were considered as possible underlying causes of her jaundice. An abdominal ultrasound was performed to exclude a bile duct obstruction and liver metastases; the examination found liver steatosis and minor cholecystolithiasis [without dilatation of the common bile duct (ductus hepatocholedochus) or the bile ducts]. Both disorders had been described in the past. In addition, neither the inferior vena cava nor the hepatic veins were dilated, and there were no signs of blood congestion found in the venous system of the liver. Coagulation parameters were also completely normal; the patient had not traveled outside the Czech Republic during the previous 5 years, there was no history of risky contacts, and no fever or abdominal pain before admission to the hospital. Infectious hepatitis A, B, and C were excluded serologically. For the previously mentioned reasons, DILI was considered as a primary possible cause of her icterus and abnormal liver function tests. During hospitalization, the patient received intravenous hydration and was put on bed rest. After consulting her oncologist, anastrozole therapy was withdrawn and her statin and metformin medications were temporarily suspended. After 5 days, there was a significant decrease in her bilirubin and liver enzyme levels and the patient was discharged home. At the 1 month post-discharge outpatient follow up, hepatic parameters had already returned to her normal long-term baseline. The time course of the patient’s bilirubin concentrations and levels of liver transaminases, alkaline phosphatase, and gamma glutamyl transferase are shown in Figure 1. The time course of the patient’s creatinine and urea concentrations are shown in Figure 2.
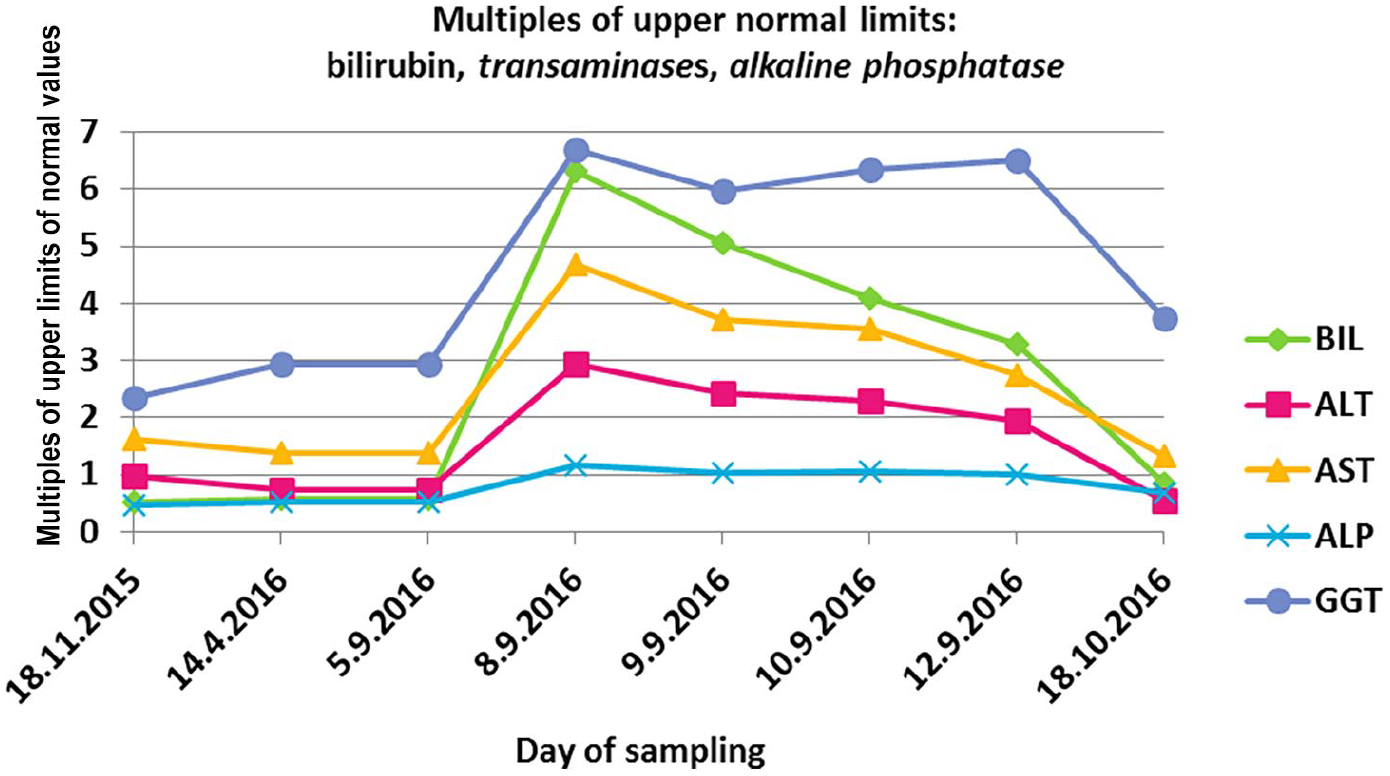
Axis Y: concentrations of bilirubin and enzymes ALT, AST, ALP, and GGT are plotted in multiples of the upper limits of the normal values. Axis X: time – dates of measurement in 2015 and 2016. Medicinal product Anaprex® containing anastrozole was started on 5 September 2016 and withdrawn on 8 September 2016.

Axis Y: concentrations of creatinine and urea in serum are plotted in multiples of the upper limits of the normal values. Axis X: time – dates of measurement of creatinine and urea concentrations in 2015 and 2016. Medicinal product Anaprex® containing anastrozole was started on 5 September 2016 and withdrawn on 8 September 2016.
Discussion
Clinical point of view
DILI
It can be difficult to accurately identify the drug causing a serious adverse drug reaction when the patient is taking multiple medications. Proving that an episode of liver injury is caused by a specific drug can be challenging because other diseases of the liver and biliary system can produce a similar clinical picture. Thus differential diagnosis of DILI requires several separate supportive assessment variables that collectively lead to a high level of certainty, including a temporal association with the time of onset, time to resolution, biochemical findings, phenotype of the hepatic injury and extrahepatic features, and the likelihood that the suspect agent is to blame based on its drug safety record.
17
The first case of anastrozole-induced hepatitis was reported in 2006.
18
To date, six more case reports have entered the literature, bringing the total number of case reports describing anastrozole hepatotoxicity to seven.19–24 If we consider how many women use anastrozole around the world, this complication is very rare. That said, anastrozole is known to be associated with the development of fatty liver disease.
25
A Chinese randomized study by Lin
In 2014 Chalasani
Host risk factors
Older age is also a risk factor for DILI since aging decreases cytochrome mediated hepatic metabolism and older patients more often present with a cholestatic pattern of liver injury compared with younger individuals.12,13,30 Patient gender may also influence the risk of DILI; for example, the immune-mediated model of DILI found more severe hepatitis in females.12,29,30 Our patient was an 81-year-old woman with advanced chronic kidney disease (CKD G4), which could have contributed to the hepatotoxic side effects by reducing renal clearance of the various medication she was taking, resulting in higher plasma levels. As such, our patient had several risk factors that enhanced susceptibility to DILI before taking her first dose of anastrozole. In our case, a liver biopsy was not performed because of the quick resolution of jaundice and the rapid improvement in her liver function tests after anastrozole was withdrawn.
Pharmacological point of view
According to the Arimidex® summary of product characteristics, changes in liver function tests with or without jaundice occurs in less than 1 in 10,000 patients. Renal elimination is not a significant route of elimination for anastrozole; thus, anastrozole clearance remains unchanged even with renal impairment. The pharmacokinetic parameters of anastrozole can be affected by drug interactions
Selected biotransformation data for drugs taken by the patient.

CYP enzymes, biotransformation enzymes belonging to the cytochrome P450 family; N/A, not applicable; SPC, summary of product characteristics; UGT, uridine diphosphate glucuronosyltransferase. Drugs highlighted in bold (anastrozole, meformin, rosuvastatin, tamoxifen) were not prescribed during hospitalization. The other drugs (not highlighted in bold) were taken before admission to the hospital and also during the hospital care.
Mechanisms of drug-induced cholestasis
A variety of medications can influence the function of transport proteins in hepatocytes, which can lead to drug-induced cholestasis. Evidence supports the hypothesis that drug-induced functional disorders in hepatic bile acid transporters can lead to intracellular accumulation of bile acids, resulting in cholestatic hepatocyte damage. Bile acids are mainly taken up by the sodium taurocholate co-transporting polypeptide transporter and excreted into the bile by the canalicular efflux transporter bile salt export pump (BSEP). Bilirubin is taken up by 1B1, an organic anion transporter. After bilirubin conjugation, bilirubin glucuronide is excreted into the bile by multidrug resistance protein 2 (MRP2) and transported into the blood by MRP3. Cholestasis or hyperbilirubinemia can be caused by the inhibition of these transporters by certain drugs.
40
However, not all drugs that inhibit BSEP cause cholestasis. This might be due to compensatory mechanisms of bile acid transport by the basolateral efflux transporters MRP3 and MRP4, which, under normal conditions, play a minor role, but can be up-regulated during cholestasis. Thus the impaired function of these transporters, by drugs or genetic predisposition, may result in cholestasis when there is also BSEP inhibition.
16
In addition, although BSEP is not directly involved in drug metabolism, its inhibition can lead to the development of harmful side effects.
40
The oncologic patient in our case report was taking several drugs that had the potential to affect the function of membrane transporters significantly involved with drug pharmacokinetics or involved in the metabolism of bilirubin or bile salts. Telmisartan, which the patient was taking for hypertension, differs from other angiotensin receptor blockers in that it has a strong potential to inhibit several ABC-transporters that are important in the pharmacokinetics: MDR1, (that is P-glycoprotein), MRP2, and BCRP.
36
MRP2 is responsible for the active transport of conjugated bilirubin into the bile and is also considered to be the primary transporter that effluxes many drug conjugates across the canalicular membrane of hepatocytes. The MRP2 export pump is also important in excreting drug metabolites and endogenous compounds, such as bilirubin, into the urine.
33
Thus inhibition of MRP2 by telmisartan could contribute to the elevation of bilirubin. In addition, telmisartan has also been identified as a BSEP inhibitor.
34
By blocking BSEP function, telmisartan could contribute to the accumulation of toxic bile acids inside hepatocytes. In one clinical study, telmisartan, by inhibiting rosuvastatin efflux and mediated by ATP binding cassette transporter G2, was shown to significantly increase systemic exposure to rosuvastatin after single and multiple doses.
41
Concomitant use of telmisartan (40 mg daily) and rosuvastatin (10 mg daily) increased the maximum plasma concentration of rosuvastatin by 76% in healthy Chinese volunteers.
41
At the time of hospital admission, the patient was taking 80 mg of telmisartan daily and 20 mg of rosuvastatin daily; rosuvastatin was suspended immediately and the dose of telmisartan was reduced to 40 mg daily on the following day. Therefore, telmisartan could have increased systemic exposure to rosuvastatin in our patient before hospital admission; however, our patient was white [ native Czech ] and the study documenting an interaction between telmisartan and rosuvastatin was conducted in Chinese volunteers. In general, statins can increase the risk of hepatic dysfunction including mild elevations of aminotransferases during therapy.42,43 Acute DILI related to rosuvastatin monotherapy is rare.43,44 In our patient, re-introduction of rosuvastatin after resolution of the DILI did not result in an increase in aminotransferases or bilirubin, thus an interaction between telmisartan and rosuvastatin was probably not associated with the hepatotoxicity and cholestasis seen after starting anastrozole. Newer angiotensin II receptor blockers such as olmesartan, telmisartan, and eprosartan have not been linked to cases of hepatotoxicity.
45
Even so, there was one interesting case in 2014 that reported a drug–drug interaction between telmisartan and fluvastatin. The patient had a single nucleotide polymorphism that resulted in the decreased function of the MRP2 biliary transporter. Consequently, creatine kinase levels were elevated after combination therapy with telmisartan and fluvastatin. Elevation of transaminase enzymes or bilirubin was not present.
46
Like rosuvastatin, tamoxifen is also a BSEP substrate. Tamoxifen inhibits both BSEP and MRP2 – two efflux transporters that move bile into the bile ducts. In addition, tamoxifen also inhibits MRP3 and MRP4, which are basolateral bile acid transporters, the inhibition of which is a known risk factor for the development of cholestasis.16,35,47 Tamoxifen has a very long half-life (4–11 days) and a significantly longer terminal
Effect of drugs on the transporters involved in pharmacokinetic processes or the handling of bilirubin and bile salts.

BCRP/ABCG2, breast cancer resistant protein/ATP-binding cassette subfamily G2; BSEP/ABCB-11, bile salt export pump/ATP-binding cassette protein subfamily B-11; MDR-1, multidrug resistance protein-1 or P-glycoprotein is an expression for ABCB1; MRP-2/ABCC2, multidrug resistance-associated protein-2/ATP-binding cassette protein subfamily C 2; MRP-3/ABCC3, multidrug resistance-associated protein-3/ATP-binding cassette protein subfamily C-3; MRP-4/ABCC4, multidrug resistance-associated protein-4/ATP-binding cassette protein subfamily C-4; NTCP, sodium taurocholate co-transporter polypeptide that is, solute carrier family 10 transporter A1; OATP1B1, organic anion transport protein 1B1 that is, SLCO 1B1, solute carrier organic anion transporter family member 1B1; OCT1, organic cation transporter, that is, SLC22A1, solute carrier family 22 transporter A1; OCTN2, organic cation/carnitine transporter.
“?” means that information regarding the effect of the drug on the transporter was not found in Drugbank database or in other literature sources specified in the right-hand column of Table 3.
Note: Drugs highlighted in bold (anastrozole, metformin, rosuvastatin and previous tamoxifen) were not prescribed in the hospital. The other drugs (not highlighted in bold) were taken before admission to the hospital and also during the hospital care.
Besides the potential contribution of impaired bile acid homeostasis, mechanisms such as the immune-mediated hypersensitivity reaction, dose, and lipophilicity of the drug itself, as well as combinations of these factors together with individual risk factors, seem to be important in the onset of acute DILI. 56 In addition, drug–drug interactions can alter a drug’s toxicity profile, which could potentially lead to hepatotoxicity. However, causality assessment in DILI cases can be challenging and the “last prescribed drug” cannot be assumed to be the only responsible agent. 49 Potential DDIs are especially important in the treatment of older patients who usually have multiple chronic conditions requiring concomitant therapies. 56 Physicians should consider the need for inter-professional co-working that would include consultancy with a clinical pharmacologist or pharmacist when investigating potential drug–drug interactions at the level of membrane drug transporters. Computer programs used to analyze drug safety may not be completely up-to-date and often not precise enough to give meaningful information on the impact of inhibitory potencies in the interactive interplay between cytochrome P450 enzymes and transporters.
Conclusion
DILI in our patient was most probably an idiosyncratic response to anastrozole since all other common causes of jaundice were excluded. This opinion is substantiated by the rapid improvement of clinical and biochemical findings after ceasing anastrozole therapy. The interplay between the patient’s individual risk factors and the properties of the drugs she had been prescribed prior to anastrozole was potentially an indirect cause of DILI. The combination of her prescribed treatments, as well as the residual tamoxifen in her system, could have contributed to the rapid development of jaundice and hepatotoxicity after being switched to anastrozole. Our research showed that several of the drugs she was taking were able to inhibit the hepatobiliary transport of drugs and drug metabolites as well as affect bilirubin homeostasis. Telmisartan and tamoxifen both block bile salt efflux pumps and MRP2 efflux transporters, which are significantly involved in the transport of bile acids and bilirubin, respectively, from hepatocytes into bile canaliculi. Breast cancer resistance protein is also inhibited by telmisartan and amlodipine. Anastrozole is a substrate for P-glycoprotein and as such, the efflux function of this transporter can be decreased by telmisartan, amlodipine, gliquidone or the previously prescribed tamoxifen. Finally, the role of genetic polymorphisms of the CYP enzymes, which determine the level of drug biotransformations, or the presence of gene polymorphisms that decrease the activity of membrane drug transporters cannot be ruled out since these genetic polymorphisms were not tested for in our patient.
Footnotes
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval statement
Ethics committee approval is not required for case reports that do not constitute research at University Hospital of the 3rd Medical Faculty, Charles University. Upon admission to hospital, the patient signed an informed consent for care at the university hospital, which includes consent to anonymous publishing of non-personal data. The anonymity of the patient was completely preserved.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the research project Progress Q35 and Progress Q28 of the Charles University, Prague.
Informed consent
Written informed consent for patient information to be published was provided by the patient.
