Abstract
Objective
To evaluate the efficacy and safety of bicyclol in patients with drug-induced liver injury (DILI) using a nationwide database.
Methods
We retrospectively analyzed the clinical data of DILI patients in the DILI-R database. Propensity score matching was performed to balance the bicyclol and control groups, and alanine aminotransferase (ALT) recovery was compared between the two groups. Factors associated with ALT recovery and safety were identified.
Results
The analysis included the data of 25,927 patients. Eighty-seven cases were included in the bicyclol group, with 932 cases in the control group. One-to-one propensity score matching created 86 matched pairs. The ALT normalization rate in the bicyclol group was significantly higher than that in the control group (50.00% vs. 24.42%), and statistical significance was found in the superiority test. After adjustment of baseline ALT levels, baseline total bilirubin levels, sex, age, acute or chronic liver diseases, and suspected drugs in the multivariate logic regression analysis, the major influencing factors for ALT recovery included the time interval between ALT tests (days) and the group factor (bicyclol treatment). There were no differences in the proportion of renal function impairment or blood abnormalities between the two groups.
Conclusions
Bicyclol is a potential candidate for DILI.
Keywords
Introduction
Drug-induced liver injury (DILI) is a major etiology of liver damage. As an increasing number of drugs enter the market, those that are either expectedly or unexpectedly metabolized into toxic agents in the human body are becoming more common. These toxic agents may induce extensive oxidative stress, inflammation, and apoptosis of hepatocytes, leading to liver failure and even death.1–3 Although the true incidence of DILI may be underestimated owing to diagnostic difficulties and underreporting, studies estimate that the incidence is between 14 and 19 cases per 100,000 people receiving prescription medication.4,5 Furthermore, DILI is the most common indication for urgent liver transplantation in the United States and Europe.6,7 Complementary and alternative medicines (CAMs) are becoming an increasingly important contributing factor to DILI, especially in Asian countries. 8
The mainstay treatment of DILI includes discontinuation of the offending drug(s) and intensive supportive care. Many agents, including N-acetylcysteine (NAC), silymarin, antioxidants, S-adenosine methionine, ursodeoxycholic acid, or a combination of these, have been used in patients with DILI and other forms of liver toxicity.9,10 N-acetylcysteine has been extensively evaluated in paracetamol-induced acute liver failure (ALF), and its potential use has been suggested in non-paracetamol drug-induced ALF. Currently, NAC is the only recommended drug to treat adults with drug-induced, early-stage acute ALF. 3 Nevertheless, a large proportion of patients do not respond to these therapies.
Bicyclol, as a liver protector, is approved by the Chinese Food and Drug Administration for patients with abnormal ALT levels caused by various liver diseases. Previous research suggests that bicyclol can not only decrease serum ALT levels but also promote liver tissue repair. 11 The underlying mechanisms of its protective effects include scavenging reactive oxygen species, inhibiting lipid peroxidation, protecting hepatic cell membranes and mitochondrial function, and inhibiting inflammatory cytokines.12–15
The efficacy and safety of bicyclol in the treatment of DILI have not yet been sufficiently investigated. One randomized controlled trial (RCT) suggested that bicyclol prevented the occurrence of anti-tuberculosis DILI in patients with underlying liver disease. 16 Additionally, Naiqiong et al. 17 reported that bicyclol was effective for statin-induced liver injury.
Although RCTs are considered the gold standard by which novel medications and interventions are evaluated, they only determine whether a given medication or intervention is effective in a specific patient population. For practicing clinicians, it is more important to know whether it will work for general patients in their particular setting. This inherent limitation can only be resolved by real-world studies. 18 Therefore, the present study uses a nationwide inpatient database (DILI-R) 19 to evaluate the efficacy and safety of oral bicyclol in patients with DILI.
Materials and methods
Data source
This retrospective study used the DILI-R database, which includes the data of 25,927 confirmed DILI cases in 308 hospitals in mainland China. 19 The database includes the following data: age, sex, history of liver disease, suspected drugs, and diagnosis. The diagnosis was further confirmed using the Roussel Uclaf Causality Assessment Method or by expert opinion (consistent with the expert opinion method of causality assessment). 20 The inclusion criteria did not include specific cutoff levels for liver chemistries. The protocol for the DILI-R study was reviewed and approved by the institutional review board at Renji Hospital of Shanghai Jiao Tong University, Shanghai, China (ClinicalTrials.gov Identifier: NCT02407964). There was no requirement for informed consent because of the retrospective study design.
Group assignment and propensity score matching
DILI patients who underwent two ALT tests with their first test showing an ALT level > the upper limit of normal (ULN) were included. Those who received only bicyclol (Beijing Union Pharmaceutical Factory, Beijing, China) for treatment after the first ALT test were defined as the bicyclol group. Those who received only supportive treatment were defined as the control group. All patients were evaluated twice for creatinine, blood urea nitrogen, hemoglobin, leukocytes, and platelets, and only those with normal results in the first test were included.
Given the differences in the baseline characteristics between patients in the two groups, propensity score matching (PSM) was used to identify a cohort of patients with similar baseline characteristics. PSM was performed as previously described. 21
Covariates included age, sex, the time interval between ALT tests, the basal ALT levels, the basal total bilirubin (TBIL) levels, history of liver disease, acute or chronic liver injury, and suspected drug categories. With intervention measures as dependent variables and the covariates as the independent variables, we assessed the propensity score using logistic regression. Matching was performed using a 1:1 matching protocol without replacement (greedy-matching algorithm), with a caliper width equal to 0.2 of the standard deviation of the logit of the propensity score. Finally, the intervention effect was evaluated between the two balanced groups.
Study endpoints22,23
Patients in the bicyclol group were evaluated for ALT before and after treatment. Patients in the control group were also assessed for ALT twice.
The primary efficacy endpoint was the ALT normalization rate, and the secondary efficacy endpoint was the ALT50% reduction rate. ALT50% reduction refers to the rate at which serum ALT decreased to half of the initial serum ALT concentration. In addition, all potential influencing factors of ALT were analyzed.
Safety was evaluated by renal function and blood test abnormalities, which were defined as values outside of the normal range.
Statistical analysis
The PSM method was used to match patients in the two groups (1:1). 24 Fisher’s exact or chi-square tests were used to compare categorical variables, and Student’s paired t-test (parametric variables) or the Mann–Whitney U test (for non-parametric variables) was performed for the comparison of continuous variables. Univariate and multivariate logistic regression analyses were used to identify the potential influencing factors for ALT normalization. All tests were two-sided, and p<0.05 was considered statistically significant. Furthermore, to assess the efficacy of treatment, the superiority test of the bicyclol group to the control group was conducted. The one-sided 97.5% confidence interval (CI) was given, and p<0.025 was considered statistically significant. All statistical analyses were performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
Propensity score matching
We examined the data of 25,927 available patients (Figure 1). Of these, 87 patients were included in the bicyclol group (received oral bicyclol), and 932 patients were included in the control group. After 1:1 PSM, 86 pairs of patients with various types of DILI (mixed type accounted for more) exhibited balance and comparable characteristics between the groups (Table 1). The median daily dose of bicyclol in the bicyclol group was 75 mg for a duration of 17.92±16.57 days (median: 12.50).
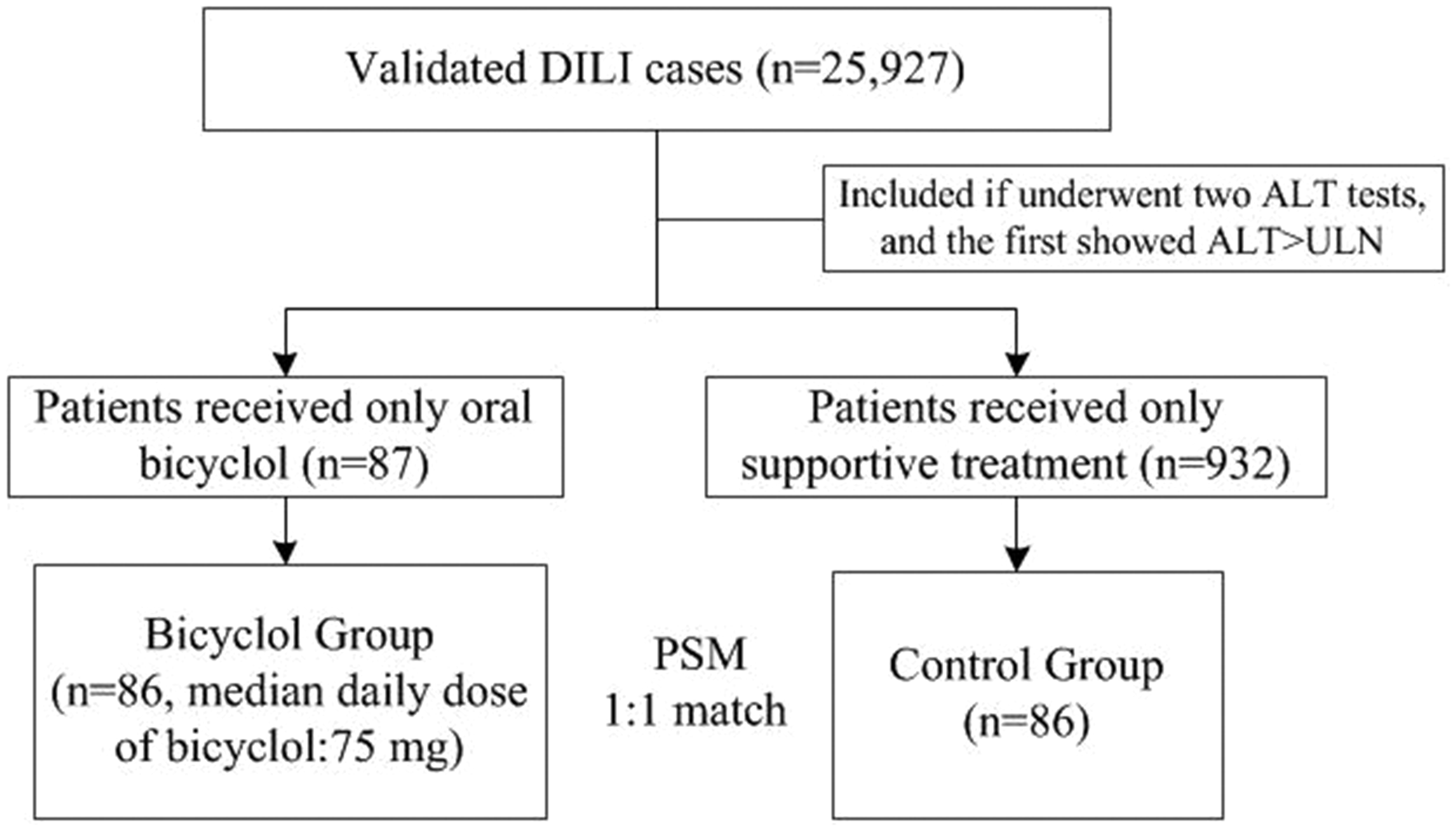
Data extraction and analysis flowchart.
Baseline characteristics before and after propensity score matching.

*The time interval between ALT tests (day)=last ALT test date−First ALT test date.
ALT, alanine aminotransferase; TBIL, total bilirubin; DILI, drug-induced liver injury.
ALT normalization rate and ALT50% reduction rate
The serum ALT levels are shown in Table 2. In the bicyclol group, the ALT normalization rate was 50.00%, which was significantly higher than the control group (24.42%, p=0.0005). Consistently, the bicyclol group exhibited a considerably higher ALT50% reduction rate (p<0.0001). The superiority test showed a boundary value of 9% for the ALT normalization rate (97.5% CI, 11.65–39.51) and 25% for the ALT50% reduction rate (97.5% CI, 25.10–56.30).
Comparison of ALT reduction rates between groups.

ALT, alanine aminotransferase; CI, confidence interval.
Influencing factors of ALT normalization
The potential influencing factors of the ALT normalization rate are shown in Table 3. In the univariate logistic regression analysis, the patients with ALT test intervals longer than 14 days demonstrated a better ALT normalization rate (odds ratio [OR], 2.19; 95% CI, 1.10–4.38; p=0.0263). As expected, bicyclol treatment significantly increased the ALT normalization rate (OR, 3.10; 95% CI, 1.62–5.92; p=0.0006). In the multivariate logic regression analysis, similar results were found. The interval between ALT tests and the grouping factor (bicyclol treatment) were two essential influencing factors (OR, 2.71; 95% CI, 1.23–5.97; p=0.0131 and OR, 3.53; 95% CI, 1.69–7.35; p=0.0008, respectively). Age was significantly correlated with ALT normalization in the univariate logistic regression analysis, but no statistical significance was found by multivariate logic regression. A sub-group analysis for the ALT normalization rate is shown in Table 4. The ALT normalization rate of the bicyclol group was higher than the control group in all sub-groups (dividing factors: sex, ALT baseline, and anti-tuberculosis drug as suspected drug or not).
Logistic regression analysis of ALT normalization.
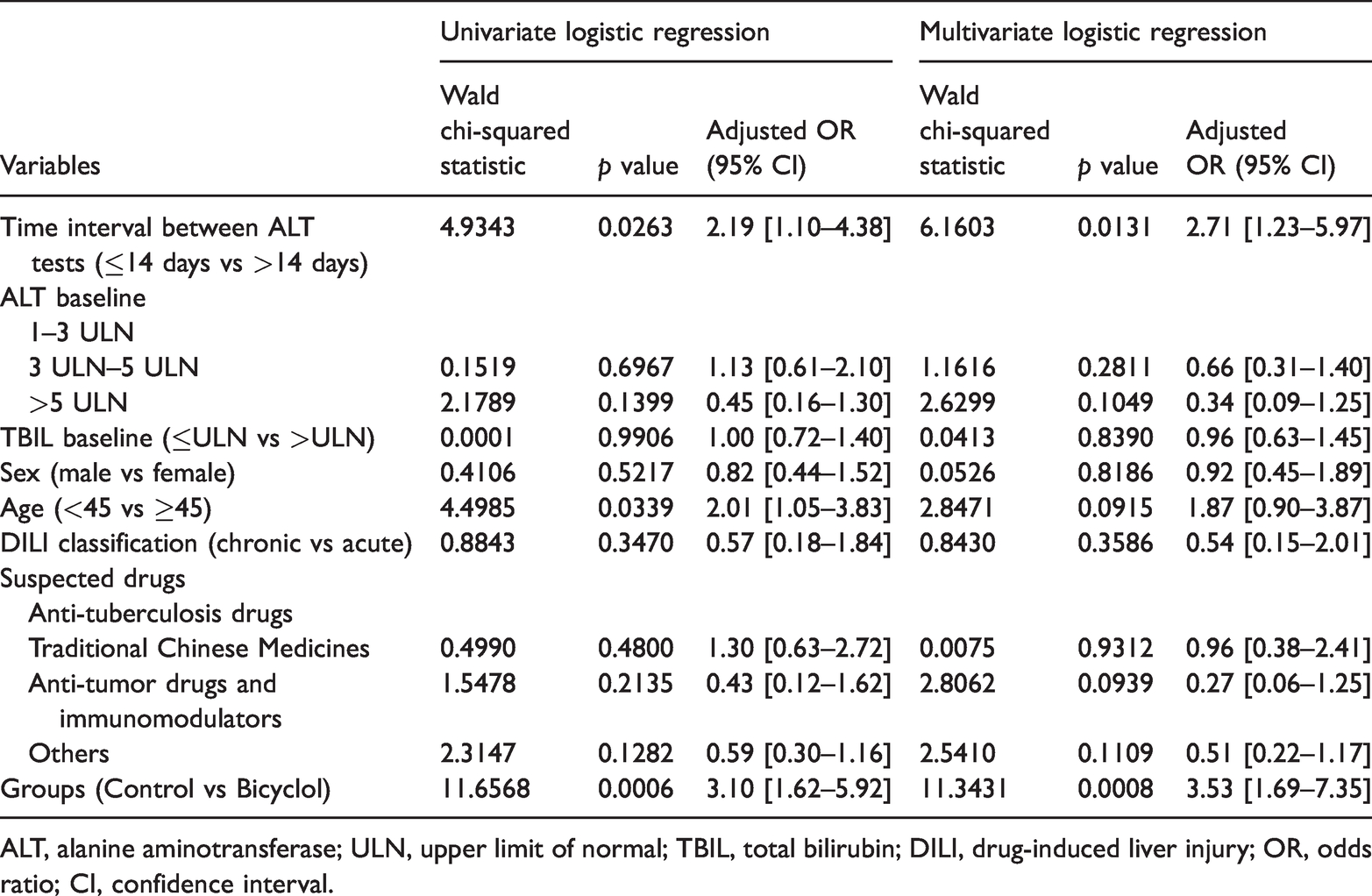
ALT, alanine aminotransferase; ULN, upper limit of normal; TBIL, total bilirubin; DILI, drug-induced liver injury; OR, odds ratio; CI, confidence interval.
Comparison of ALT normalization rates in the subgroup analysis.
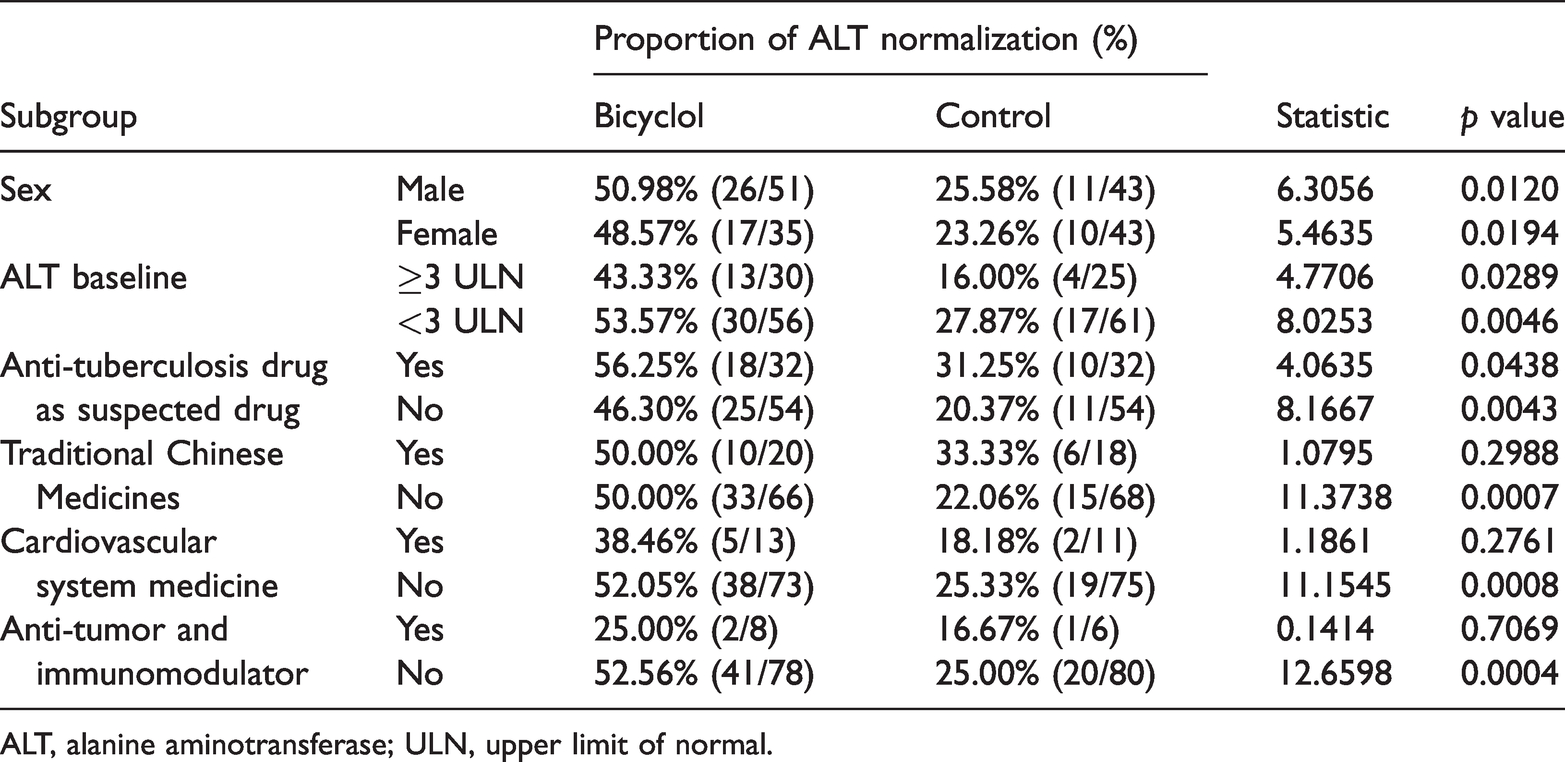
ALT, alanine aminotransferase; ULN, upper limit of normal.
Safety
As Table 5 shows, no differences in creatinine, blood urea nitrogen, hemoglobin, leukocytes, and platelet levels were found between the two groups.
Proportion of adverse events related to renal function and blood test abnormalities.

Discussion
DILI-R is a retrospective study with data from 308 medical centers in major cities across mainland China conducted to characterize DILI in hospitalized patients from 2012 to 2014, including the implicated drugs and clinical features, and estimate the incidence of DILI. 19 Using the DILI-R database with 25,927 patients, the present retrospective study demonstrates that oral bicyclol is effective for DILI, as indicated by an increase in the ALT normalization rate in patients treated with bicyclol compared with the patients who received only supportive care. There were no differences in the proportion of renal function impairment or blood abnormalities between the two groups.
Throughout mainland China, the annual incidence of DILI in the general population was estimated to be 23.80 per 100,000 people, 19 which is higher than the estimated incidences in Iceland (19.1/100,000), 4 France (13.9/100,000), 5 the United States (2.7/100,000), 25 Spain (3.42/100,000), 26 and Sweden (2.4/100,000). 27 Although NAC can reduce mortality in adult patients with ALF and grade I to II hepatic encephalopathy (including some patients with idiosyncratic DILI), 28 the efficacy of NAC for non-ALF DILI patients remains unexplored. Therefore, no treatments other than stopping the offending drug are available for patients with non-ALF DILI. However, discontinuation of the offending drug may sometimes affect the treatment of the patients’ primary diseases, such as cancer or tuberculosis. Based on our findings, bicyclol has potential for the treatment of DILI patients with elevated ALT caused by anti-tuberculosis drugs, traditional Chinese medicine, and others. Although the current data were obtained in a retrospective study, random sampling and baseline balance between the groups were achieved after PSM.
The pharmacological mechanisms underlying the protective effects of bicyclol on liver injury are multifaceted. Bicyclol may inhibit the expression/activity of key inflammatory regulatory factors, such as nuclear transcription factor-κB, tumor necrosis factor-α, interleukin-1β, transforming growth factor-β1, and inducible nitric oxide synthase. It attenuates inflammatory injury, oxidative damage, and pathological apoptosis of hepatocytes caused by reactive oxygen species.29–32 Previously, it has been suggested that bicyclol could safely and effectively treat DILI induced by multiple agents, such as anti-tuberculosis drugs,16,33 anti-immunological rejection drugs, 34 anti-rheumatic drugs, 35 anti-tumor drugs, 36 antipsychotic drugs, 37 statins, 17 and other drugs. Based on six RCTs, a meta-analysis demonstrated an efficacy rate of 91.11% in DILI patients treated with bicyclol, which was superior to the control (79.19%). 38 According to the American College of Gastroenterology Clinical Guidelines, 3 there are very limited therapies to manage DILI other than the withdrawal of the offending medication. Thus, bicyclol is a promising therapy for DILI.
Our results suggest that bicyclol is safe in terms of adverse events, which is consistent with the findings of the meta-analysis. 38 No severe adverse events were reported. However, because of the small sample sizes in all published reports, we could not detect the potentially serious adverse events occurring at a low frequency.
This study has some limitations. First, because of the retrospective nature of the present study, the details of many symptoms of DILI, such as itching, fever, nausea, jaundice, severe lethargy, and anorexia, were not available. It is unclear whether bicyclol can alleviate these symptoms and signs. Second, the retrospective characteristics of the study design leave it open to potential bias and confounding. We attempted to control this confounding using propensity score analyses, but we were unable to control for possible unmeasured variables.
Conclusions
The results of this retrospective study using a large nationwide database demonstrate that bicyclol is a potential candidate for patients with DILI. Because the present research was based on retrospective data, large-scale randomized control studies are still needed to confirm the efficacy and safety of bicyclol in the treatment of DILI.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Major Project of National Twelfth Five Plan (2012ZX09303-001) and the Major Project of National Thirteenth Five Plan (2017ZX09304016), the National Natural Science Foundation of China (NSFC 81670524), the Shanghai Shenkang Hospital Development Center (16CR2009A), and the Clinical Research Center, Shanghai Jiao Tong University School of Medicine (DLY201607).
